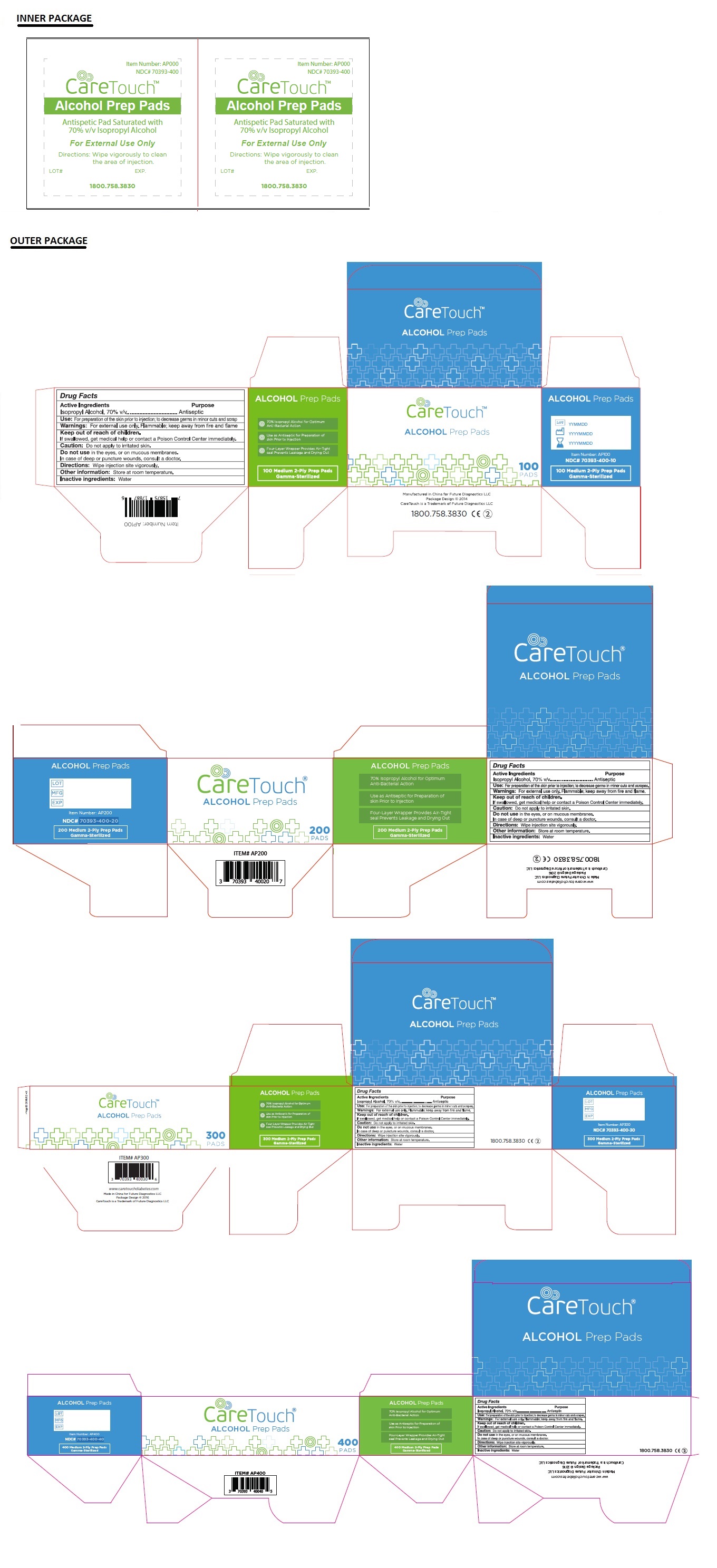
Caretouch Alcohol Prep Pad
91cfd7bf-9f42-49d5-b20d-281779d5272d
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Isopropyl Alcohol, 70% V/V
Purpose
Antiseptic
Medication Information
Warnings and Precautions
Warnings: For external use only.
Flammable; keep away from the fire and flame.
Caution: Do not apply to irritated skin
Do not use in the eyes or on the mucous membranes
In case of deep or puncture wounds, consult a doctor.
Indications and Usage
Use: For preparation of the skin prior to injection: to decrease germs in minor cuts and scrapes
Purpose
Antiseptic
Dosage and Administration
Directions: Wipe injection site vigorously.
Description
Isopropyl Alcohol, 70% V/V
Section 42229-5
One Pad Saturated with 70% Isopropyl Alcohol
- 70% Isopropyl Alcohol for Optimum Anti-Bacterial Action
- Use as Antiseptic for Preparation of skin Prior to Injection
- Four-Layer Wrapper Provides Air-Tight seal Prevents Leakage and Drying Out
Made in China for Future Diagnostics LLC
CareTouch is a Trademark of Future Diagnostics LLC
Section 44425-7
Other information: Store at room temperature
Section 51727-6
Inactive ingredient: Water
Packaging
Drug Facts
Active Ingredient
Isopropyl Alcohol, 70% V/V
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center immediately.
Structured Label Content
Indications and Usage (34067-9)
Use: For preparation of the skin prior to injection: to decrease germs in minor cuts and scrapes
Dosage and Administration (34068-7)
Directions: Wipe injection site vigorously.
Warnings and Precautions (34071-1)
Warnings: For external use only.
Flammable; keep away from the fire and flame.
Caution: Do not apply to irritated skin
Do not use in the eyes or on the mucous membranes
In case of deep or puncture wounds, consult a doctor.
Section 42229-5 (42229-5)
One Pad Saturated with 70% Isopropyl Alcohol
- 70% Isopropyl Alcohol for Optimum Anti-Bacterial Action
- Use as Antiseptic for Preparation of skin Prior to Injection
- Four-Layer Wrapper Provides Air-Tight seal Prevents Leakage and Drying Out
Made in China for Future Diagnostics LLC
CareTouch is a Trademark of Future Diagnostics LLC
Section 44425-7 (44425-7)
Other information: Store at room temperature
Section 51727-6 (51727-6)
Inactive ingredient: Water
Purpose
Antiseptic
Packaging
Drug Facts
Active Ingredient
Isopropyl Alcohol, 70% V/V
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center immediately.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:53.184714 · Updated: 2026-03-14T23:13:10.446567