paroxetine tablets usp, film coated for oral use
903e4ef1-487d-4b65-9038-b05ceaf74d19
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Contraindications
The use of MAOIs intended to treat psychiatric disorders with Paroxetine Tablets or within 14 days of stopping treatment with Paroxetine Tablets is contraindicated because of an increased risk of serotonin syndrome. The use of Paroxetine Tablets within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated (see WARNINGS and DOSAGE AND ADMINISTRATION ). Starting Paroxetine Tablets in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome (see WARNINGS and DOSAGE AND ADMINISTRATION ). Concomitant use with thioridazine is contraindicated (see WARNINGS and PRECAUTIONS ). Concomitant use in patients taking pimozide is contraindicated (see PRECAUTIONS ). Paroxetine Tablets are contraindicated in patients with a hypersensitivity to paroxetine or any of the inactive ingredients in Paroxetine Tablets.
How Supplied
Tablets: Film-coated, capsule-shaped as follows: Paroxetine Tablets USP, 40-mg beige to light brown tablets, debossed with ‘713’ on one side and ‘HH’ on the other side. NDC 71205-198-30 Bottles of 30 NDC 71205-198-90 Bottles of 90
Description
Suicidality and Antidepressant Drugs Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of Paroxetine Tablets or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Paroxetine Tablets are not approved for use in pediatric patients. (See Error! Hyperlink reference not valid., Error! Hyperlink reference not valid., and Error! Hyperlink reference not valid..)
Medication Information
Recent Major Changes
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of Paroxetine Tablets or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Paroxetine Tablets are not approved for use in pediatric patients. (See Error! Hyperlink reference not valid., Error! Hyperlink reference not valid., and Error! Hyperlink reference not valid..)
Contraindications
The use of MAOIs intended to treat psychiatric disorders with Paroxetine Tablets or within 14 days of stopping treatment with Paroxetine Tablets is contraindicated because of an increased risk of serotonin syndrome. The use of Paroxetine Tablets within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated (see WARNINGS and DOSAGE AND ADMINISTRATION ).
Starting Paroxetine Tablets in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome (see WARNINGS and DOSAGE AND ADMINISTRATION ).
Concomitant use with thioridazine is contraindicated (see WARNINGS and PRECAUTIONS ).
Concomitant use in patients taking pimozide is contraindicated (see PRECAUTIONS ).
Paroxetine Tablets are contraindicated in patients with a hypersensitivity to paroxetine or any of the inactive ingredients in Paroxetine Tablets.
How Supplied
Tablets: Film-coated, capsule-shaped as follows:
Paroxetine Tablets USP, 40-mg beige to light brown tablets, debossed with ‘713’ on one side and ‘HH’ on the other side.
NDC 71205-198-30 Bottles of 30
NDC 71205-198-90 Bottles of 90
Description
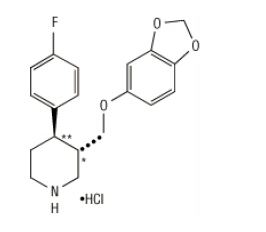
Paroxetine hydrochloride is an orally administered psychotropic drug. It is the hydrochloride salt of a phenylpiperidine compound identified chemically as (-)-trans-4R-(4'-fluorophenyl)-3S-[(3',4'-methylenedioxyphenoxy) methyl] piperidine hydrochloride hemihydrate and has the empirical formula of C19H20FNO3•HCl•1/2H2O. The molecular weight is 374.8 (329.4 as free base). The structural formula of paroxetine hydrochloride is:
Paroxetine hydrochloride, USP is an odorless, off-white powder, having a melting point range of 120° to 138°C and a solubility of 5.4 mg/mL in water.
Section 34073-7
-
Drug-Drug Interactions: In vitro drug interaction studies reveal that paroxetine inhibits CYP2D6. Clinical drug interaction studies have been performed with substrates of CYP2D6 and show that paroxetine can inhibit the metabolism of drugs metabolized by CYP2D6 including desipramine, risperidone, and atomoxetine (see Error! Hyperlink reference not valid. ).
Section 34075-2
Laboratory Tests: There are no specific laboratory tests recommended.
Section 34076-0
Information for Patients: Paroxetine Tablets should not be chewed or crushed, and should be swallowed whole.
Patients should be cautioned about the risk of serotonin syndrome with the concomitant use of Paroxetine Tablets and triptans, tramadol, or other serotonergic agents.
Patients should be advised that taking Paroxetine Tablets can cause mild pupillary dilation, which in susceptible individuals, can lead to an episode of angle closure glaucoma. Pre-existing glaucoma is almost always open-angle glaucoma because angle closure glaucoma, when diagnosed, can be treated definitively with iridectomy. Open-angle glaucoma is not a risk factor for angle closure glaucoma. Patients may wish to be examined to determine whether they are susceptible to angle closure, and have a prophylactic procedure (e.g., iridectomy), if they are susceptible.
Prescribers or other health professionals should inform patients, their families, and their caregivers about the benefits and risks associated with treatment with Paroxetine Tablets and should counsel them in its appropriate use. A patient Medication Guide is available for Paroxetine Tablets. The prescriber or health professional should instruct patients, their families, and their caregivers to read the Medication Guide and should assist them in understanding its contents. Patients should be given the opportunity to discuss the contents of the Medication Guide and to obtain answers to any questions they may have. The complete text of the Medication Guide is reprinted at the end of this document.
Patients should be advised of the following issues and asked to alert their prescriber if these occur while taking Paroxetine Tablets.
Section 34077-8
Usage in Pregnancy: Teratogenic Effects: Epidemiological studies have shown that infants exposed to paroxetine in the first trimester of pregnancy have an increased risk of congenital malformations, particularly cardiovascular malformations. The findings from these studies are summarized below:
-
•A study based on Swedish national registry data demonstrated that infants exposed to paroxetine during pregnancy (n = 815) had an increased risk of cardiovascular malformations (2% risk in paroxetine-exposed infants) compared to the entire registry population (1% risk), for an odds ratio (OR) of 1.8 (95% confidence interval 1.1 to 2.8). No increase in the risk of overall congenital malformations was seen in the paroxetine-exposed infants. The cardiac malformations in the paroxetine-exposed infants were primarily ventricular septal defects (VSDs) and atrial septal defects (ASDs). Septal defects range in severity from those that resolve spontaneously to those which require surgery.
-
•A separate retrospective cohort study from the United States (United Healthcare data) evaluated 5,956 infants of mothers dispensed antidepressants during the first trimester (n = 815 for paroxetine). This study showed a trend towards an increased risk for cardiovascular malformations for paroxetine (risk of 1.5%) compared to other antidepressants (risk of 1%), for an OR of 1.5 (95% confidence interval 0.8 to 2.9). Of the 12 paroxetine-exposed infants with cardiovascular malformations, 9 had VSDs. This study also suggested an increased risk of overall major congenital malformations including cardiovascular defects for paroxetine (4% risk) compared to other (2% risk) antidepressants (OR 1.8; 95% confidence interval 1.2 to 2.8).
