Aviane®
8f88b17a-5d0e-448a-8a60-f49c28ba6dfb
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Patients should be counseled that oral contraceptives do not protect against transmission of HIV (AIDS) and other sexually transmitted diseases (STDs) such as chlamydia, genital herpes, genital warts (human papillomavirus) , gonorrhea, hepatitis B, and syphilis.
Indications and Usage
Aviane (levonorgestrel and ethinyl estradiol tablets) is indicated for use by females of reproductive potential to prevent pregnancy. Limitations of use: The efficacy of Aviane (levonorgestrel and ethinyl estradiol tablets) in women with a body mass index (BMI) of > 35 kg/m 2 has not been adequately evaluated. In a clinical trial with Aviane (levonorgestrel and ethinyl estradiol tablets), 1,477 subjects had 7,720 cycles of use and a total of 5 pregnancies were reported. This represents an overall pregnancy rate of 0.84 per 100 woman-years. This rate includes patients who did not take the drug correctly. One or more pills were missed during 1,479 (18.8%) of the 7,870 cycles; thus all tablets were taken during 6,391 (81.2%) of the 7,870 cycles. Of the total 7,870 cycles, a total of 150 cycles were excluded from the calculation of the Pearl index due to the use of backup contraception and/or missing 3 or more consecutive pills. The mean BMI of the study population was 24 kg/m 2 . Females with a BMI greater than 30 kg/m 2 accounted for 12.1% (n=179) of the study population. Females with a BMI over 35 kg/m 2 accounted for 4.3% (n=63) of the study population.
Dosage and Administration
To achieve maximum contraceptive effectiveness, Aviane (levonorgestrel and ethinyl estradiol tablets) must be taken exactly as directed and at intervals not exceeding 24 hours. The dosage of Aviane-28 is one orange tablet daily for 21 consecutive days, followed by one light-green inert tablet daily for 7 consecutive days, according to the prescribed schedule. It is recommended that Aviane-28 tablets be taken at the same time each day. The dispenser should be kept in the wallet supplied to avoid possible fading of the pills. If the pills fade, patients should continue to take them as directed. During the first cycle of use The possibility of ovulation and conception prior to initiation of medication should be considered. The patient should be instructed to begin taking Aviane on either the first Sunday after the onset of menstruation (Sunday Start) or on Day 1 of menstruation (Day 1 Start). Sunday start: The patient is instructed to begin taking Aviane-28 on the first Sunday after the onset of menstruation. If menstruation begins on a Sunday, the first tablet (orange) is taken that day. One orange tablet should be taken daily for 21 consecutive days, followed by one light-green inert tablet daily for 7 consecutive days. Withdrawal bleeding should usually occur within 3 days following discontinuation of orange tablets and may not have finished before the next pack is started. During the first cycle, contraceptive reliance should not be placed on Aviane-28 until an orange tablet has been taken daily for 7 consecutive days, and a nonhormonal back-up method of birth control should be used during those 7 days. Day 1 start: During the first cycle of medication, the patient is instructed to begin taking Aviane-28 during the first 24 hours of her period (day one of her menstrual cycle). One orange tablet should be taken daily for 21 consecutive days, followed by one light-green inert tablet daily for 7 consecutive days. Withdrawal bleeding should usually occur within 3 days following discontinuation of orange tablets and may not have finished before the next pack is started. If medication is begun on day one of the menstrual cycle, no back-up contraception is necessary. If Aviane-28 tablets are started later than day one of the first menstrual cycle or postpartum, contraceptive reliance should not be placed on Aviane-28 tablets until after the first 7 consecutive days of administration, and a nonhormonal back-up method of birth control should be used during those 7 days. After the first cycle of use The patient begins her next and all subsequent courses of tablets on the day after taking her last light-green tablet. She should follow the same dosing schedule: 21 days on orange tablets followed by 7 days on light-green tablets. If in any cycle the patient starts tablets later than the proper day, she should protect herself against pregnancy by using a nonhormonal back-up method of birth control until she has taken an orange tablet daily for 7 consecutive days. Switching from another hormonal method of contraception When the patient is switching from a 21-day regimen of tablets, she should wait 7 days after her last tablet before she starts Aviane. She will probably experience withdrawal bleeding during that week. She should be sure that no more than 7 days pass after her previous 21-day regimen. When the patient is switching from a 28-day regimen of tablets, she should start her first pack of Aviane on the day after her last tablet. She should not wait any days between packs. The patient may switch any day from a progestin-only pill and should begin Aviane the next day. If switching from an implant or injection, the patient should start Aviane on the day of implant removal or, if using an injection, the day the next injection would be due. In switching from a progestin-only pill, injection, implant, or intrauterine system (IUS), the patient should be advised to use additional nonhormonal contraception (such as condoms) until active tablets have been taken for 7 consecutive days of the first pack. If spotting or breakthrough bleeding occurs If spotting or breakthrough bleeding occur, the patient is instructed to continue on the same regimen. This type of bleeding is usually transient and without significance; however, if the bleeding is persistent or prolonged, the patient is advised to consult her physician. Risk of pregnancy if tablets are missed While there is little likelihood of ovulation occurring if only one or two orange tablets are missed, the possibility of ovulation increases with each successive day that scheduled orange tablets are missed. Although the occurrence of pregnancy is unlikely if Aviane is taken according to directions, if withdrawal bleeding does not occur, the possibility of pregnancy must be considered. If the patient has not adhered to the prescribed schedule (missed one or more tablets or started taking them on a day later than she should have), the probability of pregnancy should be considered at the time of the first missed period and appropriate diagnostic measures taken. If the patient has adhered to the prescribed regimen and misses two consecutive periods, pregnancy should be ruled out. The risk of pregnancy increases with each active (orange) tablet missed. For additional patient instructions regarding missed tablets, see the WHAT TO DO IF YOU MISS PILLS section in the DETAILED PATIENT LABELING below. Use after pregnancy, abortion or miscarriage Aviane may be initiated no earlier than day 28 postpartum in the nonlactating mother or after a second trimester abortion due to the increased risk for thromboembolism (see C ONTRAINDICATIONS , WARNINGS , and PRECAUTIONS concerning thromboembolic disease). The patient should be advised to use additional nonhormonal contraception (such as condoms) until active tablets have been taken for 7 consecutive days. Aviane may be initiated immediately after a first trimester abortion or miscarriage. Instruct the patient to use additional nonhormonal contraception (such as condoms) until active tablets have been taken for 7 consecutive days, unless starting Aviane on the day of surgical abortion.
Contraindications
Combination oral contraceptives should not be used in women with any of the following conditions: Thrombophlebitis or thromboembolic disorders A history of deep-vein thrombophlebitis or thromboembolic disorders Cerebrovascular or coronary artery disease (current or past history) Valvular heart disease with thrombogenic complications Thrombogenic rhythm disorders Hereditary or acquired thrombophilias Prolonged immobilization (especially with major surgery) Diabetes with vascular involvement Headaches with focal neurological symptoms or migraine with aura Women with migraine who are 35 years or older Uncontrolled hypertension Known or suspected carcinoma of the breast or personal history of breast cancer Known or suspected estrogen- or progesterone sensitive malignancy Undiagnosed abnormal vaginal bleeding Cholestatic jaundice of pregnancy or jaundice with prior pill use Hepatic adenomas or carcinomas, or active liver disease Women who are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for alanine aminotransferase (ALT) elevations (see WARNINGS, Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment ) .
Adverse Reactions
An increased risk of the following serious adverse reactions (see WARNINGS section for additional information) has been associated with the use of oral contraceptives: Thromboembolic and thrombotic disorders and other vascular problems (including thrombophlebitis and venous thrombosis with or without pulmonary embolism, mesenteric thrombosis, arterial thromboembolism, myocardial infarction, cerebral hemorrhage, cerebral thrombosis), carcinoma of the reproductive organs and breasts, hepatic neoplasia (including hepatic adenomas or benign liver tumors), ocular lesions (including retinal vascular thrombosis), gallbladder disease, carbohydrate and lipid effects, elevated blood pressure, and headache including migraine. Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 2). Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 2). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use. FIGURE 2: Relevant Studies of Risk of Breast Cancer with Combined Oral Contraceptives RR = relative risk; OR = odds ratio; HR = hazard ratio. “ever COC” are females with current or past COC use; “never COC use” are females that never used COCs. The following adverse reactions associated with the use of oral CHCs were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Common adverse reactions associated with oral CHCs are headache, abdominal pain, nausea, metrorrhagia, vaginal moniliasis and pain, acne, and vaginitis. Additional adverse reactions that have been reported include the following: Eye disorder: intolerance to contact lenses, steepening of corneal curvature Gastrointestinal disorders: Abdominal bloating, vomiting General disorders and administration site conditions: Edema, fluid retention Hepatobiliary disorders: Cholestatic jaundice Psychiatric disorders: Change in libido, mood changes Reproductive system and breast disorders: Amenorrhea, breast tenderness, breast pain, breast enlargement, increased cervical mucous, change in menstrual flow, unscheduled bleeding Skin and subcutaneous tissue disorders: Acne, melasma Vascular disorders: Budd-Chiari syndrome, aggravation of varicose veins To report SUSPECTED ADVERSE EVENTS, contact Teva at 1-888-838-2872 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch for voluntary reporting of adverse reactions.
Drug Interactions
Concomitant Use with HCV Combination Therapy – Liver Enzyme Elevation Do not coadminister Aviane with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations (see WARNINGS , Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatmen t ). Changes in Contraceptive Effectiveness Associated with Coadministration of Other Products: Contraceptive effectiveness may be reduced when hormonal contraceptives are coadministered with antibiotics, anticonvulsants, and other drugs that increase the metabolism of contraceptive steroids. This could result in unintended pregnancy or breakthrough bleeding. Examples include rifampin, rifabutin, barbiturates, primidone, phenylbutazone, phenytoin, dexamethasone, carbamazepine, felbamate, oxcarbazepine, topiramate, griseofulvin, and modafinil. In such cases a back-up nonhormonal method of birth control should be considered. Several of the anti-HIV protease inhibitors have been studied with coadministration of oral combination hormonal contraceptives; significant changes (increase and decrease) in the plasma levels of the estrogen and progestin have been noted in some cases. The safety and efficacy of oral contraceptive products may be affected with coadministration of anti-HIV protease inhibitors. Healthcare providers should refer to the label of the individual anti-HIV protease inhibitors for further drug-drug interaction information. Herbal products containing St. John's Wort (Hypericum perforatum) may induce hepatic enzymes (cytochrome P 450) and p-glycoprotein transporter and may reduce the effectiveness of contraceptive steroids. This may also result in breakthrough bleeding. Increase in Plasma Levels Associated with Coadministered Drugs: Coadministration of atorvastatin and certain oral contraceptives containing ethinyl estradiol increases AUC values for ethinyl estradiol by approximately 20%. Ascorbic acid and acetaminophen increase the bioavailability of ethinyl estradiol since these drugs act as competitive inhibitors for sulfation of ethinyl estradiol in the gastrointestinal wall, a known pathway of elimination for ethinyl estradiol. CYP 3A4 inhibitors such as indinavir, itraconazole, ketoconazole, fluconazole, and troleandomycin may increase plasma hormone levels. Troleandomycin may also increase the risk of intrahepatic cholestasis during coadministration with combination oral contraceptives. Changes in Plasma Levels of Coadministered Drugs: Combination hormonal contraceptives containing some synthetic estrogens (e.g., ethinyl estradiol) may inhibit the metabolism of other compounds. Increased plasma concentrations of cyclosporin, prednisolone and other corticosteroids, and theophylline have been reported with concomitant administration of oral contraceptives. Decreased plasma concentrations of acetaminophen and increased clearance of temazepam, salicylic acid, morphine, and clofibric acid, due to induction of conjugation (particularly glucuronidation), have been noted when these drugs were administered with oral contraceptives. Concomitant use of Aviane may decrease lamotrigine exposure, which may reduce efficacy of lamotrigine. Adjust lamotrigine dosage as recommended in its Prescribing Information based on Aviane initiation or discontinuation. Concomitant use of Aviane may increase thyroid-binding globulin concentration. Monitor thyroid-stimulating hormone (TSH) level and follow the recommendation for the thyroid hormone in accordance with its Prescribing Information. The prescribing information of concomitant medications should be consulted to identify potential interactions.
