These Highlights Do Not Include All The Information Needed To Use Entecavir Tablets Safely And Effectively. See Full Prescribing Information For Entecavir Tablets.
92a5bebd-6408-463d-933c-bab8338a677e
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Severe acute exacerbations of hepatitis B have been reported in patients who have discontinued anti-hepatitis B therapy, including entecavir. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-hepatitis B therapy. If appropriate, initiation of anti-hepatitis B therapy may be warranted [see Warnings and Precautions (5.1) ] . Limited clinical experience suggests there is a potential for the development of resistance to HIV (human immunodeficiency virus) nucleoside reverse transcriptase inhibitors if entecavir is used to treat chronic hepatitis B virus (HBV) infection in patients with HIV infection that is not being treated. Therapy with entecavir is not recommended for HIV/HBV co-infected patients who are not also receiving highly active antiretroviral therapy (HAART) [see Warnings and Precautions (5.2) ] . Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogue inhibitors alone or in combination with antiretrovirals [see Warnings and Precautions (5.3) ] .
Indications and Usage
Entecavir is indicated for the treatment of chronic hepatitis B virus infection in adults and pediatric patients 2 years of age and older with evidence of active viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease.
Dosage and Administration
Nucleoside-inhibitor-treatment-naïve with compensated liver disease (greater than or equal to 16 years old): 0.5 mg once daily. ( 2.2 ) Nucleoside-inhibitor-treatment-naïve and lamivudine-experienced pediatric patients at least 2 years of age and weighing at least 10 kg: dosing is based on weight. ( 2.3 ) Lamivudine-refractory or known lamivudine or telbivudine resistance substitutions (greater than or equal to 16 years old): 1 mg once daily. ( 2.2 ) Decompensated liver disease (adults): 1 mg once daily. ( 2.2 ) Renal impairment: Dosage adjustment is recommended if creatinine clearance is less than 50 mL/min. ( 2.4 ) Entecavir tablets should be administered on an empty stomach. ( 2.1 )
Warnings and Precautions
Severe acute exacerbations of hepatitis B virus infection after discontinuation: Monitor hepatic function closely for at least several months. ( 5.1 , 6.1 ) Co-infection with HIV: Entecavir is not recommended unless the patient is also receiving HAART. ( 5.2 ) Lactic acidosis and severe hepatomegaly with steatosis: If suspected, treatment should be suspended. ( 5.3 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling: Exacerbations of hepatitis after discontinuation of treatment [see Boxed Warning , Warnings and Precautions (5.1) ] . Lactic acidosis and severe hepatomegaly with steatosis [see Boxed Warning , Warnings and Precautions (5.3) ] .
Drug Interactions
Since entecavir is primarily eliminated by the kidneys [see Clinical Pharmacology (12.3) ] , coadministration of entecavir with drugs that reduce renal function or compete for active tubular secretion may increase serum concentrations of either entecavir or the coadministered drug. Coadministration of entecavir with lamivudine, adefovir dipivoxil, or tenofovir disoproxil fumarate did not result in significant drug interactions. The effects of coadministration of entecavir with other drugs that are renally eliminated or are known to affect renal function have not been evaluated, and patients should be monitored closely for adverse events when entecavir is coadministered with such drugs.
Storage and Handling
Entecavir Tablets, USP are available in the following strengths and configurations of plastic bottles with child-resistant closures: Product Strength and Dosage Form Description Quantity NDC Number 0.5 mg film-coated tablet White round-shaped tablet, debossed with "C1" on one side. 30 tablets 42494-457-30 0.5 mg film-coated tablet White round-shaped tablet, debossed with "C1" on one side. 90 tablets 72836-005-90 1 mg film-coated tablet Pink round-shaped tablet, debossed with "C2" on one side. 30 tablets 42494-458-30
How Supplied
Entecavir Tablets, USP are available in the following strengths and configurations of plastic bottles with child-resistant closures: Product Strength and Dosage Form Description Quantity NDC Number 0.5 mg film-coated tablet White round-shaped tablet, debossed with "C1" on one side. 30 tablets 42494-457-30 0.5 mg film-coated tablet White round-shaped tablet, debossed with "C1" on one side. 90 tablets 72836-005-90 1 mg film-coated tablet Pink round-shaped tablet, debossed with "C2" on one side. 30 tablets 42494-458-30
Patient Information
Entecavir Tablets, USP Read this Patient Information before you start taking entecavir tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. What is the most important information I should know about entecavir tablets? 1. Your hepatitis B virus (HBV) infection may get worse if you stop taking entecavir tablets. This usually happens within 6 months after stopping entecavir tablets. Take entecavir tablets exactly as prescribed. Do not run out of entecavir tablets. Do not stop entecavir tablets without talking to your healthcare provider. Your healthcare provider should monitor your health and do regular blood tests to check your liver if you stop taking entecavir tablets. 2. If you have or get HIV that is not being treated with medicines while taking entecavir tablets, the HIV virus may develop resistance to certain HIV medicines and become harder to treat. You should get an HIV test before you start taking entecavir tablets and anytime after that when there is a chance you were exposed to HIV. Entecavir tablets can cause serious side effects including: 3. Lactic acidosis (buildup of acid in the blood). Some people who have taken entecavir tablets or medicines like entecavir tablets (a nucleoside analogue) have developed a serious condition called lactic acidosis. Lactic acidosis is a serious medical emergency that can cause death. Lactic acidosis must be treated in the hospital. Reports of lactic acidosis with entecavir tablets generally involved patients who were seriously ill due to their liver disease or other medical condition. Call your healthcare provider right away if you get any of the following signs or symptoms of lactic acidosis: You feel very weak or tired. You have unusual (not normal) muscle pain. You have trouble breathing. You have stomach pain with nausea and vomiting. You feel cold, especially in your arms and legs. You feel dizzy or light-headed. You have a fast or irregular heartbeat. 4. Serious liver problems. Some people who have taken medicines like entecavir tablets have developed serious liver problems called hepatotoxicity, with liver enlargement (hepatomegaly) and fat in the liver (steatosis). Hepatomegaly with steatosis is a serious medical emergency that can cause death. Call your healthcare provider right away if you get any of the following signs or symptoms of liver problems: Your skin or the white part of your eyes turns yellow (jaundice). Your urine turns dark. Your bowel movements (stools) turn light in color. You don't feel like eating food for several days or longer. You feel sick to your stomach (nausea). You have lower stomach pain. You may be more likely to get lactic acidosis or serious liver problems if you are female, very overweight, or have been taking nucleoside analogue medicines, like entecavir tablets, for a long time. What is Entecavir Tablets? Entecavir tablets is a prescription medicine used to treat chronic hepatitis B virus (HBV) in adults and children 2 years of age and older who have active liver disease. Entecavir tablets will not cure HBV. Entecavir tablets may lower the amount of HBV in the body. Entecavir tablets may lower the ability of HBV to multiply and infect new liver cells. Entecavir tablets may improve the condition of your liver. It is not known whether entecavir tablets will reduce your chances of getting liver cancer or liver damage (cirrhosis), which may be caused by chronic HBV infection. It is not known if entecavir tablets is safe and effective for use in children less than 2 years of age. What should I tell my healthcare provider before taking entecavir tablets? Before you take entecavir tablets, tell your healthcare provider if you: have kidney problems. Your entecavir tablets dose or schedule may need to be changed. have received medicine for HBV before. Some people, especially those who have already been treated with certain other medicines for HBV infection, may develop resistance to entecavir tablets. These people may have less benefit from treatment with entecavir tablets and may have worsening of hepatitis after resistant virus appears. Your healthcare provider will test the level of the hepatitis B virus in your blood regularly. have any other medical conditions. are pregnant or plan to become pregnant. It is not known if entecavir tablets will harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant. Antiretroviral Pregnancy Registry. If you take entecavir tablets while you are pregnant, talk to your healthcare provider about how you can take part in the entecavir tablets Antiretroviral Pregnancy Registry. The purpose of the pregnancy registry is to collect information about the health of you and your baby. are breastfeeding or plan to breastfeed. It is not known if entecavir tablets can pass into your breast milk. You and your healthcare provider should decide if you will take entecavir tablets or breastfeed. Tell your healthcare provider about all the medicines you take, including prescription and over-the- counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you have taken a medicine to treat HBV in the past. Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and pharmacist when you get a new medicine. How should I take Entecavir Tablets? Take entecavir tablets exactly as your healthcare provider tells you to. Your healthcare provider will tell you how much entecavir to take. Your healthcare provider will tell you when and how often to take entecavir tablets. Take entecavir tablets on an empty stomach, at least 2 hours after a meal and at least 2 hours before the next meal. Do not change your dose or stop taking entecavir tablets without talking to your healthcare provider. If you miss a dose of entecavir tablets, take it as soon as you remember and then take your next dose at its regular time. If it is almost time for your next dose, skip the missed dose. Do not take two doses at the same time. Call your healthcare provider or pharmacist if you are not sure what to do. When your supply of entecavir tablets starts to run low, call your healthcare provider or pharmacy for a refill. Do not run out of entecavir tablets. If you take too much entecavir, call your healthcare provider or go to the nearest emergency room right away. What are the possible side effects of entecavir tablets? Entecavir tablets may cause serious side effects. See " What is the most important information I should know about entecavir tablets? " The most common side effects of entecavir tablets include: headache tiredness dizziness nausea Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of entecavir tablets. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088. How should I store entecavir tablets? Store entecavir tablets at room temperature, between 68°F and 77°F (20°C and 25°C). Keep entecavir tablets in a tightly closed container. Store entecavir tablets in the original carton, and keep the carton out of the light. Safely throw away entecavir tablets that is out of date or no longer needed. Dispose of unused medicines through community take-back disposal programs when available or place entecavir tablets in an unrecognizable closed container in the household trash. Keep entecavir tablets and all medicines out of the reach of children. General information about the safe and effective use of entecavir tablets Entecavir tablets does not stop you from spreading the hepatitis B virus (HBV) to others by sex, sharing needles, or being exposed to your blood. Talk with your healthcare provider about safe sexual practices that protect your partner. Never share needles. Do not share personal items that can have blood or body fluids on them, like toothbrushes or razor blades. A shot (vaccine) is available to protect people at risk from becoming infected with HBV. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use entecavir tablets for a condition for which it was not prescribed. Do not give entecavir tablets to other people, even if they have the same symptoms you have. It may harm them. This Patient Information leaflet summarizes the most important information about entecavir tablets. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about entecavir tablets that is written for health professionals. For more information, call Conba USA Inc. at 1-609-924-0905. What are the ingredients in entecavir tablets? Active ingredient: entecavir Inactive ingredients in entecavir tablets: lactose monohydrate, microcrystalline cellulose, crospovidone, povidone, magnesium stearate. Tablet film-coat: titanium dioxide, hypromellose, polyethylene glycol 400, polysorbate 80 (0.5 mg tablet only), and iron oxide red (1 mg tablet only). Manufactured for: Cameron Pharmaceuticals Louisville, Kentucky 40245, USA Made in China This Patient Information has been approved by the U.S. Food and Drug Administration. Issued: August 2023 Brands listed are the trademarks of their respective owners.
Medication Information
Warnings and Precautions
Severe acute exacerbations of hepatitis B virus infection after discontinuation: Monitor hepatic function closely for at least several months. ( 5.1 , 6.1 ) Co-infection with HIV: Entecavir is not recommended unless the patient is also receiving HAART. ( 5.2 ) Lactic acidosis and severe hepatomegaly with steatosis: If suspected, treatment should be suspended. ( 5.3 )
Indications and Usage
Entecavir is indicated for the treatment of chronic hepatitis B virus infection in adults and pediatric patients 2 years of age and older with evidence of active viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease.
Dosage and Administration
Nucleoside-inhibitor-treatment-naïve with compensated liver disease (greater than or equal to 16 years old): 0.5 mg once daily. ( 2.2 ) Nucleoside-inhibitor-treatment-naïve and lamivudine-experienced pediatric patients at least 2 years of age and weighing at least 10 kg: dosing is based on weight. ( 2.3 ) Lamivudine-refractory or known lamivudine or telbivudine resistance substitutions (greater than or equal to 16 years old): 1 mg once daily. ( 2.2 ) Decompensated liver disease (adults): 1 mg once daily. ( 2.2 ) Renal impairment: Dosage adjustment is recommended if creatinine clearance is less than 50 mL/min. ( 2.4 ) Entecavir tablets should be administered on an empty stomach. ( 2.1 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling: Exacerbations of hepatitis after discontinuation of treatment [see Boxed Warning , Warnings and Precautions (5.1) ] . Lactic acidosis and severe hepatomegaly with steatosis [see Boxed Warning , Warnings and Precautions (5.3) ] .
Drug Interactions
Since entecavir is primarily eliminated by the kidneys [see Clinical Pharmacology (12.3) ] , coadministration of entecavir with drugs that reduce renal function or compete for active tubular secretion may increase serum concentrations of either entecavir or the coadministered drug. Coadministration of entecavir with lamivudine, adefovir dipivoxil, or tenofovir disoproxil fumarate did not result in significant drug interactions. The effects of coadministration of entecavir with other drugs that are renally eliminated or are known to affect renal function have not been evaluated, and patients should be monitored closely for adverse events when entecavir is coadministered with such drugs.
Storage and Handling
Entecavir Tablets, USP are available in the following strengths and configurations of plastic bottles with child-resistant closures: Product Strength and Dosage Form Description Quantity NDC Number 0.5 mg film-coated tablet White round-shaped tablet, debossed with "C1" on one side. 30 tablets 42494-457-30 0.5 mg film-coated tablet White round-shaped tablet, debossed with "C1" on one side. 90 tablets 72836-005-90 1 mg film-coated tablet Pink round-shaped tablet, debossed with "C2" on one side. 30 tablets 42494-458-30
How Supplied
Entecavir Tablets, USP are available in the following strengths and configurations of plastic bottles with child-resistant closures: Product Strength and Dosage Form Description Quantity NDC Number 0.5 mg film-coated tablet White round-shaped tablet, debossed with "C1" on one side. 30 tablets 42494-457-30 0.5 mg film-coated tablet White round-shaped tablet, debossed with "C1" on one side. 90 tablets 72836-005-90 1 mg film-coated tablet Pink round-shaped tablet, debossed with "C2" on one side. 30 tablets 42494-458-30
Patient Information
Entecavir Tablets, USP
Read this Patient Information before you start taking entecavir tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment.
What is the most important information I should know about entecavir tablets?
1. Your hepatitis B virus (HBV) infection may get worse if you stop taking entecavir tablets. This usually happens within 6 months after stopping entecavir tablets.
- Take entecavir tablets exactly as prescribed.
- Do not run out of entecavir tablets.
- Do not stop entecavir tablets without talking to your healthcare provider.
Your healthcare provider should monitor your health and do regular blood tests to check your liver if you stop taking entecavir tablets.
2. If you have or get HIV that is not being treated with medicines while taking entecavir tablets, the HIV virus may develop resistance to certain HIV medicines and become harder to treat. You should get an HIV test before you start taking entecavir tablets and anytime after that when there is a chance you were exposed to HIV.
Entecavir tablets can cause serious side effects including:
3. Lactic acidosis (buildup of acid in the blood). Some people who have taken entecavir tablets or medicines like entecavir tablets (a nucleoside analogue) have developed a serious condition called lactic acidosis. Lactic acidosis is a serious medical emergency that can cause death. Lactic acidosis must be treated in the hospital. Reports of lactic acidosis with entecavir tablets generally involved patients who were seriously ill due to their liver disease or other medical condition.
Call your healthcare provider right away if you get any of the following signs or symptoms of lactic acidosis:
- You feel very weak or tired.
- You have unusual (not normal) muscle pain.
- You have trouble breathing.
- You have stomach pain with nausea and vomiting.
- You feel cold, especially in your arms and legs.
- You feel dizzy or light-headed.
- You have a fast or irregular heartbeat.
4. Serious liver problems. Some people who have taken medicines like entecavir tablets have developed serious liver problems called hepatotoxicity, with liver enlargement (hepatomegaly) and fat in the liver (steatosis). Hepatomegaly with steatosis is a serious medical emergency that can cause death.
Call your healthcare provider right away if you get any of the following signs or symptoms of liver problems:
- Your skin or the white part of your eyes turns yellow (jaundice).
- Your urine turns dark.
- Your bowel movements (stools) turn light in color.
- You don't feel like eating food for several days or longer.
- You feel sick to your stomach (nausea).
- You have lower stomach pain.
- You may be more likely to get lactic acidosis or serious liver problems if you are female, very overweight, or have been taking nucleoside analogue medicines, like entecavir tablets, for a long time.
