These Highlights Do Not Include All The Information Needed To Use Moxifloxacin Ophthalmic Solution Safely And Effectively. See Full Prescribing Information For Moxifloxacin Ophthalmic Solution.
8f92f110-32d6-4d0c-a415-7a9a4cafb9b8
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2 ) 6/2020
Indications and Usage
Moxifloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms: Corynebacterium species Efficacy for this organism was studied in fewer than 10 infections. Micrococcus luteus Staphylococcus aureus Staphylococcus epidermidis Staphylococcus haemolyticus Staphylococcus hominis Staphylococcus warneri Streptococcus pneumoniae Streptococcus viridans group Acinetobacter lwoffii Haemophilus influenza Haemophilus parainfluenzae Chlamydia trachomatis
Dosage and Administration
Instill one drop in the affected eye 3 times a day for 7 days. Moxifloxacin ophthalmic solution is for topical ophthalmic use.
Warnings and Precautions
Hypersensitivity Reactions : Hypersensitivity and anaphylaxis have been reported with systemic use of moxifloxacin. ( 5.1 ) Prolonged Use: May result in overgrowth of non-susceptible organisms, including fungi. If superinfection occurs, discontinue use and institute alternative therapy. ( 5.2 ) Avoid Contact Lens Wear : Patients should not wear contact lenses if they have signs or symptoms of bacterial conjunctivitis. ( 5.3 )
Contraindications
Moxifloxacin ophthalmic solution is contraindicated in patients with a history of hypersensitivity to moxifloxacin, to other quinolones, or to any of the components in this medication.
Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in practice. The most frequently reported ocular adverse events were conjunctivitis, decreased visual acuity, dry eye, keratitis, ocular discomfort, ocular hyperemia, ocular pain, ocular pruritus, subconjunctival hemorrhage, and tearing. These events occurred in approximately 1% to 6% of patients. Non-ocular adverse events reported at a rate of 1% to 4% were fever, increased cough, infection, otitis media, pharyngitis, rash, and rhinitis.
Drug Interactions
Drug-drug interaction studies have not been conducted with moxifloxacin ophthalmic solution. In vitro studies indicate that moxifloxacin does not inhibit CYP3A4, CYP2D6, CYP2C9, CYP2C19, or CYP1A2, indicating that moxifloxacin is unlikely to alter the pharmacokinetics of drugs metabolized by these cytochrome P450 isozymes.
Storage and Handling
Moxifloxacin hydrochloride ophthalmic solution, USP is supplied as a sterile ophthalmic solution in a low-density polyethylene dropper tip bottle with a tan polypropylene cap closure. Tamper evidence is provided with a band around the neck area of the package. 3 mL in a 5 mL bottle – NDC 76420-331-03 (relabeled from NDC 0832-1410-03)
How Supplied
Moxifloxacin hydrochloride ophthalmic solution, USP is supplied as a sterile ophthalmic solution in a low-density polyethylene dropper tip bottle with a tan polypropylene cap closure. Tamper evidence is provided with a band around the neck area of the package. 3 mL in a 5 mL bottle – NDC 76420-331-03 (relabeled from NDC 0832-1410-03)
Medication Information
Warnings and Precautions
Hypersensitivity Reactions : Hypersensitivity and anaphylaxis have been reported with systemic use of moxifloxacin. ( 5.1 ) Prolonged Use: May result in overgrowth of non-susceptible organisms, including fungi. If superinfection occurs, discontinue use and institute alternative therapy. ( 5.2 ) Avoid Contact Lens Wear : Patients should not wear contact lenses if they have signs or symptoms of bacterial conjunctivitis. ( 5.3 )
Indications and Usage
Moxifloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms: Corynebacterium species Efficacy for this organism was studied in fewer than 10 infections. Micrococcus luteus Staphylococcus aureus Staphylococcus epidermidis Staphylococcus haemolyticus Staphylococcus hominis Staphylococcus warneri Streptococcus pneumoniae Streptococcus viridans group Acinetobacter lwoffii Haemophilus influenza Haemophilus parainfluenzae Chlamydia trachomatis
Dosage and Administration
Instill one drop in the affected eye 3 times a day for 7 days. Moxifloxacin ophthalmic solution is for topical ophthalmic use.