How Supplied
Aviane ® (levonorgestrel and ethinyl estradiol tablets USP), 0.10 mg/0.02 mg is available as 28 tablets packaged in cartons (NDC: 0555-9045-58) of six blister card dispensers. Each blister card dispenser contains 21 orange active tablets and 7 light-green inert tablets. Each orange tablet is round, film coated and debossed with dp on one side and 016 on the other side. Each light-green tablet is round and debossed with dp on one side and 519 on the other side. Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Keep this and all medications out of the reach of children. Brands listed are the registered trademarks of their respective owners. Teva Pharmaceuticals USA, Inc. North Wales, PA 19454 Rev. F 7/2024
Medication Information
Recent Major Changes
WARNING: CIGARETTE SMOKING AND SERIOUS CARDIOVASCULAR EVENTS Cigarette smoking increases the risk of serious cardiovascular events from combined oral-contraceptives (COC) use. This risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked. For this reason, COCs, including Aviane (levonorgestrel and ethinyl estradiol tablets), are contraindicated in women who are over 35 years of age and smoke.
Indications and Usage
Aviane (levonorgestrel and ethinyl estradiol tablets) is indicated for use by females of reproductive potential to prevent pregnancy.
Limitations of use: The efficacy of Aviane (levonorgestrel and ethinyl estradiol tablets) in women with a body mass index (BMI) of > 35 kg/m2 has not been adequately evaluated.
In a clinical trial with Aviane (levonorgestrel and ethinyl estradiol tablets), 1,477 subjects had 7,720 cycles of use and a total of 5 pregnancies were reported. This represents an overall pregnancy rate of 0.84 per 100 woman-years. This rate includes patients who did not take the drug correctly. One or more pills were missed during 1,479 (18.8%) of the 7,870 cycles; thus all tablets were taken during 6,391 (81.2%) of the 7,870 cycles. Of the total 7,870 cycles, a total of 150 cycles were excluded from the calculation of the Pearl index due to the use of backup contraception and/or missing 3 or more consecutive pills.
The mean BMI of the study population was 24 kg/m2. Females with a BMI greater than 30 kg/m2 accounted for 12.1% (n=179) of the study population. Females with a BMI over 35 kg/m2 accounted for 4.3% (n=63) of the study population.
Dosage and Administration
To achieve maximum contraceptive effectiveness, Aviane (levonorgestrel and ethinyl estradiol tablets) must be taken exactly as directed and at intervals not exceeding 24 hours. The dosage of Aviane-28 is one orange tablet daily for 21 consecutive days, followed by one light-green inert tablet daily for 7 consecutive days, according to the prescribed schedule. It is recommended that Aviane-28 tablets be taken at the same time each day.
The dispenser should be kept in the wallet supplied to avoid possible fading of the pills. If the pills fade, patients should continue to take them as directed.
During the first cycle of use
The possibility of ovulation and conception prior to initiation of medication should be considered. The patient should be instructed to begin taking Aviane on either the first Sunday after the onset of menstruation (Sunday Start) or on Day 1 of menstruation (Day 1 Start).
Sunday start:
The patient is instructed to begin taking Aviane-28 on the first Sunday after the onset of menstruation. If menstruation begins on a Sunday, the first tablet (orange) is taken that day. One orange tablet should be taken daily for 21 consecutive days, followed by one light-green inert tablet daily for 7 consecutive days. Withdrawal bleeding should usually occur within 3 days following discontinuation of orange tablets and may not have finished before the next pack is started. During the first cycle, contraceptive reliance should not be placed on Aviane-28 until an orange tablet has been taken daily for 7 consecutive days, and a nonhormonal back-up method of birth control should be used during those 7 days.
Day 1 start:
During the first cycle of medication, the patient is instructed to begin taking Aviane-28 during the first 24 hours of her period (day one of her menstrual cycle). One orange tablet should be taken daily for 21 consecutive days, followed by one light-green inert tablet daily for 7 consecutive days. Withdrawal bleeding should usually occur within 3 days following discontinuation of orange tablets and may not have finished before the next pack is started. If medication is begun on day one of the menstrual cycle, no back-up contraception is necessary. If Aviane-28 tablets are started later than day one of the first menstrual cycle or postpartum, contraceptive reliance should not be placed on Aviane-28 tablets until after the first 7 consecutive days of administration, and a nonhormonal back-up method of birth control should be used during those 7 days.
After the first cycle of use
The patient begins her next and all subsequent courses of tablets on the day after taking her last light-green tablet. She should follow the same dosing schedule: 21 days on orange tablets followed by 7 days on light-green tablets. If in any cycle the patient starts tablets later than the proper day, she should protect herself against pregnancy by using a nonhormonal back-up method of birth control until she has taken an orange tablet daily for 7 consecutive days.
Switching from another hormonal method of contraception
When the patient is switching from a 21-day regimen of tablets, she should wait 7 days after her last tablet before she starts Aviane. She will probably experience withdrawal bleeding during that week. She should be sure that no more than 7 days pass after her previous 21-day regimen. When the patient is switching from a 28-day regimen of tablets, she should start her first pack of Aviane on the day after her last tablet. She should not wait any days between packs. The patient may switch any day from a progestin-only pill and should begin Aviane the next day. If switching from an implant or injection, the patient should start Aviane on the day of implant removal or, if using an injection, the day the next injection would be due. In switching from a progestin-only pill, injection, implant, or intrauterine system (IUS), the patient should be advised to use additional nonhormonal contraception (such as condoms) until active tablets have been taken for 7 consecutive days of the first pack.
If spotting or breakthrough bleeding occurs
If spotting or breakthrough bleeding occur, the patient is instructed to continue on the same regimen. This type of bleeding is usually transient and without significance; however, if the bleeding is persistent or prolonged, the patient is advised to consult her physician.
Risk of pregnancy if tablets are missed
While there is little likelihood of ovulation occurring if only one or two orange tablets are missed, the possibility of ovulation increases with each successive day that scheduled orange tablets are missed. Although the occurrence of pregnancy is unlikely if Aviane is taken according to directions, if withdrawal bleeding does not occur, the possibility of pregnancy must be considered. If the patient has not adhered to the prescribed schedule (missed one or more tablets or started taking them on a day later than she should have), the probability of pregnancy should be considered at the time of the first missed period and appropriate diagnostic measures taken. If the patient has adhered to the prescribed regimen and misses two consecutive periods, pregnancy should be ruled out.
The risk of pregnancy increases with each active (orange) tablet missed. For additional patient instructions regarding missed tablets, see the WHAT TO DO IF YOU MISS PILLS section in the DETAILED PATIENT LABELING below.
Use after pregnancy, abortion or miscarriage
Aviane may be initiated no earlier than day 28 postpartum in the nonlactating mother or after a second trimester abortion due to the increased risk for thromboembolism (see C ONTRAINDICATIONS, WARNINGS, and PRECAUTIONS concerning thromboembolic disease). The patient should be advised to use additional nonhormonal contraception (such as condoms) until active tablets have been taken for 7 consecutive days.
Aviane may be initiated immediately after a first trimester abortion or miscarriage. Instruct the patient to use additional nonhormonal contraception (such as condoms) until active tablets have been taken for 7 consecutive days, unless starting Aviane on the day of surgical abortion.
Contraindications
Combination oral contraceptives should not be used in women with any of the following conditions:
- Thrombophlebitis or thromboembolic disorders
- A history of deep-vein thrombophlebitis or thromboembolic disorders
- Cerebrovascular or coronary artery disease (current or past history)
- Valvular heart disease with thrombogenic complications
- Thrombogenic rhythm disorders
- Hereditary or acquired thrombophilias
- Prolonged immobilization (especially with major surgery)
- Diabetes with vascular involvement
- Headaches with focal neurological symptoms or migraine with aura
- Women with migraine who are 35 years or older
- Uncontrolled hypertension
- Known or suspected carcinoma of the breast or personal history of breast cancer
- Known or suspected estrogen- or progesterone sensitive malignancy
- Undiagnosed abnormal vaginal bleeding
- Cholestatic jaundice of pregnancy or jaundice with prior pill use
- Hepatic adenomas or carcinomas, or active liver disease
Women who are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for alanine aminotransferase (ALT) elevations (see WARNINGS, Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment ).
Adverse Reactions
An increased risk of the following serious adverse reactions (see WARNINGS section for additional information) has been associated with the use of oral contraceptives:
Thromboembolic and thrombotic disorders and other vascular problems (including thrombophlebitis and venous thrombosis with or without pulmonary embolism, mesenteric thrombosis, arterial thromboembolism, myocardial infarction, cerebral hemorrhage, cerebral thrombosis), carcinoma of the reproductive organs and breasts, hepatic neoplasia (including hepatic adenomas or benign liver tumors), ocular lesions (including retinal vascular thrombosis), gallbladder disease, carbohydrate and lipid effects, elevated blood pressure, and headache including migraine.
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 2).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 2). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
FIGURE 2: Relevant Studies of Risk of Breast Cancer with Combined Oral Contraceptives
RR = relative risk; OR = odds ratio; HR = hazard ratio. “ever COC” are females with current or past COC use; “never COC use” are females that never used COCs.
The following adverse reactions associated with the use of oral CHCs were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Common adverse reactions associated with oral CHCs are headache, abdominal pain, nausea, metrorrhagia, vaginal moniliasis and pain, acne, and vaginitis.
Additional adverse reactions that have been reported include the following:
Eye disorder: intolerance to contact lenses, steepening of corneal curvature
Gastrointestinal disorders: Abdominal bloating, vomiting
General disorders and administration site conditions: Edema, fluid retention
Hepatobiliary disorders: Cholestatic jaundice
Psychiatric disorders: Change in libido, mood changes
Reproductive system and breast disorders: Amenorrhea, breast tenderness, breast pain, breast enlargement, increased cervical mucous, change in menstrual flow, unscheduled bleeding
Skin and subcutaneous tissue disorders: Acne, melasma
Vascular disorders: Budd-Chiari syndrome, aggravation of varicose veins
To report SUSPECTED ADVERSE EVENTS, contact Teva at 1-888-838-2872 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch for voluntary reporting of adverse reactions.