-
•Two large case-control studies using separate databases, each with >9,000 birth defect cases and >4,000 controls, found that maternal use of paroxetine during the first trimester of pregnancy was associated with a 2- to 3-fold increased risk of right ventricular outflow tract obstructions. In one study the odds ratio was 2.5 (95% confidence interval, 1.0 to 6.0, 7 exposed infants) and in the other study the odds ratio was 3.3 (95% confidence interval, 1.3 to 8.8, 6 exposed infants).
Other studies have found varying results as to whether there was an increased risk of overall, cardiovascular, or specific congenital malformations. A meta-analysis of epidemiological data over a 16-year period (1992 to 2008) on first trimester paroxetine use in pregnancy and congenital malformations included the above-noted studies in addition to others (n = 17 studies that included overall malformations and n = 14 studies that included cardiovascular malformations; n = 20 distinct studies). While subject to limitations, this meta-analysis suggested an increased occurrence of cardiovascular malformations (prevalence odds ratio [POR] 1.5; 95% confidence interval 1.2 to 1.9) and overall malformations (POR 1.2; 95% confidence interval 1.1 to 1.4) with paroxetine use during the first trimester. It was not possible in this meta-analysis to determine the extent to which the observed prevalence of cardiovascular malformations might have contributed to that of overall malformations, nor was it possible to determine whether any specific types of cardiovascular malformations might have contributed to the observed prevalence of all cardiovascular malformations.
If a patient becomes pregnant while taking paroxetine, she should be advised of the potential harm to the fetus. Unless the benefits of paroxetine to the mother justify continuing treatment, consideration should be given to either discontinuing paroxetine therapy or switching to another antidepressant (see PRECAUTIONS: Discontinuation of Treatment With Paroxetine Tablets). For women who intend to become pregnant or are in their first trimester of pregnancy, paroxetine should only be initiated after consideration of the other available treatment options.
Section 34078-6
Nonteratogenic Effects: Neonates exposed to Paroxetine Tablets and other SSRIs or serotonin and norepinephrine reuptake inhibitors (SNRIs), late in the third trimester have developed complications requiring prolonged hospitalization, respiratory support, and tube feeding. Such complications can arise immediately upon delivery. Reported clinical findings have included respiratory distress, cyanosis, apnea, seizures, temperature instability, feeding difficulty, vomiting, hypoglycemia, hypotonia, hypertonia, hyperreflexia, tremor, jitteriness, irritability, and constant crying. These features are consistent with either a direct toxic effect of SSRIs and SNRIs or, possibly, a drug discontinuation syndrome. It should be noted that, in some cases, the clinical picture is consistent with serotonin syndrome (see WARNINGS: Error! Hyperlink reference not valid.).
Infants exposed to SSRIs in pregnancy may have an increased risk for persistent pulmonary hypertension of the newborn (PPHN). PPHN occurs in 1 – 2 per 1,000 live births in the general population and is associated with substantial neonatal morbidity and mortality. Several recent epidemiologic studies suggest a positive statistical association between SSRI use (including Paroxetine Tablets) in pregnancy and PPHN. Other studies do not show a significant statistical association.
Physicians should also note the results of a prospective longitudinal study of 201 pregnant women with a history of major depression, who were either on antidepressants or had received antidepressants less than 12 weeks prior to their last menstrual period, and were in remission. Women who discontinued antidepressant medication during pregnancy showed a significant increase in relapse of their major depression compared to those women who remained on antidepressant medication throughout pregnancy.
When treating a pregnant woman with Paroxetine Tablets, the physician should carefully consider both the potential risks of taking an SSRI, along with the established benefits of treating depression with an antidepressant. This decision can only be made on a case by case basis (see DOSAGE AND ADMINISTRATION and ADVERSE REACTIONS: Postmarketing Reports ).
Section 34079-4
Labor and Delivery: The effect of paroxetine on labor and delivery in humans is unknown.
Section 34080-2
Nursing: Patients should be advised to notify their physician if they are breastfeeding an infant (see Error! Hyperlink reference not valid.).
Section 34081-0
Pediatric Use: Safety and effectiveness in the pediatric population have not been established (see Error! Hyperlink reference not valid.and WARNINGS: Clinical Worsening and Suicide Risk ). Three placebo-controlled trials in 752 pediatric patients with MDD have been conducted with Paroxetine Tablets, and the data were not sufficient to support a claim for use in pediatric patients. Anyone considering the use of Paroxetine Tablets in a child or adolescent must balance the potential risks with the clinical need. Decreased appetite and weight loss have been observed in association with the use of SSRIs. Consequently, regular monitoring of weight and growth should be performed in children and adolescents treated with an SSRI such as Paroxetine Tablets.
In placebo-controlled clinical trials conducted with pediatric patients, the following adverse events were reported in at least 2% of pediatric patients treated with Paroxetine Tablets and occurred at a rate at least twice that for pediatric patients receiving placebo: emotional lability (including self-harm, suicidal thoughts, attempted suicide, crying, and mood fluctuations), hostility, decreased appetite, tremor, sweating, hyperkinesia, and agitation.
Events reported upon discontinuation of treatment with Paroxetine Tablets in the pediatric clinical trials that included a taper phase regimen, which occurred in at least 2% of patients who received Paroxetine Tablets and which occurred at a rate at least twice that of placebo, were: emotional lability (including suicidal ideation, suicide attempt, mood changes, and tearfulness), nervousness, dizziness, nausea, and abdominal pain (see DOSAGE AND ADMINISTRATION: Error! Hyperlink reference not valid.).
Section 34082-8
Geriatric Use: SSRIs and SNRIs, including Paroxetine Tablets, have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse event (see Error! Hyperlink reference not valid.).
In worldwide premarketing clinical trials with Paroxetine Tablets, 17% of patients treated with Paroxetine Tablets (approximately 700) were 65 years of age or older. Pharmacokinetic studies revealed a decreased clearance in the elderly, and a lower starting dose is recommended; there were, however, no overall differences in the adverse event profile between elderly and younger patients, and effectiveness was similar in younger and older patients (see CLINICAL PHARMACOLOGY and DOSAGE AND ADMINISTRATION ).
Section 34083-6
Carcinogenesis, Mutagenesis, Impairment of Fertility: Carcinogenesis: Two-year carcinogenicity studies were conducted in rodents given paroxetine in the diet at 1 mg/kg/day, 5 mg/kg/day, and 25 mg/kg/day (mice) and 1 mg/kg/day, 5 mg/kg/day, and 20 mg/kg/day (rats). These doses are up to 2.4 (mouse) and 3.9 (rat) times the MRHD for major depressive disorder, social anxiety disorder and GAD on a mg/m2 basis. Because the MRHD for major depressive disorder is slightly less than that for OCD (50 mg versus 60 mg), the doses used in these carcinogenicity studies were only 2.0 (mouse) and 3.2 (rat) times the MRHD for OCD. There was a significantly greater number of male rats in the high-dose group with reticulum cell sarcomas (1/100, 0/50, 0/50, and 4/50 for control, low-, middle-, and high-dose groups, respectively) and a significantly increased linear trend across dose groups for the occurrence of lymphoreticular tumors in male rats. Female rats were not affected. Although there was a dose-related increase in the number of tumors in mice, there was no drug-related increase in the number of mice with tumors. The relevance of these findings to humans is unknown.