What is Entecavir Tablets?
Entecavir tablets is a prescription medicine used to treat chronic hepatitis B virus (HBV) in adults and children 2 years of age and older who have active liver disease.
- Entecavir tablets will not cure HBV.
- Entecavir tablets may lower the amount of HBV in the body.
- Entecavir tablets may lower the ability of HBV to multiply and infect new liver cells.
- Entecavir tablets may improve the condition of your liver.
- It is not known whether entecavir tablets will reduce your chances of getting liver cancer or liver damage (cirrhosis), which may be caused by chronic HBV infection.
It is not known if entecavir tablets is safe and effective for use in children less than 2 years of age.
What should I tell my healthcare provider before taking entecavir tablets?
Before you take entecavir tablets, tell your healthcare provider if you:
- have kidney problems. Your entecavir tablets dose or schedule may need to be changed.
- have received medicine for HBV before. Some people, especially those who have already been treated with certain other medicines for HBV infection, may develop resistance to entecavir tablets. These people may have less benefit from treatment with entecavir tablets and may have worsening of hepatitis after resistant virus appears. Your healthcare provider will test the level of the hepatitis B virus in your blood regularly.
- have any other medical conditions.
- are pregnant or plan to become pregnant. It is not known if entecavir tablets will harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
Antiretroviral Pregnancy Registry. If you take entecavir tablets while you are pregnant, talk to your healthcare provider about how you can take part in the entecavir tablets Antiretroviral Pregnancy Registry. The purpose of the pregnancy registry is to collect information about the health of you and your baby.
are breastfeeding or plan to breastfeed. It is not known if entecavir tablets can pass into your breast milk. You and your healthcare provider should decide if you will take entecavir tablets or breastfeed.
Tell your healthcare provider about all the medicines you take, including prescription and over-the- counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you have taken a medicine to treat HBV in the past.
Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and pharmacist when you get a new medicine.
How should I take Entecavir Tablets?
- Take entecavir tablets exactly as your healthcare provider tells you to.
- Your healthcare provider will tell you how much entecavir to take.
- Your healthcare provider will tell you when and how often to take entecavir tablets.
- Take entecavir tablets on an empty stomach, at least 2 hours after a meal and at least 2 hours before the next meal.
- Do not change your dose or stop taking entecavir tablets without talking to your healthcare provider.
- If you miss a dose of entecavir tablets, take it as soon as you remember and then take your next dose at its regular time. If it is almost time for your next dose, skip the missed dose. Do not take two doses at the same time. Call your healthcare provider or pharmacist if you are not sure what to do.
- When your supply of entecavir tablets starts to run low, call your healthcare provider or pharmacy for a refill. Do not run out of entecavir tablets.
- If you take too much entecavir, call your healthcare provider or go to the nearest emergency room right away.
What are the possible side effects of entecavir tablets?
Entecavir tablets may cause serious side effects. See "What is the most important information I should know about entecavir tablets?"
The most common side effects of entecavir tablets include:
- headache
- tiredness
- dizziness
- nausea
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of entecavir tablets. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
How should I store entecavir tablets?
- Store entecavir tablets at room temperature, between 68°F and 77°F (20°C and 25°C).
- Keep entecavir tablets in a tightly closed container.
- Store entecavir tablets in the original carton, and keep the carton out of the light.
Safely throw away entecavir tablets that is out of date or no longer needed. Dispose of unused medicines through community take-back disposal programs when available or place entecavir tablets in an unrecognizable closed container in the household trash.
Keep entecavir tablets and all medicines out of the reach of children.
General information about the safe and effective use of entecavir tablets
Entecavir tablets does not stop you from spreading the hepatitis B virus (HBV) to others by sex, sharing needles, or being exposed to your blood. Talk with your healthcare provider about safe sexual practices that protect your partner. Never share needles. Do not share personal items that can have blood or body fluids on them, like toothbrushes or razor blades. A shot (vaccine) is available to protect people at risk from becoming infected with HBV.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use entecavir tablets for a condition for which it was not prescribed. Do not give entecavir tablets to other people, even if they have the same symptoms you have. It may harm them.
This Patient Information leaflet summarizes the most important information about entecavir tablets. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about entecavir tablets that is written for health professionals.
For more information, call Conba USA Inc. at 1-609-924-0905.
What are the ingredients in entecavir tablets?
Active ingredient: entecavir
Inactive ingredients in entecavir tablets: lactose monohydrate, microcrystalline cellulose, crospovidone, povidone, magnesium stearate.
Tablet film-coat: titanium dioxide, hypromellose, polyethylene glycol 400, polysorbate 80 (0.5 mg tablet only), and iron oxide red (1 mg tablet only).
Manufactured for:
Cameron Pharmaceuticals
Louisville, Kentucky 40245, USA Made in China
This Patient Information has been approved by the U.S. Food and Drug Administration.
Issued: August 2023
Brands listed are the trademarks of their respective owners.
Description
Severe acute exacerbations of hepatitis B have been reported in patients who have discontinued anti-hepatitis B therapy, including entecavir. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-hepatitis B therapy. If appropriate, initiation of anti-hepatitis B therapy may be warranted [see Warnings and Precautions (5.1) ] . Limited clinical experience suggests there is a potential for the development of resistance to HIV (human immunodeficiency virus) nucleoside reverse transcriptase inhibitors if entecavir is used to treat chronic hepatitis B virus (HBV) infection in patients with HIV infection that is not being treated. Therapy with entecavir is not recommended for HIV/HBV co-infected patients who are not also receiving highly active antiretroviral therapy (HAART) [see Warnings and Precautions (5.2) ] . Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogue inhibitors alone or in combination with antiretrovirals [see Warnings and Precautions (5.3) ] .
Section 42229-5
Compensated Liver Disease
The recommended dose of entecavir for chronic hepatitis B virus infection in nucleoside- inhibitor-treatment-naïve adults and adolescents 16 years of age and older is 0.5 mg once daily.
The recommended dose of entecavir in adults and adolescents (at least 16 years of age) with a history of hepatitis B viremia while receiving lamivudine or known lamivudine or telbivudine resistance substitutions rtM204I/V with or without rtL180M, rtL80I/V, or rtV173L is 1 mg once daily.
Section 44425-7
Storage
Entecavir Tablets, USP should be stored in a tightly closed container at 25°C (77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Store in the outer carton to protect from light.
10 Overdosage
There is limited experience of entecavir overdosage reported in patients. Healthy subjects who received single entecavir doses up to 40 mg or multiple doses up to 20 mg/day for up to 14 days had no increase in or unexpected adverse events. If overdose occurs, the patient must be monitored for evidence of toxicity, and standard supportive treatment applied as necessary.
Following a single 1 mg dose of entecavir, a 4-hour hemodialysis session removed approximately 13% of the entecavir dose.
11 Description
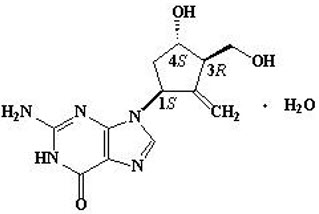
Entecavir is a guanosine nucleoside analogue with selective activity against HBV. The chemical name for entecavir is 2-amino-1,9-dihydro-9-[(1S,3R,4S)-4-hydroxy-3- (hydroxymethyl)-2-methylenecyclopentyl]-6H-purin-6-one, monohydrate. Its molecular formula is C12H15N5O3∙H2O, which corresponds to a molecular weight of 295.3. Entecavir has the following structural formula:
Entecavir is a white to off-white powder. It is slightly soluble in water (2.4 mg/mL), and the pH of the saturated solution in water is 7.9 at 25 °C ± 0.5 °C.
Entecavir film-coated tablets are available for oral administration in strengths of 0.5 mg and 1 mg of entecavir. Entecavir 0.5 mg and 1 mg film-coated tablets contain the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, crospovidone, povidone, and magnesium stearate. The tablet coating contains titanium dioxide, hypromellose, polyethylene glycol 400, polysorbate 80 (0.5 mg tablet only), and iron oxide red (1 mg tablet only).
FDA approved dissolution test specifications differ from USP.
