These Highlights Do Not Include All The Information Needed To Use Dulera Safely And Effectively. See Full Prescribing Information For Dulera.
8f399784-f257-4f31-b3dc-368bf2a1864d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
DULERA is a combination product containing a corticosteroid and a long-acting beta 2 -adrenergic agonist (LABA) indicated for: Treatment of asthma in patients 5 years of age and older. ( 1.1 ) Important Limitation of Use: Not indicated for the relief of acute bronchospasm. ( 1.1 )
Indications and Usage
DULERA is a combination product containing a corticosteroid and a long-acting beta 2 -adrenergic agonist (LABA) indicated for: Treatment of asthma in patients 5 years of age and older. ( 1.1 ) Important Limitation of Use: Not indicated for the relief of acute bronchospasm. ( 1.1 )
Dosage and Administration
For oral inhalation only. ( 2.1 ) Treatment of asthma in patients 12 years of age and older: 2 inhalations twice daily of DULERA 100 mcg/5 mcg or 200 mcg/5 mcg. Starting dosage is based on disease severity. ( 2.2 ) Treatment of asthma in patients aged 5 to less than 12 years: 2 inhalations twice daily of DULERA 50 mcg/5 mcg. ( 2.2 )
Warnings and Precautions
LABA monotherapy increases the risk of serious asthma-related events. ( 5.1 ) Deterioration of disease and acute episodes: Do not initiate in acutely deteriorating asthma or to treat acute symptoms. ( 5.2 ) Use with additional long-acting beta 2 -agonist: Do not use in combination because of risk of overdose. ( 5.3 ) Localized infections: Candida albicans infection of the mouth and throat may occur. Monitor patients periodically for signs of adverse effects on the oral cavity. After dosing, advise patients to rinse their mouth with water and spit out contents without swallowing. ( 5.4 ) Immunosuppression: Potential worsening of existing tuberculosis, fungal, bacterial, viral, or parasitic infection; or ocular herpes simplex infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. Use with caution in patients with these infections because of the potential for worsening of these infections. ( 5.5 ) Transferring patients from systemic corticosteroids: Risk of impaired adrenal function when transferring from oral steroids. Taper patients slowly from systemic corticosteroids if transferring to DULERA. ( 5.6 ) Hypercorticism and adrenal suppression: May occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue DULERA slowly. ( 5.7 ) Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir): Risk of increased systemic corticosteroid effects. Exercise caution when used with DULERA. ( 5.8 ) Paradoxical bronchospasm: Discontinue DULERA and institute alternative therapy if paradoxical bronchospasm occurs. ( 5.9 ) Patients with cardiovascular disorders: Use with caution because of beta-adrenergic stimulation. ( 5.11 ) Decreases in bone mineral density: Monitor patients with major risk factors for decreased bone mineral content. ( 5.12 ) Effects on growth: Monitor growth of pediatric patients. ( 5.13 ) Glaucoma and cataracts: Consider referral to an ophthalmologist in patients who develop ocular symptoms or use DULERA long term. ( 5.14 ) Coexisting conditions: Use with caution in patients with aneurysm, pheochromocytoma, convulsive disorders, thyrotoxicosis, diabetes mellitus, and ketoacidosis. ( 5.15 ) Hypokalemia and hyperglycemia: Be alert to hypokalemia and hyperglycemia. ( 5.16 )
Contraindications
Primary treatment of status asthmaticus or acute episodes of asthma requiring intensive measures. ( 4.1 ) Hypersensitivity to any of the ingredients of DULERA. ( 4.2 )
Adverse Reactions
LABA use may result in the following: Serious asthma-related events – hospitalizations, intubations, and death [see Warnings and Precautions (5.1) ] . Cardiovascular and central nervous system effects [see Warnings and Precautions (5.11) ]. Systemic and local corticosteroid use may result in the following: Candida albicans infection [see Warnings and Precautions (5.4) ] Immunosuppression [see Warnings and Precautions (5.5) ] Hypercorticism and adrenal suppression [see Warnings and Precautions (5.7) ] Growth effects in pediatrics [see Warnings and Precautions (5.13) ] Glaucoma and cataracts [see Warnings and Precautions (5.14) ] Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Drug Interactions
In clinical trials, concurrent administration of DULERA and other drugs, such as short-acting beta 2 -agonist and intranasal corticosteroids have not resulted in an increased frequency of adverse drug reactions. No formal drug interaction studies have been performed with DULERA. The drug interactions of the combination are expected to reflect those of the individual components.
Storage and Handling
Only use the DULERA canister with the DULERA actuator. Do not use the DULERA actuator with any other inhalation drug product. Do not use actuators from other products with the DULERA canister. Do not remove the canister from the actuator because the correct amount of medication may not be discharged; the dose counter may not function properly; reinsertion may cause the dose counter to count down by 1 and discharge a puff. The correct amount of medication in each inhalation cannot be ensured after the labeled number of actuations from the canister has been used, even though the inhaler may not feel completely empty and may continue to operate. Discard the inhaler when the labeled number of actuations has been used (the dose counter will read "0"). Store at controlled room temperature 20°C–25°C (68°F–77°F); excursions permitted to 15°C–30°C (59°F–86°F) [see USP Controlled Room Temperature]. After priming, store the inhaler with the mouthpiece down or in a horizontal position. For best results, keep the canister at room temperature before use. Shake well and remove the cap from the mouthpiece of the actuator before using. Keep out of reach of children. Avoid spraying in eyes. Contents Under Pressure: Do not puncture. Do not use or store near heat or open flame. Exposure to temperatures above 120°F may cause bursting. Never throw container into fire or incinerator.
How Supplied
DULERA is available in three strengths and supplied in the following package sizes ( Table 8 ): Table 8 Package NDC Strength Identifier (Color Band) Included on the outer carton, actuator, and canister labels. DULERA 50 mcg/5 mcg 120 inhalations 78206-125-01 Blue DULERA 100 mcg/5 mcg 120 inhalations 78206-127-01 Yellow DULERA 100 mcg/5 mcg 60 inhalations (institutional pack) 78206-127-02 Yellow DULERA 200 mcg/5 mcg 120 inhalations 78206-126-01 Purple DULERA 200 mcg/5 mcg 60 inhalations (institutional pack) 78206-126-02 Purple Each strength is supplied as a pressurized aluminum canister that has a blue plastic actuator integrated with a dose counter and a green dust cap. Each 120-inhalation canister has a net fill weight of 13 grams and each 60-inhalation canister has a net fill weight of 8.8 grams. Each canister is placed into a carton. Each carton contains 1 canister and a Patient Information leaflet. Initially the dose counter will display "64" or "124" actuations. After the initial priming with 4 actuations, the dose counter will read "60" or "120" and the inhaler is now ready for use.
Medication Information
Warnings and Precautions
LABA monotherapy increases the risk of serious asthma-related events. ( 5.1 ) Deterioration of disease and acute episodes: Do not initiate in acutely deteriorating asthma or to treat acute symptoms. ( 5.2 ) Use with additional long-acting beta 2 -agonist: Do not use in combination because of risk of overdose. ( 5.3 ) Localized infections: Candida albicans infection of the mouth and throat may occur. Monitor patients periodically for signs of adverse effects on the oral cavity. After dosing, advise patients to rinse their mouth with water and spit out contents without swallowing. ( 5.4 ) Immunosuppression: Potential worsening of existing tuberculosis, fungal, bacterial, viral, or parasitic infection; or ocular herpes simplex infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. Use with caution in patients with these infections because of the potential for worsening of these infections. ( 5.5 ) Transferring patients from systemic corticosteroids: Risk of impaired adrenal function when transferring from oral steroids. Taper patients slowly from systemic corticosteroids if transferring to DULERA. ( 5.6 ) Hypercorticism and adrenal suppression: May occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue DULERA slowly. ( 5.7 ) Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir): Risk of increased systemic corticosteroid effects. Exercise caution when used with DULERA. ( 5.8 ) Paradoxical bronchospasm: Discontinue DULERA and institute alternative therapy if paradoxical bronchospasm occurs. ( 5.9 ) Patients with cardiovascular disorders: Use with caution because of beta-adrenergic stimulation. ( 5.11 ) Decreases in bone mineral density: Monitor patients with major risk factors for decreased bone mineral content. ( 5.12 ) Effects on growth: Monitor growth of pediatric patients. ( 5.13 ) Glaucoma and cataracts: Consider referral to an ophthalmologist in patients who develop ocular symptoms or use DULERA long term. ( 5.14 ) Coexisting conditions: Use with caution in patients with aneurysm, pheochromocytoma, convulsive disorders, thyrotoxicosis, diabetes mellitus, and ketoacidosis. ( 5.15 ) Hypokalemia and hyperglycemia: Be alert to hypokalemia and hyperglycemia. ( 5.16 )
Indications and Usage
DULERA is a combination product containing a corticosteroid and a long-acting beta 2 -adrenergic agonist (LABA) indicated for: Treatment of asthma in patients 5 years of age and older. ( 1.1 ) Important Limitation of Use: Not indicated for the relief of acute bronchospasm. ( 1.1 )
Dosage and Administration
For oral inhalation only. ( 2.1 ) Treatment of asthma in patients 12 years of age and older: 2 inhalations twice daily of DULERA 100 mcg/5 mcg or 200 mcg/5 mcg. Starting dosage is based on disease severity. ( 2.2 ) Treatment of asthma in patients aged 5 to less than 12 years: 2 inhalations twice daily of DULERA 50 mcg/5 mcg. ( 2.2 )
Contraindications
Primary treatment of status asthmaticus or acute episodes of asthma requiring intensive measures. ( 4.1 ) Hypersensitivity to any of the ingredients of DULERA. ( 4.2 )
Adverse Reactions
LABA use may result in the following: Serious asthma-related events – hospitalizations, intubations, and death [see Warnings and Precautions (5.1) ] . Cardiovascular and central nervous system effects [see Warnings and Precautions (5.11) ]. Systemic and local corticosteroid use may result in the following: Candida albicans infection [see Warnings and Precautions (5.4) ] Immunosuppression [see Warnings and Precautions (5.5) ] Hypercorticism and adrenal suppression [see Warnings and Precautions (5.7) ] Growth effects in pediatrics [see Warnings and Precautions (5.13) ] Glaucoma and cataracts [see Warnings and Precautions (5.14) ] Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Drug Interactions
In clinical trials, concurrent administration of DULERA and other drugs, such as short-acting beta 2 -agonist and intranasal corticosteroids have not resulted in an increased frequency of adverse drug reactions. No formal drug interaction studies have been performed with DULERA. The drug interactions of the combination are expected to reflect those of the individual components.