Drug Interactions
Concomitant Use with HCV Combination Therapy – Liver Enzyme Elevation Do not coadminister Aviane with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations (see WARNINGS , Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatmen t ). Changes in Contraceptive Effectiveness Associated with Coadministration of Other Products: Contraceptive effectiveness may be reduced when hormonal contraceptives are coadministered with antibiotics, anticonvulsants, and other drugs that increase the metabolism of contraceptive steroids. This could result in unintended pregnancy or breakthrough bleeding. Examples include rifampin, rifabutin, barbiturates, primidone, phenylbutazone, phenytoin, dexamethasone, carbamazepine, felbamate, oxcarbazepine, topiramate, griseofulvin, and modafinil. In such cases a back-up nonhormonal method of birth control should be considered. Several of the anti-HIV protease inhibitors have been studied with coadministration of oral combination hormonal contraceptives; significant changes (increase and decrease) in the plasma levels of the estrogen and progestin have been noted in some cases. The safety and efficacy of oral contraceptive products may be affected with coadministration of anti-HIV protease inhibitors. Healthcare providers should refer to the label of the individual anti-HIV protease inhibitors for further drug-drug interaction information. Herbal products containing St. John's Wort (Hypericum perforatum) may induce hepatic enzymes (cytochrome P 450) and p-glycoprotein transporter and may reduce the effectiveness of contraceptive steroids. This may also result in breakthrough bleeding. Increase in Plasma Levels Associated with Coadministered Drugs: Coadministration of atorvastatin and certain oral contraceptives containing ethinyl estradiol increases AUC values for ethinyl estradiol by approximately 20%. Ascorbic acid and acetaminophen increase the bioavailability of ethinyl estradiol since these drugs act as competitive inhibitors for sulfation of ethinyl estradiol in the gastrointestinal wall, a known pathway of elimination for ethinyl estradiol. CYP 3A4 inhibitors such as indinavir, itraconazole, ketoconazole, fluconazole, and troleandomycin may increase plasma hormone levels. Troleandomycin may also increase the risk of intrahepatic cholestasis during coadministration with combination oral contraceptives. Changes in Plasma Levels of Coadministered Drugs: Combination hormonal contraceptives containing some synthetic estrogens (e.g., ethinyl estradiol) may inhibit the metabolism of other compounds. Increased plasma concentrations of cyclosporin, prednisolone and other corticosteroids, and theophylline have been reported with concomitant administration of oral contraceptives. Decreased plasma concentrations of acetaminophen and increased clearance of temazepam, salicylic acid, morphine, and clofibric acid, due to induction of conjugation (particularly glucuronidation), have been noted when these drugs were administered with oral contraceptives. Concomitant use of Aviane may decrease lamotrigine exposure, which may reduce efficacy of lamotrigine. Adjust lamotrigine dosage as recommended in its Prescribing Information based on Aviane initiation or discontinuation. Concomitant use of Aviane may increase thyroid-binding globulin concentration. Monitor thyroid-stimulating hormone (TSH) level and follow the recommendation for the thyroid hormone in accordance with its Prescribing Information. The prescribing information of concomitant medications should be consulted to identify potential interactions.
How Supplied
Aviane® (levonorgestrel and ethinyl estradiol tablets USP), 0.10 mg/0.02 mg is available as 28 tablets packaged in cartons (NDC: 0555-9045-58) of six blister card dispensers. Each blister card dispenser contains 21 orange active tablets and 7 light-green inert tablets. Each orange tablet is round, film coated and debossed with dp on one side and 016 on the other side. Each light-green tablet is round and debossed with dp on one side and 519 on the other side.
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Keep this and all medications out of the reach of children.
Brands listed are the registered trademarks of their respective owners.
Teva Pharmaceuticals USA, Inc.
North Wales, PA 19454
Rev. F 7/2024
Description
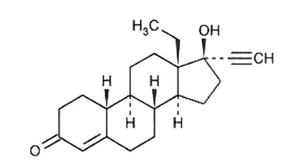
21 orange active tablets each containing 0.10 mg of levonorgestrel, USP (-)-13-Ethyl-17-hydroxy-18,19-dinor-17α-pregn-4-en-20-yn-3-one, a totally synthetic progestogen, and 0.02 mg of ethinyl estradiol, USP, (19-Nor-17α-pregna-1,3,5(10)-trien-20-yne-3,17-diol). The inactive ingredients present are: FD&C yellow no. 6 aluminum lake, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, pregelatinized corn starch, sodium starch glycolate and titanium dioxide.
7 light-green, inert tablets each containing: D&C yellow no. 10 aluminum lake, FD&C blue no. 1 aluminum lake, FD&C yellow no. 6 aluminum lake, lactose monohydrate, magnesium stearate, microcrystalline cellulose and pregelatinized corn starch.
Levonorgestrel, USP
C21H28O2 M.W. 312.45
Ethinyl Estradiol, USP
C20H24O2 M.W. 296.40
Race
Based on the pharmacokinetic study with Aviane, there are no apparent differences in pharmacokinetic parameters among women of different races.
Section 42229-5
Patients should be counseled that oral contraceptives do not protect against transmission of HIV (AIDS) and other sexually transmitted diseases (STDs) such as chlamydia, genital herpes, genital warts (human papillomavirus), gonorrhea, hepatitis B, and syphilis.
Aviane®
This product (like all oral contraceptives) is intended to prevent pregnancy. Oral contraceptives do not protect against transmission of HIV (AIDS) and other sexually transmitted diseases (STDs) such as chlamydia, genital herpes, genital warts, gonorrhea, hepatitis B, and syphilis.
Oral contraceptives, also known as “birth-control pills” or “the pill,” are taken to prevent pregnancy, and when taken correctly, have a failure rate of approximately 1.0% per year (1 pregnancy per 100 women per year of use) when used without missing any pills. The average failure rate of large numbers of pill users is approximately 5% per year (5 pregnancies per 100 women per year of use) when women who miss pills are included. For most women oral contraceptives are also free of serious or unpleasant side effects. However, forgetting to take pills considerably increases the chances of pregnancy.
For the majority of women, oral contraceptives can be taken safely. But there are some women who are at high risk of developing certain serious diseases that can be life-threatening or may cause temporary or permanent disability or death. The risks associated with taking oral contraceptives increase significantly if you:
- smoke.
- have high blood pressure, diabetes, high cholesterol, or a tendency to form blood clots.
- have or have had clotting disorders, heart attack, stroke, angina pectoris, cancer of the breast or sex organs, jaundice, malignant or benign liver tumors, or major surgery with prolonged immobilization.
- have headaches with neurological symptoms.
You should not take the pill if you take any Hepatitis C drug combination containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir. This may increase levels of the liver enzyme “alanine aminotransferase” (ALT) in the blood.
You should not take the pill if you suspect you are pregnant or have unexplained vaginal bleeding.
Although cardiovascular disease risks may be increased with oral-contraceptive use after age 40 in healthy, nonsmoking women, there are also greater potential health risks associated with pregnancy in older women.
Excretion
The elimination half-life for levonorgestrel is approximately 36 ± 13 hours at steady state. Levonorgestrel and its metabolites are primarily excreted in the urine (40% to 68%) and about 16% to 48% are excreted in feces. The elimination half-life of ethinyl estradiol is 18 ± 4.7 hours at steady state.
1. General
Patients should be counseled that oral contraceptives do not protect against transmission of HIV (AIDS) and other sexually transmitted diseases (STDs) such as chlamydia, genital herpes, genital warts, gonorrhea, hepatitis B, and syphilis.
Absorption
No specific investigation of the absolute bioavailability of Aviane in humans has been conducted. However, literature indicates that levonorgestrel is rapidly and completely absorbed after oral administration (bioavailability about 100%) and is not subject to first-pass metabolism. Ethinyl estradiol is rapidly and almost completely absorbed from the gastrointestinal tract but, due to first-pass metabolism in gut mucosa and liver, the bioavailability of ethinyl estradiol is between 38% and 48%.
After a single dose of Aviane to 22 women under fasting conditions, maximum serum concentrations of levonorgestrel are 2.8 ± 0.9 ng/mL (mean ± SD) at 1.6 ± 0.9 hours. At steady state, attained from day 19 onwards, maximum levonorgestrel concentrations of 6.0 ± 2.7 ng/mL are reached at 1.5 ± 0.5 hours after the daily dose. The minimum serum levels of levonorgestrel at steady state are 1.9 ± 1.0 ng/mL. Observed levonorgestrel concentrations increased from day 1 (single dose) to days 6 and 21 (multiple doses) by 34% and 96%, respectively (Figure 1). Unbound levonorgestrel concentrations increased from day 1 to days 6 and 21 by 25% and 83%, respectively. The kinetics of total levonorgestrel are non-linear due to an increase in binding of levonorgestrel to sex hormone binding globulin (SHBG), which is attributed to increased SHBG levels that are induced by the daily administration of ethinyl estradiol.
Following a single dose, maximum serum concentrations of ethinyl estradiol of 62 ± 21 pg/mL are reached at 1.5 ± 0.5 hours. At steady state, attained from at least day 6 onwards, maximum concentrations of ethinyl estradiol were 77 ± 30 pg/mL and were reached at 1.3 ± 0.7 hours after the daily dose. The minimum serum levels of ethinyl estradiol at steady state are 10.5 ± 5.1 pg/mL. Ethinyl estradiol concentrations did not increase from days 1 to 6, but did increase by 19% from days 1 to 21 (Figure 1).
FIGURE 1: Mean (SE) levonorgestrel and ethinyl estradiol serum concentrations in
22 subjects receiving 100 mcg levonorgestrel and 20 mcg ethinyl estradiol
TABLE 1 provides a summary of levonorgestrel and ethinyl estradiol pharmacokinetic parameters.
|
Levonorgestrel |
||||||
|
Cmax |
Tmax |
AUC |
CL/F |
Vλz/F |
SHBG |
|
|
Day |
ng/mL |
h |
ng•h/mL |
mL/h/kg |
L/kg |
nmol/L |
|
1 |
2.75 (0.88) |
1.6 (0.9) |
35.2 (12.8) |
53.7 (20.8) |
2.66 (1.09) |
57 (18) |
|
6 |
4.52 (1.79) |
1.5 (0.7) |
46.0 (18.8) |
40.8 (14.5) |
2.05 (0.86) |
81 (25) |
|
21 |
6.00 (2.65) |
1.5 (0.5) |
68.3 (32.5) |
28.4 (10.3) |
1.43 (0.62) |
93 (40) |
|
Unbound Levonorgestrel |
||||||
|
pg/mL |
h |
pg•h/mL |
L/h/kg |
L/kg |
fu% |
|
|
1 |
51.2 (12.9) |
1.6 (0.9) |
654 (201) |
2.79 (0.97) |
135.9 (41.8) |
1.92 (0.30) |
|
6 |
77.9 (22.0) |
1.5 (0.7) |
794 (240) |
2.24 (0.59) |
112.4 (40.5) |
1.80 (0.24) |
|
21 |
103.6 (36.9) |
1.5 (0.5) |
1177 (452) |
1.57 (0.49) |
78.6 (29.7) |
1.78 (0.19) |
|
Ethinyl Estradiol |
||||||
|
pg/mL |
h |
pg•h/mL |
mL/h/kg |
L/kg |
||
|
1 |
62.0 (20.5) |
1.5 (0.5) |
653 (227) |
567 (204) |
14.3 (3.7) |
|
|
6 |
76.7 (29.9) |
1.3 (0.7) |
604 (231) |
610 (196) |
15.5 (4.0) |
|
|
21 |
82.3 (33.2) |
1.4 (0.6) |
776 (308) |
486 (179) |
12.4 (4.1) |
Metabolism
Levonorgestrel:
The most important metabolic pathway occurs in the reduction of the Δ4-3-oxo group and hydroxylation at positions 2α, 1β, and 16β, followed by conjugation. Most of the metabolites that circulate in the blood are sulfates of 3α,5β-tetrahydro-levonorgestrel, while excretion occurs predominantly in the form of glucuronides. Some of the parent levonorgestrel also circulates as 17β-sulfate. Metabolic clearance rates may differ among individuals by several-fold, and this may account in part for the wide variation observed in levonorgestrel concentrations among users.