Contraindications
Moxifloxacin ophthalmic solution is contraindicated in patients with a history of hypersensitivity to moxifloxacin, to other quinolones, or to any of the components in this medication.
Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in practice. The most frequently reported ocular adverse events were conjunctivitis, decreased visual acuity, dry eye, keratitis, ocular discomfort, ocular hyperemia, ocular pain, ocular pruritus, subconjunctival hemorrhage, and tearing. These events occurred in approximately 1% to 6% of patients. Non-ocular adverse events reported at a rate of 1% to 4% were fever, increased cough, infection, otitis media, pharyngitis, rash, and rhinitis.
Drug Interactions
Drug-drug interaction studies have not been conducted with moxifloxacin ophthalmic solution. In vitro studies indicate that moxifloxacin does not inhibit CYP3A4, CYP2D6, CYP2C9, CYP2C19, or CYP1A2, indicating that moxifloxacin is unlikely to alter the pharmacokinetics of drugs metabolized by these cytochrome P450 isozymes.
Storage and Handling
Moxifloxacin hydrochloride ophthalmic solution, USP is supplied as a sterile ophthalmic solution in a low-density polyethylene dropper tip bottle with a tan polypropylene cap closure. Tamper evidence is provided with a band around the neck area of the package. 3 mL in a 5 mL bottle – NDC 76420-331-03 (relabeled from NDC 0832-1410-03)
How Supplied
Moxifloxacin hydrochloride ophthalmic solution, USP is supplied as a sterile ophthalmic solution in a low-density polyethylene dropper tip bottle with a tan polypropylene cap closure. Tamper evidence is provided with a band around the neck area of the package. 3 mL in a 5 mL bottle – NDC 76420-331-03 (relabeled from NDC 0832-1410-03)
Description
Dosage and Administration ( 2 ) 6/2020
Section 42229-5
Risk Summary
There are no adequate and well-controlled studies with moxifloxacin ophthalmic solution in pregnant women to inform any drug-associated risks.
Oral administration of moxifloxacin to pregnant rats and monkeys and intravenously to pregnant rabbits during the period of organogenesis did not produce adverse maternal or fetal effects at clinically relevant doses. Oral administration of moxifloxacin to pregnant rats during late gestation through lactation did not produce adverse maternal, fetal or neonatal effects at clinically relevant doses (see Data).
Section 43683-2
| Dosage and Administration ( 2) | 6/2020 |
Section 44425-7
Storage: Store at 2°C to 25°C (36°F to 77°F).
11 Description
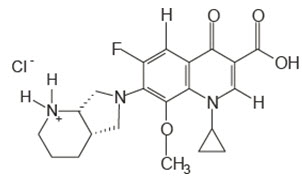
Moxifloxacin ophthalmic solution, USP 0.5% is a sterile solution for topical ophthalmic use. Moxifloxacin hydrochloride is an 8-methoxy fluoroquinolone anti-infective, with a diazabicyclononyl ring at the C7 position. Chemical Name: 1-Cyclopropyl-6-fluoro-1,4-dihydro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolol[3,4b]pyridin6-yl]-4-oxo-3-quinolinecarboxylic acid, monohydrochloride. The molecular formula for moxifloxacin hydrochloride, USP is C 21H 24FN 3O 4∙HCl and its molecular weight is 437.9 g/mol. The chemical structure is presented below.
Moxifloxacin hydrochloride is a slightly yellow to yellow crystalline powder.
Each mL of moxifloxacin ophthalmic solution contains 5.45 mg moxifloxacin hydrochloride, equivalent to 5 mg moxifloxacin base. Moxifloxacin ophthalmic solution contains: Active: Moxifloxacin 0.5% (5 mg/mL); Inactives: Boric acid, purified water and sodium chloride. May also contain hydrochloric acid/sodium hydroxide to adjust pH to approximately 6.8.