Storage and Handling
Only use the DULERA canister with the DULERA actuator. Do not use the DULERA actuator with any other inhalation drug product. Do not use actuators from other products with the DULERA canister. Do not remove the canister from the actuator because the correct amount of medication may not be discharged; the dose counter may not function properly; reinsertion may cause the dose counter to count down by 1 and discharge a puff. The correct amount of medication in each inhalation cannot be ensured after the labeled number of actuations from the canister has been used, even though the inhaler may not feel completely empty and may continue to operate. Discard the inhaler when the labeled number of actuations has been used (the dose counter will read "0"). Store at controlled room temperature 20°C–25°C (68°F–77°F); excursions permitted to 15°C–30°C (59°F–86°F) [see USP Controlled Room Temperature]. After priming, store the inhaler with the mouthpiece down or in a horizontal position. For best results, keep the canister at room temperature before use. Shake well and remove the cap from the mouthpiece of the actuator before using. Keep out of reach of children. Avoid spraying in eyes. Contents Under Pressure: Do not puncture. Do not use or store near heat or open flame. Exposure to temperatures above 120°F may cause bursting. Never throw container into fire or incinerator.
How Supplied
DULERA is available in three strengths and supplied in the following package sizes ( Table 8 ): Table 8 Package NDC Strength Identifier (Color Band) Included on the outer carton, actuator, and canister labels. DULERA 50 mcg/5 mcg 120 inhalations 78206-125-01 Blue DULERA 100 mcg/5 mcg 120 inhalations 78206-127-01 Yellow DULERA 100 mcg/5 mcg 60 inhalations (institutional pack) 78206-127-02 Yellow DULERA 200 mcg/5 mcg 120 inhalations 78206-126-01 Purple DULERA 200 mcg/5 mcg 60 inhalations (institutional pack) 78206-126-02 Purple Each strength is supplied as a pressurized aluminum canister that has a blue plastic actuator integrated with a dose counter and a green dust cap. Each 120-inhalation canister has a net fill weight of 13 grams and each 60-inhalation canister has a net fill weight of 8.8 grams. Each canister is placed into a carton. Each carton contains 1 canister and a Patient Information leaflet. Initially the dose counter will display "64" or "124" actuations. After the initial priming with 4 actuations, the dose counter will read "60" or "120" and the inhaler is now ready for use.
Description
DULERA is a combination product containing a corticosteroid and a long-acting beta 2 -adrenergic agonist (LABA) indicated for: Treatment of asthma in patients 5 years of age and older. ( 1.1 ) Important Limitation of Use: Not indicated for the relief of acute bronchospasm. ( 1.1 )
Section 42229-5
Important Limitation of Use:
- DULERA is NOT indicated for the relief of acute bronchospasm.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: 6/2025 | |||
|
PATIENT INFORMATION
DULERA® [dew-LAIR-ah] 50 mcg/5 mcg (mometasone furoate 50 mcg and formoterol fumarate dihydrate 5 mcg) Inhalation Aerosol DULERA® 100 mcg/5 mcg (mometasone furoate 100 mcg and formoterol fumarate dihydrate 5 mcg) Inhalation Aerosol DULERA® 200 mcg/5 mcg (mometasone furoate 200 mcg and formoterol fumarate dihydrate 5 mcg) Inhalation Aerosol |
||||
| Read the Patient Information leaflet that comes with DULERA ® before you start using it and each time you get a refill. There may be new information. The Patient Information leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment. | ||||
What is DULERA?
|
||||
Do not use DULERA:
|
||||
Before and during treatment with DULERA, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take including prescription and over-the-counter medicines, vitamins, and herbal supplements. DULERA and certain other medicines may interact with each other. This may cause serious side effects. |
||||
|
How should I use DULERA? See the step-by-step instructions for using DULERA at the end of this Patient Information leaflet. Do not use DULERA unless your healthcare provider has taught you and you understand how to use it. Ask your healthcare provider or pharmacist if you have any questions.
|
||||
|
What are the possible side effects of DULERA? DULERA may cause serious side effects, including:
|
||||
|
|
|
||
|
||||
|
|
|||
|
||||
|
|
|||
|
||||
The most common side effects reported while using DULERA include:
These are not all the side effects of DULERA. Ask your healthcare provider or pharmacist for more information. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Organon LLC., a subsidiary of Organon & Co., at 1-844-674-3200. |
||||
How should I store DULERA?
|
||||
|
General Information about the safe and effective use of DULERA.
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information leaflet. Do not use DULERA for a condition for which it was not prescribed. Do not give your DULERA to other people, even if they have the same condition. It may harm them. You can ask your healthcare provider or pharmacist for information about DULERA that was written for healthcare professionals. |
||||
|
What are the ingredients in DULERA?
Active ingredients: mometasone furoate and formoterol fumarate dihydrate Inactive ingredients: hydrofluoroalkane (HFA-227), anhydrous alcohol and oleic acid |
||||
|
For more information about DULERA:
Go to www.DULERA.com or call 1-844-674-3200. |
Section 51945-4
PRINCIPAL DISPLAY PANEL - 100 mcg/5 mcg Canister Carton
NDC 78206-127-01
Dulera
®
(mometasone furoate and
formoterol fumarate dihydrate)
Inhalation Aerosol
100 mcg/5 mcg
per actuation
For oral inhalation only
SHAKE WELL BEFORE USING.
Dulera canister to be used with Dulera actuator only.
Rx only
120 Metered Actuations
Net Wt. 13g
7.4 Diuretics
Concomitant treatment with diuretics may potentiate the possible hypokalemic effect of adrenergic agonists. The ECG changes and/or hypokalemia that may result from the administration of non-potassium-sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dose of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the coadministration of DULERA with non-potassium-sparing diuretics.
11 Description
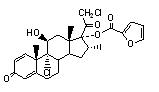
DULERA 50 mcg/5 mcg, DULERA 100 mcg/5 mcg, and DULERA 200 mcg/5 mcg are combinations of mometasone furoate and formoterol fumarate dihydrate for oral inhalation only.
One active component of DULERA is mometasone furoate, a corticosteroid having the chemical name 9,21-dichloro-11(Beta),17-dihydroxy-16 (alpha)-methylpregna-1,4-diene-3,20-dione 17-(2-furoate) with the following chemical structure:
Mometasone furoate is a white powder with an empirical formula of C27H30Cl2O6, and molecular weight 521.44. It is practically insoluble in water; slightly soluble in methanol, ethanol, and isopropanol; soluble in acetone.
One active component of DULERA is formoterol fumarate dihydrate, a racemate. Formoterol fumarate dihydrate is a selective beta2-adrenergic bronchodilator having the chemical name of (±)-2-hydroxy-5-[(1RS)-1-hydroxy-2-[[(1RS)-2-(4-methoxyphenyl)-1-methylethyl]-amino]ethyl]formanilide fumarate dihydrate with the following chemical structure:
Formoterol fumarate dihydrate has a molecular weight of 840.9, and its empirical formula is (C19H24N2O4)2•C4H4O4•2H2O. Formoterol fumarate dihydrate is a white to yellowish powder, which is freely soluble in glacial acetic acid, soluble in methanol, sparingly soluble in ethanol and isopropanol, slightly soluble in water, and practically insoluble in acetone, ethyl acetate, and diethyl ether.
DULERA 50 mcg/5 mcg, 100 mcg/5 mcg, and 200 mcg/5 mcg are each formulated as a hydrofluoroalkane (HFA-227; 1, 1, 1, 2, 3, 3, 3-heptafluoropropane) propelled pressurized metered dose inhaler containing sufficient amount of drug for 60 or 120 inhalations [see How Supplied/Storage and Handling (16)]. After priming, each actuation of the inhaler delivers 60, 115, or 225 mcg of mometasone furoate and 5.5 mcg of formoterol fumarate dihydrate in 69.6 mg of suspension from the valve and delivers 50, 100, or 200 mcg of mometasone furoate and 5 mcg of formoterol fumarate dihydrate from the actuator. The actual amount of drug delivered to the lung may depend on patient factors, such as the coordination between actuation of the device and inspiration through the delivery system. DULERA also contains anhydrous alcohol as a cosolvent and oleic acid as a surfactant.
DULERA should be primed before using for the first time by releasing 4 test sprays into the air, away from the face, shaking well before each spray. In cases where the inhaler has not been used for more than 5 days, prime the inhaler again by releasing 4 test sprays into the air, away from the face, shaking well before each spray.
16.1 How Supplied
DULERA is available in three strengths and supplied in the following package sizes (Table 8):
| Package | NDC | Strength Identifier (Color Band) Included on the outer carton, actuator, and canister labels.
|
|---|---|---|
| DULERA 50 mcg/5 mcg 120 inhalations |
78206-125-01 | Blue |
| DULERA 100 mcg/5 mcg 120 inhalations |
78206-127-01 | Yellow |
| DULERA 100 mcg/5 mcg 60 inhalations (institutional pack) |
78206-127-02 | Yellow |
| DULERA 200 mcg/5 mcg 120 inhalations |
78206-126-01 | Purple |
| DULERA 200 mcg/5 mcg 60 inhalations (institutional pack) |
78206-126-02 | Purple |
Each strength is supplied as a pressurized aluminum canister that has a blue plastic actuator integrated with a dose counter and a green dust cap. Each 120-inhalation canister has a net fill weight of 13 grams and each 60-inhalation canister has a net fill weight of 8.8 grams. Each canister is placed into a carton. Each carton contains 1 canister and a Patient Information leaflet.