Ethinyl estradiol:
Cytochrome P450 enzymes (CYP3A4) in the liver are responsible for the 2-hydroxylation that is the major oxidative reaction. The 2-hydroxy metabolite is further transformed by methylation and glucuronidation prior to urinary and fecal excretion. Levels of Cytochrome P450 (CYP3A) vary widely among individuals and can explain the variation in rates of ethinyl estradiol 2-hydroxylation. Ethinyl estradiol is excreted in the urine and feces as glucuronide and sulfate conjugates, and undergoes enterohepatic circulation.
Overdosage
Symptoms of oral contraceptive overdosage in adults and children may include nausea, vomiting, and drowsiness/fatigue; withdrawal bleeding may occur in females. There is no specific antidote and further treatment of overdose, if necessary, is directed to the symptoms.
10. Headache
Aviane is contraindicated in females who have headaches with focal neurological symptoms or have migraine headaches with aura, and in women over age 35 years who have migraine head aches with or without aura.
The onset or exacerbation of migraine or development of headache with a new pattern that is recurrent, persistent, or severe requires discontinuation of oral contraceptives and evaluation of the cause. Consider discontinuation of Aviane if there is an increased frequency or severity of migraines during CHC use (which may be prodromal of a cerebrovascular event; see WARNINGS and CONTRAINDICATIONS).
13. Chloasma
Chloasma may occur, especially in women with a history of chloasma gravidarum. Advise women with a history of chloasma to avoid exposure to the sun or ultraviolet radiation while taking Aviane.
Distribution
Levonorgestrel in serum is primarily bound to SHBG. Ethinyl estradiol is about 97% bound to plasma albumin. Ethinyl estradiol does not bind to SHBG, but induces SHBG synthesis.
12. Pregnancy
Risk Summary
There is no use for contraception in pregnancy; therefore, Aviane should be discontinued during pregnancy. Epidemiologic studies and meta-analyses have not found an increased risk of genital or nongenital birth defects (including cardiac anomalies and limb-reduction defects) following exposure to COCs before conception or during early pregnancy.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4 percent and 15 to 20 percent,
respectively.
Data
Human Data
Epidemiologic studies and meta-analyses have not found an increased risk of genital or nongenital birth defects (including cardiac anomalies and limb-reduction defects) following exposure to COCs before conception or during early pregnancy.
13. Lactation
Contraceptive hormones and metabolites are present in human milk. CHCs can reduce milk production in breastfeeding females. This reduction can occur at any time but is less likely to occur once breastfeeding is well-established. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Aviane and any potential adverse effects on the breastfed child from Aviane or from the underlying maternal condition.
6. depression
Monitor females with a history of depression and discontinue Aviane if depression recurs to a serious degree. Data on the association of CHCs with onset of depression or exacerbation of existing depression are limited.
Mode of Action
Combination oral contraceptives prevent pregnancy primarily by suppressing ovulation.
14. Pediatric Use
Safety and efficacy of Aviane tablets have been established in women of reproductive age. Safety and efficacy are expected to be the same for postpubertal adolescents under the age of 16 and for users 16 years and older. Use of Aviane before menarche is not indicated.
15. Geriatric Use
Aviane has not been studied in women over 65 years of age and is not indicated in this population.
4. Liver Function
If jaundice develops in any woman receiving such drugs, the medication should be discontinued. Steroid hormones may be poorly metabolized in patients with impaired liver function.
5. Ocular Lesions
There have been clinical case reports of retinal thrombosis associated with the use of oral contraceptives that may lead to partial or complete loss of vision. Oral contraceptives should be discontinued if there is unexplained partial or complete loss of vision; onset of proptosis or diplopia; papilledema; or retinal vascular lesions. Appropriate diagnostic and therapeutic measures should be undertaken immediately.
7. Contact Lenses
Contact-lens wearers who develop visual changes or changes in lens tolerance should be assessed by an ophthalmologist.
11. Carcinogenesis
See WARNINGS section.
3. Lipid Disorders
Women who are being treated for hyperlipidemias should be followed closely if they elect to use oral contraceptives. Some progestogens may elevate LDL levels and may render the control of hyperlipidemias more difficult (see WARNINGS ).
A small proportion of women will have adverse lipid changes while taking oral contraceptives. Nonhormonal contraception should be considered in women with uncontrolled dyslipidemias. Persistent hypertriglyceridemia may occur in a small population of combination oral contraceptive users. Elevations of plasma triglycerides may lead to pancreatitis and other complications.
5. Fluid Retention
Oral contraceptives may cause some degree of fluid retention. They should be prescribed with caution, and only with careful monitoring, in patients with conditions which might be aggravated by fluid retention.
16. Body Mass Index
The safety and effectiveness of Aviane in females with a BMI > 35 kg/m2 have not been adequately evaluated.
8. Gastrointestinal
Diarrhea and/or vomiting may reduce hormone absorption resulting in decreased serum concentrations. If a patient vomits or has diarrhea after taking an active tablet, instruct the patient to not take an additional active tablet on that day and to continue the regimen the next day as prescribed. In case of vomiting or diarrhea that continues for 48 hours or greater, instruct the patient to contact the health care provider and use back-up or alternative contraception until active tablets have been taken for 7 consecutive days after vomiting and diarrhea have resolved.
Renal Insufficiency
No formal studies have evaluated the effect of renal disease on the disposition of Aviane.
3. Hepatic Neoplasia
Benign hepatic adenomas are associated with oral-contraceptive use, although the incidence of these benign tumors is rare in the United States. Indirect calculations have estimated the attributable risk to be in the range of 3.3 cases/100,000 for users, a risk that increases after four or more years of use. Rupture of rare, benign, hepatic adenomas may cause death through intra-abdominal hemorrhage.
Studies from Britain have shown an increased risk of developing hepatocellular carcinoma in long-term (>8 years) oral-contraceptive users. However, these cancers are extremely rare in the U.S. and the attributable risk (the excess incidence) of liver cancers in oral-contraceptive users approaches less than one per million users.
9. Drug Interactions
Concomitant Use with HCV Combination Therapy – Liver Enzyme Elevation
Do not coadminister Aviane with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations (see WARNINGS , Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatmen t).
Changes in Contraceptive Effectiveness Associated with Coadministration of Other Products:
Contraceptive effectiveness may be reduced when hormonal contraceptives are coadministered with antibiotics, anticonvulsants, and other drugs that increase the metabolism of contraceptive steroids. This could result in unintended pregnancy or breakthrough bleeding. Examples include rifampin, rifabutin, barbiturates, primidone, phenylbutazone, phenytoin, dexamethasone, carbamazepine, felbamate, oxcarbazepine, topiramate, griseofulvin, and modafinil. In such cases a back-up nonhormonal method of birth control should be considered.
Several of the anti-HIV protease inhibitors have been studied with coadministration of oral combination hormonal contraceptives; significant changes (increase and decrease) in the plasma levels of the estrogen and progestin have been noted in some cases. The safety and efficacy of oral contraceptive products may be affected with coadministration of anti-HIV protease inhibitors. Healthcare providers should refer to the label of the individual anti-HIV protease inhibitors for further drug-drug interaction information.
Herbal products containing St. John's Wort (Hypericum perforatum) may induce hepatic enzymes (cytochrome P 450) and p-glycoprotein transporter and may reduce the effectiveness of contraceptive steroids. This may also result in breakthrough bleeding.
Increase in Plasma Levels Associated with Coadministered Drugs:
Coadministration of atorvastatin and certain oral contraceptives containing ethinyl estradiol increases AUC values for ethinyl estradiol by approximately 20%. Ascorbic acid and acetaminophen increase the bioavailability of ethinyl estradiol since these drugs act as competitive inhibitors for sulfation of ethinyl estradiol in the gastrointestinal wall, a known pathway of elimination for ethinyl estradiol. CYP 3A4 inhibitors such as indinavir, itraconazole, ketoconazole, fluconazole, and troleandomycin may increase plasma hormone levels. Troleandomycin may also increase the risk of intrahepatic cholestasis during coadministration with combination oral contraceptives.
Changes in Plasma Levels of Coadministered Drugs:
Combination hormonal contraceptives containing some synthetic estrogens (e.g., ethinyl estradiol) may inhibit the metabolism of other compounds. Increased plasma concentrations of cyclosporin, prednisolone and other corticosteroids, and theophylline have been reported with concomitant administration of oral contraceptives. Decreased plasma concentrations of acetaminophen and increased clearance of temazepam, salicylic acid, morphine, and clofibric acid, due to induction of conjugation (particularly glucuronidation), have been noted when these drugs were administered with oral contraceptives.
Concomitant use of Aviane may decrease lamotrigine exposure, which may reduce efficacy of lamotrigine. Adjust lamotrigine dosage as recommended in its Prescribing Information based on Aviane initiation or discontinuation.
Concomitant use of Aviane may increase thyroid-binding globulin concentration. Monitor thyroid-stimulating hormone (TSH) level and follow the recommendation for the thyroid hormone in accordance with its Prescribing Information.
The prescribing information of concomitant medications should be consulted to identify potential interactions.
Hepatic Insufficiency
No formal studies have evaluated the effect of hepatic disease on the disposition of Aviane. However, steroid hormones may be poorly metabolized in patients with impaired liver function.
2. Malignant Neoplasms
Breast Cancer
Aviane is contraindicated in females who currently have or have had breast cancer because breast cancer may be hormonally sensitive (see
CONTRAINDICATIONS
). Epidemiology studies have not found a consistent association between use of combined oral contraceptives (COCs) and breast cancer risk. Studies do not show an association between ever (current or past) use of COCs and risk of breast cancer. However, some studies report a small increase in the risk of breast cancer among current or recent users (<6 months since last use) and current users with longer duration of COC use (see
ADVERSE REACTIONS
).
Cervical Cancer
Some studies suggest that oral contraceptive use has been associated with an increase in the risk of cervical intraepithelial neoplasia or invasive cervical cancer in some populations of women. However, there continues to be controversy about the extent to which such findings may be due to differences in sexual behavior and other factors.
7. Gallbladder Disease
Combination oral contraceptives may worsen existing gallbladder disease and may accelerate the development of this disease in previously asymptomatic women. Earlier studies have reported an increased lifetime relative risk of gallbladder surgery in users of oral contraceptives and estrogens. More recent studies, however, have shown that the relative risk of developing gallbladder disease among oral-contraceptive users may be minimal. The recent findings of minimal risk may be related to the use of oral-contraceptive formulations containing lower hormonal doses of estrogens and progestogens. A past history of COC-related cholestasis predicts an increased risk with subsequent COC use. Women with a history of pregnancy-related cholestasis may be at an increased risk for COC related cholestasis.
