Methyl Salicylate 25% Cream
8f0ba6e7-8aa4-443e-99ae-a255085ddfec
34390-5
HUMAN OTC DRUG LABEL
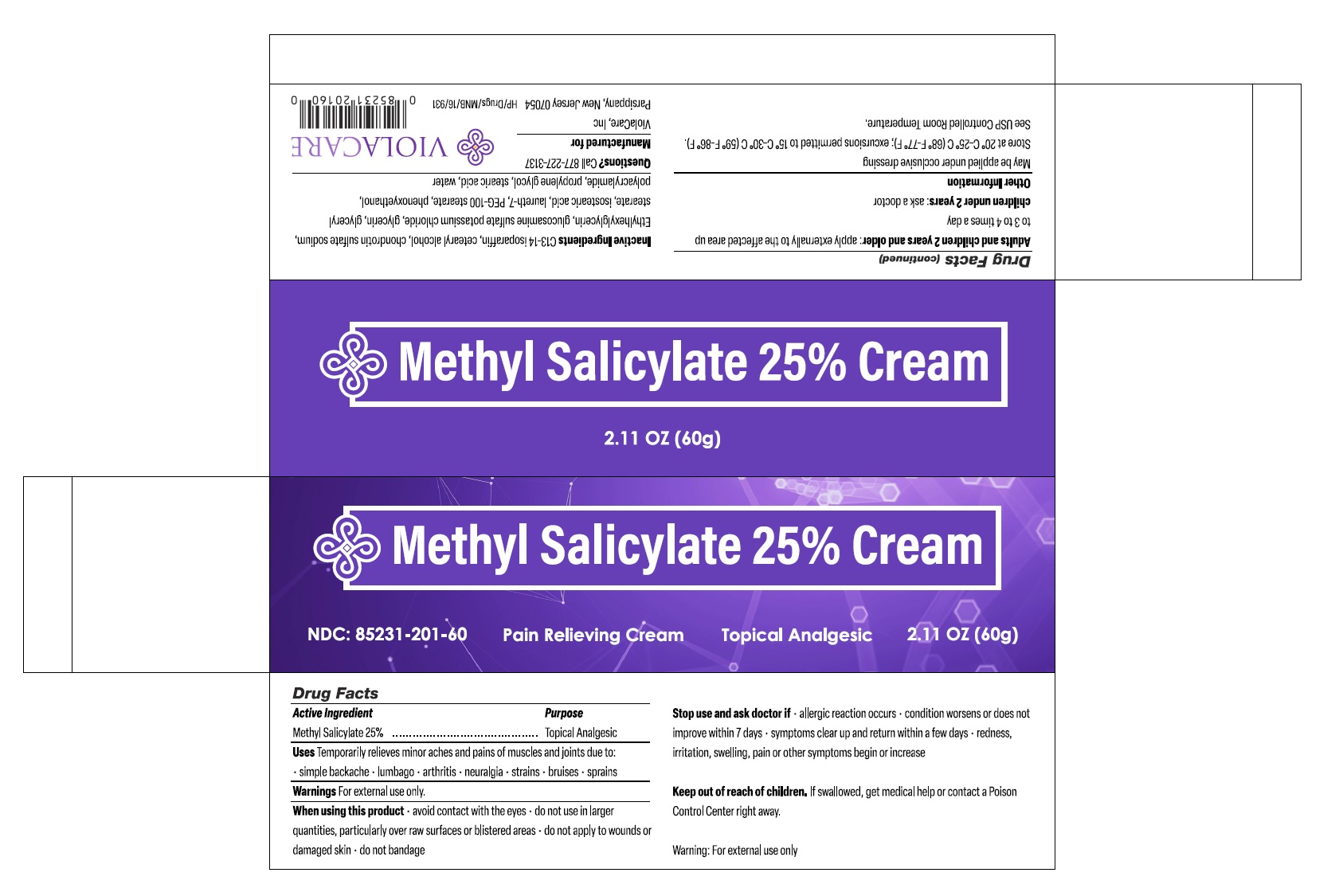
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Methyl Salicylate 25%........................................Topical Analgesic
Purpose
Methyl Salicylate 25%........................................Topical Analgesic
Medication Information
Purpose
Methyl Salicylate 25%........................................Topical Analgesic
Dosage and Administration
Adults and children 2 years and older: apply externally to the affected area up to 3 to 4 times a day
children under 2 years: ask a doctor
Description
Methyl Salicylate 25%........................................Topical Analgesic
Uses
Temporarily relieves minor aches and pains of muscles and joints due to:
- simple backache
- lumbago
- arthritis
- neuralgia
- strains
- bruises
- sprains
Section 53413-1
Questions?Call 877-227-3137
Section 55105-1
Warnings
For external use only.