Initially the dose counter will display "64" or "124" actuations. After the initial priming with 4 actuations, the dose counter will read "60" or "120" and the inhaler is now ready for use.
5.4 Local Effects
In clinical trials, the development of localized infections of the mouth and pharynx with Candida albicans have occurred in patients treated with DULERA. If oropharyngeal candidiasis develops, treat with appropriate local or systemic (i.e., oral) antifungal therapy while remaining on treatment with DULERA therapy, but at times therapy with DULERA may need to be interrupted. To reduce the risk of oropharyngeal candidiasis, after dosing with DULERA, advise patients to rinse their mouth with water and spit out the contents without swallowing.
8.4 Pediatric Use
The safety and effectiveness of DULERA have been established in patients 12 years of age and older in 3 clinical trials up to 52 weeks in duration. In the 3 clinical trials, 101 patients 12 to 17 years of age were treated with DULERA. Patients in this age-group demonstrated efficacy results similar to those observed in patients 18 years of age and older. There were no obvious differences in the type or frequency of adverse reactions reported in this age group compared to patients 18 years of age and older. Similar efficacy and safety results were observed in an additional 22 patients 12 to 17 years of age who were treated with DULERA in another clinical trial.
The safety and effectiveness of DULERA 50 mcg/5 mcg, two inhalations twice daily, have been established in patients with asthma aged 5 to less than 12 years in clinical trials up to 24 weeks of treatment duration. Patients in this age group demonstrated efficacy and safety results similar to those observed in patients aged 12 years and older who were treated with DULERA [see Adverse Reactions (6.1) and Clinical Studies (14.1)].
The safety and effectiveness of DULERA have not been established in children younger than 5 years of age.
Controlled clinical studies have shown that inhaled corticosteroids may cause a reduction in growth velocity in pediatric patients. In these studies, the mean reduction in growth velocity was approximately 1 cm per year (range 0.3 to 1.8 per year) and appears to depend upon dose and duration of exposure. This effect was observed in the absence of laboratory evidence of hypothalamic-pituitary-adrenal (HPA) axis suppression, suggesting that growth velocity is a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA axis function. The long-term effects of this reduction in growth velocity associated with orally inhaled corticosteroids, including the impact on final adult height, are unknown. The potential for "catch up" growth following discontinuation of treatment with orally inhaled corticosteroids has not been adequately studied.
The growth of children and adolescents receiving orally inhaled corticosteroids, including DULERA, should be monitored routinely (e.g., via stadiometry). If a child or adolescent on any corticosteroid appears to have growth suppression, the possibility that he/she is particularly sensitive to this effect should be considered. The potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the risks associated with alternative therapies. To minimize the systemic effects of orally inhaled corticosteroids, including DULERA, each patient should be titrated to his/her lowest effective dose [see Dosage and Administration (2.2)].
8.5 Geriatric Use
A total of 77 patients 65 years of age and older (11 of whom were 75 years and older) have been treated with DULERA in 3 clinical trials up to 52 weeks in duration. Similar efficacy and safety results were observed in an additional 28 patients 65 years of age and older who were treated with DULERA in another clinical trial. No overall differences in safety or effectiveness were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out. As with other products containing beta2-agonists, special caution should be observed when using DULERA in geriatric patients who have concomitant cardiovascular disease that could be adversely affected by beta2-agonists. Based on available data for DULERA or its active components, no adjustment of dosage of DULERA in geriatric patients is warranted.
4 Contraindications
6 Adverse Reactions
LABA use may result in the following:
- Serious asthma-related events – hospitalizations, intubations, and death [see Warnings and Precautions (5.1)].
- Cardiovascular and central nervous system effects [see Warnings and Precautions (5.11)].
Systemic and local corticosteroid use may result in the following:
- Candida albicans infection [see Warnings and Precautions (5.4)]
- Immunosuppression [see Warnings and Precautions (5.5)]
- Hypercorticism and adrenal suppression [see Warnings and Precautions (5.7)]
- Growth effects in pediatrics [see Warnings and Precautions (5.13)]
- Glaucoma and cataracts [see Warnings and Precautions (5.14)]
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7 Drug Interactions
In clinical trials, concurrent administration of DULERA and other drugs, such as short-acting beta2-agonist and intranasal corticosteroids have not resulted in an increased frequency of adverse drug reactions. No formal drug interaction studies have been performed with DULERA. The drug interactions of the combination are expected to reflect those of the individual components.
4.2 Hypersensitivity
DULERA is contraindicated in patients with known hypersensitivity to mometasone furoate, formoterol fumarate, or any of the ingredients in DULERA [see Warnings and Precautions (5.10)].
5.13 Effect On Growth
Orally inhaled corticosteroids, including DULERA, may cause a reduction in growth velocity when administered to pediatric patients. Monitor the growth of pediatric patients receiving DULERA routinely (e.g., via stadiometry). To minimize the systemic effects of orally inhaled corticosteroids, including DULERA, titrate each patient's dose to the lowest dosage that effectively controls his/her symptoms [see Use in Specific Populations (8.4)].
5.5 Immunosuppression
Persons who are using drugs that suppress the immune system are more susceptible to infections than healthy individuals.
Chickenpox and measles, for example, can have a more serious or even fatal course in susceptible children or adults using corticosteroids. In such children or adults who have not had these diseases or who are not properly immunized, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) or pooled intravenous immunoglobulin (IVIG) may be indicated. If exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the respective package inserts for complete VZIG and IG prescribing information.) If chickenpox develops, treatment with antiviral agents may be considered.
DULERA should be used with caution, if at all, in patients with active or quiescent tuberculosis infection of the respiratory tract, untreated systemic fungal, bacterial, viral, or parasitic infections; or ocular herpes simplex.
7.2 Adrenergic Agents
If additional adrenergic drugs are to be administered by any route, they should be used with caution because the pharmacologically predictable sympathetic effects of formoterol, a component of DULERA, may be potentiated.
2.2 Recommended Dosage
Administer DULERA as two inhalations twice daily every day (morning and evening) by the orally inhaled route. Shake well prior to each inhalation. Individual patients may experience a variable time to onset and degree of symptom relief. If symptoms arise between doses, use an inhaled short-acting beta2-agonist for immediate relief. Improvement in lung function following administration of DULERA can occur within 5 minutes of treatment, although the maximum benefit may not be achieved for 1 week or longer after beginning treatment.
4.1 Status Asthmaticus
DULERA is contraindicated in the primary treatment of status asthmaticus or other acute episodes of asthma where intensive measures are required.
8.6 Hepatic Impairment
Concentrations of mometasone furoate appear to increase with severity of hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage
1.1 Treatment of Asthma
DULERA is indicated for the twice-daily treatment of asthma in patients 5 years of age and older. DULERA should be used for patients not adequately controlled on a long-term asthma-control medication such as an inhaled corticosteroid (ICS) or whose disease warrants initiation of treatment with both an ICS and long-acting beta2-adrenergic agonist (LABA).
7.3 Xanthine Derivatives
Concomitant treatment with xanthine derivatives may potentiate any hypokalemic effect of formoterol, a component of DULERA.
16.2 Storage and Handling
Only use the DULERA canister with the DULERA actuator. Do not use the DULERA actuator with any other inhalation drug product. Do not use actuators from other products with the DULERA canister.
Do not remove the canister from the actuator because the correct amount of medication may not be discharged; the dose counter may not function properly; reinsertion may cause the dose counter to count down by 1 and discharge a puff.
The correct amount of medication in each inhalation cannot be ensured after the labeled number of actuations from the canister has been used, even though the inhaler may not feel completely empty and may continue to operate. Discard the inhaler when the labeled number of actuations has been used (the dose counter will read "0").
Store at controlled room temperature 20°C–25°C (68°F–77°F); excursions permitted to 15°C–30°C (59°F–86°F) [see USP Controlled Room Temperature].
After priming, store the inhaler with the mouthpiece down or in a horizontal position.
For best results, keep the canister at room temperature before use. Shake well and remove the cap from the mouthpiece of the actuator before using. Keep out of reach of children. Avoid spraying in eyes.
Contents Under Pressure: Do not puncture. Do not use or store near heat or open flame. Exposure to temperatures above 120°F may cause bursting. Never throw container into fire or incinerator.
5 Warnings and Precautions
- LABA monotherapy increases the risk of serious asthma-related events. (5.1)
- Deterioration of disease and acute episodes: Do not initiate in acutely deteriorating asthma or to treat acute symptoms. (5.2)
- Use with additional long-acting beta2-agonist: Do not use in combination because of risk of overdose. (5.3)
- Localized infections: Candida albicans infection of the mouth and throat may occur. Monitor patients periodically for signs of adverse effects on the oral cavity. After dosing, advise patients to rinse their mouth with water and spit out contents without swallowing. (5.4)
- Immunosuppression: Potential worsening of existing tuberculosis, fungal, bacterial, viral, or parasitic infection; or ocular herpes simplex infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. Use with caution in patients with these infections because of the potential for worsening of these infections. (5.5)
- Transferring patients from systemic corticosteroids: Risk of impaired adrenal function when transferring from oral steroids. Taper patients slowly from systemic corticosteroids if transferring to DULERA. (5.6)
- Hypercorticism and adrenal suppression: May occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue DULERA slowly. (5.7)
- Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir): Risk of increased systemic corticosteroid effects. Exercise caution when used with DULERA. (5.8)
- Paradoxical bronchospasm: Discontinue DULERA and institute alternative therapy if paradoxical bronchospasm occurs. (5.9)
- Patients with cardiovascular disorders: Use with caution because of beta-adrenergic stimulation. (5.11)
- Decreases in bone mineral density: Monitor patients with major risk factors for decreased bone mineral content. (5.12)
- Effects on growth: Monitor growth of pediatric patients. (5.13)
- Glaucoma and cataracts: Consider referral to an ophthalmologist in patients who develop ocular symptoms or use DULERA long term. (5.14)
- Coexisting conditions: Use with caution in patients with aneurysm, pheochromocytoma, convulsive disorders, thyrotoxicosis, diabetes mellitus, and ketoacidosis. (5.15)
- Hypokalemia and hyperglycemia: Be alert to hypokalemia and hyperglycemia. (5.16)
5.15 Coexisting Conditions
DULERA, like other medications containing sympathomimetic amines, should be used with caution in patients with aneurysm, pheochromocytoma, convulsive disorders, or thyrotoxicosis; and in patients who are unusually responsive to sympathomimetic amines. Doses of the related beta2-agonist albuterol, when administered intravenously, have been reported to aggravate preexisting diabetes mellitus and ketoacidosis.