8.4 Pediatric Use
Entecavir was evaluated in two clinical trials of pediatric subjects 2 years of age and older with HBeAg-positive chronic HBV infection and compensated liver disease. The exposure of entecavir in nucleoside-inhibitor-treatment-naïve and lamivudine-experienced pediatric subjects 2 years of age and older with HBeAg-positive chronic HBV infection and compensated liver disease receiving 0.015 mg/kg (up to 0.5 mg once daily) or 0.03 mg/kg (up to 1 mg once daily), respectively, was evaluated in Study AI463028. Safety and efficacy of the selected dose in treatment-naïve pediatric subjects were confirmed in Study AI463189, a randomized, placebo-controlled treatment trial [see Indications and Usage (1), Dosage and Administration (2.3), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)].
There are limited data available on the use of entecavir in lamivudine-experienced pediatric patients; entecavir should be used in these patients only if the potential benefit justifies the potential risk to the child. Since some pediatric patients may require long-term or even lifetime management of chronic active hepatitis B, consideration should be given to the impact of entecavir on future treatment options [see Microbiology (12.4)].
The efficacy and safety of entecavir have not been established in patients less than 2 years of age. Use of entecavir in this age group has not been evaluated because treatment of HBV in this age group is rarely required.
8.5 Geriatric Use
Clinical studies of entecavir did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects. Entecavir is substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.4)].
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are discussed in other sections of the labeling:
- Exacerbations of hepatitis after discontinuation of treatment [see Boxed Warning, Warnings and Precautions (5.1)].
- Lactic acidosis and severe hepatomegaly with steatosis [see Boxed Warning, Warnings and Precautions (5.3)].
7 Drug Interactions
Since entecavir is primarily eliminated by the kidneys [see Clinical Pharmacology (12.3)], coadministration of entecavir with drugs that reduce renal function or compete for active tubular secretion may increase serum concentrations of either entecavir or the coadministered drug.
Coadministration of entecavir with lamivudine, adefovir dipivoxil, or tenofovir disoproxil fumarate did not result in significant drug interactions. The effects of coadministration of entecavir with other drugs that are renally eliminated or are known to affect renal function have not been evaluated, and patients should be monitored closely for adverse events when entecavir is coadministered with such drugs.
2.4 Renal Impairment
In adult subjects with renal impairment, the apparent oral clearance of entecavir decreased as creatinine clearance decreased [see Clinical Pharmacology (12.3)]. Dosage adjustment is recommended for patients with creatinine clearance less than 50 mL/min, including patients on hemodialysis or continuous ambulatory peritoneal dialysis (CAPD), as shown in Table 2. The once-daily dosing regimens are preferred.
| Creatinine Clearance (mL/min) | Usual Dose (0.5 mg) | Lamivudine-Refractory or Decompensated Liver Disease (1mg) |
|---|---|---|
| 50 or greater | 0.5 mg once daily | 1 mg once daily |
| 30 to less than 50 | 0.25 mg once daily For doses less than 0.5 mg, Baraclude Oral Solution is recommended.
OR 0.5 mg every 48 hours |
0.5 mg once daily OR 1 mg every 48 hours |
| 10 to less than 30 | 0.15 mg once daily
OR 0.5 mg every 72 hours |
0.3 mg once daily
OR 1 mg every 72 hours |
| Less than 10 Hemodialysis If administered on a hemodialysis day, administer entecavir after the hemodialysis session. or CAPD |
0.05 mg once daily
OR 0.5 mg every 7 days |
0.1 mg once daily
OR 1 mg every 7 days |
Although there are insufficient data to recommend a specific dose adjustment of entecavir in pediatric patients with renal impairment, a reduction in the dose or an increase in the dosing interval similar to adjustments for adults should be considered.
8.7 Renal Impairment
Dosage adjustment of entecavir is recommended for patients with creatinine clearance less than 50 mL/min, including patients on hemodialysis or CAPD [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The single- and multiple-dose pharmacokinetics of entecavir were evaluated in healthy subjects and subjects with chronic hepatitis B virus infection.
2.5 Hepatic Impairment
No dosage adjustment is necessary for patients with hepatic impairment.
1 Indications and Usage
Entecavir is indicated for the treatment of chronic hepatitis B virus infection in adults and pediatric patients 2 years of age and older with evidence of active viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease.
2.6 Duration of Therapy
The optimal duration of treatment with entecavir for patients with chronic hepatitis B virus infection and the relationship between treatment and long-term outcomes such as cirrhosis and hepatocellular carcinoma are unknown.
12.1 Mechanism of Action
Entecavir is an antiviral drug against hepatitis B virus [see Microbiology (12.4)].
8.6 Racial/ethnic Groups
There are no significant racial differences in entecavir pharmacokinetics. The safety and efficacy of entecavir 0.5 mg once daily were assessed in a single-arm, open-label trial of HBeAg-positive or -negative, nucleoside-inhibitor-naïve, Black/African American (n=40) and Hispanic (n=6) subjects with chronic HBV infection. In this trial, 76% of subjects were male, the mean age was 42 years, 57% were HBeAg-positive, the mean baseline HBV DNA was 7.0 log10 IU/mL, and the mean baseline ALT was 162 U/L. At Week 48 of treatment, 32 of 46 (70%) subjects had HBV DNA <50 IU/mL (approximately 300 copies/mL), 31 of 46 (67%) subjects had ALT normalization (≤1 × ULN), and 12 of 26 (46%) HBeAg-positive subjects had HBe seroconversion. Safety data were similar to those observed in the larger controlled clinical trials.
Because of low enrollment, safety and efficacy have not been established in the US Hispanic population.
5 Warnings and Precautions
- Severe acute exacerbations of hepatitis B virus infection after discontinuation: Monitor hepatic function closely for at least several months. (5.1, 6.1)
- Co-infection with HIV: Entecavir is not recommended unless the patient is also receiving HAART. (5.2)
Lactic acidosis and severe hepatomegaly with steatosis: If suspected, treatment should be suspended. (5.3)
2 Dosage and Administration
- Nucleoside-inhibitor-treatment-naïve with compensated liver disease (greater than or equal to 16 years old): 0.5 mg once daily. (2.2)
- Nucleoside-inhibitor-treatment-naïve and lamivudine-experienced pediatric patients at least 2 years of age and weighing at least 10 kg: dosing is based on weight. (2.3)
- Lamivudine-refractory or known lamivudine or telbivudine resistance substitutions (greater than or equal to 16 years old): 1 mg once daily. (2.2)
- Decompensated liver disease (adults): 1 mg once daily. (2.2)
- Renal impairment: Dosage adjustment is recommended if creatinine clearance is less than 50 mL/min. (2.4)
Entecavir tablets should be administered on an empty stomach. (2.1)
2.1 Timing of Administration
Entecavir tablets should be administered on an empty stomach (at least 2 hours after a meal and 2 hours before the next meal).
3 Dosage Forms and Strengths
- Entecavir Tablets, USP, 0.5 mg, are white round-shaped, film-coated tablets, debossed with "C1" on one side.
- Entecavir Tablets, USP, 1 mg, are pink round-shaped, film-coated tablet, debossed with "C2" on one side.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
8 Use in Specific Populations
Liver transplant recipients: Limited data on safety and efficacy are available. (8.8)
8.8 Liver Transplant Recipients
The safety and efficacy of entecavir were assessed in a single-arm, open-label trial in 65 subjects who received a liver transplant for complications of chronic HBV infection. Eligible subjects who had HBV DNA less than 172 IU/mL (approximately 1000 copies/mL) at the time of transplant were treated with entecavir 1 mg once daily in addition to usual post-transplantation management, including hepatitis B immune globulin. The trial population was 82% male, 39% Caucasian, and 37% Asian, with a mean age of 49 years; 89% of subjects had HBeAg-negative disease at the time of transplant.
Four of the 65 subjects received 4 weeks or less of entecavir (2 deaths, 1 retransplantation, and 1 protocol violation) and were not considered evaluable. Of the 61 subjects who received more than 4 weeks of entecavir, 60 received hepatitis B immune globulin post-transplant. Fifty-three subjects (82% of all 65 subjects treated) completed the trial and had HBV DNA measurements at or after 72 weeks treatment post-transplant. All 53 subjects had HBV DNA <50 IU/mL (approximately 300 copies/mL). Eight evaluable subjects did not have HBV DNA data available at 72 weeks, including 3 subjects who died prior to study completion. No subjects had HBV DNA values ≥50 IU/mL while receiving entecavir (plus hepatitis B immune globulin). All 61 evaluable subjects lost HBsAg post-transplant; 2 of these subjects experienced recurrence of measurable HBsAg without recurrence of HBV viremia. This trial was not designed to determine whether addition of entecavir to hepatitis B immune globulin decreased the proportion of subjects with measurable HBV DNA post- transplant compared to hepatitis B immune globulin alone.
