These Highlights Do Not Include All The Information Needed To Use Orenitram Safely And Effectively. See Full Prescribing Information For Orenitram.
8ed2003a-c801-411e-831e-d06079bb0d7c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration ( 2.1 ) 08/2023
Indications and Usage
Orenitram is a prostacyclin mimetic indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1): To delay disease progression and to improve exercise capacity. The studies that established effectiveness included predominately patients with WHO functional class II-III symptoms and etiologies of idiopathic or heritable PAH (66%) or PAH associated with connective tissue disease (26%). ( 1.1 )
Dosage and Administration
Give with food. Swallow tablets whole; use only intact tablets. ( 2.1 ) Starting dose: 0.125 mg TID or 0.25 mg BID. ( 2.1 ) Titrate by 0.125 mg TID or by 0.25 mg or 0.5 mg BID, not more frequently than every 3 to 4 days as tolerated. The maximum daily dose is 120 mg. ( 2.1 ) If transitioning from intravenous (IV) or subcutaneous (SC) Remodulin ® , the Orenitram dose should be increased while simultaneously decreasing the IV/SC infusion rate. ( 2.2 ) Mild hepatic impairment (Child Pugh Class A): Initiate at 0.125 mg BID. Increment at 0.125 mg BID not more frequently than every 3 to 4 days. ( 2.3 ) Avoid use in patients with moderate hepatic impairment. ( 2.3 )
Warnings and Precautions
Do not abruptly discontinue dosing. ( 2.5 , 5.1 ) In patients with diverticulosis, Orenitram tablets can become lodged in a diverticulum. ( 5.2 )
Contraindications
Severe hepatic impairment (Child Pugh Class C) [see Use In Specific Populations (8.6) and Clinical Pharmacology (12.3) ] .
Adverse Reactions
Most common adverse reactions (incidence >10%) reported in clinical studies in patients treated with Orenitram compared with placebo are headache, diarrhea, nausea, vomiting, jaw pain, and flushing. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact United Therapeutics Corp. at 1-866-458-6479 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
When co-administered with strong CYP2C8 inhibitors the initial dose is 0.125 mg BID with 0.125 mg BID dose increments not more frequently than every 3 to 4 days. ( 2.4 , 7.1 )
How Supplied
Orenitram is an 8-mm round biconvex tablet with strength identified by color and print and supplied as follows: Strength Color Printing on Tablets NDC # 100-Count Bottle NDC # 10-Count Bottle NDC # 10-Count Carton (Unit-Dose Blister) 0.125 mg White UT 0.125 66302-300-01 66302-300-10 66302-300-02 0.25 mg Green UT 0.25 66302-302-01 66302-302-10 66302-302-02 1 mg Yellow UT 1 66302-310-01 66302-310-10 66302-310-02 2.5 mg Pink UT 2.5 66302-325-01 66302-325-10 66302-325-02 5 mg Red UT 5 66302-350-01 66302-350-10 66302-350-02 Titration Kits for Orenitram are supplied in the following configurations: Titration Kit Blister Configurations NDC # Month 1 containing four weekly cartons Seven daily wallet blister packs in each weekly carton containing the following total number of tablets: Week 1 (twenty-one × 0.125 mg tablets) Week 2 (forty-two × 0.125 mg tablets) Week 3 (twenty-one × 0.125 mg tablets and twenty-one × 0.25 mg tablets) Week 4 (forty-two × 0.125 mg tablets and twenty-one × 0.25 mg tablets) 66302-361-28 Month 2 containing four weekly cartons Seven daily wallet blister packs in each weekly carton containing the following total number of tablets: Week 5 (twenty-one × 0.125 mg tablets and forty-two × 0.25 mg tablets) Week 6 (forty-two × 0.125 mg tablets and forty-two × 0.25 mg tablets) Week 7 (twenty-one × 0.125 mg tablets and sixty-three × 0.25 mg tablets) Week 8 (forty-two × 0.125 mg tablets and sixty-three × 0.25 mg tablets) 66302-362-56 Month 3 containing four weekly cartons Seven daily wallet blister packs in each weekly carton containing the following total number of tablets: Week 9 (twenty-one × 0.125 mg tablets and twenty-one × 1 mg tablets) Week 10 (forty-two × 0.125 mg tablets and twenty-one × 1 mg tablets) Week 11 (twenty-one × 0.125 mg tablets, twenty-one × 0.25 mg tablets, and twenty-one × 1 mg tablets) Week 12 (forty-two × 0.125 mg tablets, twenty-one × 0.25 mg tablets, and twenty-one × 1 mg tablets) 66302-363-84