Section 34086-9
Alcohol: Although Paroxetine Tablets do not increase the impairment of mental and motor skills caused by alcohol, patients should be advised to avoid alcohol while taking Paroxetine Tablets.
Section 34090-1
-
Other Clinical Pharmacology Information: Specific Populations: Renal and Liver Disease: Increased plasma concentrations of paroxetine occur in subjects with renal and hepatic impairment. The mean plasma concentrations in patients with creatinine clearance below 30 mL/min. were approximately 4 times greater than seen in normal volunteers. Patients with creatinine clearance of 30 mL/min to 60 mL/min. and patients with hepatic functional impairment had about a 2-fold increase in plasma concentrations (AUC, C max ).
The initial dosage should therefore be reduced in patients with severe renal or hepatic impairment, and upward titration, if necessary, should be at increased intervals (see DOSAGE AND ADMINISTRATION).
Section 42228-7
Pregnancy: Patients should be advised to notify their physician if they become pregnant or intend to become pregnant during therapy (see WARNINGS: Error! Hyperlink reference not valid.: Error! Hyperlink reference not valid. and Error! Hyperlink reference not valid.).
Section 42229-5
Tablets: Each film-coated tablet contains paroxetine hydrochloride equivalent to paroxetine as follows: 40 mg– beige to light brown. Inactive ingredients consist of hypromellose, glyceryl behenate, lactose monohydrate, magnesium stearate, titanium dioxide, polyethylene glycols, iron oxide yellow and iron oxide red.
Section 43681-6
Pharmacodynamics: The efficacy of paroxetine in the treatment of major depressive disorder, social anxiety disorder, obsessive compulsive disorder (OCD), panic disorder (PD) and generalized anxiety disorder (GAD) is presumed to be linked to potentiation of serotonergic activity in the central nervous system resulting from inhibition of neuronal reuptake of serotonin (5-hydroxy-tryptamine, 5-HT). Studies at clinically relevant doses in humans have demonstrated that paroxetine blocks the uptake of serotonin into human platelets. In vitro studies in animals also suggest that paroxetine is a potent and highly selective inhibitor of neuronal serotonin reuptake and has only very weak effects on norepinephrine and dopamine neuronal reuptake. In vitro radioligand binding studies indicate that paroxetine has little affinity for muscarinic, alpha1-, alpha2-, beta-adrenergic-, dopamine (D2-, 5-HT1-, 5-HT2-, and histamine (H1)-receptors; antagonism of muscarinic, histaminergic, and alpha1- receptors has been associated with various anticholinergic, sedative, and cardiovascular effects for other psychotropic drugs.
Because the relative potencies of paroxetine’s major metabolites are at most 1/50 of the parent compound, they are essentially inactive.
Section 43682-4
Pharmacokinetics: Paroxetine hydrochloride is completely absorbed after oral dosing of a solution of the hydrochloride salt. The mean elimination half-life is approximately 21 hours (CV 32%) after oral dosing of 30 mg tablets of Paroxetine Tablets daily for 30 days. Paroxetine is extensively metabolized and the metabolites are considered to be inactive. Nonlinearity in pharmacokinetics is observed with increasing doses. Paroxetine metabolism is mediated in part by CYP2D6, and the metabolites are primarily excreted in the urine and to some extent in the feces. Pharmacokinetic behavior of paroxetine has not been evaluated in subjects who are deficient in CYP2D6 (poor metabolizers).
In a meta-analysis of paroxetine from 4 studies done in healthy volunteers following multiple dosing of 20 mg/day to 40 mg/day, males did not exhibit a significantly lower Cmax or AUC than females.
Section 44425-7
Store tablets at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
You may report side effects to Solco Healthcare US, LLC at 1-866-257-2597 or FDA at 1-800-FDA-1088.
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang 317024, China
Distributed by:
Solco Healthcare US, LLC.
Cranbury, NJ 08512, USA
Repackaged by:
Proficient Rx LP
Thousand Oaks, CA 91320
Issue Date: 10/2014
07490-01
Section 51945-4
PRINCIPAL DISPLAY PANEL - Container Label - 40 mg container label image
NDC 71205-198-90 Rx only
Paroxetine
Tablets, USP
40 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
90 Tablets
Precautions
-
General: Activation of Mania/Hypomania: During premarketing testing, hypomania or mania occurred in approximately 1.0% of unipolar patients treated with Paroxetine Tablets compared to 1.1% of active-control and 0.3% of placebo-treated unipolar patients. In a subset of patients classified as bipolar, the rate of manic episodes was 2.2% for Paroxetine Tablets and 11.6% for the combined active-control groups. As with all drugs effective in the treatment of major depressive disorder, Paroxetine Tablets should be used cautiously in patients with a history of mania.
Medication Guide
Paroxetine Tablets USP
(PA-rox-eh-tine)
Read the Medication Guide that comes with Paroxetine Tablets before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or treatment. Talk with your healthcare provider if there is something you do not understand or want to learn more about.
What is the most important information I should know about Paroxetine Tablets?
Paroxetine Tablets and other antidepressant medicines may cause serious side effects, including:
1. Suicidal thoughts or actions:
-
•Paroxetine Tablets and other antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers, or young adults within the first few months of treatment or when the dose is changed.
-
•Depression or other serious mental illnesses are the most important causes of suicidal thoughts or actions.
-
•Watch for these changes and call your healthcare provider right away if you notice:
-
oNew or sudden changes in mood, behavior, actions, thoughts, or feelings, especially if severe.
-
oPay particular attention to such changes when Paroxetine Tablets are started or when the dose is changed.
-
Keep all follow-up visits with your healthcare provider and call between visits if you are worried about symptoms.
Call your healthcare provider right away if you have any of the following symptoms, or call 911 if an emergency, especially if they are new, worse, or worry you:
-
•attempts to commit suicide
-
•acting on dangerous impulses
-
•acting aggressive or violent
-
•thoughts about suicide or dying
-
•new or worse depression
-
•new or worse anxiety or panic attacks
-
•feeling agitated, restless, angry, or irritable
-
•trouble sleeping
-
•an increase in activity or talking more than what is normal for you
-
•other unusual changes in behavior or mood
Call your healthcare provider right away if you have any of the following symptoms, or call 911 if an emergency. Paroxetine Tablets may be associated with these serious side effects:
2. Serotonin Syndrome or Neuroleptic Malignant Syndrome-like reactions. This condition can be life-threatening and may include:
-
1.agitation, hallucinations, coma, or other changes in mental status
-
2.coordination problems or muscle twitching (overactive reflexes)
-
3.racing heartbeat, high or low blood pressure
-
4.sweating or fever
-
5.nausea, vomiting, or diarrhea
-
6.muscle rigidity
3. Visual problems
-
1.eye pain
-
2.changes in vision
-
3.swelling or redness in or around the eye
Only some people are at risk for these problems. You may want to undergo an eye examination to see if you are at risk and receive preventative treatment if you are.