If entecavir treatment is determined to be necessary for a liver transplant recipient who has received or is receiving an immunosuppressant that may affect renal function, such as cyclosporine or tacrolimus, renal function must be carefully monitored both before and during treatment with entecavir [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
17 Patient Counseling Information
See FDA-approved patient labeling (Patient Information).
14.2 Outcomes in Pediatric Subjects
The pharmacokinetics, safety and antiviral activity of entecavir in pediatric subjects were initially assessed in Study AI463028. Twenty-four treatment-naïve and 19 lamivudine-experienced HBeAg-positive pediatric subjects 2 to less than 18 years of age with compensated chronic hepatitis B virus infection and elevated ALT were treated with entecavir 0.015 mg/kg (up to 0.5 mg) or 0.03 mg/kg (up to 1 mg) once daily. Fifty-eight percent (14/24) of treatment-naïve subjects and 47% (9/19) of lamivudine-experienced subjects achieved HBV DNA <50 IU/mL at Week 48 and ALT normalized in 83% (20/24) of treatment- naïve and 95% (18/19) of lamivudine-experienced subjects.
Safety and antiviral efficacy were confirmed in Study AI463189, a study of entecavir among 180 nucleoside-inhibitor-treatment-naïve pediatric subjects 2 to less than 18 years of age with HBeAg-positive chronic hepatitis B infection, compensated liver disease, and elevated ALT. Subjects were randomized 2:1 to receive blinded treatment with entecavir 0.015 mg/kg up to 0.5 mg/day (N=120) or placebo (N=60). The randomization was stratified by age group (2 to 6 years; >6 to 12 years; and >12 to <18 years). Baseline demographics and HBV disease characteristics were comparable between the 2 treatment arms and across age cohorts. At study entry, the mean HBV DNA was 8.1 log10 IU/mL and mean ALT was 103 U/L. The primary efficacy endpoint was a composite of HBeAg seroconversion and serum HBV DNA <50 IU/mL at Week 48 assessed in the first 123 subjects reaching 48 weeks of blinded treatment. Twenty-four percent (20/82) of subjects in the entecavir-treated group and 2% (1/41) of subjects in the placebo-treated group met the primary endpoint. Forty-six percent (38/82) of entecavir-treated subjects and 2% (1/41) of placebo-treated subjects achieved HBV DNA <50 IU/mL at Week 48. ALT normalization occurred in 67% (55/82) of entecavir- treated subjects and 22% (9/41) of placebo-treated subjects; 24% (20/82) of entecavir-treated subjects and 12% (5/41) of placebo-treated subjects had HBeAg seroconversion.
16 How Supplied/storage and Handling
Entecavir Tablets, USP are available in the following strengths and configurations of plastic bottles with child-resistant closures:
| Product Strength and Dosage Form | Description | Quantity | NDC Number |
|---|---|---|---|
| 0.5 mg film-coated tablet | White round-shaped tablet, debossed with "C1" on one side. | 30 tablets | 42494-457-30 |
| 0.5 mg film-coated tablet | White round-shaped tablet, debossed with "C1" on one side. | 90 tablets | 72836-005-90 |
| 1 mg film-coated tablet | Pink round-shaped tablet, debossed with "C2" on one side. | 30 tablets | 42494-458-30 |
5.2 Patients Co Infected With Hiv and Hbv
Entecavir has not been evaluated in HIV/HBV co-infected patients who were not simultaneously receiving effective HIV treatment. Limited clinical experience suggests there is a potential for the development of resistance to HIV nucleoside reverse transcriptase inhibitors if entecavir is used to treat chronic hepatitis B virus infection in patients with HIV infection that is not being treated [see Microbiology (12.4)]. Therefore, therapy with entecavir is not recommended for HIV/HBV co-infected patients who are not also receiving HAART. Before initiating entecavir therapy, HIV antibody testing should be offered to all patients. Entecavir has not been studied as a treatment for HIV infection and is not recommended for this use.
2.3 Recommended Dosage in Pediatric Patients
Table 1 describes the recommended dose of entecavir for pediatric patients 2 years of age or older and weighing at least 10 kg. The oral solution should be used for patients with body weight up to 30 kg.
| Recommended Once-Daily Dose of Oral Solution (mL) | ||
|---|---|---|
| Body Weight (kg) | Treatment-Naïve Patients Children with body weight greater than 30 kg should receive 10 mL (0.5 mg) of oral solution or one 0.5 mg tablet once daily.
|
Lamivudine-Experienced Patients Children with body weight greater than 30 kg should receive 20 mL (1 mg) of oral solution or one 1 mg tablet once daily.
|
| 10 to 11 | 3 | 6 |
| greater than 11 to 14 | 4 | 8 |
| greater than 14 to 17 | 5 | 10 |
| greater than 17 to 20 | 6 | 12 |
| greater than 20 to 23 | 7 | 14 |
| greater than 23 to 26 | 8 | 16 |
| greater than 26 to 30 | 9 | 18 |
| greater than 30 | 10 | 20 |
5.1 Severe Acute Exacerbations of Hepatitis B
Severe acute exacerbations of hepatitis B have been reported in patients who have discontinued anti- hepatitis B therapy, including entecavir [see Adverse Reactions (6.1)]. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-hepatitis B therapy. If appropriate, initiation of anti-hepatitis B therapy may be warranted.
Principal Display Panel 1 Mg Tablet Bottle Carton
NDC 42494-458-30
Rx Only
Entecavir
Tablets, USP
1 mg
30 Tablets
CAMERON
PHARMACEUTICALS™
Principal Display Panel 0.5 Mg Tablet Bottle Carton
NDC 42494-457-30
Rx Only
Entecavir
Tablets, USP
0.5 mg
30 Tablets
CAMERON
PHARMACEUTICALS™
5.3 Lactic Acidosis and Severe Hepatomegaly With Steatosis
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogue inhibitors, including entecavir, alone or in combination with antiretrovirals. A majority of these cases have been in women. Obesity and prolonged nucleoside inhibitor exposure may be risk factors. Particular caution should be exercised when administering nucleoside analogue inhibitors to any patient with known risk factors for liver disease; however, cases have also been reported in patients with no known risk factors.
Lactic acidosis with entecavir use has been reported, often in association with hepatic decompensation, other serious medical conditions, or drug exposures. Patients with decompensated liver disease may be at higher risk for lactic acidosis. Treatment with entecavir should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
Warning: Severe Acute Exacerbations of Hepatitis B, Patients Co Infected With Hiv and Hbv, and Lactic Acidosis and Hepatomegaly
Severe acute exacerbations of hepatitis B have been reported in patients who have discontinued anti-hepatitis B therapy, including entecavir. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-hepatitis B therapy. If appropriate, initiation of anti-hepatitis B therapy may be warranted [see Warnings and Precautions (5.1)].
Limited clinical experience suggests there is a potential for the development of resistance to HIV (human immunodeficiency virus) nucleoside reverse transcriptase inhibitors if entecavir is used to treat chronic hepatitis B virus (HBV) infection in patients with HIV infection that is not being treated. Therapy with entecavir is not recommended for HIV/HBV co-infected patients who are not also receiving highly active antiretroviral therapy (HAART) [see Warnings and Precautions (5.2)].
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogue inhibitors alone or in combination with antiretrovirals [see Warnings and Precautions (5.3)].
Structured Label Content
Section 42229-5 (42229-5)
Compensated Liver Disease
The recommended dose of entecavir for chronic hepatitis B virus infection in nucleoside- inhibitor-treatment-naïve adults and adolescents 16 years of age and older is 0.5 mg once daily.
The recommended dose of entecavir in adults and adolescents (at least 16 years of age) with a history of hepatitis B viremia while receiving lamivudine or known lamivudine or telbivudine resistance substitutions rtM204I/V with or without rtL180M, rtL80I/V, or rtV173L is 1 mg once daily.
Section 44425-7 (44425-7)
Storage
Entecavir Tablets, USP should be stored in a tightly closed container at 25°C (77°F); excursions permitted between 15°C and 30°C (59°F and 86°F) [see USP Controlled Room Temperature]. Store in the outer carton to protect from light.
10 Overdosage (10 OVERDOSAGE)
There is limited experience of entecavir overdosage reported in patients. Healthy subjects who received single entecavir doses up to 40 mg or multiple doses up to 20 mg/day for up to 14 days had no increase in or unexpected adverse events. If overdose occurs, the patient must be monitored for evidence of toxicity, and standard supportive treatment applied as necessary.