Medication Information
Warnings and Precautions
Do not abruptly discontinue dosing. ( 2.5 , 5.1 ) In patients with diverticulosis, Orenitram tablets can become lodged in a diverticulum. ( 5.2 )
Indications and Usage
Orenitram is a prostacyclin mimetic indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1): To delay disease progression and to improve exercise capacity. The studies that established effectiveness included predominately patients with WHO functional class II-III symptoms and etiologies of idiopathic or heritable PAH (66%) or PAH associated with connective tissue disease (26%). ( 1.1 )
Dosage and Administration
Give with food. Swallow tablets whole; use only intact tablets. ( 2.1 ) Starting dose: 0.125 mg TID or 0.25 mg BID. ( 2.1 ) Titrate by 0.125 mg TID or by 0.25 mg or 0.5 mg BID, not more frequently than every 3 to 4 days as tolerated. The maximum daily dose is 120 mg. ( 2.1 ) If transitioning from intravenous (IV) or subcutaneous (SC) Remodulin ® , the Orenitram dose should be increased while simultaneously decreasing the IV/SC infusion rate. ( 2.2 ) Mild hepatic impairment (Child Pugh Class A): Initiate at 0.125 mg BID. Increment at 0.125 mg BID not more frequently than every 3 to 4 days. ( 2.3 ) Avoid use in patients with moderate hepatic impairment. ( 2.3 )
Contraindications
Severe hepatic impairment (Child Pugh Class C) [see Use In Specific Populations (8.6) and Clinical Pharmacology (12.3) ] .
Adverse Reactions
Most common adverse reactions (incidence >10%) reported in clinical studies in patients treated with Orenitram compared with placebo are headache, diarrhea, nausea, vomiting, jaw pain, and flushing. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact United Therapeutics Corp. at 1-866-458-6479 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
When co-administered with strong CYP2C8 inhibitors the initial dose is 0.125 mg BID with 0.125 mg BID dose increments not more frequently than every 3 to 4 days. ( 2.4 , 7.1 )
How Supplied
Orenitram is an 8-mm round biconvex tablet with strength identified by color and print and supplied as follows: Strength Color Printing on Tablets NDC # 100-Count Bottle NDC # 10-Count Bottle NDC # 10-Count Carton (Unit-Dose Blister) 0.125 mg White UT 0.125 66302-300-01 66302-300-10 66302-300-02 0.25 mg Green UT 0.25 66302-302-01 66302-302-10 66302-302-02 1 mg Yellow UT 1 66302-310-01 66302-310-10 66302-310-02 2.5 mg Pink UT 2.5 66302-325-01 66302-325-10 66302-325-02 5 mg Red UT 5 66302-350-01 66302-350-10 66302-350-02 Titration Kits for Orenitram are supplied in the following configurations: Titration Kit Blister Configurations NDC # Month 1 containing four weekly cartons Seven daily wallet blister packs in each weekly carton containing the following total number of tablets: Week 1 (twenty-one × 0.125 mg tablets) Week 2 (forty-two × 0.125 mg tablets) Week 3 (twenty-one × 0.125 mg tablets and twenty-one × 0.25 mg tablets) Week 4 (forty-two × 0.125 mg tablets and twenty-one × 0.25 mg tablets) 66302-361-28 Month 2 containing four weekly cartons Seven daily wallet blister packs in each weekly carton containing the following total number of tablets: Week 5 (twenty-one × 0.125 mg tablets and forty-two × 0.25 mg tablets) Week 6 (forty-two × 0.125 mg tablets and forty-two × 0.25 mg tablets) Week 7 (twenty-one × 0.125 mg tablets and sixty-three × 0.25 mg tablets) Week 8 (forty-two × 0.125 mg tablets and sixty-three × 0.25 mg tablets) 66302-362-56 Month 3 containing four weekly cartons Seven daily wallet blister packs in each weekly carton containing the following total number of tablets: Week 9 (twenty-one × 0.125 mg tablets and twenty-one × 1 mg tablets) Week 10 (forty-two × 0.125 mg tablets and twenty-one × 1 mg tablets) Week 11 (twenty-one × 0.125 mg tablets, twenty-one × 0.25 mg tablets, and twenty-one × 1 mg tablets) Week 12 (forty-two × 0.125 mg tablets, twenty-one × 0.25 mg tablets, and twenty-one × 1 mg tablets) 66302-363-84