4. Severe allergic reactions:
-
1.trouble breathing
-
2.swelling of the face, tongue, eyes, or mouth
-
3.rash, itchy welts (hives), or blisters, alone or with fever or joint pain
5. Abnormal bleeding: Paroxetine Tablets and other antidepressant medicines may increase your risk of bleeding or bruising, especially if you take the blood thinner warfarin (Coumadin"®", Jantoven"®"), a non-steroidal anti-inflammatory drug (NSAIDs, like ibuprofen or naproxen), or aspirin.
6. Seizures or convulsions
7. Manic episodes:
-
1.greatly increased energy
-
2.severe trouble sleeping
-
3.racing thoughts
-
4.reckless behavior
-
5.unusually grand ideas
-
6.excessive happiness or irritability
-
7.talking more or faster than usual
8. Changes in appetite or weight. Children and adolescents should have height and weight monitored during treatment.
9. Low salt (sodium) levels in the blood. Elderly people may be at greater risk for this. Symptoms may include:
-
1.headache
-
2.weakness or feeling unsteady
-
3.confusion, problems concentrating or thinking, or memory problems
Do not stop Paroxetine Tablets without first talking to your healthcare provider. Stopping Paroxetine Tablets too quickly may cause serious symptoms including:
-
1.anxiety, irritability, high or low mood, feeling restless, or changes in sleep habits
-
2.headache, sweating, nausea, dizziness
-
3.electric shock-like sensations, shaking, confusion
What are Paroxetine Tablets?
Paroxetine Tablets are prescription medicines used to treat depression. It is important to talk with your healthcare provider about the risks of treating depression and also the risks of not treating it. You should discuss all treatment choices with your healthcare provider. Paroxetine Tablets are also used to treat:
-
1.Major Depressive Disorder (MDD)
-
2.Obsessive Compulsive Disorder (OCD)
-
3.Panic Disorder
-
4.Social Anxiety Disorder
-
5.Generalized Anxiety Disorder (GAD)
Talk to your healthcare provider if you do not think that your condition is getting better with treatment using Paroxetine Tablets.
Who should not take Paroxetine Tablets?
Do not take Paroxetine Tablets if you:
-
1.are allergic to paroxetine or any of the ingredients in Paroxetine Tablets. See the end of this Medication Guide for a complete list of ingredients in Paroxetine Tablets.
-
2.take a monoamine oxidase inhibitor (MAOI). Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including the antibiotic linezolid.
-
1.Do not take an MAOI within 2 weeks of stopping Paroxetine Tablets unless directed to do so by your physician.
-
2.Do not start Paroxetine Tablets if you stopped taking an MAOI in the last 2 weeks unless directed to do so by your physician.
-
3.People who take Paroxetine Tablets close in time to an MAOI may have serious or even life-threatening side effects. Get medical help right away if you have any of these symptoms:
-
1.high fever
-
2.uncontrolled muscle spasms
-
3.stiff muscles
-
4.rapid changes in heart rate or blood pressure
-
5.confusion
-
6.loss of consciousness (pass out)
-
-
4.take MELLARIL ®* (thioridazine). Do not take MELLARIL ®* together with Paroxetine Tablets because this can cause serious heart rhythm problems or sudden death.
-
5.take the antipsychotic medicine pimozide (ORAP ®* ) because this can cause serious heart problems.
-
-
What should I tell my healthcare provider before taking Paroxetine Tablets? Ask if you are not sure.
Tell your healthcare provider about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal supplements. Paroxetine Tablets and some medicines may interact with each other, may not work as well, or may cause serious side effects.
Your healthcare provider or pharmacist can tell you if it is safe to take Paroxetine Tablets with your other medicines. Do not start or stop any medicine while taking Paroxetine Tablets without talking to your healthcare provider first.
|
If you take Paroxetine Tablets, you should not take any other medicines that contain paroxetine, including paroxetine extended-release tablets and PEXEVA®* (paroxetine mesylate). |
How should I take Paroxetine Tablets?
-
1.Take Paroxetine Tablets exactly as prescribed. Your healthcare provider may need to change the dose of Paroxetine Tablets until it is the right dose for you.
-
2.Paroxetine Tablets may be taken with or without food.
-
3.If you miss a dose of Paroxetine Tablets, take the missed dose as soon as you remember. If it is almost time for the next dose, skip the missed dose and take your next dose at the regular time. Do not take two doses of Paroxetine Tablets at the same time.
-
4.If you take too many Paroxetine Tablets call your healthcare provider or poison control center right away, or get emergency treatment.
-
5.Do not stop taking Paroxetine Tablets suddenly without talking to your doctor (unless you have symptoms of a severe allergic reaction). If you need to stop taking Paroxetine Tablets, your healthcare provider can tell you how to safely stop taking it.
What should I avoid while taking Paroxetine Tablets?
Paroxetine Tablets can cause sleepiness or may affect your ability to make decisions, think clearly, or react quickly. You should not drive, operate heavy machinery, or do other dangerous activities until you know how Paroxetine Tablets affect you. Do not drink alcohol while using Paroxetine Tablets.
What are possible side effects of Paroxetine Tablets?
Paroxetine Tablets may cause serious side effects, including all of those described in the section entitled “What is the most important information I should know about Paroxetine Tablets?”
Common possible side effects in people who take Paroxetine Tablets include:
-
1.nausea
-
2.sleepiness
-
3.weakness
-
4.dizziness
-
5.feeling anxious or trouble sleeping
-
6.sexual problems
-
7.sweating
-
8.shaking
-
9.not feeling hungry
-
10.dry mouth
-
11.constipation
-
12.infection
-
13.yawning
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Paroxetine Tablets. For more information, ask your healthcare provider or pharmacist.
CALL YOUR DOCTOR FOR MEDICAL ADVICE ABOUT SIDE EFFECTS. YOU MAY REPORT SIDE EFFECTS TO SOLCO HEALTHCARE US, LLC AT 1-866-257-2597 OR TO THE FDA AT 1-800-FDA1088 or 1-800-332-1088.
How should I store Paroxetine Tablets?
-
1.Store Paroxetine Tablets at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature].
-
2.Keep Paroxetine Tablets away from light.
-
3.Keep bottle of Paroxetine Tablets closed tightly.
Keep Paroxetine Tablets and all medicines out of the reach of children.
General information about Paroxetine Tablets
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Paroxetine Tablets for a condition for which it was not prescribed. Do not give Paroxetine Tablets to other people, even if they have the same condition. It may harm them.
This Medication Guide summarizes the most important information about Paroxetine Tablets. If you would like more information, talk with your healthcare provider. You may ask your healthcare provider or pharmacist for information about Paroxetine Tablets that is written for healthcare professionals.
What are the ingredients in Paroxetine Tablets?