Following a single 1 mg dose of entecavir, a 4-hour hemodialysis session removed approximately 13% of the entecavir dose.
11 Description (11 DESCRIPTION)
Entecavir is a guanosine nucleoside analogue with selective activity against HBV. The chemical name for entecavir is 2-amino-1,9-dihydro-9-[(1S,3R,4S)-4-hydroxy-3- (hydroxymethyl)-2-methylenecyclopentyl]-6H-purin-6-one, monohydrate. Its molecular formula is C12H15N5O3∙H2O, which corresponds to a molecular weight of 295.3. Entecavir has the following structural formula:
Entecavir is a white to off-white powder. It is slightly soluble in water (2.4 mg/mL), and the pH of the saturated solution in water is 7.9 at 25 °C ± 0.5 °C.
Entecavir film-coated tablets are available for oral administration in strengths of 0.5 mg and 1 mg of entecavir. Entecavir 0.5 mg and 1 mg film-coated tablets contain the following inactive ingredients: lactose monohydrate, microcrystalline cellulose, crospovidone, povidone, and magnesium stearate. The tablet coating contains titanium dioxide, hypromellose, polyethylene glycol 400, polysorbate 80 (0.5 mg tablet only), and iron oxide red (1 mg tablet only).
FDA approved dissolution test specifications differ from USP.
8.4 Pediatric Use
Entecavir was evaluated in two clinical trials of pediatric subjects 2 years of age and older with HBeAg-positive chronic HBV infection and compensated liver disease. The exposure of entecavir in nucleoside-inhibitor-treatment-naïve and lamivudine-experienced pediatric subjects 2 years of age and older with HBeAg-positive chronic HBV infection and compensated liver disease receiving 0.015 mg/kg (up to 0.5 mg once daily) or 0.03 mg/kg (up to 1 mg once daily), respectively, was evaluated in Study AI463028. Safety and efficacy of the selected dose in treatment-naïve pediatric subjects were confirmed in Study AI463189, a randomized, placebo-controlled treatment trial [see Indications and Usage (1), Dosage and Administration (2.3), Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)].
There are limited data available on the use of entecavir in lamivudine-experienced pediatric patients; entecavir should be used in these patients only if the potential benefit justifies the potential risk to the child. Since some pediatric patients may require long-term or even lifetime management of chronic active hepatitis B, consideration should be given to the impact of entecavir on future treatment options [see Microbiology (12.4)].
The efficacy and safety of entecavir have not been established in patients less than 2 years of age. Use of entecavir in this age group has not been evaluated because treatment of HBV in this age group is rarely required.
8.5 Geriatric Use
Clinical studies of entecavir did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects. Entecavir is substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.4)].
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in other sections of the labeling:
- Exacerbations of hepatitis after discontinuation of treatment [see Boxed Warning, Warnings and Precautions (5.1)].
- Lactic acidosis and severe hepatomegaly with steatosis [see Boxed Warning, Warnings and Precautions (5.3)].
7 Drug Interactions (7 DRUG INTERACTIONS)
Since entecavir is primarily eliminated by the kidneys [see Clinical Pharmacology (12.3)], coadministration of entecavir with drugs that reduce renal function or compete for active tubular secretion may increase serum concentrations of either entecavir or the coadministered drug.
Coadministration of entecavir with lamivudine, adefovir dipivoxil, or tenofovir disoproxil fumarate did not result in significant drug interactions. The effects of coadministration of entecavir with other drugs that are renally eliminated or are known to affect renal function have not been evaluated, and patients should be monitored closely for adverse events when entecavir is coadministered with such drugs.
Patient Information
Entecavir Tablets, USP
Read this Patient Information before you start taking entecavir tablets and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment.
What is the most important information I should know about entecavir tablets?
1. Your hepatitis B virus (HBV) infection may get worse if you stop taking entecavir tablets. This usually happens within 6 months after stopping entecavir tablets.
- Take entecavir tablets exactly as prescribed.
- Do not run out of entecavir tablets.
- Do not stop entecavir tablets without talking to your healthcare provider.
Your healthcare provider should monitor your health and do regular blood tests to check your liver if you stop taking entecavir tablets.
2. If you have or get HIV that is not being treated with medicines while taking entecavir tablets, the HIV virus may develop resistance to certain HIV medicines and become harder to treat. You should get an HIV test before you start taking entecavir tablets and anytime after that when there is a chance you were exposed to HIV.
Entecavir tablets can cause serious side effects including:
3. Lactic acidosis (buildup of acid in the blood). Some people who have taken entecavir tablets or medicines like entecavir tablets (a nucleoside analogue) have developed a serious condition called lactic acidosis. Lactic acidosis is a serious medical emergency that can cause death. Lactic acidosis must be treated in the hospital. Reports of lactic acidosis with entecavir tablets generally involved patients who were seriously ill due to their liver disease or other medical condition.
Call your healthcare provider right away if you get any of the following signs or symptoms of lactic acidosis:
- You feel very weak or tired.
- You have unusual (not normal) muscle pain.
- You have trouble breathing.
- You have stomach pain with nausea and vomiting.
- You feel cold, especially in your arms and legs.
- You feel dizzy or light-headed.
- You have a fast or irregular heartbeat.
4. Serious liver problems. Some people who have taken medicines like entecavir tablets have developed serious liver problems called hepatotoxicity, with liver enlargement (hepatomegaly) and fat in the liver (steatosis). Hepatomegaly with steatosis is a serious medical emergency that can cause death.
Call your healthcare provider right away if you get any of the following signs or symptoms of liver problems:
- Your skin or the white part of your eyes turns yellow (jaundice).
- Your urine turns dark.
- Your bowel movements (stools) turn light in color.
- You don't feel like eating food for several days or longer.
- You feel sick to your stomach (nausea).
- You have lower stomach pain.
- You may be more likely to get lactic acidosis or serious liver problems if you are female, very overweight, or have been taking nucleoside analogue medicines, like entecavir tablets, for a long time.
What is Entecavir Tablets?
Entecavir tablets is a prescription medicine used to treat chronic hepatitis B virus (HBV) in adults and children 2 years of age and older who have active liver disease.
- Entecavir tablets will not cure HBV.
- Entecavir tablets may lower the amount of HBV in the body.
- Entecavir tablets may lower the ability of HBV to multiply and infect new liver cells.
- Entecavir tablets may improve the condition of your liver.
- It is not known whether entecavir tablets will reduce your chances of getting liver cancer or liver damage (cirrhosis), which may be caused by chronic HBV infection.
It is not known if entecavir tablets is safe and effective for use in children less than 2 years of age.
What should I tell my healthcare provider before taking entecavir tablets?
Before you take entecavir tablets, tell your healthcare provider if you:
- have kidney problems. Your entecavir tablets dose or schedule may need to be changed.
- have received medicine for HBV before. Some people, especially those who have already been treated with certain other medicines for HBV infection, may develop resistance to entecavir tablets. These people may have less benefit from treatment with entecavir tablets and may have worsening of hepatitis after resistant virus appears. Your healthcare provider will test the level of the hepatitis B virus in your blood regularly.
- have any other medical conditions.
- are pregnant or plan to become pregnant. It is not known if entecavir tablets will harm your unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
Antiretroviral Pregnancy Registry. If you take entecavir tablets while you are pregnant, talk to your healthcare provider about how you can take part in the entecavir tablets Antiretroviral Pregnancy Registry. The purpose of the pregnancy registry is to collect information about the health of you and your baby.
are breastfeeding or plan to breastfeed. It is not known if entecavir tablets can pass into your breast milk. You and your healthcare provider should decide if you will take entecavir tablets or breastfeed.
Tell your healthcare provider about all the medicines you take, including prescription and over-the- counter medicines, vitamins, and herbal supplements. Especially tell your healthcare provider if you have taken a medicine to treat HBV in the past.
Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and pharmacist when you get a new medicine.
How should I take Entecavir Tablets?
- Take entecavir tablets exactly as your healthcare provider tells you to.
- Your healthcare provider will tell you how much entecavir to take.
- Your healthcare provider will tell you when and how often to take entecavir tablets.
- Take entecavir tablets on an empty stomach, at least 2 hours after a meal and at least 2 hours before the next meal.
- Do not change your dose or stop taking entecavir tablets without talking to your healthcare provider.