Description
Dosage and Administration ( 2.1 ) 08/2023
Section 42229-5
Risk Summary
Limited published data from case reports with Orenitram use in pregnant women are not sufficient to assess for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. There are risks to the mother and the fetus associated with pulmonary arterial hypertension (see Clinical Considerations). Animal reproductive studies with treprostinil diolamine administered orally have shown an adverse effect on the fetus. In rats, administration of treprostinil to pregnant rats during the period of organogenesis at doses ≥10 mg/kg/day (approximately 15 times the human exposure at the dose of 3.5 mg BID on an AUC basis) resulted in decreased pregnancy rate, increased post-implantation loss, and decreased fetal viability and growth. In rabbits, teratogenicity and decreased fetal viability and growth were observed at doses ≥1.5 mg/kg/day (approximately 7 times the human exposure at the dose of 3.5 mg BID on an AUC basis) (see Animal Data).
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 02/2023 |
|
Patient Information Orenitram(®) (oh-REN-i-tram) (treprostinil) extended-release tablets |
|
What is Orenitram?
|
|
| Do not take Orenitram if you have severe liver problems. | |
Before taking Orenitram, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take another medicine that contains treprostinil, such as Remodulin®, Tyvaso®, or Tyvaso DPI®. |
|
How should I take Orenitram?
|
|
|
What are the possible side effects of Orenitram? Orenitram can cause serious side effects, including worsening of PAH symptoms.
|
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of Orenitram. For more information, ask your healthcare provider or pharmacist. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store Orenitram?
|
|
|
General information about the safe and effective use of Orenitram.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. Do not use Orenitram for a condition for which it was not prescribed. Do not give Orenitram to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about Orenitram that is written for health professionals. |
|
|
What are the ingredients in Orenitram?
Active ingredient: treprostinil diolamine Inactive ingredients: xylitol, maltodextrin, sodium lauryl sulfate, magnesium stearate, cellulose acetate, triethyl citrate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, and talc. In addition, tablets may contain colorants FD&C Blue #2, iron oxide yellow, and iron oxide red. The imprint ink contains shellac glaze, ethanol, isopropyl alcohol, iron oxide black, n-butyl alcohol, and propylene glycol. United Therapeutics Corp., Research Triangle Park, NC 27709 USA Copyright 2023, United Therapeutics Corp. All rights reserved. ORENITRAM is a registered trademark of United Therapeutics Corp. For more information, go to www.orenitram.com or call 1-877-864-8437. |
Section 43683-2
| Dosage and Administration (2.1) | 08/2023 |
16.2 Storage
Store at 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]. Keep out of reach of children.
10 Overdosage
Signs and symptoms of overdose with Orenitram during clinical trials reflect its dose-limiting pharmacologic effects and include severe headache, nausea, vomiting, diarrhea, and hypotension. Treat supportively.