Drug Drug Interactions
See PRECAUTIONS - Drug Interactions
12. Hereditary Angioedema
In women with hereditary angioedema, exogenous estrogens may induce or exacerbate symptoms of angioedema.
9. Elevated Blood Pressure
Aviane is contraindicated in women with uncontrolled hypertension or hypertension with vascular disease (see CONTRAINDICATIONS ). For all females, including those with well-controlled hypertension, monitor blood pressure at routine visits and stop Aviane if blood pressure rises significantly.
An increase in blood pressure has been reported in females using CHCs, and this increase is more likely in older women with extended duration of use. The effect of CHCs on blood pressure may vary according to the progestin in the CHC.
11. Bleeding Irregularities
Unscheduled bleeding and spotting are sometimes encountered in patients on oral contraceptives, especially during the first three months of use. The type and dose of progestogen may be important. If bleeding persists or recurs, nonhormonal causes should be considered and adequate diagnostic measures taken to rule out malignancy or pregnancy in the event of unscheduled bleeding, as in the case of any abnormal vaginal bleeding. If pathology has been excluded, time or a change to another formulation may solve the problem. In the event of amenorrhea, pregnancy should be ruled out.
Some women may encounter post-pill amenorrhea or oligomenorrhea (possibly with anovulation), especially when such a condition was preexistent.
In the clinical trial with levonorgestrel 0.1 mg and ethinyl estradiol 0.02 mg tablets, unscheduled bleeding was defined as bleeding or spotting that occurred:
- During cycle 1 on pill-pack Days 4 to 21, inclusive of a 28-day cycle.
- In subsequent cycles, on Days 5 to 21 inclusive or on pill-pack Days 1 to 4 inclusive if preceded by 2 consecutive days without bleeding or spotting.
Based on subject diaries, the proportion of subjects reporting unscheduled bleeding or spotting per 28-day cycle decreased over time: 30.5% at Cycle 1 versus 18.2% at Cycle 12.
14. Effect On Binding Globulins
The estrogen component of COCs may raise the serum concentrations of thyroxine-binding globulin, sex hormone-binding globulin, and cortisol-binding globulin. Monitor thyroid-stimulating hormone (TSH) levels for females receiving Aviane and thyroid hormone replacement therapy concomitantly. Follow the recommendation for the thyroid hormone in accordance with its Prescribing Information.
17. Information for the Patient
See Patient Labeling printed below.
2. Physical Examination and Follow Up
A periodic personal and family medical history and complete physical examination are appropriate for all women, including women using oral contraceptives. The physical examination, however, may be deferred until after initiation of oral contraceptives if requested by the woman and judged appropriate by the clinician. The physical examination should include special reference to blood pressure, breasts, abdomen, pelvic organs, and relevant laboratory tests. In case of undiagnosed, persistent, or recurrent abnormal vaginal bleeding, appropriate diagnostic measures should be conducted to rule out malignancy. Women with a strong family history of breast cancer or who have breast nodules should be monitored with particular care.
10. Interactions With Laboratory Tests
Certain endocrine- and liver-function tests and blood components may be affected by oral contraceptives:
a. Increased prothrombin and factors VII, VIII, IX, and X; decreased antithrombin 3; increased norepinephrine-induced platelet aggregability.
b. Increased thyroid-binding globulin (TBG) leading to increased circulating total thyroid hormone, as measured by protein-bound iodine (PBI), T4 by column or by radioimmunoassay.
c. Other binding proteins may be elevated in serum i.e., corticosteroid binding globulin (CBG), sex hormone-binding globulins (SHBG) leading to increased levels of total circulating corticosteroids and sex steroids respectively.
d. Triglycerides may be increased and levels of various other lipids and lipoproteins may be affected.
e. Glucose tolerance may be decreased.
f. Serum folate levels may be depressed by oral-contraceptive therapy. This may be of clinical significance if a woman becomes pregnant shortly after discontinuing oral contraceptives.
Package/label Display Panel, Part 1 of 2
NDC 0555-9045-58
6 Blister Cards, 28 Tablets Each
Aviane®
(levonorgestrel and ethinyl
estradiol tablets USP)
0.10 mg/0.02 mg
Contains 6 blister cards, each containing 28 tablets. Twenty-one
orange tablets, each containing 0.10 mg levonorgestrel, USP with
0.02 mg ethinyl estradiol, USP and seven light-green inert tablets.
Rx only
SHAPING
WOMEN’S HEALTH®
TEVA
Package/label Display Panel, Part 2 of 2
8. Carbohydrate and Lipid Metabolic Effects
Oral contraceptives have been shown to cause glucose intolerance in a significant percentage of users. Carefully monitor females with prediabetes and diabetes who are using Aviane. Aviane may decrease glucose tolerance.
A small proportion of women will have persistent hypertriglyceridemia while on COCs. As discussed earlier (see WARNINGS and PRECAUTIONS ), changes in serum triglycerides and lipoprotein levels have been reported in oral-contraceptive users.
1. Thromboembolic Disorders and Other Vascular Problems
Cardiovascular and Cerebrovascular Events
Use of CHCs increases the risk of cardiovascular events and cerebrovascular events, such as myocardial infarction and stroke. The risk of these events with CHC use is greater in females with concomitant risk factors: age 35 years and older, smoking, hypertension, dyslipidemia, diabetes, or obesity. The risk increases with increasing age and with increasing number of cigarettes smoked.
Venous Thromboembolism
Use of CHCs also increases the risk of VTE, such as deep vein thrombosis and pulmonary embolism. The rate of VTE in females using COCs has been estimated to be 3 to 9 cases per 10,000 woman-years. The VTE risk should be considered in the context of relevant subpopulations of females of reproductive potential who are not taking CHCs ( ADVERSE REACTIONS ).
Risk factors for VTE with CHC use include smoking, obesity, family history of VTE, and prolonged immobilization, in addition to other factors that contraindicate use of CHCs (see CONTRAINDICATIONS ). The presence of multiple risk factors for VTE with CHC use may further increase the risk. The risk of VTE is highest during the first year of CHC use and when restarting hormonal contraception after a break of four weeks or longer. The risk of VTE returns to baseline approximately 3 months after CHC use is discontinued.
Postpartum Venous Thromboembolism
The risk of VTE is increased during the first six weeks postpartum. The risk is highest up to four weeks postpartum but remains higher than baseline until at least six weeks postpartum. The presence of multiple risk factors for VTE may further increase the risk. Obstetric complications may extend the elevated risk up to 12 weeks postpartum.
6. Oral Contraceptive Use Before Or During Early Pregnancy
Extensive epidemiological studies have revealed no increased risk of birth defects in infants born to women who have used oral contraceptives prior to pregnancy. Studies also do not suggest a teratogenic effect, particularly insofar as cardiac anomalies and limb-reduction defects are concerned, when taken inadvertently during early pregnancy (see CONTRAINDICATIONS).
It is recommended that for any patient who has missed two consecutive periods, pregnancy should be ruled out. If the patient has not adhered to the prescribed schedule, the possibility of pregnancy should be considered at the time of the first missed period. Oral-contraceptive use should be discontinued if pregnancy is confirmed.
4. Risk of Liver Enzyme Elevations With Concomitant Hepatitis C Treatment
During clinical trials with the Hepatitis C combination drug regimen that contains ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN), including some cases greater than 20 times the ULN, were significantly more frequent in women using ethinyl estradiol-containing medications such as COCs. Discontinue Aviane prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir, with or without dasabuvir (see CONTRAINDICATIONS). Aviane can be restarted approximately 2 weeks following completion of treatment with the combination drug regimen.
Structured Label Content
Race
Based on the pharmacokinetic study with Aviane, there are no apparent differences in pharmacokinetic parameters among women of different races.
Recent Major Changes (34066-1)
WARNING: CIGARETTE SMOKING AND SERIOUS CARDIOVASCULAR EVENTS Cigarette smoking increases the risk of serious cardiovascular events from combined oral-contraceptives (COC) use. This risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked. For this reason, COCs, including Aviane (levonorgestrel and ethinyl estradiol tablets), are contraindicated in women who are over 35 years of age and smoke.
Section 42229-5 (42229-5)
Patients should be counseled that oral contraceptives do not protect against transmission of HIV (AIDS) and other sexually transmitted diseases (STDs) such as chlamydia, genital herpes, genital warts (human papillomavirus), gonorrhea, hepatitis B, and syphilis.
Aviane®
This product (like all oral contraceptives) is intended to prevent pregnancy. Oral contraceptives do not protect against transmission of HIV (AIDS) and other sexually transmitted diseases (STDs) such as chlamydia, genital herpes, genital warts, gonorrhea, hepatitis B, and syphilis.
Oral contraceptives, also known as “birth-control pills” or “the pill,” are taken to prevent pregnancy, and when taken correctly, have a failure rate of approximately 1.0% per year (1 pregnancy per 100 women per year of use) when used without missing any pills. The average failure rate of large numbers of pill users is approximately 5% per year (5 pregnancies per 100 women per year of use) when women who miss pills are included. For most women oral contraceptives are also free of serious or unpleasant side effects. However, forgetting to take pills considerably increases the chances of pregnancy.
For the majority of women, oral contraceptives can be taken safely. But there are some women who are at high risk of developing certain serious diseases that can be life-threatening or may cause temporary or permanent disability or death. The risks associated with taking oral contraceptives increase significantly if you:
- smoke.
- have high blood pressure, diabetes, high cholesterol, or a tendency to form blood clots.
- have or have had clotting disorders, heart attack, stroke, angina pectoris, cancer of the breast or sex organs, jaundice, malignant or benign liver tumors, or major surgery with prolonged immobilization.
- have headaches with neurological symptoms.
You should not take the pill if you take any Hepatitis C drug combination containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir. This may increase levels of the liver enzyme “alanine aminotransferase” (ALT) in the blood.
You should not take the pill if you suspect you are pregnant or have unexplained vaginal bleeding.
Although cardiovascular disease risks may be increased with oral-contraceptive use after age 40 in healthy, nonsmoking women, there are also greater potential health risks associated with pregnancy in older women.
Excretion
The elimination half-life for levonorgestrel is approximately 36 ± 13 hours at steady state. Levonorgestrel and its metabolites are primarily excreted in the urine (40% to 68%) and about 16% to 48% are excreted in feces. The elimination half-life of ethinyl estradiol is 18 ± 4.7 hours at steady state.
1. General
Patients should be counseled that oral contraceptives do not protect against transmission of HIV (AIDS) and other sexually transmitted diseases (STDs) such as chlamydia, genital herpes, genital warts, gonorrhea, hepatitis B, and syphilis.
Absorption
No specific investigation of the absolute bioavailability of Aviane in humans has been conducted. However, literature indicates that levonorgestrel is rapidly and completely absorbed after oral administration (bioavailability about 100%) and is not subject to first-pass metabolism. Ethinyl estradiol is rapidly and almost completely absorbed from the gastrointestinal tract but, due to first-pass metabolism in gut mucosa and liver, the bioavailability of ethinyl estradiol is between 38% and 48%.