- If you miss a dose of entecavir tablets, take it as soon as you remember and then take your next dose at its regular time. If it is almost time for your next dose, skip the missed dose. Do not take two doses at the same time. Call your healthcare provider or pharmacist if you are not sure what to do.
- When your supply of entecavir tablets starts to run low, call your healthcare provider or pharmacy for a refill. Do not run out of entecavir tablets.
- If you take too much entecavir, call your healthcare provider or go to the nearest emergency room right away.
What are the possible side effects of entecavir tablets?
Entecavir tablets may cause serious side effects. See "What is the most important information I should know about entecavir tablets?"
The most common side effects of entecavir tablets include:
- headache
- tiredness
- dizziness
- nausea
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of entecavir tablets. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
How should I store entecavir tablets?
- Store entecavir tablets at room temperature, between 68°F and 77°F (20°C and 25°C).
- Keep entecavir tablets in a tightly closed container.
- Store entecavir tablets in the original carton, and keep the carton out of the light.
Safely throw away entecavir tablets that is out of date or no longer needed. Dispose of unused medicines through community take-back disposal programs when available or place entecavir tablets in an unrecognizable closed container in the household trash.
Keep entecavir tablets and all medicines out of the reach of children.
General information about the safe and effective use of entecavir tablets
Entecavir tablets does not stop you from spreading the hepatitis B virus (HBV) to others by sex, sharing needles, or being exposed to your blood. Talk with your healthcare provider about safe sexual practices that protect your partner. Never share needles. Do not share personal items that can have blood or body fluids on them, like toothbrushes or razor blades. A shot (vaccine) is available to protect people at risk from becoming infected with HBV.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use entecavir tablets for a condition for which it was not prescribed. Do not give entecavir tablets to other people, even if they have the same symptoms you have. It may harm them.
This Patient Information leaflet summarizes the most important information about entecavir tablets. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about entecavir tablets that is written for health professionals.
For more information, call Conba USA Inc. at 1-609-924-0905.
What are the ingredients in entecavir tablets?
Active ingredient: entecavir
Inactive ingredients in entecavir tablets: lactose monohydrate, microcrystalline cellulose, crospovidone, povidone, magnesium stearate.
Tablet film-coat: titanium dioxide, hypromellose, polyethylene glycol 400, polysorbate 80 (0.5 mg tablet only), and iron oxide red (1 mg tablet only).
Manufactured for:
Cameron Pharmaceuticals
Louisville, Kentucky 40245, USA Made in China
This Patient Information has been approved by the U.S. Food and Drug Administration.
Issued: August 2023
Brands listed are the trademarks of their respective owners.
2.4 Renal Impairment
In adult subjects with renal impairment, the apparent oral clearance of entecavir decreased as creatinine clearance decreased [see Clinical Pharmacology (12.3)]. Dosage adjustment is recommended for patients with creatinine clearance less than 50 mL/min, including patients on hemodialysis or continuous ambulatory peritoneal dialysis (CAPD), as shown in Table 2. The once-daily dosing regimens are preferred.
| Creatinine Clearance (mL/min) | Usual Dose (0.5 mg) | Lamivudine-Refractory or Decompensated Liver Disease (1mg) |
|---|---|---|
| 50 or greater | 0.5 mg once daily | 1 mg once daily |
| 30 to less than 50 | 0.25 mg once daily For doses less than 0.5 mg, Baraclude Oral Solution is recommended.
OR 0.5 mg every 48 hours |
0.5 mg once daily OR 1 mg every 48 hours |
| 10 to less than 30 | 0.15 mg once daily
OR 0.5 mg every 72 hours |
0.3 mg once daily
OR 1 mg every 72 hours |
| Less than 10 Hemodialysis If administered on a hemodialysis day, administer entecavir after the hemodialysis session. or CAPD |
0.05 mg once daily
OR 0.5 mg every 7 days |
0.1 mg once daily
OR 1 mg every 7 days |
Although there are insufficient data to recommend a specific dose adjustment of entecavir in pediatric patients with renal impairment, a reduction in the dose or an increase in the dosing interval similar to adjustments for adults should be considered.
8.7 Renal Impairment
Dosage adjustment of entecavir is recommended for patients with creatinine clearance less than 50 mL/min, including patients on hemodialysis or CAPD [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The single- and multiple-dose pharmacokinetics of entecavir were evaluated in healthy subjects and subjects with chronic hepatitis B virus infection.
2.5 Hepatic Impairment
No dosage adjustment is necessary for patients with hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Entecavir is indicated for the treatment of chronic hepatitis B virus infection in adults and pediatric patients 2 years of age and older with evidence of active viral replication and either evidence of persistent elevations in serum aminotransferases (ALT or AST) or histologically active disease.
2.6 Duration of Therapy
The optimal duration of treatment with entecavir for patients with chronic hepatitis B virus infection and the relationship between treatment and long-term outcomes such as cirrhosis and hepatocellular carcinoma are unknown.
12.1 Mechanism of Action
Entecavir is an antiviral drug against hepatitis B virus [see Microbiology (12.4)].
8.6 Racial/ethnic Groups (8.6 Racial/Ethnic Groups)
There are no significant racial differences in entecavir pharmacokinetics. The safety and efficacy of entecavir 0.5 mg once daily were assessed in a single-arm, open-label trial of HBeAg-positive or -negative, nucleoside-inhibitor-naïve, Black/African American (n=40) and Hispanic (n=6) subjects with chronic HBV infection. In this trial, 76% of subjects were male, the mean age was 42 years, 57% were HBeAg-positive, the mean baseline HBV DNA was 7.0 log10 IU/mL, and the mean baseline ALT was 162 U/L. At Week 48 of treatment, 32 of 46 (70%) subjects had HBV DNA <50 IU/mL (approximately 300 copies/mL), 31 of 46 (67%) subjects had ALT normalization (≤1 × ULN), and 12 of 26 (46%) HBeAg-positive subjects had HBe seroconversion. Safety data were similar to those observed in the larger controlled clinical trials.
Because of low enrollment, safety and efficacy have not been established in the US Hispanic population.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Severe acute exacerbations of hepatitis B virus infection after discontinuation: Monitor hepatic function closely for at least several months. (5.1, 6.1)
- Co-infection with HIV: Entecavir is not recommended unless the patient is also receiving HAART. (5.2)
Lactic acidosis and severe hepatomegaly with steatosis: If suspected, treatment should be suspended. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Nucleoside-inhibitor-treatment-naïve with compensated liver disease (greater than or equal to 16 years old): 0.5 mg once daily. (2.2)
- Nucleoside-inhibitor-treatment-naïve and lamivudine-experienced pediatric patients at least 2 years of age and weighing at least 10 kg: dosing is based on weight. (2.3)
- Lamivudine-refractory or known lamivudine or telbivudine resistance substitutions (greater than or equal to 16 years old): 1 mg once daily. (2.2)
- Decompensated liver disease (adults): 1 mg once daily. (2.2)
- Renal impairment: Dosage adjustment is recommended if creatinine clearance is less than 50 mL/min. (2.4)
Entecavir tablets should be administered on an empty stomach. (2.1)
2.1 Timing of Administration
Entecavir tablets should be administered on an empty stomach (at least 2 hours after a meal and 2 hours before the next meal).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Entecavir Tablets, USP, 0.5 mg, are white round-shaped, film-coated tablets, debossed with "C1" on one side.
- Entecavir Tablets, USP, 1 mg, are pink round-shaped, film-coated tablet, debossed with "C2" on one side.
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Liver transplant recipients: Limited data on safety and efficacy are available. (8.8)
8.8 Liver Transplant Recipients
The safety and efficacy of entecavir were assessed in a single-arm, open-label trial in 65 subjects who received a liver transplant for complications of chronic HBV infection. Eligible subjects who had HBV DNA less than 172 IU/mL (approximately 1000 copies/mL) at the time of transplant were treated with entecavir 1 mg once daily in addition to usual post-transplantation management, including hepatitis B immune globulin. The trial population was 82% male, 39% Caucasian, and 37% Asian, with a mean age of 49 years; 89% of subjects had HBeAg-negative disease at the time of transplant.