11 Description
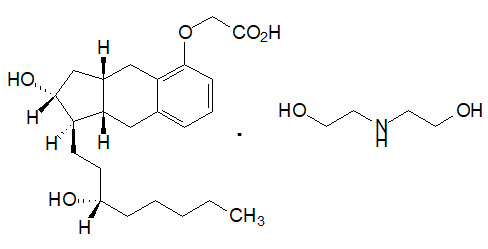
Orenitram is an extended-release osmotic tablet for oral administration. Orenitram is formulated as the diolamine salt of treprostinil, a tricyclic benzindene analogue of prostacyclin. The chemical name is Acetic acid, 2-[[(1R,2R,3aS,9aS)-2,3,3a,4,9,9a-hexahydro-2-hydroxy-1-[(3S)-3-hydroxyoctyl]-1H-benz[f]inden-5-yl]oxy]-, complexed with 2,2'-iminobis[ethanol] (1:1). The molecular formula is C23H34O5∙C4H11NO2, the molecular weight is 495.65, and it has the following structural formula:
Orenitram tablets are formulated in five strengths, which contain 0.125 mg of treprostinil (equivalent to 0.159 mg treprostinil diolamine), 0.25 mg of treprostinil (equivalent to 0.317 mg treprostinil diolamine), 1 mg of treprostinil (equivalent to 1.27 mg treprostinil diolamine), 2.5 mg of treprostinil (equivalent to 3.17 mg treprostinil diolamine), or 5 mg of treprostinil (equivalent to 6.35 mg treprostinil diolamine). The formulations also contain xylitol, maltodextrin, sodium lauryl sulfate, magnesium stearate, cellulose acetate, triethyl citrate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, and talc. In addition, tablets may contain colorants FD&C Blue #2, iron oxide yellow, and iron oxide red. The imprint ink contains shellac glaze, ethanol, isopropyl alcohol USP, iron oxide black, n-butyl alcohol, and propylene glycol.
Orenitram is designed to release treprostinil at a near zero-order rate using an osmotic tablet technology. The tablet core is coated with a semi-permeable membrane and has a laser-drilled aperture through the membrane. Upon contact with water (e.g., after ingestion), the core tablet absorbs water through the semi-permeable membrane. The water dissolves the water-soluble treprostinil diolamine and the water-soluble osmotic excipients, which creates hydrostatic pressure within the membrane, eventually forcing the drug across the membrane at a controlled rate.
16.1 How Supplied
Orenitram is an 8-mm round biconvex tablet with strength identified by color and print and supplied as follows:
| Strength | Color | Printing on Tablets | NDC # 100-Count Bottle |
NDC # 10-Count Bottle |
NDC # 10-Count Carton (Unit-Dose Blister) |
|---|---|---|---|---|---|
| 0.125 mg | White | UT 0.125 | 66302-300-01 | 66302-300-10 | 66302-300-02 |
| 0.25 mg | Green | UT 0.25 | 66302-302-01 | 66302-302-10 | 66302-302-02 |
| 1 mg | Yellow | UT 1 | 66302-310-01 | 66302-310-10 | 66302-310-02 |
| 2.5 mg | Pink | UT 2.5 | 66302-325-01 | 66302-325-10 | 66302-325-02 |
| 5 mg | Red | UT 5 | 66302-350-01 | 66302-350-10 | 66302-350-02 |
Titration Kits for Orenitram are supplied in the following configurations:
| Titration Kit | Blister Configurations | NDC # |
|---|---|---|
| Month 1 containing four weekly cartons | Seven daily wallet blister packs in each weekly carton containing the following total number of tablets:
|
66302-361-28 |
| Month 2 containing four weekly cartons | Seven daily wallet blister packs in each weekly carton containing the following total number of tablets:
|
66302-362-56 |
| Month 3 containing four weekly cartons | Seven daily wallet blister packs in each weekly carton containing the following total number of tablets:
|
66302-363-84 |
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Use of Orenitram in patients aged 65 years and over demonstrated slightly higher absolute and relative adverse event rates compared to younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications
Severe hepatic impairment (Child Pugh Class C) [see Use In Specific Populations (8.6) and Clinical Pharmacology (12.3)].
6 Adverse Reactions
Most common adverse reactions (incidence >10%) reported in clinical studies in patients treated with Orenitram compared with placebo are headache, diarrhea, nausea, vomiting, jaw pain, and flushing. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact United Therapeutics Corp. at 1-866-458-6479 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
12.2 Pharmacodynamics
In a clinical trial of 240 healthy adult volunteers, single doses of inhaled treprostinil 54 µg (the target clinical dose) and 84 µg (supratherapeutic inhalation dose) prolonged the corrected QTc interval by approximately 10 msec. The QTc effect dissipated rapidly as the concentration of treprostinil decreased. Orenitram has not been evaluated in a thorough QTc study.
