Nystatin Tablets Usp
8fd35f97-5405-2160-ca0f-35034ecd5a92
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
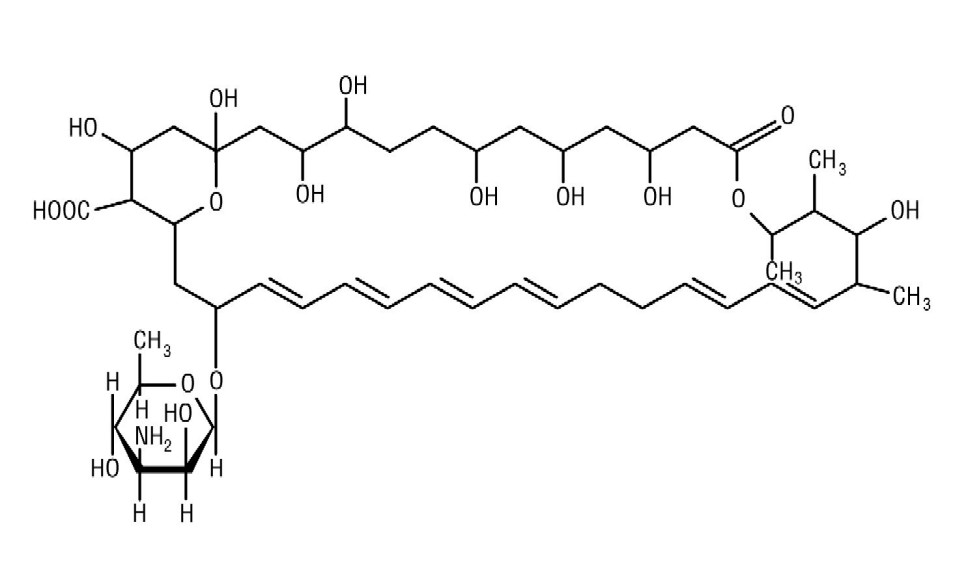
Nystatin, USP is an antimycotic polyene antibiotic obtained from Streptomyces noursei . Its structural formula: C 47 H 75 NO 17 M.W. 926.13 Nystatin tablets USP contain the inactive ingredients: Corn Starch, Povidone, Compressible Sugar, Microcrystalline Cellulose, Sodium Starch Glycolate, Talc, Magnesium Stearate, Purified Water, and Coloring.
Indications and Usage
Nystatin tablets are intended for the treatment of non-esophageal mucus membrane gastrointestinal candidiasis.
Dosage and Administration
The usual therapeutic dosage is one to two tablets (500,000 to 1,000,000 units nystatin) three times daily. Treatment should generally be continued for at least 48 hours after clinical cure to prevent relapse.
Contraindications
Nystatin Tablets are contraindicated in patients with a history of hypersensitivity to any of their components.
Adverse Reactions
Nystatin is well tolerated even with prolonged therapy. Oral irritation and sensitization have been reported (see PRECAUTIONS, General ).
How Supplied
Nystatin Tablets USP, 500,000 Units are round, convex, brown, film-coated tablet debossed with 93 on one side and 983 on the reverse and are packaged in bottles of 90 tablets (NDC 42291-651-90). Store at 20° and 25°C (68° and 77°F) [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required). Keep tightly closed. KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN. Manufactured For: AvKARE Pulaski, TN 38478 Mfg. Rev. 11/22 AV Rev. 09/25(M)
Medication Information
Indications and Usage
Nystatin tablets are intended for the treatment of non-esophageal mucus membrane gastrointestinal candidiasis.
Dosage and Administration
The usual therapeutic dosage is one to two tablets (500,000 to 1,000,000 units nystatin) three times daily. Treatment should generally be continued for at least 48 hours after clinical cure to prevent relapse.
Contraindications
Nystatin Tablets are contraindicated in patients with a history of hypersensitivity to any of their components.
Adverse Reactions
Nystatin is well tolerated even with prolonged therapy. Oral irritation and sensitization have been reported (see PRECAUTIONS, General).
How Supplied
Nystatin Tablets USP, 500,000 Units are round, convex, brown, film-coated tablet debossed with 93 on one side and 983 on the reverse and are packaged in bottles of 90 tablets (NDC 42291-651-90).
Store at 20° and 25°C (68° and 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
Keep tightly closed.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Manufactured For:
AvKARE
Pulaski, TN 38478
Mfg. Rev. 11/22
AV Rev. 09/25(M)
Description
Nystatin, USP is an antimycotic polyene antibiotic obtained from Streptomyces noursei. Its structural formula:
C 47H 75NO 17 M.W. 926.13
Nystatin tablets USP contain the inactive ingredients: Corn Starch, Povidone, Compressible Sugar, Microcrystalline Cellulose, Sodium Starch Glycolate, Talc, Magnesium Stearate, Purified Water, and Coloring.
Other
Tachycardia, bronchospasm, facial swelling, and nonspecific myalgia have also been rarely reported.
To report SUSPECTED ADVERSE REACTIONS contact AvKARE at 1-855-361-3993; email [email protected]; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
General
This medication is not to be used for the treatment of systemic mycoses. Discontinue treatment if sensitization or irritation is reported during use.