Moxifloxacin ophthalmic solution, USP is an isotonic solution with an osmolality of approximately 290 mOsm/kg.
12.4 Microbiology
The antibacterial action of moxifloxacin results from inhibition of the topoisomerase II (DNA gyrase) and topoisomerase IV. DNA gyrase is an essential enzyme that is involved in the replication, transcription and repair of bacterial DNA. Topoisomerase IV is an enzyme known to play a key role in the partitioning of the chromosomal DNA during bacterial cell division.
The mechanism of action for quinolones, including moxifloxacin, is different from that of macrolides, aminoglycosides, or tetracyclines. Therefore, moxifloxacin may be active against pathogens that are resistant to these antibiotics and these antibiotics may be active against pathogens that are resistant to moxifloxacin. There is no cross-resistance between moxifloxacin and the aforementioned classes of antibiotics. Cross-resistance has been observed between systemic moxifloxacin and some other quinolones.
In vitroresistance to moxifloxacin develops via multiple-step mutations. Resistance to moxifloxacin occurs in vitroat a general frequency of between 1.8 × 10 -9to less than 1 × 10 -11for gram-positive bacteria.
Moxifloxacin has been shown to be active against most strains of the following microorganisms, both in vitroand in clinical infections as described in the Indications and Usage section:
8.4 Pediatric Use
The safety and effectiveness of moxifloxacin ophthalmic solution have been established in all ages. Use of moxifloxacin ophthalmic solution is supported by evidence from adequate and well controlled studies of moxifloxacin ophthalmic solution in adults, children, and neonates [see Clinical Studies (14)] .
There is no evidence that the ophthalmic administration of moxifloxacin ophthalmic solution has any effect on weight bearing joints, even though oral administration of some quinolones has been shown to cause arthropathy in immature animals.
8.5 Geriatric Use
No overall differences in safety and effectiveness have been observed between elderly and younger patients.
14 Clinical Studies
In two randomized, double-masked, multicenter, controlled clinical trials in which patients were dosed 3 times a day for 4 days, moxifloxacin ophthalmic solution produced clinical cures on Day 5 to 6 in 66% to 69% of patients treated for bacterial conjunctivitis. Microbiological success rates for the eradication of baseline pathogens ranged from 84% to 94%.
In a randomized, double-masked, multicenter, parallel-group clinical trial of pediatric patients with bacterial conjunctivitis between birth and 31 days of age, patients were dosed with moxifloxacin ophthalmic or another anti-infective agent. Clinical outcomes for the trial demonstrated a clinical cure rate of 80% at Day 9 and a microbiological eradication success rate of 92% at Day 9.
Please note that microbiologic eradication does not always correlate with clinical outcome in anti-infective trials.
4 Contraindications
Moxifloxacin ophthalmic solution is contraindicated in patients with a history of hypersensitivity to moxifloxacin, to other quinolones, or to any of the components in this medication.
6 Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most frequently reported ocular adverse events were conjunctivitis, decreased visual acuity, dry eye, keratitis, ocular discomfort, ocular hyperemia, ocular pain, ocular pruritus, subconjunctival hemorrhage, and tearing. These events occurred in approximately 1% to 6% of patients.
Non-ocular adverse events reported at a rate of 1% to 4% were fever, increased cough, infection, otitis media, pharyngitis, rash, and rhinitis.
7 Drug Interactions
Drug-drug interaction studies have not been conducted with moxifloxacin ophthalmic solution. In vitrostudies indicate that moxifloxacin does not inhibit CYP3A4, CYP2D6, CYP2C9, CYP2C19, or CYP1A2, indicating that moxifloxacin is unlikely to alter the pharmacokinetics of drugs metabolized by these cytochrome P450 isozymes.