After a single dose of Aviane to 22 women under fasting conditions, maximum serum concentrations of levonorgestrel are 2.8 ± 0.9 ng/mL (mean ± SD) at 1.6 ± 0.9 hours. At steady state, attained from day 19 onwards, maximum levonorgestrel concentrations of 6.0 ± 2.7 ng/mL are reached at 1.5 ± 0.5 hours after the daily dose. The minimum serum levels of levonorgestrel at steady state are 1.9 ± 1.0 ng/mL. Observed levonorgestrel concentrations increased from day 1 (single dose) to days 6 and 21 (multiple doses) by 34% and 96%, respectively (Figure 1). Unbound levonorgestrel concentrations increased from day 1 to days 6 and 21 by 25% and 83%, respectively. The kinetics of total levonorgestrel are non-linear due to an increase in binding of levonorgestrel to sex hormone binding globulin (SHBG), which is attributed to increased SHBG levels that are induced by the daily administration of ethinyl estradiol.
Following a single dose, maximum serum concentrations of ethinyl estradiol of 62 ± 21 pg/mL are reached at 1.5 ± 0.5 hours. At steady state, attained from at least day 6 onwards, maximum concentrations of ethinyl estradiol were 77 ± 30 pg/mL and were reached at 1.3 ± 0.7 hours after the daily dose. The minimum serum levels of ethinyl estradiol at steady state are 10.5 ± 5.1 pg/mL. Ethinyl estradiol concentrations did not increase from days 1 to 6, but did increase by 19% from days 1 to 21 (Figure 1).
FIGURE 1: Mean (SE) levonorgestrel and ethinyl estradiol serum concentrations in
22 subjects receiving 100 mcg levonorgestrel and 20 mcg ethinyl estradiol
TABLE 1 provides a summary of levonorgestrel and ethinyl estradiol pharmacokinetic parameters.
|
Levonorgestrel |
||||||
|
Cmax |
Tmax |
AUC |
CL/F |
Vλz/F |
SHBG |
|
|
Day |
ng/mL |
h |
ng•h/mL |
mL/h/kg |
L/kg |
nmol/L |
|
1 |
2.75 (0.88) |
1.6 (0.9) |
35.2 (12.8) |
53.7 (20.8) |
2.66 (1.09) |
57 (18) |
|
6 |
4.52 (1.79) |
1.5 (0.7) |
46.0 (18.8) |
40.8 (14.5) |
2.05 (0.86) |
81 (25) |
|
21 |
6.00 (2.65) |
1.5 (0.5) |
68.3 (32.5) |
28.4 (10.3) |
1.43 (0.62) |
93 (40) |
|
Unbound Levonorgestrel |
||||||
|
pg/mL |
h |
pg•h/mL |
L/h/kg |
L/kg |
fu% |
|
|
1 |
51.2 (12.9) |
1.6 (0.9) |
654 (201) |
2.79 (0.97) |
135.9 (41.8) |
1.92 (0.30) |
|
6 |
77.9 (22.0) |
1.5 (0.7) |
794 (240) |
2.24 (0.59) |
112.4 (40.5) |
1.80 (0.24) |
|
21 |
103.6 (36.9) |
1.5 (0.5) |
1177 (452) |
1.57 (0.49) |
78.6 (29.7) |
1.78 (0.19) |
|
Ethinyl Estradiol |
||||||
|
pg/mL |
h |
pg•h/mL |
mL/h/kg |
L/kg |
||
|
1 |
62.0 (20.5) |
1.5 (0.5) |
653 (227) |
567 (204) |
14.3 (3.7) |
|
|
6 |
76.7 (29.9) |
1.3 (0.7) |
604 (231) |
610 (196) |
15.5 (4.0) |
|
|
21 |
82.3 (33.2) |
1.4 (0.6) |
776 (308) |
486 (179) |
12.4 (4.1) |
Metabolism
Levonorgestrel:
The most important metabolic pathway occurs in the reduction of the Δ4-3-oxo group and hydroxylation at positions 2α, 1β, and 16β, followed by conjugation. Most of the metabolites that circulate in the blood are sulfates of 3α,5β-tetrahydro-levonorgestrel, while excretion occurs predominantly in the form of glucuronides. Some of the parent levonorgestrel also circulates as 17β-sulfate. Metabolic clearance rates may differ among individuals by several-fold, and this may account in part for the wide variation observed in levonorgestrel concentrations among users.
Ethinyl estradiol:
Cytochrome P450 enzymes (CYP3A4) in the liver are responsible for the 2-hydroxylation that is the major oxidative reaction. The 2-hydroxy metabolite is further transformed by methylation and glucuronidation prior to urinary and fecal excretion. Levels of Cytochrome P450 (CYP3A) vary widely among individuals and can explain the variation in rates of ethinyl estradiol 2-hydroxylation. Ethinyl estradiol is excreted in the urine and feces as glucuronide and sulfate conjugates, and undergoes enterohepatic circulation.
Overdosage (OVERDOSAGE)
Symptoms of oral contraceptive overdosage in adults and children may include nausea, vomiting, and drowsiness/fatigue; withdrawal bleeding may occur in females. There is no specific antidote and further treatment of overdose, if necessary, is directed to the symptoms.
Description (DESCRIPTION)
21 orange active tablets each containing 0.10 mg of levonorgestrel, USP (-)-13-Ethyl-17-hydroxy-18,19-dinor-17α-pregn-4-en-20-yn-3-one, a totally synthetic progestogen, and 0.02 mg of ethinyl estradiol, USP, (19-Nor-17α-pregna-1,3,5(10)-trien-20-yne-3,17-diol). The inactive ingredients present are: FD&C yellow no. 6 aluminum lake, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, pregelatinized corn starch, sodium starch glycolate and titanium dioxide.
7 light-green, inert tablets each containing: D&C yellow no. 10 aluminum lake, FD&C blue no. 1 aluminum lake, FD&C yellow no. 6 aluminum lake, lactose monohydrate, magnesium stearate, microcrystalline cellulose and pregelatinized corn starch.
Levonorgestrel, USP
C21H28O2 M.W. 312.45
Ethinyl Estradiol, USP
C20H24O2 M.W. 296.40
10. Headache
Aviane is contraindicated in females who have headaches with focal neurological symptoms or have migraine headaches with aura, and in women over age 35 years who have migraine head aches with or without aura.
The onset or exacerbation of migraine or development of headache with a new pattern that is recurrent, persistent, or severe requires discontinuation of oral contraceptives and evaluation of the cause. Consider discontinuation of Aviane if there is an increased frequency or severity of migraines during CHC use (which may be prodromal of a cerebrovascular event; see WARNINGS and CONTRAINDICATIONS).
13. Chloasma
Chloasma may occur, especially in women with a history of chloasma gravidarum. Advise women with a history of chloasma to avoid exposure to the sun or ultraviolet radiation while taking Aviane.
Distribution
Levonorgestrel in serum is primarily bound to SHBG. Ethinyl estradiol is about 97% bound to plasma albumin. Ethinyl estradiol does not bind to SHBG, but induces SHBG synthesis.
How Supplied (HOW SUPPLIED)
Aviane® (levonorgestrel and ethinyl estradiol tablets USP), 0.10 mg/0.02 mg is available as 28 tablets packaged in cartons (NDC: 0555-9045-58) of six blister card dispensers. Each blister card dispenser contains 21 orange active tablets and 7 light-green inert tablets. Each orange tablet is round, film coated and debossed with dp on one side and 016 on the other side. Each light-green tablet is round and debossed with dp on one side and 519 on the other side.
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Keep this and all medications out of the reach of children.
Brands listed are the registered trademarks of their respective owners.
Teva Pharmaceuticals USA, Inc.
North Wales, PA 19454
Rev. F 7/2024
12. Pregnancy
Risk Summary
There is no use for contraception in pregnancy; therefore, Aviane should be discontinued during pregnancy. Epidemiologic studies and meta-analyses have not found an increased risk of genital or nongenital birth defects (including cardiac anomalies and limb-reduction defects) following exposure to COCs before conception or during early pregnancy.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4 percent and 15 to 20 percent,
respectively.
Data
Human Data
Epidemiologic studies and meta-analyses have not found an increased risk of genital or nongenital birth defects (including cardiac anomalies and limb-reduction defects) following exposure to COCs before conception or during early pregnancy.
13. Lactation
Contraceptive hormones and metabolites are present in human milk. CHCs can reduce milk production in breastfeeding females. This reduction can occur at any time but is less likely to occur once breastfeeding is well-established. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Aviane and any potential adverse effects on the breastfed child from Aviane or from the underlying maternal condition.
6. depression (6. Depression)
Monitor females with a history of depression and discontinue Aviane if depression recurs to a serious degree. Data on the association of CHCs with onset of depression or exacerbation of existing depression are limited.
Mode of Action
Combination oral contraceptives prevent pregnancy primarily by suppressing ovulation.
14. Pediatric Use
Safety and efficacy of Aviane tablets have been established in women of reproductive age. Safety and efficacy are expected to be the same for postpubertal adolescents under the age of 16 and for users 16 years and older. Use of Aviane before menarche is not indicated.
15. Geriatric Use
Aviane has not been studied in women over 65 years of age and is not indicated in this population.
4. Liver Function
If jaundice develops in any woman receiving such drugs, the medication should be discontinued. Steroid hormones may be poorly metabolized in patients with impaired liver function.
5. Ocular Lesions
There have been clinical case reports of retinal thrombosis associated with the use of oral contraceptives that may lead to partial or complete loss of vision. Oral contraceptives should be discontinued if there is unexplained partial or complete loss of vision; onset of proptosis or diplopia; papilledema; or retinal vascular lesions. Appropriate diagnostic and therapeutic measures should be undertaken immediately.
7. Contact Lenses
Contact-lens wearers who develop visual changes or changes in lens tolerance should be assessed by an ophthalmologist.
Adverse Reactions (ADVERSE REACTIONS)
An increased risk of the following serious adverse reactions (see WARNINGS section for additional information) has been associated with the use of oral contraceptives:
Thromboembolic and thrombotic disorders and other vascular problems (including thrombophlebitis and venous thrombosis with or without pulmonary embolism, mesenteric thrombosis, arterial thromboembolism, myocardial infarction, cerebral hemorrhage, cerebral thrombosis), carcinoma of the reproductive organs and breasts, hepatic neoplasia (including hepatic adenomas or benign liver tumors), ocular lesions (including retinal vascular thrombosis), gallbladder disease, carbohydrate and lipid effects, elevated blood pressure, and headache including migraine.
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 2).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 2). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
FIGURE 2: Relevant Studies of Risk of Breast Cancer with Combined Oral Contraceptives
RR = relative risk; OR = odds ratio; HR = hazard ratio. “ever COC” are females with current or past COC use; “never COC use” are females that never used COCs.
The following adverse reactions associated with the use of oral CHCs were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Common adverse reactions associated with oral CHCs are headache, abdominal pain, nausea, metrorrhagia, vaginal moniliasis and pain, acne, and vaginitis.