Drug Facts
Other Information
May be applied under occlusive dressing
Store at 20° C-25° C (68° F-77° F); excursions permitted to 15° C-30° C (59° F-86° F). See USP Controlled Room Temperature.
Inactive Ingredients
C13-14 isoparaffin, cetearyl alcohol, chondrotin sulfate sodium, Ethylhexylglycerin, glucosamine sulfate potassium chloride, glycerin, glyceryl stearate, isostearic acid, laureth-7, PEG-100 stearate, phenoxyethanol, polyacrylamide, propylene glycol, stearic acid, water
Principal Display Panel
Methyl Salicylate 25% Cream - NDC 85231-201-60 - Pain Relieving Cream - Topical Analgesic - 2.11 OZ (60g)
Carton Label
Methyl Salicylate 25% Cream - NDC 85231-201-60 - Pain Relieving Cream - Topical Analgesic - 2.11 OZ (60g)
Box Label
Manufactured For:
ViolaCare, Inc.
Parsippany, New Jersey, 07054
When Using This Product
- avoid contact with the eyes
- do not use in larger quantities, particularly over raw surfaces or blistered areas
- do not apply to wounds or damaged skin
- do not bandage
Stop Use and Ask Doctor If
- allergic reaction occurs
- condition worsens or does not improve within 7 days
- symptoms clear up and return within a few days
- redness, irritation, swelling, pain or other symptoms begin or increase
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Active Ingredient purpose
Methyl Salicylate 25%........................................Topical Analgesic
Structured Label Content
Uses
Temporarily relieves minor aches and pains of muscles and joints due to:
- simple backache
- lumbago
- arthritis
- neuralgia
- strains
- bruises
- sprains
Dosage and Administration (34068-7)
Adults and children 2 years and older: apply externally to the affected area up to 3 to 4 times a day
children under 2 years: ask a doctor
Section 53413-1 (53413-1)
Questions?Call 877-227-3137
Section 55105-1 (55105-1)
Warnings
For external use only.
Drug Facts
Other Information
May be applied under occlusive dressing
Store at 20° C-25° C (68° F-77° F); excursions permitted to 15° C-30° C (59° F-86° F). See USP Controlled Room Temperature.
Inactive Ingredients
C13-14 isoparaffin, cetearyl alcohol, chondrotin sulfate sodium, Ethylhexylglycerin, glucosamine sulfate potassium chloride, glycerin, glyceryl stearate, isostearic acid, laureth-7, PEG-100 stearate, phenoxyethanol, polyacrylamide, propylene glycol, stearic acid, water
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Methyl Salicylate 25% Cream - NDC 85231-201-60 - Pain Relieving Cream - Topical Analgesic - 2.11 OZ (60g)
Carton Label
Methyl Salicylate 25% Cream - NDC 85231-201-60 - Pain Relieving Cream - Topical Analgesic - 2.11 OZ (60g)
Box Label
Manufactured For:
ViolaCare, Inc.
Parsippany, New Jersey, 07054
When Using This Product (When using this product)
- avoid contact with the eyes
- do not use in larger quantities, particularly over raw surfaces or blistered areas
- do not apply to wounds or damaged skin
- do not bandage
Stop Use and Ask Doctor If (Stop use and ask doctor if)
- allergic reaction occurs
- condition worsens or does not improve within 7 days
- symptoms clear up and return within a few days
- redness, irritation, swelling, pain or other symptoms begin or increase
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Active Ingredient purpose (Active Ingredient Purpose)
Methyl Salicylate 25%........................................Topical Analgesic
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:31.703574 · Updated: 2026-03-14T23:09:21.323328