Active ingredient: paroxetine hydrochloride
Inactive ingredients in tablets: hypromellose, glyceryl behenate, lactose monohydrate, magnesium stearate, titanium dioxide, polyethylene glycols, iron oxide yellow, and iron oxide red.
*: Brand names listed are trademarks of their respective owners and are not trademarks of Solco Healthcare US, LLC.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Structured Label Content
Recent Major Changes (34066-1)
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies of major depressive disorder (MDD) and other psychiatric disorders. Anyone considering the use of Paroxetine Tablets or any other antidepressant in a child, adolescent, or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Paroxetine Tablets are not approved for use in pediatric patients. (See Error! Hyperlink reference not valid., Error! Hyperlink reference not valid., and Error! Hyperlink reference not valid..)
Contraindications (CONTRAINDICATIONS)
The use of MAOIs intended to treat psychiatric disorders with Paroxetine Tablets or within 14 days of stopping treatment with Paroxetine Tablets is contraindicated because of an increased risk of serotonin syndrome. The use of Paroxetine Tablets within 14 days of stopping an MAOI intended to treat psychiatric disorders is also contraindicated (see WARNINGS and DOSAGE AND ADMINISTRATION ).
Starting Paroxetine Tablets in a patient who is being treated with MAOIs such as linezolid or intravenous methylene blue is also contraindicated because of an increased risk of serotonin syndrome (see WARNINGS and DOSAGE AND ADMINISTRATION ).
Concomitant use with thioridazine is contraindicated (see WARNINGS and PRECAUTIONS ).
Concomitant use in patients taking pimozide is contraindicated (see PRECAUTIONS ).
Paroxetine Tablets are contraindicated in patients with a hypersensitivity to paroxetine or any of the inactive ingredients in Paroxetine Tablets.
How Supplied (HOW SUPPLIED)
Tablets: Film-coated, capsule-shaped as follows:
Paroxetine Tablets USP, 40-mg beige to light brown tablets, debossed with ‘713’ on one side and ‘HH’ on the other side.
NDC 71205-198-30 Bottles of 30
NDC 71205-198-90 Bottles of 90
Description (DESCRIPTION)
Paroxetine hydrochloride is an orally administered psychotropic drug. It is the hydrochloride salt of a phenylpiperidine compound identified chemically as (-)-trans-4R-(4'-fluorophenyl)-3S-[(3',4'-methylenedioxyphenoxy) methyl] piperidine hydrochloride hemihydrate and has the empirical formula of C19H20FNO3•HCl•1/2H2O. The molecular weight is 374.8 (329.4 as free base). The structural formula of paroxetine hydrochloride is:
Paroxetine hydrochloride, USP is an odorless, off-white powder, having a melting point range of 120° to 138°C and a solubility of 5.4 mg/mL in water.
Section 34073-7 (34073-7)
-
Drug-Drug Interactions: In vitro drug interaction studies reveal that paroxetine inhibits CYP2D6. Clinical drug interaction studies have been performed with substrates of CYP2D6 and show that paroxetine can inhibit the metabolism of drugs metabolized by CYP2D6 including desipramine, risperidone, and atomoxetine (see Error! Hyperlink reference not valid. ).
Section 34075-2 (34075-2)
Laboratory Tests: There are no specific laboratory tests recommended.
Section 34076-0 (34076-0)
Information for Patients: Paroxetine Tablets should not be chewed or crushed, and should be swallowed whole.
Patients should be cautioned about the risk of serotonin syndrome with the concomitant use of Paroxetine Tablets and triptans, tramadol, or other serotonergic agents.
Patients should be advised that taking Paroxetine Tablets can cause mild pupillary dilation, which in susceptible individuals, can lead to an episode of angle closure glaucoma. Pre-existing glaucoma is almost always open-angle glaucoma because angle closure glaucoma, when diagnosed, can be treated definitively with iridectomy. Open-angle glaucoma is not a risk factor for angle closure glaucoma. Patients may wish to be examined to determine whether they are susceptible to angle closure, and have a prophylactic procedure (e.g., iridectomy), if they are susceptible.
Prescribers or other health professionals should inform patients, their families, and their caregivers about the benefits and risks associated with treatment with Paroxetine Tablets and should counsel them in its appropriate use. A patient Medication Guide is available for Paroxetine Tablets. The prescriber or health professional should instruct patients, their families, and their caregivers to read the Medication Guide and should assist them in understanding its contents. Patients should be given the opportunity to discuss the contents of the Medication Guide and to obtain answers to any questions they may have. The complete text of the Medication Guide is reprinted at the end of this document.
Patients should be advised of the following issues and asked to alert their prescriber if these occur while taking Paroxetine Tablets.
Section 34077-8 (34077-8)
Usage in Pregnancy: Teratogenic Effects: Epidemiological studies have shown that infants exposed to paroxetine in the first trimester of pregnancy have an increased risk of congenital malformations, particularly cardiovascular malformations. The findings from these studies are summarized below:
-
•A study based on Swedish national registry data demonstrated that infants exposed to paroxetine during pregnancy (n = 815) had an increased risk of cardiovascular malformations (2% risk in paroxetine-exposed infants) compared to the entire registry population (1% risk), for an odds ratio (OR) of 1.8 (95% confidence interval 1.1 to 2.8). No increase in the risk of overall congenital malformations was seen in the paroxetine-exposed infants. The cardiac malformations in the paroxetine-exposed infants were primarily ventricular septal defects (VSDs) and atrial septal defects (ASDs). Septal defects range in severity from those that resolve spontaneously to those which require surgery.
-
•A separate retrospective cohort study from the United States (United Healthcare data) evaluated 5,956 infants of mothers dispensed antidepressants during the first trimester (n = 815 for paroxetine). This study showed a trend towards an increased risk for cardiovascular malformations for paroxetine (risk of 1.5%) compared to other antidepressants (risk of 1%), for an OR of 1.5 (95% confidence interval 0.8 to 2.9). Of the 12 paroxetine-exposed infants with cardiovascular malformations, 9 had VSDs. This study also suggested an increased risk of overall major congenital malformations including cardiovascular defects for paroxetine (4% risk) compared to other (2% risk) antidepressants (OR 1.8; 95% confidence interval 1.2 to 2.8).
-
•Two large case-control studies using separate databases, each with >9,000 birth defect cases and >4,000 controls, found that maternal use of paroxetine during the first trimester of pregnancy was associated with a 2- to 3-fold increased risk of right ventricular outflow tract obstructions. In one study the odds ratio was 2.5 (95% confidence interval, 1.0 to 6.0, 7 exposed infants) and in the other study the odds ratio was 3.3 (95% confidence interval, 1.3 to 8.8, 6 exposed infants).