2 Dosage and Administration
For oral inhalation only. (2.1)
Treatment of asthma in patients 12 years of age and older: 2 inhalations twice daily of DULERA 100 mcg/5 mcg or 200 mcg/5 mcg. Starting dosage is based on disease severity. (2.2)
Treatment of asthma in patients aged 5 to less than 12 years: 2 inhalations twice daily of DULERA 50 mcg/5 mcg. (2.2)
5.14 Glaucoma and Cataracts
Glaucoma, increased intraocular pressure, and cataracts have been reported following the use of long-term administration of inhaled corticosteroids, including mometasone furoate, a component of DULERA. Consider referral to an ophthalmologist in patients who develop ocular symptoms or use DULERA long term [see Adverse Reactions (6)].
3 Dosage Forms and Strengths
DULERA is a pressurized metered dose inhaler (MDI) that is available in 2 strengths (100 mcg/5 mcg or 200 mcg/5 mcg) for adult and adolescent patients aged 12 and older and 1 strength (50 mcg/5 mcg) for pediatric patients aged 5 to less than 12 years.
DULERA 50 mcg/5 mcg delivers 50 mcg of mometasone furoate and 5 mcg of formoterol fumarate dihydrate per actuation.
DULERA 100 mcg/5 mcg delivers 100 mcg of mometasone furoate and 5 mcg of formoterol fumarate dihydrate per actuation.
DULERA 200 mcg/5 mcg delivers 200 mcg of mometasone furoate and 5 mcg of formoterol fumarate dihydrate per actuation.
Each strength of DULERA is supplied with a blue colored actuator and green dust cap [see How Supplied/Storage and Handling (16.1)].
6.2 Postmarketing Experience
The following adverse reactions have been reported during post-approval use of DULERA or post-approval use with inhaled mometasone furoate or inhaled formoterol fumarate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac disorders: angina pectoris, cardiac arrhythmias, e.g., atrial fibrillation, ventricular extrasystoles, tachyarrhythmia
Eye disorders: vision blurred [see Warnings and Precautions (5.14)]
Immune system disorders: immediate and delayed hypersensitivity reactions including anaphylactic reaction, angioedema, severe hypotension, rash, pruritus
Investigations: electrocardiogram QT prolonged, blood pressure increased (including hypertension)
Metabolism and nutrition disorders: hypokalemia, hyperglycemia
Respiratory, thoracic and mediastinal disorders: asthma aggravation, which may include cough, dyspnea, wheezing and bronchospasm
7.7 Halogenated Hydrocarbons
There is an elevated risk of arrhythmias in patients receiving concomitant anesthesia with halogenated hydrocarbons.
8 Use in Specific Populations
- Hepatic impairment: Monitor patients for signs of increased drug exposure. (8.6)
2.1 Administration Information
Administer DULERA as two inhalations twice daily every day (morning and evening) by the orally inhaled route (see Patient Instructions for Use in the Patient Information leaflet). Do not use more than two inhalations twice daily of the prescribed strength of DULERA as some patients are more likely to experience adverse effects with higher doses of formoterol. If symptoms arise between doses, an inhaled short-acting beta2-agonist should be taken for immediate relief. Shake well prior to each inhalation. After each dose, advise patients to rinse their mouth with water and, without swallowing, spit out the contents to help reduce the risk of oropharyngeal candidiasis.
Remove the cap from the mouthpiece of the actuator before using DULERA.
Prime DULERA before using for the first time by releasing 4 test sprays into the air, away from the face, shaking well before each spray. In cases where the inhaler has not been used for more than 5 days, prime the inhaler again by releasing 4 test sprays into the air, away from the face, shaking well before each spray.
Only use the DULERA canister with the DULERA actuator. Do not use the DULERA actuator with any other inhalation drug product. Do not use actuators from other products with the DULERA canister.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
5.16 Hypokalemia and Hyperglycemia
Beta2-agonist medications may produce significant hypokalemia in some patients, possibly through intracellular shunting, which has the potential to produce adverse cardiovascular effects. The decrease in serum potassium is usually transient, not requiring supplementation. Clinically significant changes in blood glucose and/or serum potassium were seen infrequently during clinical studies with DULERA at recommended doses.
7.1 Inhibitors of Cytochrome P450 3a4
The main route of metabolism of corticosteroids, including mometasone furoate, a component of DULERA, is via cytochrome P450 (CYP) isoenzyme 3A4 (CYP3A4). After oral administration of ketoconazole, a strong inhibitor of CYP3A4, the mean plasma concentration of orally inhaled mometasone furoate increased. Concomitant administration of CYP3A4 inhibitors may inhibit the metabolism of, and increase the systemic exposure to, mometasone furoate and potentially increase the risk for systemic corticosteroid side effects. Caution should be exercised when considering the coadministration of DULERA with long-term ketoconazole and other known strong CYP3A4 inhibitors (e.g., ritonavir, cobicistat-containing products, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, telithromycin) [see Warnings and Precautions (5.8) and Clinical Pharmacology (12.3)]. Consider the benefit of coadministration versus the potential risk of systemic corticosteroid effects, in which case patients should be monitored for systemic corticosteroid side effects.
5.12 Reduction in Bone Mineral Density
Decreases in bone mineral density (BMD) have been observed with long-term administration of products containing inhaled corticosteroids, including mometasone furoate, one of the components of DULERA. The clinical significance of small changes in BMD with regard to long-term outcomes, such as fracture, is unknown. Patients with major risk factors for decreased bone mineral content, such as prolonged immobilization, family history of osteoporosis, or chronic use of drugs that can reduce bone mass (e.g., anticonvulsants and corticosteroids) should be monitored and treated with established standards of care.
In a 2-year double-blind study in 103 male and female asthma patients 18 to 50 years of age previously maintained on bronchodilator therapy (Baseline FEV1 85%–88% predicted), treatment with mometasone furoate dry powder inhaler (DPI) 200 mcg twice daily resulted in significant reductions in lumbar spine (LS) BMD at the end of the treatment period compared to placebo. The mean change from Baseline to Endpoint in the lumbar spine BMD was -0.015 (-1.43%) for the mometasone furoate group compared to 0.002 (0.25%) for the placebo group. In another 2-year double-blind study in 87 male and female asthma patients 18 to 50 years of age previously maintained on bronchodilator therapy (Baseline FEV1 82%–83% predicted), treatment with mometasone furoate 400 mcg twice daily demonstrated no statistically significant changes in lumbar spine BMD at the end of the treatment period compared to placebo. The mean change from Baseline to Endpoint in the lumbar spine BMD was -0.018 (-1.57%) for the mometasone furoate group compared to -0.006 (-0.43%) for the placebo group.
7.6 Beta Adrenergic Receptor Antagonists
Beta-adrenergic receptor antagonists (beta-blockers) and formoterol may inhibit the effect of each other when administered concurrently. Beta-blockers not only block the therapeutic effects of beta2-agonists, such as formoterol, a component of DULERA, but may produce severe bronchospasm in patients with asthma. Therefore, patients with asthma should not normally be treated with beta-blockers. However, under certain circumstances, e.g., as prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-blockers in patients with asthma. In this setting, cardioselective beta-blockers could be considered, although they should be administered with caution.
5.10 Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions may occur after administration of DULERA, as demonstrated by cases of urticaria, flushing, allergic dermatitis, and bronchospasm.
5.7 Hypercorticism and Adrenal Suppression
Mometasone furoate, a component of DULERA, will often help control asthma symptoms with less suppression of HPA function than therapeutically equivalent oral doses of prednisone. Since mometasone furoate is absorbed into the circulation and can be systemically active at higher doses, the beneficial effects of DULERA in minimizing HPA dysfunction may be expected only when recommended dosages are not exceeded and individual patients are titrated to the lowest effective dose.
Because of the possibility of systemic absorption of inhaled corticosteroids, patients treated with DULERA should be observed carefully for any evidence of systemic corticosteroid effects. Particular care should be taken in observing patients postoperatively or during periods of stress for evidence of inadequate adrenal response.
It is possible that systemic corticosteroid effects such as hypercorticism and adrenal suppression (including adrenal crisis) may appear in a small number of patients, particularly when mometasone furoate is administered at higher than recommended doses over prolonged periods of time. If such effects occur, the dosage of DULERA should be reduced slowly, consistent with accepted procedures for reducing systemic corticosteroids and for management of asthma symptoms.
5.2 Deterioration of Disease and Acute Episodes
DULERA should not be initiated in patients during rapidly deteriorating or potentially life-threatening episodes of asthma. DULERA has not been studied in patients with acutely deteriorating asthma. The initiation of DULERA in this setting is not appropriate.
Increasing use of inhaled, short-acting beta2-agonists is a marker of deteriorating asthma. In this situation, the patient requires immediate re-evaluation with reassessment of the treatment regimen, giving special consideration to the possible need for replacing the current strength of DULERA with a higher strength, adding additional inhaled corticosteroid, or initiating systemic corticosteroids. Patients should not use more than 2 inhalations twice daily (morning and evening) of DULERA.
DULERA is not indicated for the relief of acute symptoms, i.e., as rescue therapy for the treatment of acute episodes of bronchospasm. An inhaled, short-acting beta2-agonist, not DULERA, should be used to relieve acute symptoms such as shortness of breath.