12.3 Pharmacokinetics
Plasma concentrations of moxifloxacin were measured in healthy adult male and female subjects who received bilateral topical ocular doses of moxifloxacin ophthalmic solution 3 times a day. The mean steady-state C max(2.7 ng/mL) and AUC 0-00(41.9 ng∙hr/mL) values were 1,600 and 1,100 times lower than the mean C maxand AUC reported after therapeutic 400 mg doses of moxifloxacin. The plasma half-life of moxifloxacin was estimated to be 13 hours.
1 Indications and Usage
Moxifloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms:
Corynebacteriumspecies
Micrococcus luteus
Staphylococcus aureus
Staphylococcus epidermidis
Staphylococcus haemolyticus
Staphylococcus hominis
Staphylococcus warneri
Streptococcus pneumoniae
Streptococcus viridans group
Acinetobacter lwoffii
Haemophilus influenza
Haemophilus parainfluenzae
Chlamydia trachomatis
Principal Display Panel
12.1 Mechanism of Action
Moxifloxacin is a member of the fluoroquinolone class of anti-infective drugs [see Microbiology (12.4)] .
5 Warnings and Precautions
- Hypersensitivity Reactions: Hypersensitivity and anaphylaxis have been reported with systemic use of moxifloxacin. ( 5.1)
- Prolonged Use:May result in overgrowth of non-susceptible organisms, including fungi. If superinfection occurs, discontinue use and institute alternative therapy. ( 5.2)
- Avoid Contact Lens Wear: Patients should not wear contact lenses if they have signs or symptoms of bacterial conjunctivitis. ( 5.3)
2 Dosage and Administration
Instill one drop in the affected eye 3 times a day for 7 days. Moxifloxacin ophthalmic solution is for topical ophthalmic use.
3 Dosage Forms and Strengths
Ophthalmic solution containing moxifloxacin 0.5%.
5.1 Hypersensitivity Reactions
In patients receiving systemically administered quinolones, including moxifloxacin, serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported, some following the first dose. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, angioedema (including laryngeal, pharyngeal or facial edema), airway obstruction, dyspnea, urticaria, and itching. If an allergic reaction to moxifloxacin occurs, discontinue use of the drug. Serious acute hypersensitivity reactions may require immediate emergency treatment. Oxygen and airway management should be administered as clinically indicated.
5.3 Avoidance of Contact Lens Wear
Patients should be advised not to wear contact lenses if they have signs or symptoms of bacterial conjunctivitis.
16 How Supplied/storage and Handling
Moxifloxacin hydrochloride ophthalmic solution, USP is supplied as a sterile ophthalmic solution in a low-density polyethylene dropper tip bottle with a tan polypropylene cap closure. Tamper evidence is provided with a band around the neck area of the package.
3 mL in a 5 mL bottle – NDC 76420-331-03 (relabeled from NDC 0832-1410-03)
5.2 Growth of Resistant Organisms With Prolonged Use
As with other anti-infectives, prolonged use may result in overgrowth of non-susceptible organisms, including fungi. If superinfection occurs, discontinue use and institute alternative therapy. Whenever clinical judgment dictates, the patient should be examined with the aid of magnification, such as slit-lamp biomicroscopy, and, where appropriate, fluorescein staining.
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
There are no adequate and well-controlled studies with moxifloxacin ophthalmic solution in pregnant women to inform any drug-associated risks.
Oral administration of moxifloxacin to pregnant rats and monkeys and intravenously to pregnant rabbits during the period of organogenesis did not produce adverse maternal or fetal effects at clinically relevant doses. Oral administration of moxifloxacin to pregnant rats during late gestation through lactation did not produce adverse maternal, fetal or neonatal effects at clinically relevant doses (see Data).
Section 43683-2 (43683-2)
| Dosage and Administration ( 2) | 6/2020 |
Section 44425-7 (44425-7)
Storage: Store at 2°C to 25°C (36°F to 77°F).