Other studies have found varying results as to whether there was an increased risk of overall, cardiovascular, or specific congenital malformations. A meta-analysis of epidemiological data over a 16-year period (1992 to 2008) on first trimester paroxetine use in pregnancy and congenital malformations included the above-noted studies in addition to others (n = 17 studies that included overall malformations and n = 14 studies that included cardiovascular malformations; n = 20 distinct studies). While subject to limitations, this meta-analysis suggested an increased occurrence of cardiovascular malformations (prevalence odds ratio [POR] 1.5; 95% confidence interval 1.2 to 1.9) and overall malformations (POR 1.2; 95% confidence interval 1.1 to 1.4) with paroxetine use during the first trimester. It was not possible in this meta-analysis to determine the extent to which the observed prevalence of cardiovascular malformations might have contributed to that of overall malformations, nor was it possible to determine whether any specific types of cardiovascular malformations might have contributed to the observed prevalence of all cardiovascular malformations.
If a patient becomes pregnant while taking paroxetine, she should be advised of the potential harm to the fetus. Unless the benefits of paroxetine to the mother justify continuing treatment, consideration should be given to either discontinuing paroxetine therapy or switching to another antidepressant (see PRECAUTIONS: Discontinuation of Treatment With Paroxetine Tablets). For women who intend to become pregnant or are in their first trimester of pregnancy, paroxetine should only be initiated after consideration of the other available treatment options.
Section 34078-6 (34078-6)
Nonteratogenic Effects: Neonates exposed to Paroxetine Tablets and other SSRIs or serotonin and norepinephrine reuptake inhibitors (SNRIs), late in the third trimester have developed complications requiring prolonged hospitalization, respiratory support, and tube feeding. Such complications can arise immediately upon delivery. Reported clinical findings have included respiratory distress, cyanosis, apnea, seizures, temperature instability, feeding difficulty, vomiting, hypoglycemia, hypotonia, hypertonia, hyperreflexia, tremor, jitteriness, irritability, and constant crying. These features are consistent with either a direct toxic effect of SSRIs and SNRIs or, possibly, a drug discontinuation syndrome. It should be noted that, in some cases, the clinical picture is consistent with serotonin syndrome (see WARNINGS: Error! Hyperlink reference not valid.).
Infants exposed to SSRIs in pregnancy may have an increased risk for persistent pulmonary hypertension of the newborn (PPHN). PPHN occurs in 1 – 2 per 1,000 live births in the general population and is associated with substantial neonatal morbidity and mortality. Several recent epidemiologic studies suggest a positive statistical association between SSRI use (including Paroxetine Tablets) in pregnancy and PPHN. Other studies do not show a significant statistical association.
Physicians should also note the results of a prospective longitudinal study of 201 pregnant women with a history of major depression, who were either on antidepressants or had received antidepressants less than 12 weeks prior to their last menstrual period, and were in remission. Women who discontinued antidepressant medication during pregnancy showed a significant increase in relapse of their major depression compared to those women who remained on antidepressant medication throughout pregnancy.
When treating a pregnant woman with Paroxetine Tablets, the physician should carefully consider both the potential risks of taking an SSRI, along with the established benefits of treating depression with an antidepressant. This decision can only be made on a case by case basis (see DOSAGE AND ADMINISTRATION and ADVERSE REACTIONS: Postmarketing Reports ).
Section 34079-4 (34079-4)
Labor and Delivery: The effect of paroxetine on labor and delivery in humans is unknown.
Section 34080-2 (34080-2)
Nursing: Patients should be advised to notify their physician if they are breastfeeding an infant (see Error! Hyperlink reference not valid.).
Section 34081-0 (34081-0)
Pediatric Use: Safety and effectiveness in the pediatric population have not been established (see Error! Hyperlink reference not valid.and WARNINGS: Clinical Worsening and Suicide Risk ). Three placebo-controlled trials in 752 pediatric patients with MDD have been conducted with Paroxetine Tablets, and the data were not sufficient to support a claim for use in pediatric patients. Anyone considering the use of Paroxetine Tablets in a child or adolescent must balance the potential risks with the clinical need. Decreased appetite and weight loss have been observed in association with the use of SSRIs. Consequently, regular monitoring of weight and growth should be performed in children and adolescents treated with an SSRI such as Paroxetine Tablets.
In placebo-controlled clinical trials conducted with pediatric patients, the following adverse events were reported in at least 2% of pediatric patients treated with Paroxetine Tablets and occurred at a rate at least twice that for pediatric patients receiving placebo: emotional lability (including self-harm, suicidal thoughts, attempted suicide, crying, and mood fluctuations), hostility, decreased appetite, tremor, sweating, hyperkinesia, and agitation.
Events reported upon discontinuation of treatment with Paroxetine Tablets in the pediatric clinical trials that included a taper phase regimen, which occurred in at least 2% of patients who received Paroxetine Tablets and which occurred at a rate at least twice that of placebo, were: emotional lability (including suicidal ideation, suicide attempt, mood changes, and tearfulness), nervousness, dizziness, nausea, and abdominal pain (see DOSAGE AND ADMINISTRATION: Error! Hyperlink reference not valid.).
Section 34082-8 (34082-8)
Geriatric Use: SSRIs and SNRIs, including Paroxetine Tablets, have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse event (see Error! Hyperlink reference not valid.).
In worldwide premarketing clinical trials with Paroxetine Tablets, 17% of patients treated with Paroxetine Tablets (approximately 700) were 65 years of age or older. Pharmacokinetic studies revealed a decreased clearance in the elderly, and a lower starting dose is recommended; there were, however, no overall differences in the adverse event profile between elderly and younger patients, and effectiveness was similar in younger and older patients (see CLINICAL PHARMACOLOGY and DOSAGE AND ADMINISTRATION ).
Section 34083-6 (34083-6)
Carcinogenesis, Mutagenesis, Impairment of Fertility: Carcinogenesis: Two-year carcinogenicity studies were conducted in rodents given paroxetine in the diet at 1 mg/kg/day, 5 mg/kg/day, and 25 mg/kg/day (mice) and 1 mg/kg/day, 5 mg/kg/day, and 20 mg/kg/day (rats). These doses are up to 2.4 (mouse) and 3.9 (rat) times the MRHD for major depressive disorder, social anxiety disorder and GAD on a mg/m2 basis. Because the MRHD for major depressive disorder is slightly less than that for OCD (50 mg versus 60 mg), the doses used in these carcinogenicity studies were only 2.0 (mouse) and 3.2 (rat) times the MRHD for OCD. There was a significantly greater number of male rats in the high-dose group with reticulum cell sarcomas (1/100, 0/50, 0/50, and 4/50 for control, low-, middle-, and high-dose groups, respectively) and a significantly increased linear trend across dose groups for the occurrence of lymphoreticular tumors in male rats. Female rats were not affected. Although there was a dose-related increase in the number of tumors in mice, there was no drug-related increase in the number of mice with tumors. The relevance of these findings to humans is unknown.
Section 34086-9 (34086-9)
Alcohol: Although Paroxetine Tablets do not increase the impairment of mental and motor skills caused by alcohol, patients should be advised to avoid alcohol while taking Paroxetine Tablets.