12.3 Pharmacokinetics
In patients with PAH, pharmacokinetics of treprostinil is dose-proportional for systemic exposure (AUC0-t) over the dose range from 0.5 to 15 mg BID. Upon repeat administration with a BID regimen, the accumulation in the systemic exposures to treprostinil is minimal and results in a peak-to-trough ratio of approximately 7. However, a TID regimen will reduce the peak-to-trough fluctuations to approximately 2.5 for the same total daily dose.
2.1 Recommended Dosing
Take Orenitram with food. Swallow Orenitram tablets whole; do not crush, split, or chew.
The recommended starting dose of Orenitram is 0.125 mg three times daily (TID) with food, taken approximately 8 hours apart or 0.25 mg twice daily (BID) with food, taken approximately 12 hours apart.
Titrate by 0.125 mg TID or 0.25 or 0.5 mg BID not more frequently than every 3 to 4 days. Increase the dose to the highest tolerated dose. The recommended maximum daily dose is 120 mg.
If dose increments are not tolerated, consider titrating slower. If intolerable pharmacologic effects occur, decrease the dose in increments of 0.125 mg TID or 0.25 mg BID. Avoid abrupt discontinuation [see Warnings and Precautions (5.1)].
1 Indications and Usage
Orenitram is a prostacyclin mimetic indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1):
- To delay disease progression and to improve exercise capacity. The studies that established effectiveness included predominately patients with WHO functional class II-III symptoms and etiologies of idiopathic or heritable PAH (66%) or PAH associated with connective tissue disease (26%). (1.1)
12.1 Mechanism of Action
The major pharmacologic actions of treprostinil are direct vasodilation of pulmonary and systemic arterial vascular beds, inhibition of platelet aggregation, and inhibition of smooth muscle cell proliferation.
5 Warnings and Precautions
2 Dosage and Administration
- Give with food. Swallow tablets whole; use only intact tablets. (2.1)
- Starting dose: 0.125 mg TID or 0.25 mg BID. (2.1)
- Titrate by 0.125 mg TID or by 0.25 mg or 0.5 mg BID, not more frequently than every 3 to 4 days as tolerated. The maximum daily dose is 120 mg. (2.1)
- If transitioning from intravenous (IV) or subcutaneous (SC) Remodulin®, the Orenitram dose should be increased while simultaneously decreasing the IV/SC infusion rate. (2.2)
- Mild hepatic impairment (Child Pugh Class A): Initiate at 0.125 mg BID. Increment at 0.125 mg BID not more frequently than every 3 to 4 days. (2.3)
- Avoid use in patients with moderate hepatic impairment. (2.3)
3 Dosage Forms and Strengths
Orenitram (treprostinil) extended-release tablets are available in the following five strengths:
-
-0.125 mg [White tablet imprinted with UT 0.125]
-
-0.25 mg [Green tablet imprinted with UT 0.25]
-
-1 mg [Yellow tablet imprinted with UT 1]
-
-2.5 mg [Pink tablet imprinted with UT 2.5]
-
-5 mg [Red tablet imprinted with UT 5]
6.2 Post Marketing Experience
The following adverse reactions have been identified during postapproval use of Orenitram: dizziness, dyspepsia, vomiting, myalgia, and arthralgia. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In a 12-week, placebo-controlled, monotherapy study (Study 1; WHO Group 1; functional class II-III), and an event-driven, placebo-controlled, combination therapy study (Study 4; WHO Group 1; functional class I-III), the most commonly reported adverse reactions that occurred in patients receiving Orenitram included: headache, diarrhea, nausea, and flushing.
Orenitram patients in Study 1 (N=151) had access to 0.25 mg tablets at randomization. Approximately 91% of such patients in Study 1 experienced an adverse reaction, but only 4% discontinued therapy for an adverse reaction (compared to 3% receiving placebo). Study 4 enrolled a total of 690 patients, 346 received Orenitram and 344 received placebo. Overall, 19% of patients treated with Orenitram discontinued treatment in Study 4 due to an adverse event (compared to 4% of patients receiving placebo). The exposure to Orenitram in Study 4 was up to 5.1 years with a median duration of exposure of 1.2 years. Table 1 summarizes adverse events with rates at least 5% higher on Orenitram therapy than on placebo that were reported in either Study 1 or 4.