11 Description (11 DESCRIPTION)
Moxifloxacin ophthalmic solution, USP 0.5% is a sterile solution for topical ophthalmic use. Moxifloxacin hydrochloride is an 8-methoxy fluoroquinolone anti-infective, with a diazabicyclononyl ring at the C7 position. Chemical Name: 1-Cyclopropyl-6-fluoro-1,4-dihydro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolol[3,4b]pyridin6-yl]-4-oxo-3-quinolinecarboxylic acid, monohydrochloride. The molecular formula for moxifloxacin hydrochloride, USP is C 21H 24FN 3O 4∙HCl and its molecular weight is 437.9 g/mol. The chemical structure is presented below.
Moxifloxacin hydrochloride is a slightly yellow to yellow crystalline powder.
Each mL of moxifloxacin ophthalmic solution contains 5.45 mg moxifloxacin hydrochloride, equivalent to 5 mg moxifloxacin base. Moxifloxacin ophthalmic solution contains: Active: Moxifloxacin 0.5% (5 mg/mL); Inactives: Boric acid, purified water and sodium chloride. May also contain hydrochloric acid/sodium hydroxide to adjust pH to approximately 6.8.
Moxifloxacin ophthalmic solution, USP is an isotonic solution with an osmolality of approximately 290 mOsm/kg.
12.4 Microbiology
The antibacterial action of moxifloxacin results from inhibition of the topoisomerase II (DNA gyrase) and topoisomerase IV. DNA gyrase is an essential enzyme that is involved in the replication, transcription and repair of bacterial DNA. Topoisomerase IV is an enzyme known to play a key role in the partitioning of the chromosomal DNA during bacterial cell division.
The mechanism of action for quinolones, including moxifloxacin, is different from that of macrolides, aminoglycosides, or tetracyclines. Therefore, moxifloxacin may be active against pathogens that are resistant to these antibiotics and these antibiotics may be active against pathogens that are resistant to moxifloxacin. There is no cross-resistance between moxifloxacin and the aforementioned classes of antibiotics. Cross-resistance has been observed between systemic moxifloxacin and some other quinolones.
In vitroresistance to moxifloxacin develops via multiple-step mutations. Resistance to moxifloxacin occurs in vitroat a general frequency of between 1.8 × 10 -9to less than 1 × 10 -11for gram-positive bacteria.
Moxifloxacin has been shown to be active against most strains of the following microorganisms, both in vitroand in clinical infections as described in the Indications and Usage section:
8.4 Pediatric Use
The safety and effectiveness of moxifloxacin ophthalmic solution have been established in all ages. Use of moxifloxacin ophthalmic solution is supported by evidence from adequate and well controlled studies of moxifloxacin ophthalmic solution in adults, children, and neonates [see Clinical Studies (14)] .
There is no evidence that the ophthalmic administration of moxifloxacin ophthalmic solution has any effect on weight bearing joints, even though oral administration of some quinolones has been shown to cause arthropathy in immature animals.
8.5 Geriatric Use
No overall differences in safety and effectiveness have been observed between elderly and younger patients.
14 Clinical Studies (14 CLINICAL STUDIES)
In two randomized, double-masked, multicenter, controlled clinical trials in which patients were dosed 3 times a day for 4 days, moxifloxacin ophthalmic solution produced clinical cures on Day 5 to 6 in 66% to 69% of patients treated for bacterial conjunctivitis. Microbiological success rates for the eradication of baseline pathogens ranged from 84% to 94%.
In a randomized, double-masked, multicenter, parallel-group clinical trial of pediatric patients with bacterial conjunctivitis between birth and 31 days of age, patients were dosed with moxifloxacin ophthalmic or another anti-infective agent. Clinical outcomes for the trial demonstrated a clinical cure rate of 80% at Day 9 and a microbiological eradication success rate of 92% at Day 9.
Please note that microbiologic eradication does not always correlate with clinical outcome in anti-infective trials.
4 Contraindications (4 CONTRAINDICATIONS)
Moxifloxacin ophthalmic solution is contraindicated in patients with a history of hypersensitivity to moxifloxacin, to other quinolones, or to any of the components in this medication.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most frequently reported ocular adverse events were conjunctivitis, decreased visual acuity, dry eye, keratitis, ocular discomfort, ocular hyperemia, ocular pain, ocular pruritus, subconjunctival hemorrhage, and tearing. These events occurred in approximately 1% to 6% of patients.
