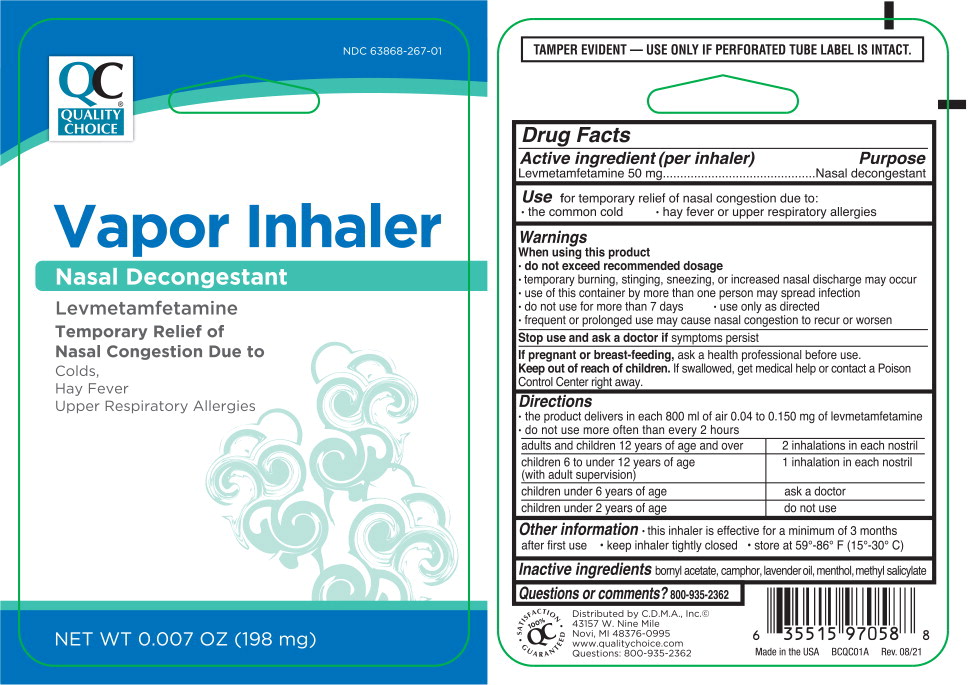
Drug Facts
904b7e21-cce5-4b96-afc1-b36f532f98ac
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Levmetamfetamine 50 mg
Purpose
Nasal decongestant
Medication Information
Purpose
Nasal decongestant
Description
Levmetamfetamine 50 mg
Use
for temporary relief of nasal congestion due to:
- the common cold
- hay fever or upper respiratory allergies
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if symptoms persist
Section 50570-1
- do not use for more than 7 days
- use only as directed
- frequent or prolonged use may cause nasal congestion to recur or worsen
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
- the product delivers in each 800 ml of air 0.04 to 0.150 mg of levmetamfetamine
- do not use more often than every 2 hours
| adults and children 12 years of age and over | 2 inhalations in each nostril |
| children 6 to under 12 years of age (with adult supervision) | 1 inhalation in each nostril |
| children under 6 years of age | ask a doctor |
| children under 2 years of age | do not use |
Other Information
- this inhaler is effective for a minimum of 3 months after first use
- keep inhaler tightly closed
- store at 59°-86° F (15°-30° C)
Inactive Ingredients
bornyl acetate, camphor, lavender oil, menthol, methyl salicylate
Questions Or Comments?
800-935-2362
When Using This Product
- do not exceed recommended dosage
- temporary burning, stinging, sneezing, or increased nasal discharge may occur
- use of this container by more than one person may spread infection
Active Ingredient (per Inhaler)
Levmetamfetamine 50 mg
Principal Display Panel – 198 Mg Blister Pack Label
NDC 63868-267-01
QC ®
QUALITY
CHOICE
Vapor Inhaler
Nasal Decongestant
Levmetamfetamine
Temporary Relief of
Nasal Congestion Due to
Colds,
Hay Fever
Upper Respiratory Allergies
NET WT 0.007 OZ (198 mg)
Structured Label Content
Use
for temporary relief of nasal congestion due to:
- the common cold
- hay fever or upper respiratory allergies
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if symptoms persist
Section 50570-1 (50570-1)
- do not use for more than 7 days
- use only as directed
- frequent or prolonged use may cause nasal congestion to recur or worsen
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Nasal decongestant
Directions
- the product delivers in each 800 ml of air 0.04 to 0.150 mg of levmetamfetamine
- do not use more often than every 2 hours
| adults and children 12 years of age and over | 2 inhalations in each nostril |
| children 6 to under 12 years of age (with adult supervision) | 1 inhalation in each nostril |
| children under 6 years of age | ask a doctor |
| children under 2 years of age | do not use |
Other Information (Other information)
- this inhaler is effective for a minimum of 3 months after first use
- keep inhaler tightly closed
- store at 59°-86° F (15°-30° C)
Inactive Ingredients (Inactive ingredients)
bornyl acetate, camphor, lavender oil, menthol, methyl salicylate
Questions Or Comments? (Questions or comments?)
800-935-2362
When Using This Product (When using this product)
- do not exceed recommended dosage
- temporary burning, stinging, sneezing, or increased nasal discharge may occur
- use of this container by more than one person may spread infection
Active Ingredient (per Inhaler) (Active ingredient (per inhaler))
Levmetamfetamine 50 mg
Principal Display Panel – 198 Mg Blister Pack Label (Principal Display Panel – 198 mg Blister Pack Label)
NDC 63868-267-01
QC ®
QUALITY
CHOICE
Vapor Inhaler
Nasal Decongestant
Levmetamfetamine
Temporary Relief of
Nasal Congestion Due to
Colds,
Hay Fever
Upper Respiratory Allergies
NET WT 0.007 OZ (198 mg)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:02.301673 · Updated: 2026-03-14T23:06:18.731306