Additional adverse reactions that have been reported include the following:
Eye disorder: intolerance to contact lenses, steepening of corneal curvature
Gastrointestinal disorders: Abdominal bloating, vomiting
General disorders and administration site conditions: Edema, fluid retention
Hepatobiliary disorders: Cholestatic jaundice
Psychiatric disorders: Change in libido, mood changes
Reproductive system and breast disorders: Amenorrhea, breast tenderness, breast pain, breast enlargement, increased cervical mucous, change in menstrual flow, unscheduled bleeding
Skin and subcutaneous tissue disorders: Acne, melasma
Vascular disorders: Budd-Chiari syndrome, aggravation of varicose veins
To report SUSPECTED ADVERSE EVENTS, contact Teva at 1-888-838-2872 or FDA at 1-800-FDA-1088 or http://www.fda.gov/medwatch for voluntary reporting of adverse reactions.
Contraindications (CONTRAINDICATIONS)
Combination oral contraceptives should not be used in women with any of the following conditions:
- Thrombophlebitis or thromboembolic disorders
- A history of deep-vein thrombophlebitis or thromboembolic disorders
- Cerebrovascular or coronary artery disease (current or past history)
- Valvular heart disease with thrombogenic complications
- Thrombogenic rhythm disorders
- Hereditary or acquired thrombophilias
- Prolonged immobilization (especially with major surgery)
- Diabetes with vascular involvement
- Headaches with focal neurological symptoms or migraine with aura
- Women with migraine who are 35 years or older
- Uncontrolled hypertension
- Known or suspected carcinoma of the breast or personal history of breast cancer
- Known or suspected estrogen- or progesterone sensitive malignancy
- Undiagnosed abnormal vaginal bleeding
- Cholestatic jaundice of pregnancy or jaundice with prior pill use
- Hepatic adenomas or carcinomas, or active liver disease
Women who are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for alanine aminotransferase (ALT) elevations (see WARNINGS, Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment ).
11. Carcinogenesis
See WARNINGS section.
3. Lipid Disorders
Women who are being treated for hyperlipidemias should be followed closely if they elect to use oral contraceptives. Some progestogens may elevate LDL levels and may render the control of hyperlipidemias more difficult (see WARNINGS ).
A small proportion of women will have adverse lipid changes while taking oral contraceptives. Nonhormonal contraception should be considered in women with uncontrolled dyslipidemias. Persistent hypertriglyceridemia may occur in a small population of combination oral contraceptive users. Elevations of plasma triglycerides may lead to pancreatitis and other complications.
5. Fluid Retention
Oral contraceptives may cause some degree of fluid retention. They should be prescribed with caution, and only with careful monitoring, in patients with conditions which might be aggravated by fluid retention.
16. Body Mass Index
The safety and effectiveness of Aviane in females with a BMI > 35 kg/m2 have not been adequately evaluated.
8. Gastrointestinal
Diarrhea and/or vomiting may reduce hormone absorption resulting in decreased serum concentrations. If a patient vomits or has diarrhea after taking an active tablet, instruct the patient to not take an additional active tablet on that day and to continue the regimen the next day as prescribed. In case of vomiting or diarrhea that continues for 48 hours or greater, instruct the patient to contact the health care provider and use back-up or alternative contraception until active tablets have been taken for 7 consecutive days after vomiting and diarrhea have resolved.
Renal Insufficiency
No formal studies have evaluated the effect of renal disease on the disposition of Aviane.
3. Hepatic Neoplasia
Benign hepatic adenomas are associated with oral-contraceptive use, although the incidence of these benign tumors is rare in the United States. Indirect calculations have estimated the attributable risk to be in the range of 3.3 cases/100,000 for users, a risk that increases after four or more years of use. Rupture of rare, benign, hepatic adenomas may cause death through intra-abdominal hemorrhage.
Studies from Britain have shown an increased risk of developing hepatocellular carcinoma in long-term (>8 years) oral-contraceptive users. However, these cancers are extremely rare in the U.S. and the attributable risk (the excess incidence) of liver cancers in oral-contraceptive users approaches less than one per million users.
9. Drug Interactions
Concomitant Use with HCV Combination Therapy – Liver Enzyme Elevation
Do not coadminister Aviane with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations (see WARNINGS , Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatmen t).
Changes in Contraceptive Effectiveness Associated with Coadministration of Other Products:
Contraceptive effectiveness may be reduced when hormonal contraceptives are coadministered with antibiotics, anticonvulsants, and other drugs that increase the metabolism of contraceptive steroids. This could result in unintended pregnancy or breakthrough bleeding. Examples include rifampin, rifabutin, barbiturates, primidone, phenylbutazone, phenytoin, dexamethasone, carbamazepine, felbamate, oxcarbazepine, topiramate, griseofulvin, and modafinil. In such cases a back-up nonhormonal method of birth control should be considered.
Several of the anti-HIV protease inhibitors have been studied with coadministration of oral combination hormonal contraceptives; significant changes (increase and decrease) in the plasma levels of the estrogen and progestin have been noted in some cases. The safety and efficacy of oral contraceptive products may be affected with coadministration of anti-HIV protease inhibitors. Healthcare providers should refer to the label of the individual anti-HIV protease inhibitors for further drug-drug interaction information.
Herbal products containing St. John's Wort (Hypericum perforatum) may induce hepatic enzymes (cytochrome P 450) and p-glycoprotein transporter and may reduce the effectiveness of contraceptive steroids. This may also result in breakthrough bleeding.
Increase in Plasma Levels Associated with Coadministered Drugs:
Coadministration of atorvastatin and certain oral contraceptives containing ethinyl estradiol increases AUC values for ethinyl estradiol by approximately 20%. Ascorbic acid and acetaminophen increase the bioavailability of ethinyl estradiol since these drugs act as competitive inhibitors for sulfation of ethinyl estradiol in the gastrointestinal wall, a known pathway of elimination for ethinyl estradiol. CYP 3A4 inhibitors such as indinavir, itraconazole, ketoconazole, fluconazole, and troleandomycin may increase plasma hormone levels. Troleandomycin may also increase the risk of intrahepatic cholestasis during coadministration with combination oral contraceptives.
Changes in Plasma Levels of Coadministered Drugs:
Combination hormonal contraceptives containing some synthetic estrogens (e.g., ethinyl estradiol) may inhibit the metabolism of other compounds. Increased plasma concentrations of cyclosporin, prednisolone and other corticosteroids, and theophylline have been reported with concomitant administration of oral contraceptives. Decreased plasma concentrations of acetaminophen and increased clearance of temazepam, salicylic acid, morphine, and clofibric acid, due to induction of conjugation (particularly glucuronidation), have been noted when these drugs were administered with oral contraceptives.
Concomitant use of Aviane may decrease lamotrigine exposure, which may reduce efficacy of lamotrigine. Adjust lamotrigine dosage as recommended in its Prescribing Information based on Aviane initiation or discontinuation.
Concomitant use of Aviane may increase thyroid-binding globulin concentration. Monitor thyroid-stimulating hormone (TSH) level and follow the recommendation for the thyroid hormone in accordance with its Prescribing Information.
The prescribing information of concomitant medications should be consulted to identify potential interactions.
Hepatic Insufficiency
No formal studies have evaluated the effect of hepatic disease on the disposition of Aviane. However, steroid hormones may be poorly metabolized in patients with impaired liver function.
Indications and Usage (INDICATIONS AND USAGE)
Aviane (levonorgestrel and ethinyl estradiol tablets) is indicated for use by females of reproductive potential to prevent pregnancy.
Limitations of use: The efficacy of Aviane (levonorgestrel and ethinyl estradiol tablets) in women with a body mass index (BMI) of > 35 kg/m2 has not been adequately evaluated.
In a clinical trial with Aviane (levonorgestrel and ethinyl estradiol tablets), 1,477 subjects had 7,720 cycles of use and a total of 5 pregnancies were reported. This represents an overall pregnancy rate of 0.84 per 100 woman-years. This rate includes patients who did not take the drug correctly. One or more pills were missed during 1,479 (18.8%) of the 7,870 cycles; thus all tablets were taken during 6,391 (81.2%) of the 7,870 cycles. Of the total 7,870 cycles, a total of 150 cycles were excluded from the calculation of the Pearl index due to the use of backup contraception and/or missing 3 or more consecutive pills.
The mean BMI of the study population was 24 kg/m2. Females with a BMI greater than 30 kg/m2 accounted for 12.1% (n=179) of the study population. Females with a BMI over 35 kg/m2 accounted for 4.3% (n=63) of the study population.
2. Malignant Neoplasms
Breast Cancer
Aviane is contraindicated in females who currently have or have had breast cancer because breast cancer may be hormonally sensitive (see
CONTRAINDICATIONS
). Epidemiology studies have not found a consistent association between use of combined oral contraceptives (COCs) and breast cancer risk. Studies do not show an association between ever (current or past) use of COCs and risk of breast cancer. However, some studies report a small increase in the risk of breast cancer among current or recent users (<6 months since last use) and current users with longer duration of COC use (see
ADVERSE REACTIONS
).
Cervical Cancer
Some studies suggest that oral contraceptive use has been associated with an increase in the risk of cervical intraepithelial neoplasia or invasive cervical cancer in some populations of women. However, there continues to be controversy about the extent to which such findings may be due to differences in sexual behavior and other factors.
7. Gallbladder Disease
Combination oral contraceptives may worsen existing gallbladder disease and may accelerate the development of this disease in previously asymptomatic women. Earlier studies have reported an increased lifetime relative risk of gallbladder surgery in users of oral contraceptives and estrogens. More recent studies, however, have shown that the relative risk of developing gallbladder disease among oral-contraceptive users may be minimal. The recent findings of minimal risk may be related to the use of oral-contraceptive formulations containing lower hormonal doses of estrogens and progestogens. A past history of COC-related cholestasis predicts an increased risk with subsequent COC use. Women with a history of pregnancy-related cholestasis may be at an increased risk for COC related cholestasis.
Drug Drug Interactions (Drug-Drug Interactions)
See PRECAUTIONS - Drug Interactions
12. Hereditary Angioedema
In women with hereditary angioedema, exogenous estrogens may induce or exacerbate symptoms of angioedema.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
To achieve maximum contraceptive effectiveness, Aviane (levonorgestrel and ethinyl estradiol tablets) must be taken exactly as directed and at intervals not exceeding 24 hours. The dosage of Aviane-28 is one orange tablet daily for 21 consecutive days, followed by one light-green inert tablet daily for 7 consecutive days, according to the prescribed schedule. It is recommended that Aviane-28 tablets be taken at the same time each day.
The dispenser should be kept in the wallet supplied to avoid possible fading of the pills. If the pills fade, patients should continue to take them as directed.
During the first cycle of use
The possibility of ovulation and conception prior to initiation of medication should be considered. The patient should be instructed to begin taking Aviane on either the first Sunday after the onset of menstruation (Sunday Start) or on Day 1 of menstruation (Day 1 Start).
Sunday start:
The patient is instructed to begin taking Aviane-28 on the first Sunday after the onset of menstruation. If menstruation begins on a Sunday, the first tablet (orange) is taken that day. One orange tablet should be taken daily for 21 consecutive days, followed by one light-green inert tablet daily for 7 consecutive days. Withdrawal bleeding should usually occur within 3 days following discontinuation of orange tablets and may not have finished before the next pack is started. During the first cycle, contraceptive reliance should not be placed on Aviane-28 until an orange tablet has been taken daily for 7 consecutive days, and a nonhormonal back-up method of birth control should be used during those 7 days.