When beginning treatment with DULERA, patients who have been taking oral or inhaled, short-acting beta2-agonists on a regular basis (e.g., 4 times a day) should be instructed to discontinue the regular use of these drugs.
5.11 Cardiovascular and Central Nervous System Effects
Excessive beta-adrenergic stimulation has been associated with seizures, angina, hypertension or hypotension, tachycardia with rates up to 200 beats/min, arrhythmias, nervousness, headache, tremor, palpitation, nausea, dizziness, fatigue, malaise, and insomnia. Therefore, DULERA should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension.
Formoterol fumarate, a component of DULERA, can produce a clinically significant cardiovascular effect in some patients as measured by pulse rate, blood pressure, and/or symptoms. Although such effects are uncommon after administration of DULERA at recommended doses, if they occur, the drug may need to be discontinued. In addition, beta-agonists have been reported to produce ECG changes, such as flattening of the T wave, prolongation of the QTc interval, and ST segment depression. The clinical significance of these findings is unknown. Fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs.
5.9 Paradoxical Bronchospasm and Upper Airway Symptoms
DULERA may produce inhalation induced bronchospasm with an immediate increase in wheezing after dosing that may be life-threatening. If inhalation induced bronchospasm occurs, it should be treated immediately with an inhaled, short-acting bronchodilator. DULERA should be discontinued immediately and alternative therapy instituted.
Principal Display Panel 50 Mcg/5 Mcg Canister Carton
NDC 78206-125-01
Dulera
®
(mometasone furoate and
formoterol fumarate dihydrate)
Inhalation Aerosol
50 mcg/5 mcg
per actuation
For oral inhalation only
SHAKE WELL BEFORE USING.
Dulera canister to be used with Dulera actuator only.
Rx only
120 Metered Actuations
Net Wt. 13g
5.6 Transferring Patients From Systemic Corticosteroid Therapy
Particular care is needed for patients who are transferred from systemically active corticosteroids to DULERA because deaths due to adrenal insufficiency have occurred in asthmatic patients during and after transfer from systemic corticosteroids to less systemically available inhaled corticosteroids. After withdrawal from systemic corticosteroids, a number of months are required for recovery of hypothalamic-pituitary-adrenal (HPA) function.
Patients who have been previously maintained on 20 mg or more per day of prednisone (or its equivalent) may be most susceptible, particularly when their systemic corticosteroids have been almost completely withdrawn. During this period of HPA suppression, patients may exhibit signs and symptoms of adrenal insufficiency when exposed to trauma, surgery, or infection (particularly gastroenteritis) or other conditions associated with severe electrolyte loss. Although DULERA may improve control of asthma symptoms during these episodes, in recommended doses it supplies less than normal physiological amounts of corticosteroid systemically and does NOT provide the mineralocorticoid activity necessary for coping with these emergencies.
During periods of stress or severe asthma attack, patients who have been withdrawn from systemic corticosteroids should be instructed to resume oral corticosteroids (in large doses) immediately and to contact their physicians for further instruction. These patients should also be instructed to carry a medical identification card indicating that they may need supplementary systemic corticosteroids during periods of stress or severe asthma attack.
Patients requiring systemic corticosteroids should be weaned slowly from systemic corticosteroid use after transferring to DULERA. Lung function (FEV1 or PEF), beta-agonist use, and asthma symptoms should be carefully monitored during withdrawal of systemic corticosteroids. In addition to monitoring asthma signs and symptoms, patients should be observed for signs and symptoms of adrenal insufficiency such as fatigue, lassitude, weakness, nausea and vomiting, and hypotension.
Transfer of patients from systemic corticosteroid therapy to DULERA may unmask allergic conditions previously suppressed by the systemic corticosteroid therapy, e.g., rhinitis, conjunctivitis, eczema, arthritis, and eosinophilic conditions.
During withdrawal from oral corticosteroids, some patients may experience symptoms of systemically active corticosteroid withdrawal, e.g., joint and/or muscular pain, lassitude, and depression, despite maintenance or even improvement of respiratory function.
5.3 Excessive Use of Dulera and Use With Other Long Acting Beta2
As with other inhaled drugs containing beta2-adrenergic agents, DULERA should not be used more often than recommended, at higher doses than recommended, or in conjunction with other medications containing long-acting beta2-agonists, as an overdose may result. Clinically significant cardiovascular effects and fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs. Patients using DULERA should not use an additional long-acting beta2-agonist (e.g., salmeterol, formoterol fumarate, arformoterol tartrate) for any reason, including prevention of exercise-induced bronchospasm (EIB) or the treatment of asthma.
5.8 Drug Interactions With Strong Cytochrome P450 3a4 Inhibitors
Caution should be exercised when considering the coadministration of DULERA with ketoconazole, and other known strong CYP3A4 inhibitors (e.g., ritonavir, cobicistat-containing products, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, telithromycin) because adverse effects related to increased systemic exposure to mometasone furoate may occur [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
5.1 Serious Asthma Related Events – Hospitalizations, Intubations, and Death
Use of LABA as monotherapy (without ICS) for asthma is associated with an increased risk of asthma-related death [see Salmeterol Multicenter Asthma Research Trial (SMART)]. Available data from controlled clinical trials also suggest that use of LABA as monotherapy increases the risk of asthma-related hospitalization in pediatric and adolescent patients. These findings are considered a class effect of LABA monotherapy. When LABA are used in fixed-dose combination with ICS, data from large clinical trials do not show a significant increase in the risk of serious asthma-related events (hospitalizations, intubations, death) compared to ICS alone [see Serious Asthma-Related Events with ICS/LABA].
7.5 Monoamine Oxidase Inhibitors, Tricyclic Antidepressants, and Drugs Known to Prolong the Qtc Interval
DULERA should be administered with caution to patients being treated with monoamine oxidase inhibitors, tricyclic antidepressants, macrolides, or drugs known to prolong the QTc interval or within 2 weeks of discontinuation of such agents, because the action of formoterol, a component of DULERA, on the cardiovascular system may be potentiated by these agents. Drugs that are known to prolong the QTc interval have an increased risk of ventricular arrhythmias.
Structured Label Content
Section 42229-5 (42229-5)
Important Limitation of Use:
- DULERA is NOT indicated for the relief of acute bronchospasm.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: 6/2025 | |||
|
PATIENT INFORMATION
DULERA® [dew-LAIR-ah] 50 mcg/5 mcg (mometasone furoate 50 mcg and formoterol fumarate dihydrate 5 mcg) Inhalation Aerosol DULERA® 100 mcg/5 mcg (mometasone furoate 100 mcg and formoterol fumarate dihydrate 5 mcg) Inhalation Aerosol DULERA® 200 mcg/5 mcg (mometasone furoate 200 mcg and formoterol fumarate dihydrate 5 mcg) Inhalation Aerosol |
||||
| Read the Patient Information leaflet that comes with DULERA ® before you start using it and each time you get a refill. There may be new information. The Patient Information leaflet does not take the place of talking to your healthcare provider about your medical condition or treatment. | ||||
What is DULERA?
|
||||
Do not use DULERA:
|
||||
Before and during treatment with DULERA, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take including prescription and over-the-counter medicines, vitamins, and herbal supplements. DULERA and certain other medicines may interact with each other. This may cause serious side effects. |
||||
|
How should I use DULERA? See the step-by-step instructions for using DULERA at the end of this Patient Information leaflet. Do not use DULERA unless your healthcare provider has taught you and you understand how to use it. Ask your healthcare provider or pharmacist if you have any questions.
|
||||
|
What are the possible side effects of DULERA? DULERA may cause serious side effects, including:
|
||||
|
|
|
||
|
||||
|
|
|||
|
||||
|
|
|||
|
||||
The most common side effects reported while using DULERA include:
These are not all the side effects of DULERA. Ask your healthcare provider or pharmacist for more information. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Organon LLC., a subsidiary of Organon & Co., at 1-844-674-3200. |
||||
How should I store DULERA?
|
||||
|
General Information about the safe and effective use of DULERA.
Medicines are sometimes prescribed for purposes other than those listed in the Patient Information leaflet. Do not use DULERA for a condition for which it was not prescribed. Do not give your DULERA to other people, even if they have the same condition. It may harm them. You can ask your healthcare provider or pharmacist for information about DULERA that was written for healthcare professionals. |
||||
|
What are the ingredients in DULERA?
Active ingredients: mometasone furoate and formoterol fumarate dihydrate Inactive ingredients: hydrofluoroalkane (HFA-227), anhydrous alcohol and oleic acid |
||||
|
For more information about DULERA:
Go to www.DULERA.com or call 1-844-674-3200. |
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL - 100 mcg/5 mcg Canister Carton
NDC 78206-127-01
Dulera
®
(mometasone furoate and
formoterol fumarate dihydrate)
Inhalation Aerosol
100 mcg/5 mcg
per actuation
For oral inhalation only
SHAKE WELL BEFORE USING.
Dulera canister to be used with Dulera actuator only.
Rx only
120 Metered Actuations
Net Wt. 13g
7.4 Diuretics
Concomitant treatment with diuretics may potentiate the possible hypokalemic effect of adrenergic agonists. The ECG changes and/or hypokalemia that may result from the administration of non-potassium-sparing diuretics (such as loop or thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dose of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the coadministration of DULERA with non-potassium-sparing diuretics.
11 Description (11 DESCRIPTION)
DULERA 50 mcg/5 mcg, DULERA 100 mcg/5 mcg, and DULERA 200 mcg/5 mcg are combinations of mometasone furoate and formoterol fumarate dihydrate for oral inhalation only.
One active component of DULERA is mometasone furoate, a corticosteroid having the chemical name 9,21-dichloro-11(Beta),17-dihydroxy-16 (alpha)-methylpregna-1,4-diene-3,20-dione 17-(2-furoate) with the following chemical structure:
Mometasone furoate is a white powder with an empirical formula of C27H30Cl2O6, and molecular weight 521.44. It is practically insoluble in water; slightly soluble in methanol, ethanol, and isopropanol; soluble in acetone.