Section 34090-1 (34090-1)
-
Other Clinical Pharmacology Information: Specific Populations: Renal and Liver Disease: Increased plasma concentrations of paroxetine occur in subjects with renal and hepatic impairment. The mean plasma concentrations in patients with creatinine clearance below 30 mL/min. were approximately 4 times greater than seen in normal volunteers. Patients with creatinine clearance of 30 mL/min to 60 mL/min. and patients with hepatic functional impairment had about a 2-fold increase in plasma concentrations (AUC, C max ).
The initial dosage should therefore be reduced in patients with severe renal or hepatic impairment, and upward titration, if necessary, should be at increased intervals (see DOSAGE AND ADMINISTRATION).
Section 42228-7 (42228-7)
Pregnancy: Patients should be advised to notify their physician if they become pregnant or intend to become pregnant during therapy (see WARNINGS: Error! Hyperlink reference not valid.: Error! Hyperlink reference not valid. and Error! Hyperlink reference not valid.).
Section 42229-5 (42229-5)
Tablets: Each film-coated tablet contains paroxetine hydrochloride equivalent to paroxetine as follows: 40 mg– beige to light brown. Inactive ingredients consist of hypromellose, glyceryl behenate, lactose monohydrate, magnesium stearate, titanium dioxide, polyethylene glycols, iron oxide yellow and iron oxide red.
Section 43681-6 (43681-6)
Pharmacodynamics: The efficacy of paroxetine in the treatment of major depressive disorder, social anxiety disorder, obsessive compulsive disorder (OCD), panic disorder (PD) and generalized anxiety disorder (GAD) is presumed to be linked to potentiation of serotonergic activity in the central nervous system resulting from inhibition of neuronal reuptake of serotonin (5-hydroxy-tryptamine, 5-HT). Studies at clinically relevant doses in humans have demonstrated that paroxetine blocks the uptake of serotonin into human platelets. In vitro studies in animals also suggest that paroxetine is a potent and highly selective inhibitor of neuronal serotonin reuptake and has only very weak effects on norepinephrine and dopamine neuronal reuptake. In vitro radioligand binding studies indicate that paroxetine has little affinity for muscarinic, alpha1-, alpha2-, beta-adrenergic-, dopamine (D2-, 5-HT1-, 5-HT2-, and histamine (H1)-receptors; antagonism of muscarinic, histaminergic, and alpha1- receptors has been associated with various anticholinergic, sedative, and cardiovascular effects for other psychotropic drugs.
Because the relative potencies of paroxetine’s major metabolites are at most 1/50 of the parent compound, they are essentially inactive.
Section 43682-4 (43682-4)
Pharmacokinetics: Paroxetine hydrochloride is completely absorbed after oral dosing of a solution of the hydrochloride salt. The mean elimination half-life is approximately 21 hours (CV 32%) after oral dosing of 30 mg tablets of Paroxetine Tablets daily for 30 days. Paroxetine is extensively metabolized and the metabolites are considered to be inactive. Nonlinearity in pharmacokinetics is observed with increasing doses. Paroxetine metabolism is mediated in part by CYP2D6, and the metabolites are primarily excreted in the urine and to some extent in the feces. Pharmacokinetic behavior of paroxetine has not been evaluated in subjects who are deficient in CYP2D6 (poor metabolizers).
In a meta-analysis of paroxetine from 4 studies done in healthy volunteers following multiple dosing of 20 mg/day to 40 mg/day, males did not exhibit a significantly lower Cmax or AUC than females.
Section 44425-7 (44425-7)
Store tablets at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
You may report side effects to Solco Healthcare US, LLC at 1-866-257-2597 or FDA at 1-800-FDA-1088.
Manufactured by:
Zhejiang Huahai Pharmaceutical Co., Ltd.
Xunqiao, Linhai, Zhejiang 317024, China
Distributed by:
Solco Healthcare US, LLC.
Cranbury, NJ 08512, USA
Repackaged by:
Proficient Rx LP
Thousand Oaks, CA 91320
Issue Date: 10/2014
07490-01
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL - Container Label - 40 mg container label image
NDC 71205-198-90 Rx only
Paroxetine
Tablets, USP
40 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
90 Tablets
Precautions (PRECAUTIONS)
-
General: Activation of Mania/Hypomania: During premarketing testing, hypomania or mania occurred in approximately 1.0% of unipolar patients treated with Paroxetine Tablets compared to 1.1% of active-control and 0.3% of placebo-treated unipolar patients. In a subset of patients classified as bipolar, the rate of manic episodes was 2.2% for Paroxetine Tablets and 11.6% for the combined active-control groups. As with all drugs effective in the treatment of major depressive disorder, Paroxetine Tablets should be used cautiously in patients with a history of mania.
Medication Guide
Paroxetine Tablets USP
(PA-rox-eh-tine)
Read the Medication Guide that comes with Paroxetine Tablets before you start taking it and each time you get a refill. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or treatment. Talk with your healthcare provider if there is something you do not understand or want to learn more about.
What is the most important information I should know about Paroxetine Tablets?
Paroxetine Tablets and other antidepressant medicines may cause serious side effects, including:
1. Suicidal thoughts or actions:
-
•Paroxetine Tablets and other antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers, or young adults within the first few months of treatment or when the dose is changed.
-
•Depression or other serious mental illnesses are the most important causes of suicidal thoughts or actions.
-
•Watch for these changes and call your healthcare provider right away if you notice:
-
oNew or sudden changes in mood, behavior, actions, thoughts, or feelings, especially if severe.
-
oPay particular attention to such changes when Paroxetine Tablets are started or when the dose is changed.
-
Keep all follow-up visits with your healthcare provider and call between visits if you are worried about symptoms.
Call your healthcare provider right away if you have any of the following symptoms, or call 911 if an emergency, especially if they are new, worse, or worry you:
-
•attempts to commit suicide
-
•acting on dangerous impulses
-
•acting aggressive or violent
-
•thoughts about suicide or dying
-
•new or worse depression
-
•new or worse anxiety or panic attacks
-
•feeling agitated, restless, angry, or irritable
-
•trouble sleeping
-
•an increase in activity or talking more than what is normal for you
-
•other unusual changes in behavior or mood
Call your healthcare provider right away if you have any of the following symptoms, or call 911 if an emergency. Paroxetine Tablets may be associated with these serious side effects:
2. Serotonin Syndrome or Neuroleptic Malignant Syndrome-like reactions. This condition can be life-threatening and may include:
-
1.agitation, hallucinations, coma, or other changes in mental status
-
2.coordination problems or muscle twitching (overactive reflexes)
-
3.racing heartbeat, high or low blood pressure
-
4.sweating or fever
-
5.nausea, vomiting, or diarrhea
-
6.muscle rigidity
3. Visual problems
-
1.eye pain
-
2.changes in vision
-
3.swelling or redness in or around the eye
Only some people are at risk for these problems. You may want to undergo an eye examination to see if you are at risk and receive preventative treatment if you are.