| Reaction | Study 1 N=228 Includes all subjects in the Primary Analysis Population
|
Study 4 N=690 |
||
|---|---|---|---|---|
| Orenitram n=151 |
Placebo n=77 |
Orenitram n=346 |
Placebo n=344 |
|
| Headache | 63% | 19% | 75% | 35% |
| Diarrhea | 30% | 16% | 69% | 29% |
| Nausea | 30% | 18% | 40% | 23% |
| Vomiting | 17% | 16% | 36% | 10% |
| Flushing | 15% | 6% | 45% | 8% |
| Pain in jaw | 11% | 4% | 18% | 3% |
| Pain in extremity | 14% | 8% | 18% | 9% |
| Hypokalemia | 9% | 3% | 4% | 3% |
| Abdominal discomfort | 6% | 0% | 8% | 4% |
| Upper abdominal pain | 5% | 3% | 12% | 5% |
Orenitram was studied in a long-term, open-label, extension study in which 824 patients were dosed for a mean duration of approximately 2 years. About 70% of patients continued treatment with Orenitram for at least a year. The mean dose was 4.2 mg BID at one year. The adverse reactions were similar to those observed in the placebo-controlled trials.
The safety of Orenitram was also evaluated in an open-label study transitioning patients from Remodulin. The safety profile during this study was similar to that observed in the three pivotal studies.
17 Patient Counseling Information
See FDA-approved patient labeling (Patient Information).
Tell patients:
- Abrupt discontinuation of therapy could result in worsening of PAH symptoms.
- Take Orenitram with food.
- Swallow Orenitram tablets whole. Do not split, chew, crush, or break. Do not take a tablet that is damaged or broken.
- The biologically inert components of the tablet remain intact during gastrointestinal transit and are eliminated in the feces as an insoluble shell.
8.7 Patients With Renal Impairment
No dose adjustments are required in patients with renal impairment. Orenitram is not removed by dialysis [see Clinical Pharmacology (12.3)].
1.1 Pulmonary Arterial Hypertension
Orenitram is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to delay disease progression and to improve exercise capacity.
The studies that established effectiveness included predominately patients with WHO functional class II-III symptoms and etiologies of idiopathic or heritable PAH (66%) or PAH associated with connective tissue disease (26%).
8.6 Patients With Hepatic Impairment
There is a marked increase in the systemic exposure to treprostinil in hepatically impaired patients [see Dosage and Administration (2.3), Contraindications (4), and Clinical Pharmacology (12.3)].
2.5 Interruptions and Discontinuation
If a dose of medication is missed, the patient should take the missed dose as soon as possible, with food. If a patient misses two or more doses, restart at a lower dose and re-titrate.
In the event of a planned short-term treatment interruption for patients unable to take oral medications, consider a temporary infusion of subcutaneous or intravenous treprostinil. To calculate the total daily dose (mg) of treprostinil for the parenteral route use the following equation:
| Remodulin (ng/kg/min) = | 139 × Orenitram total daily dose (mg) |
| weight (kg) |
When discontinuing Orenitram, reduce the dose in steps of 0.5 to 1 mg per day [see Warnings and Precautions (5.1)].
5.2 Use in Patients With Blind End Pouches
The tablet shell does not dissolve. In patients with diverticulosis, Orenitram tablets can lodge in a diverticulum.
7.1 Effect of Cyp2c8 Inhibitors On Treprostinil
Co-administration of Orenitram and the CYP2C8 enzyme inhibitor gemfibrozil in healthy adult volunteers increases exposure to treprostinil. Reduce the starting dose of Orenitram to 0.125 mg BID and use 0.125 mg BID increments not more frequently than every 3 to 4 days [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
5.1 Worsening Pah Symptoms Upon Abrupt Withdrawal
Abrupt discontinuation or sudden large reductions in dosage of Orenitram may result in worsening of PAH symptoms.
2.4 Dose Adjustment for Use With Cyp2c8 Inhibitors
When co-administered with strong CYP2C8 inhibitors (e.g., gemfibrozil) the initial dose is 0.125 mg BID with 0.125 mg BID dose increments not more frequently than every 3 to 4 days.