Overdosage
Oral doses of nystatin in excess of five million units daily have caused nausea and gastrointestinal upset. There have been no reports of serious toxic effects of superinfections (see CLINICAL PHARMACOLOGY, Pharmacokinetics).
Dermatologic
Rash, including urticaria has been reported rarely. Stevens-Johnson syndrome has been reported very rarely.
Microbiology
Nystatin is both fungistatic and fungicidal in vitro against a wide variety of yeasts and yeast like fungi. Candida albicans demonstrates no significant resistance to nystatin in vitro on repeated subculture in increasing levels of nystatin; other Candida species become quite resistant. Generally, resistance does not develop in vivo. Nystatin acts by binding to sterols in the cell membrane of susceptible Candida species with a resultant change in membrane permeability allowing leakage of intracellular components. Nystatin exhibits no appreciable activity against bacteria, protozoa, or viruses.
Nursing Mothers
It is not known whether nystatin is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when nystatin is administered to a nursing woman.
Gastrointestinal
Diarrhea (including one case of bloody diarrhea), nausea, vomiting, gastrointestinal upset/disturbances.
Pharmacokinetics
Gastrointestinal absorption of nystatin is insignificant. Most orally administered nystatin is passed unchanged in the stool. In patients with renal insufficiency receiving oral therapy with conventional dosage forms, significant plasma concentrations of nystatin may occasionally occur.
Pregnancy Category C
Animal reproduction studies have not been conducted with nystatin. It is also not known whether nystatin can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Nystatin should be given to a pregnant woman only if clearly needed.
Package Label.principal Display Panel
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to evaluate carcinogenic potential. There also have been no studies to determine mutagenicity or whether this medication affects fertility in males or females.
Structured Label Content
Other
Tachycardia, bronchospasm, facial swelling, and nonspecific myalgia have also been rarely reported.
To report SUSPECTED ADVERSE REACTIONS contact AvKARE at 1-855-361-3993; email [email protected]; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
General
This medication is not to be used for the treatment of systemic mycoses. Discontinue treatment if sensitization or irritation is reported during use.
Overdosage (OVERDOSAGE)
Oral doses of nystatin in excess of five million units daily have caused nausea and gastrointestinal upset. There have been no reports of serious toxic effects of superinfections (see CLINICAL PHARMACOLOGY, Pharmacokinetics).
Description (DESCRIPTION)
Nystatin, USP is an antimycotic polyene antibiotic obtained from Streptomyces noursei. Its structural formula:
C 47H 75NO 17 M.W. 926.13
Nystatin tablets USP contain the inactive ingredients: Corn Starch, Povidone, Compressible Sugar, Microcrystalline Cellulose, Sodium Starch Glycolate, Talc, Magnesium Stearate, Purified Water, and Coloring.
Dermatologic
Rash, including urticaria has been reported rarely. Stevens-Johnson syndrome has been reported very rarely.
How Supplied (HOW SUPPLIED)
Nystatin Tablets USP, 500,000 Units are round, convex, brown, film-coated tablet debossed with 93 on one side and 983 on the reverse and are packaged in bottles of 90 tablets (NDC 42291-651-90).
Store at 20° and 25°C (68° and 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
Keep tightly closed.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Manufactured For:
AvKARE
Pulaski, TN 38478
Mfg. Rev. 11/22
AV Rev. 09/25(M)
Microbiology
Nystatin is both fungistatic and fungicidal in vitro against a wide variety of yeasts and yeast like fungi. Candida albicans demonstrates no significant resistance to nystatin in vitro on repeated subculture in increasing levels of nystatin; other Candida species become quite resistant. Generally, resistance does not develop in vivo. Nystatin acts by binding to sterols in the cell membrane of susceptible Candida species with a resultant change in membrane permeability allowing leakage of intracellular components. Nystatin exhibits no appreciable activity against bacteria, protozoa, or viruses.
Nursing Mothers
It is not known whether nystatin is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when nystatin is administered to a nursing woman.
Gastrointestinal
Diarrhea (including one case of bloody diarrhea), nausea, vomiting, gastrointestinal upset/disturbances.
Pharmacokinetics
Gastrointestinal absorption of nystatin is insignificant. Most orally administered nystatin is passed unchanged in the stool. In patients with renal insufficiency receiving oral therapy with conventional dosage forms, significant plasma concentrations of nystatin may occasionally occur.
Adverse Reactions (ADVERSE REACTIONS)
Nystatin is well tolerated even with prolonged therapy. Oral irritation and sensitization have been reported (see PRECAUTIONS, General).
Contraindications (CONTRAINDICATIONS)
Nystatin Tablets are contraindicated in patients with a history of hypersensitivity to any of their components.
Pregnancy Category C
Animal reproduction studies have not been conducted with nystatin. It is also not known whether nystatin can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Nystatin should be given to a pregnant woman only if clearly needed.
Indications and Usage (INDICATIONS AND USAGE)
Nystatin tablets are intended for the treatment of non-esophageal mucus membrane gastrointestinal candidiasis.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
The usual therapeutic dosage is one to two tablets (500,000 to 1,000,000 units nystatin) three times daily. Treatment should generally be continued for at least 48 hours after clinical cure to prevent relapse.
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to evaluate carcinogenic potential. There also have been no studies to determine mutagenicity or whether this medication affects fertility in males or females.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:40.501139 · Updated: 2026-03-14T22:34:06.521153