Non-ocular adverse events reported at a rate of 1% to 4% were fever, increased cough, infection, otitis media, pharyngitis, rash, and rhinitis.
7 Drug Interactions (7 DRUG INTERACTIONS)
Drug-drug interaction studies have not been conducted with moxifloxacin ophthalmic solution. In vitrostudies indicate that moxifloxacin does not inhibit CYP3A4, CYP2D6, CYP2C9, CYP2C19, or CYP1A2, indicating that moxifloxacin is unlikely to alter the pharmacokinetics of drugs metabolized by these cytochrome P450 isozymes.
12.3 Pharmacokinetics
Plasma concentrations of moxifloxacin were measured in healthy adult male and female subjects who received bilateral topical ocular doses of moxifloxacin ophthalmic solution 3 times a day. The mean steady-state C max(2.7 ng/mL) and AUC 0-00(41.9 ng∙hr/mL) values were 1,600 and 1,100 times lower than the mean C maxand AUC reported after therapeutic 400 mg doses of moxifloxacin. The plasma half-life of moxifloxacin was estimated to be 13 hours.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Moxifloxacin ophthalmic solution is indicated for the treatment of bacterial conjunctivitis caused by susceptible strains of the following organisms:
Corynebacteriumspecies
Micrococcus luteus
Staphylococcus aureus
Staphylococcus epidermidis
Staphylococcus haemolyticus
Staphylococcus hominis
Staphylococcus warneri
Streptococcus pneumoniae
Streptococcus viridans group
Acinetobacter lwoffii
Haemophilus influenza
Haemophilus parainfluenzae
Chlamydia trachomatis
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
12.1 Mechanism of Action
Moxifloxacin is a member of the fluoroquinolone class of anti-infective drugs [see Microbiology (12.4)] .
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions: Hypersensitivity and anaphylaxis have been reported with systemic use of moxifloxacin. ( 5.1)
- Prolonged Use:May result in overgrowth of non-susceptible organisms, including fungi. If superinfection occurs, discontinue use and institute alternative therapy. ( 5.2)
- Avoid Contact Lens Wear: Patients should not wear contact lenses if they have signs or symptoms of bacterial conjunctivitis. ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Instill one drop in the affected eye 3 times a day for 7 days. Moxifloxacin ophthalmic solution is for topical ophthalmic use.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Ophthalmic solution containing moxifloxacin 0.5%.
5.1 Hypersensitivity Reactions
In patients receiving systemically administered quinolones, including moxifloxacin, serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported, some following the first dose. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, angioedema (including laryngeal, pharyngeal or facial edema), airway obstruction, dyspnea, urticaria, and itching. If an allergic reaction to moxifloxacin occurs, discontinue use of the drug. Serious acute hypersensitivity reactions may require immediate emergency treatment. Oxygen and airway management should be administered as clinically indicated.
5.3 Avoidance of Contact Lens Wear
Patients should be advised not to wear contact lenses if they have signs or symptoms of bacterial conjunctivitis.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Moxifloxacin hydrochloride ophthalmic solution, USP is supplied as a sterile ophthalmic solution in a low-density polyethylene dropper tip bottle with a tan polypropylene cap closure. Tamper evidence is provided with a band around the neck area of the package.
3 mL in a 5 mL bottle – NDC 76420-331-03 (relabeled from NDC 0832-1410-03)
5.2 Growth of Resistant Organisms With Prolonged Use
As with other anti-infectives, prolonged use may result in overgrowth of non-susceptible organisms, including fungi. If superinfection occurs, discontinue use and institute alternative therapy. Whenever clinical judgment dictates, the patient should be examined with the aid of magnification, such as slit-lamp biomicroscopy, and, where appropriate, fluorescein staining.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:46.196041 · Updated: 2026-03-14T22:25:35.146821