One active component of DULERA is formoterol fumarate dihydrate, a racemate. Formoterol fumarate dihydrate is a selective beta2-adrenergic bronchodilator having the chemical name of (±)-2-hydroxy-5-[(1RS)-1-hydroxy-2-[[(1RS)-2-(4-methoxyphenyl)-1-methylethyl]-amino]ethyl]formanilide fumarate dihydrate with the following chemical structure:
Formoterol fumarate dihydrate has a molecular weight of 840.9, and its empirical formula is (C19H24N2O4)2•C4H4O4•2H2O. Formoterol fumarate dihydrate is a white to yellowish powder, which is freely soluble in glacial acetic acid, soluble in methanol, sparingly soluble in ethanol and isopropanol, slightly soluble in water, and practically insoluble in acetone, ethyl acetate, and diethyl ether.
DULERA 50 mcg/5 mcg, 100 mcg/5 mcg, and 200 mcg/5 mcg are each formulated as a hydrofluoroalkane (HFA-227; 1, 1, 1, 2, 3, 3, 3-heptafluoropropane) propelled pressurized metered dose inhaler containing sufficient amount of drug for 60 or 120 inhalations [see How Supplied/Storage and Handling (16)]. After priming, each actuation of the inhaler delivers 60, 115, or 225 mcg of mometasone furoate and 5.5 mcg of formoterol fumarate dihydrate in 69.6 mg of suspension from the valve and delivers 50, 100, or 200 mcg of mometasone furoate and 5 mcg of formoterol fumarate dihydrate from the actuator. The actual amount of drug delivered to the lung may depend on patient factors, such as the coordination between actuation of the device and inspiration through the delivery system. DULERA also contains anhydrous alcohol as a cosolvent and oleic acid as a surfactant.
DULERA should be primed before using for the first time by releasing 4 test sprays into the air, away from the face, shaking well before each spray. In cases where the inhaler has not been used for more than 5 days, prime the inhaler again by releasing 4 test sprays into the air, away from the face, shaking well before each spray.
16.1 How Supplied
DULERA is available in three strengths and supplied in the following package sizes (Table 8):
| Package | NDC | Strength Identifier (Color Band) Included on the outer carton, actuator, and canister labels.
|
|---|---|---|
| DULERA 50 mcg/5 mcg 120 inhalations |
78206-125-01 | Blue |
| DULERA 100 mcg/5 mcg 120 inhalations |
78206-127-01 | Yellow |
| DULERA 100 mcg/5 mcg 60 inhalations (institutional pack) |
78206-127-02 | Yellow |
| DULERA 200 mcg/5 mcg 120 inhalations |
78206-126-01 | Purple |
| DULERA 200 mcg/5 mcg 60 inhalations (institutional pack) |
78206-126-02 | Purple |
Each strength is supplied as a pressurized aluminum canister that has a blue plastic actuator integrated with a dose counter and a green dust cap. Each 120-inhalation canister has a net fill weight of 13 grams and each 60-inhalation canister has a net fill weight of 8.8 grams. Each canister is placed into a carton. Each carton contains 1 canister and a Patient Information leaflet.
Initially the dose counter will display "64" or "124" actuations. After the initial priming with 4 actuations, the dose counter will read "60" or "120" and the inhaler is now ready for use.
5.4 Local Effects
In clinical trials, the development of localized infections of the mouth and pharynx with Candida albicans have occurred in patients treated with DULERA. If oropharyngeal candidiasis develops, treat with appropriate local or systemic (i.e., oral) antifungal therapy while remaining on treatment with DULERA therapy, but at times therapy with DULERA may need to be interrupted. To reduce the risk of oropharyngeal candidiasis, after dosing with DULERA, advise patients to rinse their mouth with water and spit out the contents without swallowing.
8.4 Pediatric Use
The safety and effectiveness of DULERA have been established in patients 12 years of age and older in 3 clinical trials up to 52 weeks in duration. In the 3 clinical trials, 101 patients 12 to 17 years of age were treated with DULERA. Patients in this age-group demonstrated efficacy results similar to those observed in patients 18 years of age and older. There were no obvious differences in the type or frequency of adverse reactions reported in this age group compared to patients 18 years of age and older. Similar efficacy and safety results were observed in an additional 22 patients 12 to 17 years of age who were treated with DULERA in another clinical trial.
The safety and effectiveness of DULERA 50 mcg/5 mcg, two inhalations twice daily, have been established in patients with asthma aged 5 to less than 12 years in clinical trials up to 24 weeks of treatment duration. Patients in this age group demonstrated efficacy and safety results similar to those observed in patients aged 12 years and older who were treated with DULERA [see Adverse Reactions (6.1) and Clinical Studies (14.1)].
The safety and effectiveness of DULERA have not been established in children younger than 5 years of age.
Controlled clinical studies have shown that inhaled corticosteroids may cause a reduction in growth velocity in pediatric patients. In these studies, the mean reduction in growth velocity was approximately 1 cm per year (range 0.3 to 1.8 per year) and appears to depend upon dose and duration of exposure. This effect was observed in the absence of laboratory evidence of hypothalamic-pituitary-adrenal (HPA) axis suppression, suggesting that growth velocity is a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA axis function. The long-term effects of this reduction in growth velocity associated with orally inhaled corticosteroids, including the impact on final adult height, are unknown. The potential for "catch up" growth following discontinuation of treatment with orally inhaled corticosteroids has not been adequately studied.
The growth of children and adolescents receiving orally inhaled corticosteroids, including DULERA, should be monitored routinely (e.g., via stadiometry). If a child or adolescent on any corticosteroid appears to have growth suppression, the possibility that he/she is particularly sensitive to this effect should be considered. The potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the risks associated with alternative therapies. To minimize the systemic effects of orally inhaled corticosteroids, including DULERA, each patient should be titrated to his/her lowest effective dose [see Dosage and Administration (2.2)].
8.5 Geriatric Use
A total of 77 patients 65 years of age and older (11 of whom were 75 years and older) have been treated with DULERA in 3 clinical trials up to 52 weeks in duration. Similar efficacy and safety results were observed in an additional 28 patients 65 years of age and older who were treated with DULERA in another clinical trial. No overall differences in safety or effectiveness were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out. As with other products containing beta2-agonists, special caution should be observed when using DULERA in geriatric patients who have concomitant cardiovascular disease that could be adversely affected by beta2-agonists. Based on available data for DULERA or its active components, no adjustment of dosage of DULERA in geriatric patients is warranted.
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
LABA use may result in the following:
- Serious asthma-related events – hospitalizations, intubations, and death [see Warnings and Precautions (5.1)].
- Cardiovascular and central nervous system effects [see Warnings and Precautions (5.11)].
Systemic and local corticosteroid use may result in the following:
- Candida albicans infection [see Warnings and Precautions (5.4)]
- Immunosuppression [see Warnings and Precautions (5.5)]
- Hypercorticism and adrenal suppression [see Warnings and Precautions (5.7)]
- Growth effects in pediatrics [see Warnings and Precautions (5.13)]
- Glaucoma and cataracts [see Warnings and Precautions (5.14)]
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7 Drug Interactions (7 DRUG INTERACTIONS)
In clinical trials, concurrent administration of DULERA and other drugs, such as short-acting beta2-agonist and intranasal corticosteroids have not resulted in an increased frequency of adverse drug reactions. No formal drug interaction studies have been performed with DULERA. The drug interactions of the combination are expected to reflect those of the individual components.
4.2 Hypersensitivity
DULERA is contraindicated in patients with known hypersensitivity to mometasone furoate, formoterol fumarate, or any of the ingredients in DULERA [see Warnings and Precautions (5.10)].
5.13 Effect On Growth (5.13 Effect on Growth)
Orally inhaled corticosteroids, including DULERA, may cause a reduction in growth velocity when administered to pediatric patients. Monitor the growth of pediatric patients receiving DULERA routinely (e.g., via stadiometry). To minimize the systemic effects of orally inhaled corticosteroids, including DULERA, titrate each patient's dose to the lowest dosage that effectively controls his/her symptoms [see Use in Specific Populations (8.4)].
5.5 Immunosuppression
Persons who are using drugs that suppress the immune system are more susceptible to infections than healthy individuals.
Chickenpox and measles, for example, can have a more serious or even fatal course in susceptible children or adults using corticosteroids. In such children or adults who have not had these diseases or who are not properly immunized, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) or pooled intravenous immunoglobulin (IVIG) may be indicated. If exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the respective package inserts for complete VZIG and IG prescribing information.) If chickenpox develops, treatment with antiviral agents may be considered.
DULERA should be used with caution, if at all, in patients with active or quiescent tuberculosis infection of the respiratory tract, untreated systemic fungal, bacterial, viral, or parasitic infections; or ocular herpes simplex.
7.2 Adrenergic Agents
If additional adrenergic drugs are to be administered by any route, they should be used with caution because the pharmacologically predictable sympathetic effects of formoterol, a component of DULERA, may be potentiated.
2.2 Recommended Dosage
Administer DULERA as two inhalations twice daily every day (morning and evening) by the orally inhaled route. Shake well prior to each inhalation. Individual patients may experience a variable time to onset and degree of symptom relief. If symptoms arise between doses, use an inhaled short-acting beta2-agonist for immediate relief. Improvement in lung function following administration of DULERA can occur within 5 minutes of treatment, although the maximum benefit may not be achieved for 1 week or longer after beginning treatment.
4.1 Status Asthmaticus
DULERA is contraindicated in the primary treatment of status asthmaticus or other acute episodes of asthma where intensive measures are required.
8.6 Hepatic Impairment
Concentrations of mometasone furoate appear to increase with severity of hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
1.1 Treatment of Asthma
DULERA is indicated for the twice-daily treatment of asthma in patients 5 years of age and older. DULERA should be used for patients not adequately controlled on a long-term asthma-control medication such as an inhaled corticosteroid (ICS) or whose disease warrants initiation of treatment with both an ICS and long-acting beta2-adrenergic agonist (LABA).
