No-weak-nails
9158cd3f-4c27-40e9-bc07-fd560375c3a1
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
To improve nails
Medication Information
Purpose
To improve nails
Description
Drug Facts
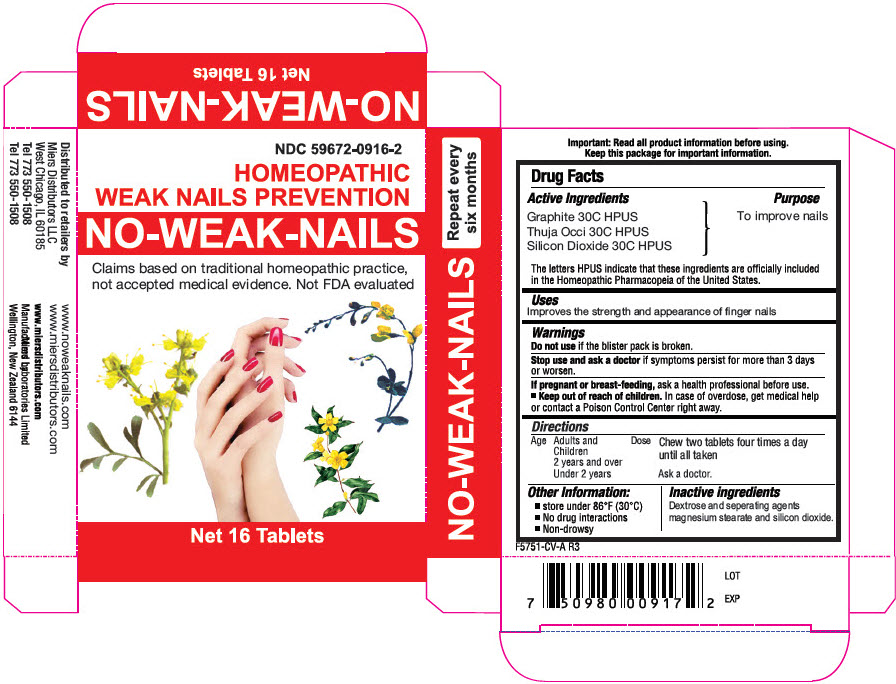
Uses
Improves the strength and appearance of finger nails
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if symptoms persist for more than 3 days or worsen.
Section 50569-3
Ask a doctor before use in children under 2 years of age.
Section 50570-1
Do not use if the blister pack is broken.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
| Age | Dose |
|---|---|
| Adults and Children 2 years and over | Chew two tablets four times a day for two days until all taken. |
| Under 2 years | Ask a doctor. |
Other Information
- Store under 86°F (30°C)
- No drug interactions
- Non-drowsy
Active Ingredients
Graphite 30C HPUS
Thuja Occi 30C HPUS
Silicon Dioxide 30C HPUS
The letters HPUS indicate that these ingredients are officially included in the Homeopathic Pharmacopeia of the United States.
Inactive Ingredients
Dextrose and separating agents magnesium stearate and silicon dioxide
Principal Display Panel 16 Tablet Blister Pack Carton
NDC 59672-0916-2
HOMEOPATHIC
WEAK NAILS PREVENTION
NO-WEAK-NAILS
Claims based on traditional homeopathic practice,
not accepted medical evidence. Not FDA evaluated
Net 16 Tablets
Structured Label Content
Uses
Improves the strength and appearance of finger nails
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if symptoms persist for more than 3 days or worsen.
Section 50569-3 (50569-3)
Ask a doctor before use in children under 2 years of age.
Section 50570-1 (50570-1)
Do not use if the blister pack is broken.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
To improve nails
Directions
| Age | Dose |
|---|---|
| Adults and Children 2 years and over | Chew two tablets four times a day for two days until all taken. |
| Under 2 years | Ask a doctor. |
Other Information
- Store under 86°F (30°C)
- No drug interactions
- Non-drowsy
Active Ingredients
Graphite 30C HPUS
Thuja Occi 30C HPUS
Silicon Dioxide 30C HPUS
The letters HPUS indicate that these ingredients are officially included in the Homeopathic Pharmacopeia of the United States.
Inactive Ingredients (Inactive ingredients)
Dextrose and separating agents magnesium stearate and silicon dioxide
Principal Display Panel 16 Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 16 Tablet Blister Pack Carton)
NDC 59672-0916-2
HOMEOPATHIC
WEAK NAILS PREVENTION
NO-WEAK-NAILS
Claims based on traditional homeopathic practice,
not accepted medical evidence. Not FDA evaluated
Net 16 Tablets
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:11:48.511351 · Updated: 2026-03-14T23:17:09.961113