Principal Display Panel 1 Mg Tablet Bottle Label
NDC 66302-310-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
1 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
Principal Display Panel 5 Mg Tablet Bottle Label
NDC 66302-350-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
5 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
Principal Display Panel 2.5 Mg Tablet Bottle Label
NDC 66302-325-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
2.5 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
Principal Display Panel 0.25 Mg Tablet Bottle Label
NDC 66302-302-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
0.25 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
Principal Display Panel 0.125 Mg Tablet Bottle Label
NDC 66302-300-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
0.125 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
14.1 Clinical Trials in Pulmonary Arterial Hypertension
Four multicenter, randomized, double-blind studies were conducted and compared Orenitram to placebo in a total of 349 (Study 1), 350 (Study 2), 310 (Study 3), and 690 (Study 4) patients with PAH.
2.3 Dose Adjustment in Patients With Hepatic Impairment
In patients with mild hepatic impairment (Child Pugh Class A) start at 0.125 mg BID with 0.125 mg BID dose increments not more frequently than every 3 to 4 days. Avoid use of Orenitram in patients with moderate hepatic impairment (Child Pugh Class B). Orenitram is contraindicated in patients with severe hepatic impairment (Child Pugh Class C) due to increases in systemic exposure [see Contraindications (4), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Treprostinil diolamine did not demonstrate any carcinogenic effects in mouse or rat carcinogenicity studies. Oral administration of treprostinil diolamine to Tg.rasH2 mice at 0, 5, 10, and 20 mg/kg/day in males and 0, 3, 7.5, and 15 mg/kg/day in females daily for 26 weeks did not significantly increase the incidence of tumors. Oral administration of treprostinil diolamine to Sprague Dawley rats at 0, 1, 3, and 10 mg/kg/day daily for 104 weeks did not significantly increase the incidence of tumors. The exposures obtained at the highest dose levels used in males and females are about 13- and 18-fold, respectively, the human exposure at the dose of 3.5 mg BID on an AUC basis.
In vitro genotoxicity studies with high doses of treprostinil did not demonstrate any mutagenic or clastogenic effects. Treprostinil diolamine was tested in vivo in a rat micronucleus assay and did not induce an increased incidence of micronucleated polychromatic erythrocytes.
In rats, treatment with treprostinil diolamine had no effect on reproductive performance or sperm motility at doses up to 10 mg/kg/day. The exposures at this dose level are about 6- (male) to 11- (female) fold the human exposure at the dose of 3.5 mg BID on an AUC basis.
Principal Display Panel 1 Mg Tablet Blister Pack Carton
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
1 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-310-02
Principal Display Panel 5 Mg Tablet Blister Pack Carton
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
5 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-350-02
Principal Display Panel 2.5 Mg Tablet Blister Pack Carton
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
2.5 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-325-02
Principal Display Panel 0.25 Mg Tablet Blister Pack Carton
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
0.25 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-302-02
Principal Display Panel 0.125 Mg Tablet Blister Pack Carton
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
0.125 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-300-02
Principal Display Panel 0.125 Mg 0.25 Mg Titration Kit Carton Month 1
Rx Only
NDC 66302-361-28
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
Titration Kit
Month 1
0.125 mg
per tablet
0.25 mg
per tablet
Swallow whole.
Do not split, chew, crush, or break tablets.
Kit Contains:
168 tablets (126 x 0.125 mg per tablet and
42 x 0.25 mg per tablet).
Monthly carton contains 4 individual weekly cartons
each with 7 daily wallet blister packs.
Principal Display Panel 0.125 Mg 0.25 Mg Titration Kit Carton Month 2
Rx Only
NDC 66302-362-56
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
Titration Kit
Month 2
0.125 mg
per tablet
0.25 mg
per tablet
Swallow whole.
Do not split, chew, crush, or break tablets.
Kit Contains:
336 tablets (126 x 0.125 mg per tablet and
210 x 0.25 mg per tablet).
Monthly carton contains 4 individual weekly cartons
each with 7 daily wallet blister packs.
Principal Display Panel 0.125 Mg 0.25 Mg 1 Mg Titration Kit Carton Month 3
Rx Only
NDC 66302-363-84
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
Titration Kit
Month 3
0.125 mg
per tablet
0.25 mg
per tablet
1 mg
per tablet
Swallow whole.