7.3 Xanthine Derivatives
Concomitant treatment with xanthine derivatives may potentiate any hypokalemic effect of formoterol, a component of DULERA.
16.2 Storage and Handling
Only use the DULERA canister with the DULERA actuator. Do not use the DULERA actuator with any other inhalation drug product. Do not use actuators from other products with the DULERA canister.
Do not remove the canister from the actuator because the correct amount of medication may not be discharged; the dose counter may not function properly; reinsertion may cause the dose counter to count down by 1 and discharge a puff.
The correct amount of medication in each inhalation cannot be ensured after the labeled number of actuations from the canister has been used, even though the inhaler may not feel completely empty and may continue to operate. Discard the inhaler when the labeled number of actuations has been used (the dose counter will read "0").
Store at controlled room temperature 20°C–25°C (68°F–77°F); excursions permitted to 15°C–30°C (59°F–86°F) [see USP Controlled Room Temperature].
After priming, store the inhaler with the mouthpiece down or in a horizontal position.
For best results, keep the canister at room temperature before use. Shake well and remove the cap from the mouthpiece of the actuator before using. Keep out of reach of children. Avoid spraying in eyes.
Contents Under Pressure: Do not puncture. Do not use or store near heat or open flame. Exposure to temperatures above 120°F may cause bursting. Never throw container into fire or incinerator.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- LABA monotherapy increases the risk of serious asthma-related events. (5.1)
- Deterioration of disease and acute episodes: Do not initiate in acutely deteriorating asthma or to treat acute symptoms. (5.2)
- Use with additional long-acting beta2-agonist: Do not use in combination because of risk of overdose. (5.3)
- Localized infections: Candida albicans infection of the mouth and throat may occur. Monitor patients periodically for signs of adverse effects on the oral cavity. After dosing, advise patients to rinse their mouth with water and spit out contents without swallowing. (5.4)
- Immunosuppression: Potential worsening of existing tuberculosis, fungal, bacterial, viral, or parasitic infection; or ocular herpes simplex infections. More serious or even fatal course of chickenpox or measles can occur in susceptible patients. Use with caution in patients with these infections because of the potential for worsening of these infections. (5.5)
- Transferring patients from systemic corticosteroids: Risk of impaired adrenal function when transferring from oral steroids. Taper patients slowly from systemic corticosteroids if transferring to DULERA. (5.6)
- Hypercorticism and adrenal suppression: May occur with very high dosages or at the regular dosage in susceptible individuals. If such changes occur, discontinue DULERA slowly. (5.7)
- Strong cytochrome P450 3A4 inhibitors (e.g., ritonavir): Risk of increased systemic corticosteroid effects. Exercise caution when used with DULERA. (5.8)
- Paradoxical bronchospasm: Discontinue DULERA and institute alternative therapy if paradoxical bronchospasm occurs. (5.9)
- Patients with cardiovascular disorders: Use with caution because of beta-adrenergic stimulation. (5.11)
- Decreases in bone mineral density: Monitor patients with major risk factors for decreased bone mineral content. (5.12)
- Effects on growth: Monitor growth of pediatric patients. (5.13)
- Glaucoma and cataracts: Consider referral to an ophthalmologist in patients who develop ocular symptoms or use DULERA long term. (5.14)
- Coexisting conditions: Use with caution in patients with aneurysm, pheochromocytoma, convulsive disorders, thyrotoxicosis, diabetes mellitus, and ketoacidosis. (5.15)
- Hypokalemia and hyperglycemia: Be alert to hypokalemia and hyperglycemia. (5.16)
5.15 Coexisting Conditions
DULERA, like other medications containing sympathomimetic amines, should be used with caution in patients with aneurysm, pheochromocytoma, convulsive disorders, or thyrotoxicosis; and in patients who are unusually responsive to sympathomimetic amines. Doses of the related beta2-agonist albuterol, when administered intravenously, have been reported to aggravate preexisting diabetes mellitus and ketoacidosis.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
For oral inhalation only. (2.1)
Treatment of asthma in patients 12 years of age and older: 2 inhalations twice daily of DULERA 100 mcg/5 mcg or 200 mcg/5 mcg. Starting dosage is based on disease severity. (2.2)
Treatment of asthma in patients aged 5 to less than 12 years: 2 inhalations twice daily of DULERA 50 mcg/5 mcg. (2.2)
5.14 Glaucoma and Cataracts
Glaucoma, increased intraocular pressure, and cataracts have been reported following the use of long-term administration of inhaled corticosteroids, including mometasone furoate, a component of DULERA. Consider referral to an ophthalmologist in patients who develop ocular symptoms or use DULERA long term [see Adverse Reactions (6)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
DULERA is a pressurized metered dose inhaler (MDI) that is available in 2 strengths (100 mcg/5 mcg or 200 mcg/5 mcg) for adult and adolescent patients aged 12 and older and 1 strength (50 mcg/5 mcg) for pediatric patients aged 5 to less than 12 years.
DULERA 50 mcg/5 mcg delivers 50 mcg of mometasone furoate and 5 mcg of formoterol fumarate dihydrate per actuation.
DULERA 100 mcg/5 mcg delivers 100 mcg of mometasone furoate and 5 mcg of formoterol fumarate dihydrate per actuation.
DULERA 200 mcg/5 mcg delivers 200 mcg of mometasone furoate and 5 mcg of formoterol fumarate dihydrate per actuation.
Each strength of DULERA is supplied with a blue colored actuator and green dust cap [see How Supplied/Storage and Handling (16.1)].
6.2 Postmarketing Experience
The following adverse reactions have been reported during post-approval use of DULERA or post-approval use with inhaled mometasone furoate or inhaled formoterol fumarate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac disorders: angina pectoris, cardiac arrhythmias, e.g., atrial fibrillation, ventricular extrasystoles, tachyarrhythmia
Eye disorders: vision blurred [see Warnings and Precautions (5.14)]
Immune system disorders: immediate and delayed hypersensitivity reactions including anaphylactic reaction, angioedema, severe hypotension, rash, pruritus
Investigations: electrocardiogram QT prolonged, blood pressure increased (including hypertension)
Metabolism and nutrition disorders: hypokalemia, hyperglycemia
Respiratory, thoracic and mediastinal disorders: asthma aggravation, which may include cough, dyspnea, wheezing and bronchospasm
7.7 Halogenated Hydrocarbons
There is an elevated risk of arrhythmias in patients receiving concomitant anesthesia with halogenated hydrocarbons.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Hepatic impairment: Monitor patients for signs of increased drug exposure. (8.6)
2.1 Administration Information
Administer DULERA as two inhalations twice daily every day (morning and evening) by the orally inhaled route (see Patient Instructions for Use in the Patient Information leaflet). Do not use more than two inhalations twice daily of the prescribed strength of DULERA as some patients are more likely to experience adverse effects with higher doses of formoterol. If symptoms arise between doses, an inhaled short-acting beta2-agonist should be taken for immediate relief. Shake well prior to each inhalation. After each dose, advise patients to rinse their mouth with water and, without swallowing, spit out the contents to help reduce the risk of oropharyngeal candidiasis.
Remove the cap from the mouthpiece of the actuator before using DULERA.
Prime DULERA before using for the first time by releasing 4 test sprays into the air, away from the face, shaking well before each spray. In cases where the inhaler has not been used for more than 5 days, prime the inhaler again by releasing 4 test sprays into the air, away from the face, shaking well before each spray.
Only use the DULERA canister with the DULERA actuator. Do not use the DULERA actuator with any other inhalation drug product. Do not use actuators from other products with the DULERA canister.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
5.16 Hypokalemia and Hyperglycemia
Beta2-agonist medications may produce significant hypokalemia in some patients, possibly through intracellular shunting, which has the potential to produce adverse cardiovascular effects. The decrease in serum potassium is usually transient, not requiring supplementation. Clinically significant changes in blood glucose and/or serum potassium were seen infrequently during clinical studies with DULERA at recommended doses.
7.1 Inhibitors of Cytochrome P450 3a4 (7.1 Inhibitors of Cytochrome P450 3A4)
The main route of metabolism of corticosteroids, including mometasone furoate, a component of DULERA, is via cytochrome P450 (CYP) isoenzyme 3A4 (CYP3A4). After oral administration of ketoconazole, a strong inhibitor of CYP3A4, the mean plasma concentration of orally inhaled mometasone furoate increased. Concomitant administration of CYP3A4 inhibitors may inhibit the metabolism of, and increase the systemic exposure to, mometasone furoate and potentially increase the risk for systemic corticosteroid side effects. Caution should be exercised when considering the coadministration of DULERA with long-term ketoconazole and other known strong CYP3A4 inhibitors (e.g., ritonavir, cobicistat-containing products, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, telithromycin) [see Warnings and Precautions (5.8) and Clinical Pharmacology (12.3)]. Consider the benefit of coadministration versus the potential risk of systemic corticosteroid effects, in which case patients should be monitored for systemic corticosteroid side effects.
5.12 Reduction in Bone Mineral Density
Decreases in bone mineral density (BMD) have been observed with long-term administration of products containing inhaled corticosteroids, including mometasone furoate, one of the components of DULERA. The clinical significance of small changes in BMD with regard to long-term outcomes, such as fracture, is unknown. Patients with major risk factors for decreased bone mineral content, such as prolonged immobilization, family history of osteoporosis, or chronic use of drugs that can reduce bone mass (e.g., anticonvulsants and corticosteroids) should be monitored and treated with established standards of care.