4. Severe allergic reactions:
-
1.trouble breathing
-
2.swelling of the face, tongue, eyes, or mouth
-
3.rash, itchy welts (hives), or blisters, alone or with fever or joint pain
5. Abnormal bleeding: Paroxetine Tablets and other antidepressant medicines may increase your risk of bleeding or bruising, especially if you take the blood thinner warfarin (Coumadin"®", Jantoven"®"), a non-steroidal anti-inflammatory drug (NSAIDs, like ibuprofen or naproxen), or aspirin.
6. Seizures or convulsions
7. Manic episodes:
-
1.greatly increased energy
-
2.severe trouble sleeping
-
3.racing thoughts
-
4.reckless behavior
-
5.unusually grand ideas
-
6.excessive happiness or irritability
-
7.talking more or faster than usual
8. Changes in appetite or weight. Children and adolescents should have height and weight monitored during treatment.
9. Low salt (sodium) levels in the blood. Elderly people may be at greater risk for this. Symptoms may include:
-
1.headache
-
2.weakness or feeling unsteady
-
3.confusion, problems concentrating or thinking, or memory problems
Do not stop Paroxetine Tablets without first talking to your healthcare provider. Stopping Paroxetine Tablets too quickly may cause serious symptoms including:
-
1.anxiety, irritability, high or low mood, feeling restless, or changes in sleep habits
-
2.headache, sweating, nausea, dizziness
-
3.electric shock-like sensations, shaking, confusion
What are Paroxetine Tablets?
Paroxetine Tablets are prescription medicines used to treat depression. It is important to talk with your healthcare provider about the risks of treating depression and also the risks of not treating it. You should discuss all treatment choices with your healthcare provider. Paroxetine Tablets are also used to treat:
-
1.Major Depressive Disorder (MDD)
-
2.Obsessive Compulsive Disorder (OCD)
-
3.Panic Disorder
-
4.Social Anxiety Disorder
-
5.Generalized Anxiety Disorder (GAD)
Talk to your healthcare provider if you do not think that your condition is getting better with treatment using Paroxetine Tablets.
Who should not take Paroxetine Tablets?
Do not take Paroxetine Tablets if you:
-
1.are allergic to paroxetine or any of the ingredients in Paroxetine Tablets. See the end of this Medication Guide for a complete list of ingredients in Paroxetine Tablets.
-
2.take a monoamine oxidase inhibitor (MAOI). Ask your healthcare provider or pharmacist if you are not sure if you take an MAOI, including the antibiotic linezolid.
-
1.Do not take an MAOI within 2 weeks of stopping Paroxetine Tablets unless directed to do so by your physician.
-
2.Do not start Paroxetine Tablets if you stopped taking an MAOI in the last 2 weeks unless directed to do so by your physician.
-
3.People who take Paroxetine Tablets close in time to an MAOI may have serious or even life-threatening side effects. Get medical help right away if you have any of these symptoms:
-
1.high fever
-
2.uncontrolled muscle spasms
-
3.stiff muscles
-
4.rapid changes in heart rate or blood pressure
-
5.confusion
-
6.loss of consciousness (pass out)
-
-
4.take MELLARIL ®* (thioridazine). Do not take MELLARIL ®* together with Paroxetine Tablets because this can cause serious heart rhythm problems or sudden death.
-
5.take the antipsychotic medicine pimozide (ORAP ®* ) because this can cause serious heart problems.
-
-
What should I tell my healthcare provider before taking Paroxetine Tablets? Ask if you are not sure.
Tell your healthcare provider about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal supplements. Paroxetine Tablets and some medicines may interact with each other, may not work as well, or may cause serious side effects.
Your healthcare provider or pharmacist can tell you if it is safe to take Paroxetine Tablets with your other medicines. Do not start or stop any medicine while taking Paroxetine Tablets without talking to your healthcare provider first.
|
If you take Paroxetine Tablets, you should not take any other medicines that contain paroxetine, including paroxetine extended-release tablets and PEXEVA®* (paroxetine mesylate). |
How should I take Paroxetine Tablets?
-
1.Take Paroxetine Tablets exactly as prescribed. Your healthcare provider may need to change the dose of Paroxetine Tablets until it is the right dose for you.
-
2.Paroxetine Tablets may be taken with or without food.
-
3.If you miss a dose of Paroxetine Tablets, take the missed dose as soon as you remember. If it is almost time for the next dose, skip the missed dose and take your next dose at the regular time. Do not take two doses of Paroxetine Tablets at the same time.
-
4.If you take too many Paroxetine Tablets call your healthcare provider or poison control center right away, or get emergency treatment.
-
5.Do not stop taking Paroxetine Tablets suddenly without talking to your doctor (unless you have symptoms of a severe allergic reaction). If you need to stop taking Paroxetine Tablets, your healthcare provider can tell you how to safely stop taking it.
What should I avoid while taking Paroxetine Tablets?
Paroxetine Tablets can cause sleepiness or may affect your ability to make decisions, think clearly, or react quickly. You should not drive, operate heavy machinery, or do other dangerous activities until you know how Paroxetine Tablets affect you. Do not drink alcohol while using Paroxetine Tablets.
What are possible side effects of Paroxetine Tablets?
Paroxetine Tablets may cause serious side effects, including all of those described in the section entitled “What is the most important information I should know about Paroxetine Tablets?”
Common possible side effects in people who take Paroxetine Tablets include:
-
1.nausea
-
2.sleepiness
-
3.weakness
-
4.dizziness
-
5.feeling anxious or trouble sleeping
-
6.sexual problems
-
7.sweating
-
8.shaking
-
9.not feeling hungry
-
10.dry mouth
-
11.constipation
-
12.infection
-
13.yawning
Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Paroxetine Tablets. For more information, ask your healthcare provider or pharmacist.
CALL YOUR DOCTOR FOR MEDICAL ADVICE ABOUT SIDE EFFECTS. YOU MAY REPORT SIDE EFFECTS TO SOLCO HEALTHCARE US, LLC AT 1-866-257-2597 OR TO THE FDA AT 1-800-FDA1088 or 1-800-332-1088.
How should I store Paroxetine Tablets?
-
1.Store Paroxetine Tablets at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature].
-
2.Keep Paroxetine Tablets away from light.
-
3.Keep bottle of Paroxetine Tablets closed tightly.
Keep Paroxetine Tablets and all medicines out of the reach of children.
General information about Paroxetine Tablets
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use Paroxetine Tablets for a condition for which it was not prescribed. Do not give Paroxetine Tablets to other people, even if they have the same condition. It may harm them.
This Medication Guide summarizes the most important information about Paroxetine Tablets. If you would like more information, talk with your healthcare provider. You may ask your healthcare provider or pharmacist for information about Paroxetine Tablets that is written for healthcare professionals.
What are the ingredients in Paroxetine Tablets?
Active ingredient: paroxetine hydrochloride
Inactive ingredients in tablets: hypromellose, glyceryl behenate, lactose monohydrate, magnesium stearate, titanium dioxide, polyethylene glycols, iron oxide yellow, and iron oxide red.
*: Brand names listed are trademarks of their respective owners and are not trademarks of Solco Healthcare US, LLC.
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:05.345857 · Updated: 2026-03-14T22:04:55.765860