Day 1 start:
During the first cycle of medication, the patient is instructed to begin taking Aviane-28 during the first 24 hours of her period (day one of her menstrual cycle). One orange tablet should be taken daily for 21 consecutive days, followed by one light-green inert tablet daily for 7 consecutive days. Withdrawal bleeding should usually occur within 3 days following discontinuation of orange tablets and may not have finished before the next pack is started. If medication is begun on day one of the menstrual cycle, no back-up contraception is necessary. If Aviane-28 tablets are started later than day one of the first menstrual cycle or postpartum, contraceptive reliance should not be placed on Aviane-28 tablets until after the first 7 consecutive days of administration, and a nonhormonal back-up method of birth control should be used during those 7 days.
After the first cycle of use
The patient begins her next and all subsequent courses of tablets on the day after taking her last light-green tablet. She should follow the same dosing schedule: 21 days on orange tablets followed by 7 days on light-green tablets. If in any cycle the patient starts tablets later than the proper day, she should protect herself against pregnancy by using a nonhormonal back-up method of birth control until she has taken an orange tablet daily for 7 consecutive days.
Switching from another hormonal method of contraception
When the patient is switching from a 21-day regimen of tablets, she should wait 7 days after her last tablet before she starts Aviane. She will probably experience withdrawal bleeding during that week. She should be sure that no more than 7 days pass after her previous 21-day regimen. When the patient is switching from a 28-day regimen of tablets, she should start her first pack of Aviane on the day after her last tablet. She should not wait any days between packs. The patient may switch any day from a progestin-only pill and should begin Aviane the next day. If switching from an implant or injection, the patient should start Aviane on the day of implant removal or, if using an injection, the day the next injection would be due. In switching from a progestin-only pill, injection, implant, or intrauterine system (IUS), the patient should be advised to use additional nonhormonal contraception (such as condoms) until active tablets have been taken for 7 consecutive days of the first pack.
If spotting or breakthrough bleeding occurs
If spotting or breakthrough bleeding occur, the patient is instructed to continue on the same regimen. This type of bleeding is usually transient and without significance; however, if the bleeding is persistent or prolonged, the patient is advised to consult her physician.
Risk of pregnancy if tablets are missed
While there is little likelihood of ovulation occurring if only one or two orange tablets are missed, the possibility of ovulation increases with each successive day that scheduled orange tablets are missed. Although the occurrence of pregnancy is unlikely if Aviane is taken according to directions, if withdrawal bleeding does not occur, the possibility of pregnancy must be considered. If the patient has not adhered to the prescribed schedule (missed one or more tablets or started taking them on a day later than she should have), the probability of pregnancy should be considered at the time of the first missed period and appropriate diagnostic measures taken. If the patient has adhered to the prescribed regimen and misses two consecutive periods, pregnancy should be ruled out.
The risk of pregnancy increases with each active (orange) tablet missed. For additional patient instructions regarding missed tablets, see the WHAT TO DO IF YOU MISS PILLS section in the DETAILED PATIENT LABELING below.
Use after pregnancy, abortion or miscarriage
Aviane may be initiated no earlier than day 28 postpartum in the nonlactating mother or after a second trimester abortion due to the increased risk for thromboembolism (see C ONTRAINDICATIONS, WARNINGS, and PRECAUTIONS concerning thromboembolic disease). The patient should be advised to use additional nonhormonal contraception (such as condoms) until active tablets have been taken for 7 consecutive days.
Aviane may be initiated immediately after a first trimester abortion or miscarriage. Instruct the patient to use additional nonhormonal contraception (such as condoms) until active tablets have been taken for 7 consecutive days, unless starting Aviane on the day of surgical abortion.
9. Elevated Blood Pressure
Aviane is contraindicated in women with uncontrolled hypertension or hypertension with vascular disease (see CONTRAINDICATIONS ). For all females, including those with well-controlled hypertension, monitor blood pressure at routine visits and stop Aviane if blood pressure rises significantly.
An increase in blood pressure has been reported in females using CHCs, and this increase is more likely in older women with extended duration of use. The effect of CHCs on blood pressure may vary according to the progestin in the CHC.
11. Bleeding Irregularities
Unscheduled bleeding and spotting are sometimes encountered in patients on oral contraceptives, especially during the first three months of use. The type and dose of progestogen may be important. If bleeding persists or recurs, nonhormonal causes should be considered and adequate diagnostic measures taken to rule out malignancy or pregnancy in the event of unscheduled bleeding, as in the case of any abnormal vaginal bleeding. If pathology has been excluded, time or a change to another formulation may solve the problem. In the event of amenorrhea, pregnancy should be ruled out.
Some women may encounter post-pill amenorrhea or oligomenorrhea (possibly with anovulation), especially when such a condition was preexistent.
In the clinical trial with levonorgestrel 0.1 mg and ethinyl estradiol 0.02 mg tablets, unscheduled bleeding was defined as bleeding or spotting that occurred:
- During cycle 1 on pill-pack Days 4 to 21, inclusive of a 28-day cycle.
- In subsequent cycles, on Days 5 to 21 inclusive or on pill-pack Days 1 to 4 inclusive if preceded by 2 consecutive days without bleeding or spotting.
Based on subject diaries, the proportion of subjects reporting unscheduled bleeding or spotting per 28-day cycle decreased over time: 30.5% at Cycle 1 versus 18.2% at Cycle 12.
14. Effect On Binding Globulins (14. Effect on Binding Globulins)
The estrogen component of COCs may raise the serum concentrations of thyroxine-binding globulin, sex hormone-binding globulin, and cortisol-binding globulin. Monitor thyroid-stimulating hormone (TSH) levels for females receiving Aviane and thyroid hormone replacement therapy concomitantly. Follow the recommendation for the thyroid hormone in accordance with its Prescribing Information.
17. Information for the Patient
See Patient Labeling printed below.
2. Physical Examination and Follow Up (2. Physical Examination and Follow-Up)
A periodic personal and family medical history and complete physical examination are appropriate for all women, including women using oral contraceptives. The physical examination, however, may be deferred until after initiation of oral contraceptives if requested by the woman and judged appropriate by the clinician. The physical examination should include special reference to blood pressure, breasts, abdomen, pelvic organs, and relevant laboratory tests. In case of undiagnosed, persistent, or recurrent abnormal vaginal bleeding, appropriate diagnostic measures should be conducted to rule out malignancy. Women with a strong family history of breast cancer or who have breast nodules should be monitored with particular care.
10. Interactions With Laboratory Tests (10. Interactions with Laboratory Tests)
Certain endocrine- and liver-function tests and blood components may be affected by oral contraceptives:
a. Increased prothrombin and factors VII, VIII, IX, and X; decreased antithrombin 3; increased norepinephrine-induced platelet aggregability.
b. Increased thyroid-binding globulin (TBG) leading to increased circulating total thyroid hormone, as measured by protein-bound iodine (PBI), T4 by column or by radioimmunoassay.
c. Other binding proteins may be elevated in serum i.e., corticosteroid binding globulin (CBG), sex hormone-binding globulins (SHBG) leading to increased levels of total circulating corticosteroids and sex steroids respectively.
d. Triglycerides may be increased and levels of various other lipids and lipoproteins may be affected.
e. Glucose tolerance may be decreased.
f. Serum folate levels may be depressed by oral-contraceptive therapy. This may be of clinical significance if a woman becomes pregnant shortly after discontinuing oral contraceptives.
Package/label Display Panel, Part 1 of 2 (Package/Label Display Panel, Part 1 of 2)
NDC 0555-9045-58
6 Blister Cards, 28 Tablets Each
Aviane®
(levonorgestrel and ethinyl
estradiol tablets USP)
0.10 mg/0.02 mg
Contains 6 blister cards, each containing 28 tablets. Twenty-one
orange tablets, each containing 0.10 mg levonorgestrel, USP with
0.02 mg ethinyl estradiol, USP and seven light-green inert tablets.
Rx only
SHAPING
WOMEN’S HEALTH®
TEVA
Package/label Display Panel, Part 2 of 2 (Package/Label Display Panel, Part 2 of 2)
8. Carbohydrate and Lipid Metabolic Effects
Oral contraceptives have been shown to cause glucose intolerance in a significant percentage of users. Carefully monitor females with prediabetes and diabetes who are using Aviane. Aviane may decrease glucose tolerance.
A small proportion of women will have persistent hypertriglyceridemia while on COCs. As discussed earlier (see WARNINGS and PRECAUTIONS ), changes in serum triglycerides and lipoprotein levels have been reported in oral-contraceptive users.
1. Thromboembolic Disorders and Other Vascular Problems
Cardiovascular and Cerebrovascular Events
Use of CHCs increases the risk of cardiovascular events and cerebrovascular events, such as myocardial infarction and stroke. The risk of these events with CHC use is greater in females with concomitant risk factors: age 35 years and older, smoking, hypertension, dyslipidemia, diabetes, or obesity. The risk increases with increasing age and with increasing number of cigarettes smoked.
Venous Thromboembolism
Use of CHCs also increases the risk of VTE, such as deep vein thrombosis and pulmonary embolism. The rate of VTE in females using COCs has been estimated to be 3 to 9 cases per 10,000 woman-years. The VTE risk should be considered in the context of relevant subpopulations of females of reproductive potential who are not taking CHCs ( ADVERSE REACTIONS ).
Risk factors for VTE with CHC use include smoking, obesity, family history of VTE, and prolonged immobilization, in addition to other factors that contraindicate use of CHCs (see CONTRAINDICATIONS ). The presence of multiple risk factors for VTE with CHC use may further increase the risk. The risk of VTE is highest during the first year of CHC use and when restarting hormonal contraception after a break of four weeks or longer. The risk of VTE returns to baseline approximately 3 months after CHC use is discontinued.
Postpartum Venous Thromboembolism
The risk of VTE is increased during the first six weeks postpartum. The risk is highest up to four weeks postpartum but remains higher than baseline until at least six weeks postpartum. The presence of multiple risk factors for VTE may further increase the risk. Obstetric complications may extend the elevated risk up to 12 weeks postpartum.
6. Oral Contraceptive Use Before Or During Early Pregnancy (6. Oral-Contraceptive Use Before or During Early Pregnancy)
Extensive epidemiological studies have revealed no increased risk of birth defects in infants born to women who have used oral contraceptives prior to pregnancy. Studies also do not suggest a teratogenic effect, particularly insofar as cardiac anomalies and limb-reduction defects are concerned, when taken inadvertently during early pregnancy (see CONTRAINDICATIONS).
It is recommended that for any patient who has missed two consecutive periods, pregnancy should be ruled out. If the patient has not adhered to the prescribed schedule, the possibility of pregnancy should be considered at the time of the first missed period. Oral-contraceptive use should be discontinued if pregnancy is confirmed.
4. Risk of Liver Enzyme Elevations With Concomitant Hepatitis C Treatment (4. Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment)
During clinical trials with the Hepatitis C combination drug regimen that contains ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN), including some cases greater than 20 times the ULN, were significantly more frequent in women using ethinyl estradiol-containing medications such as COCs. Discontinue Aviane prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir, with or without dasabuvir (see CONTRAINDICATIONS). Aviane can be restarted approximately 2 weeks following completion of treatment with the combination drug regimen.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:42:41.528315 · Updated: 2026-03-14T22:10:30.418145