In a 2-year double-blind study in 103 male and female asthma patients 18 to 50 years of age previously maintained on bronchodilator therapy (Baseline FEV1 85%–88% predicted), treatment with mometasone furoate dry powder inhaler (DPI) 200 mcg twice daily resulted in significant reductions in lumbar spine (LS) BMD at the end of the treatment period compared to placebo. The mean change from Baseline to Endpoint in the lumbar spine BMD was -0.015 (-1.43%) for the mometasone furoate group compared to 0.002 (0.25%) for the placebo group. In another 2-year double-blind study in 87 male and female asthma patients 18 to 50 years of age previously maintained on bronchodilator therapy (Baseline FEV1 82%–83% predicted), treatment with mometasone furoate 400 mcg twice daily demonstrated no statistically significant changes in lumbar spine BMD at the end of the treatment period compared to placebo. The mean change from Baseline to Endpoint in the lumbar spine BMD was -0.018 (-1.57%) for the mometasone furoate group compared to -0.006 (-0.43%) for the placebo group.
7.6 Beta Adrenergic Receptor Antagonists (7.6 Beta-Adrenergic Receptor Antagonists)
Beta-adrenergic receptor antagonists (beta-blockers) and formoterol may inhibit the effect of each other when administered concurrently. Beta-blockers not only block the therapeutic effects of beta2-agonists, such as formoterol, a component of DULERA, but may produce severe bronchospasm in patients with asthma. Therefore, patients with asthma should not normally be treated with beta-blockers. However, under certain circumstances, e.g., as prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-blockers in patients with asthma. In this setting, cardioselective beta-blockers could be considered, although they should be administered with caution.
5.10 Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions may occur after administration of DULERA, as demonstrated by cases of urticaria, flushing, allergic dermatitis, and bronchospasm.
5.7 Hypercorticism and Adrenal Suppression
Mometasone furoate, a component of DULERA, will often help control asthma symptoms with less suppression of HPA function than therapeutically equivalent oral doses of prednisone. Since mometasone furoate is absorbed into the circulation and can be systemically active at higher doses, the beneficial effects of DULERA in minimizing HPA dysfunction may be expected only when recommended dosages are not exceeded and individual patients are titrated to the lowest effective dose.
Because of the possibility of systemic absorption of inhaled corticosteroids, patients treated with DULERA should be observed carefully for any evidence of systemic corticosteroid effects. Particular care should be taken in observing patients postoperatively or during periods of stress for evidence of inadequate adrenal response.
It is possible that systemic corticosteroid effects such as hypercorticism and adrenal suppression (including adrenal crisis) may appear in a small number of patients, particularly when mometasone furoate is administered at higher than recommended doses over prolonged periods of time. If such effects occur, the dosage of DULERA should be reduced slowly, consistent with accepted procedures for reducing systemic corticosteroids and for management of asthma symptoms.
5.2 Deterioration of Disease and Acute Episodes
DULERA should not be initiated in patients during rapidly deteriorating or potentially life-threatening episodes of asthma. DULERA has not been studied in patients with acutely deteriorating asthma. The initiation of DULERA in this setting is not appropriate.
Increasing use of inhaled, short-acting beta2-agonists is a marker of deteriorating asthma. In this situation, the patient requires immediate re-evaluation with reassessment of the treatment regimen, giving special consideration to the possible need for replacing the current strength of DULERA with a higher strength, adding additional inhaled corticosteroid, or initiating systemic corticosteroids. Patients should not use more than 2 inhalations twice daily (morning and evening) of DULERA.
DULERA is not indicated for the relief of acute symptoms, i.e., as rescue therapy for the treatment of acute episodes of bronchospasm. An inhaled, short-acting beta2-agonist, not DULERA, should be used to relieve acute symptoms such as shortness of breath.
When beginning treatment with DULERA, patients who have been taking oral or inhaled, short-acting beta2-agonists on a regular basis (e.g., 4 times a day) should be instructed to discontinue the regular use of these drugs.
5.11 Cardiovascular and Central Nervous System Effects
Excessive beta-adrenergic stimulation has been associated with seizures, angina, hypertension or hypotension, tachycardia with rates up to 200 beats/min, arrhythmias, nervousness, headache, tremor, palpitation, nausea, dizziness, fatigue, malaise, and insomnia. Therefore, DULERA should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension.
Formoterol fumarate, a component of DULERA, can produce a clinically significant cardiovascular effect in some patients as measured by pulse rate, blood pressure, and/or symptoms. Although such effects are uncommon after administration of DULERA at recommended doses, if they occur, the drug may need to be discontinued. In addition, beta-agonists have been reported to produce ECG changes, such as flattening of the T wave, prolongation of the QTc interval, and ST segment depression. The clinical significance of these findings is unknown. Fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs.
5.9 Paradoxical Bronchospasm and Upper Airway Symptoms
DULERA may produce inhalation induced bronchospasm with an immediate increase in wheezing after dosing that may be life-threatening. If inhalation induced bronchospasm occurs, it should be treated immediately with an inhaled, short-acting bronchodilator. DULERA should be discontinued immediately and alternative therapy instituted.
Principal Display Panel 50 Mcg/5 Mcg Canister Carton (PRINCIPAL DISPLAY PANEL - 50 mcg/5 mcg Canister Carton)
NDC 78206-125-01
Dulera
®
(mometasone furoate and
formoterol fumarate dihydrate)
Inhalation Aerosol
50 mcg/5 mcg
per actuation
For oral inhalation only
SHAKE WELL BEFORE USING.
Dulera canister to be used with Dulera actuator only.
Rx only
120 Metered Actuations
Net Wt. 13g
5.6 Transferring Patients From Systemic Corticosteroid Therapy (5.6 Transferring Patients from Systemic Corticosteroid Therapy)
Particular care is needed for patients who are transferred from systemically active corticosteroids to DULERA because deaths due to adrenal insufficiency have occurred in asthmatic patients during and after transfer from systemic corticosteroids to less systemically available inhaled corticosteroids. After withdrawal from systemic corticosteroids, a number of months are required for recovery of hypothalamic-pituitary-adrenal (HPA) function.
Patients who have been previously maintained on 20 mg or more per day of prednisone (or its equivalent) may be most susceptible, particularly when their systemic corticosteroids have been almost completely withdrawn. During this period of HPA suppression, patients may exhibit signs and symptoms of adrenal insufficiency when exposed to trauma, surgery, or infection (particularly gastroenteritis) or other conditions associated with severe electrolyte loss. Although DULERA may improve control of asthma symptoms during these episodes, in recommended doses it supplies less than normal physiological amounts of corticosteroid systemically and does NOT provide the mineralocorticoid activity necessary for coping with these emergencies.
During periods of stress or severe asthma attack, patients who have been withdrawn from systemic corticosteroids should be instructed to resume oral corticosteroids (in large doses) immediately and to contact their physicians for further instruction. These patients should also be instructed to carry a medical identification card indicating that they may need supplementary systemic corticosteroids during periods of stress or severe asthma attack.
Patients requiring systemic corticosteroids should be weaned slowly from systemic corticosteroid use after transferring to DULERA. Lung function (FEV1 or PEF), beta-agonist use, and asthma symptoms should be carefully monitored during withdrawal of systemic corticosteroids. In addition to monitoring asthma signs and symptoms, patients should be observed for signs and symptoms of adrenal insufficiency such as fatigue, lassitude, weakness, nausea and vomiting, and hypotension.
Transfer of patients from systemic corticosteroid therapy to DULERA may unmask allergic conditions previously suppressed by the systemic corticosteroid therapy, e.g., rhinitis, conjunctivitis, eczema, arthritis, and eosinophilic conditions.
During withdrawal from oral corticosteroids, some patients may experience symptoms of systemically active corticosteroid withdrawal, e.g., joint and/or muscular pain, lassitude, and depression, despite maintenance or even improvement of respiratory function.
5.3 Excessive Use of Dulera and Use With Other Long Acting Beta2 (5.3 Excessive Use of DULERA and Use with Other Long-Acting Beta2)
As with other inhaled drugs containing beta2-adrenergic agents, DULERA should not be used more often than recommended, at higher doses than recommended, or in conjunction with other medications containing long-acting beta2-agonists, as an overdose may result. Clinically significant cardiovascular effects and fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs. Patients using DULERA should not use an additional long-acting beta2-agonist (e.g., salmeterol, formoterol fumarate, arformoterol tartrate) for any reason, including prevention of exercise-induced bronchospasm (EIB) or the treatment of asthma.
5.8 Drug Interactions With Strong Cytochrome P450 3a4 Inhibitors (5.8 Drug Interactions with Strong Cytochrome P450 3A4 Inhibitors)
Caution should be exercised when considering the coadministration of DULERA with ketoconazole, and other known strong CYP3A4 inhibitors (e.g., ritonavir, cobicistat-containing products, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, telithromycin) because adverse effects related to increased systemic exposure to mometasone furoate may occur [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
5.1 Serious Asthma Related Events – Hospitalizations, Intubations, and Death (5.1 Serious Asthma-Related Events – Hospitalizations, Intubations, and Death)
Use of LABA as monotherapy (without ICS) for asthma is associated with an increased risk of asthma-related death [see Salmeterol Multicenter Asthma Research Trial (SMART)]. Available data from controlled clinical trials also suggest that use of LABA as monotherapy increases the risk of asthma-related hospitalization in pediatric and adolescent patients. These findings are considered a class effect of LABA monotherapy. When LABA are used in fixed-dose combination with ICS, data from large clinical trials do not show a significant increase in the risk of serious asthma-related events (hospitalizations, intubations, death) compared to ICS alone [see Serious Asthma-Related Events with ICS/LABA].
7.5 Monoamine Oxidase Inhibitors, Tricyclic Antidepressants, and Drugs Known to Prolong the Qtc Interval (7.5 Monoamine Oxidase Inhibitors, Tricyclic Antidepressants, and Drugs Known to Prolong the QTc Interval)
DULERA should be administered with caution to patients being treated with monoamine oxidase inhibitors, tricyclic antidepressants, macrolides, or drugs known to prolong the QTc interval or within 2 weeks of discontinuation of such agents, because the action of formoterol, a component of DULERA, on the cardiovascular system may be potentiated by these agents. Drugs that are known to prolong the QTc interval have an increased risk of ventricular arrhythmias.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:53:00.413909 · Updated: 2026-03-14T22:45:20.597363