Four of the 65 subjects received 4 weeks or less of entecavir (2 deaths, 1 retransplantation, and 1 protocol violation) and were not considered evaluable. Of the 61 subjects who received more than 4 weeks of entecavir, 60 received hepatitis B immune globulin post-transplant. Fifty-three subjects (82% of all 65 subjects treated) completed the trial and had HBV DNA measurements at or after 72 weeks treatment post-transplant. All 53 subjects had HBV DNA <50 IU/mL (approximately 300 copies/mL). Eight evaluable subjects did not have HBV DNA data available at 72 weeks, including 3 subjects who died prior to study completion. No subjects had HBV DNA values ≥50 IU/mL while receiving entecavir (plus hepatitis B immune globulin). All 61 evaluable subjects lost HBsAg post-transplant; 2 of these subjects experienced recurrence of measurable HBsAg without recurrence of HBV viremia. This trial was not designed to determine whether addition of entecavir to hepatitis B immune globulin decreased the proportion of subjects with measurable HBV DNA post- transplant compared to hepatitis B immune globulin alone.
If entecavir treatment is determined to be necessary for a liver transplant recipient who has received or is receiving an immunosuppressant that may affect renal function, such as cyclosporine or tacrolimus, renal function must be carefully monitored both before and during treatment with entecavir [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA-approved patient labeling (Patient Information).
14.2 Outcomes in Pediatric Subjects
The pharmacokinetics, safety and antiviral activity of entecavir in pediatric subjects were initially assessed in Study AI463028. Twenty-four treatment-naïve and 19 lamivudine-experienced HBeAg-positive pediatric subjects 2 to less than 18 years of age with compensated chronic hepatitis B virus infection and elevated ALT were treated with entecavir 0.015 mg/kg (up to 0.5 mg) or 0.03 mg/kg (up to 1 mg) once daily. Fifty-eight percent (14/24) of treatment-naïve subjects and 47% (9/19) of lamivudine-experienced subjects achieved HBV DNA <50 IU/mL at Week 48 and ALT normalized in 83% (20/24) of treatment- naïve and 95% (18/19) of lamivudine-experienced subjects.
Safety and antiviral efficacy were confirmed in Study AI463189, a study of entecavir among 180 nucleoside-inhibitor-treatment-naïve pediatric subjects 2 to less than 18 years of age with HBeAg-positive chronic hepatitis B infection, compensated liver disease, and elevated ALT. Subjects were randomized 2:1 to receive blinded treatment with entecavir 0.015 mg/kg up to 0.5 mg/day (N=120) or placebo (N=60). The randomization was stratified by age group (2 to 6 years; >6 to 12 years; and >12 to <18 years). Baseline demographics and HBV disease characteristics were comparable between the 2 treatment arms and across age cohorts. At study entry, the mean HBV DNA was 8.1 log10 IU/mL and mean ALT was 103 U/L. The primary efficacy endpoint was a composite of HBeAg seroconversion and serum HBV DNA <50 IU/mL at Week 48 assessed in the first 123 subjects reaching 48 weeks of blinded treatment. Twenty-four percent (20/82) of subjects in the entecavir-treated group and 2% (1/41) of subjects in the placebo-treated group met the primary endpoint. Forty-six percent (38/82) of entecavir-treated subjects and 2% (1/41) of placebo-treated subjects achieved HBV DNA <50 IU/mL at Week 48. ALT normalization occurred in 67% (55/82) of entecavir- treated subjects and 22% (9/41) of placebo-treated subjects; 24% (20/82) of entecavir-treated subjects and 12% (5/41) of placebo-treated subjects had HBeAg seroconversion.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Entecavir Tablets, USP are available in the following strengths and configurations of plastic bottles with child-resistant closures:
| Product Strength and Dosage Form | Description | Quantity | NDC Number |
|---|---|---|---|
| 0.5 mg film-coated tablet | White round-shaped tablet, debossed with "C1" on one side. | 30 tablets | 42494-457-30 |
| 0.5 mg film-coated tablet | White round-shaped tablet, debossed with "C1" on one side. | 90 tablets | 72836-005-90 |
| 1 mg film-coated tablet | Pink round-shaped tablet, debossed with "C2" on one side. | 30 tablets | 42494-458-30 |
5.2 Patients Co Infected With Hiv and Hbv (5.2 Patients Co-infected with HIV and HBV)
Entecavir has not been evaluated in HIV/HBV co-infected patients who were not simultaneously receiving effective HIV treatment. Limited clinical experience suggests there is a potential for the development of resistance to HIV nucleoside reverse transcriptase inhibitors if entecavir is used to treat chronic hepatitis B virus infection in patients with HIV infection that is not being treated [see Microbiology (12.4)]. Therefore, therapy with entecavir is not recommended for HIV/HBV co-infected patients who are not also receiving HAART. Before initiating entecavir therapy, HIV antibody testing should be offered to all patients. Entecavir has not been studied as a treatment for HIV infection and is not recommended for this use.
2.3 Recommended Dosage in Pediatric Patients
Table 1 describes the recommended dose of entecavir for pediatric patients 2 years of age or older and weighing at least 10 kg. The oral solution should be used for patients with body weight up to 30 kg.
| Recommended Once-Daily Dose of Oral Solution (mL) | ||
|---|---|---|
| Body Weight (kg) | Treatment-Naïve Patients Children with body weight greater than 30 kg should receive 10 mL (0.5 mg) of oral solution or one 0.5 mg tablet once daily.
|
Lamivudine-Experienced Patients Children with body weight greater than 30 kg should receive 20 mL (1 mg) of oral solution or one 1 mg tablet once daily.
|
| 10 to 11 | 3 | 6 |
| greater than 11 to 14 | 4 | 8 |
| greater than 14 to 17 | 5 | 10 |
| greater than 17 to 20 | 6 | 12 |
| greater than 20 to 23 | 7 | 14 |
| greater than 23 to 26 | 8 | 16 |
| greater than 26 to 30 | 9 | 18 |
| greater than 30 | 10 | 20 |
5.1 Severe Acute Exacerbations of Hepatitis B
Severe acute exacerbations of hepatitis B have been reported in patients who have discontinued anti- hepatitis B therapy, including entecavir [see Adverse Reactions (6.1)]. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-hepatitis B therapy. If appropriate, initiation of anti-hepatitis B therapy may be warranted.
Principal Display Panel 1 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Carton)
NDC 42494-458-30
Rx Only
Entecavir
Tablets, USP
1 mg
30 Tablets
CAMERON
PHARMACEUTICALS™
Principal Display Panel 0.5 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 0.5 mg Tablet Bottle Carton)
NDC 42494-457-30
Rx Only
Entecavir
Tablets, USP
0.5 mg
30 Tablets
CAMERON
PHARMACEUTICALS™
5.3 Lactic Acidosis and Severe Hepatomegaly With Steatosis (5.3 Lactic Acidosis and Severe Hepatomegaly with Steatosis)
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogue inhibitors, including entecavir, alone or in combination with antiretrovirals. A majority of these cases have been in women. Obesity and prolonged nucleoside inhibitor exposure may be risk factors. Particular caution should be exercised when administering nucleoside analogue inhibitors to any patient with known risk factors for liver disease; however, cases have also been reported in patients with no known risk factors.
Lactic acidosis with entecavir use has been reported, often in association with hepatic decompensation, other serious medical conditions, or drug exposures. Patients with decompensated liver disease may be at higher risk for lactic acidosis. Treatment with entecavir should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
Warning: Severe Acute Exacerbations of Hepatitis B, Patients Co Infected With Hiv and Hbv, and Lactic Acidosis and Hepatomegaly (WARNING: SEVERE ACUTE EXACERBATIONS OF HEPATITIS B, PATIENTS CO- INFECTED WITH HIV AND HBV, AND LACTIC ACIDOSIS AND HEPATOMEGALY)
Severe acute exacerbations of hepatitis B have been reported in patients who have discontinued anti-hepatitis B therapy, including entecavir. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who discontinue anti-hepatitis B therapy. If appropriate, initiation of anti-hepatitis B therapy may be warranted [see Warnings and Precautions (5.1)].
Limited clinical experience suggests there is a potential for the development of resistance to HIV (human immunodeficiency virus) nucleoside reverse transcriptase inhibitors if entecavir is used to treat chronic hepatitis B virus (HBV) infection in patients with HIV infection that is not being treated. Therapy with entecavir is not recommended for HIV/HBV co-infected patients who are not also receiving highly active antiretroviral therapy (HAART) [see Warnings and Precautions (5.2)].
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogue inhibitors alone or in combination with antiretrovirals [see Warnings and Precautions (5.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:33.435039 · Updated: 2026-03-14T22:27:27.393048