Do not split, chew, crush, or break tablets.
Kit Contains:
252 tablets (126 x 0.125 mg per tablet,
42 x 0.25 mg per tablet, and 84 × 1 mg per tablet).
Monthly carton contains 4 individual weekly cartons
each with 7 daily wallet blister packs.
2.2 Transitioning From Subcutaneous Or Intravenous Routes of Administration of Treprostinil
Decrease the dose of Remodulin while simultaneously increasing the dose of Orenitram. The dose of Remodulin can be reduced up to 30 ng/kg/min per day and the dose of Orenitram simultaneously increased up to 6 mg per day (2 mg TID) if tolerated. The following equation can be used to estimate a target total daily dose of Orenitram in mg using a patient's dose of intravenous (IV)/subcutaneous (SC) treprostinil (in ng/kg/min) and weight (in kg).
Orenitram total daily dose (mg) = 0.0072 × Remodulin dose (ng/kg/min) × weight (kg)
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
Limited published data from case reports with Orenitram use in pregnant women are not sufficient to assess for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. There are risks to the mother and the fetus associated with pulmonary arterial hypertension (see Clinical Considerations). Animal reproductive studies with treprostinil diolamine administered orally have shown an adverse effect on the fetus. In rats, administration of treprostinil to pregnant rats during the period of organogenesis at doses ≥10 mg/kg/day (approximately 15 times the human exposure at the dose of 3.5 mg BID on an AUC basis) resulted in decreased pregnancy rate, increased post-implantation loss, and decreased fetal viability and growth. In rabbits, teratogenicity and decreased fetal viability and growth were observed at doses ≥1.5 mg/kg/day (approximately 7 times the human exposure at the dose of 3.5 mg BID on an AUC basis) (see Animal Data).
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 02/2023 |
|
Patient Information Orenitram(®) (oh-REN-i-tram) (treprostinil) extended-release tablets |
|
What is Orenitram?
|
|
| Do not take Orenitram if you have severe liver problems. | |
Before taking Orenitram, tell your healthcare provider about all of your medical conditions, including if you:
Especially tell your healthcare provider if you take another medicine that contains treprostinil, such as Remodulin®, Tyvaso®, or Tyvaso DPI®. |
|
How should I take Orenitram?
|
|
|
What are the possible side effects of Orenitram? Orenitram can cause serious side effects, including worsening of PAH symptoms.
|
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of Orenitram. For more information, ask your healthcare provider or pharmacist. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store Orenitram?
|
|
|
General information about the safe and effective use of Orenitram.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. Do not use Orenitram for a condition for which it was not prescribed. Do not give Orenitram to other people, even if they have the same symptoms that you have. It may harm them. You can ask your healthcare provider or pharmacist for information about Orenitram that is written for health professionals. |
|
|
What are the ingredients in Orenitram?
Active ingredient: treprostinil diolamine Inactive ingredients: xylitol, maltodextrin, sodium lauryl sulfate, magnesium stearate, cellulose acetate, triethyl citrate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, and talc. In addition, tablets may contain colorants FD&C Blue #2, iron oxide yellow, and iron oxide red. The imprint ink contains shellac glaze, ethanol, isopropyl alcohol, iron oxide black, n-butyl alcohol, and propylene glycol. United Therapeutics Corp., Research Triangle Park, NC 27709 USA Copyright 2023, United Therapeutics Corp. All rights reserved. ORENITRAM is a registered trademark of United Therapeutics Corp. For more information, go to www.orenitram.com or call 1-877-864-8437. |
Section 43683-2 (43683-2)
| Dosage and Administration (2.1) | 08/2023 |
16.2 Storage
Store at 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature]. Keep out of reach of children.
10 Overdosage (10 OVERDOSAGE)
Signs and symptoms of overdose with Orenitram during clinical trials reflect its dose-limiting pharmacologic effects and include severe headache, nausea, vomiting, diarrhea, and hypotension. Treat supportively.
11 Description (11 DESCRIPTION)
Orenitram is an extended-release osmotic tablet for oral administration. Orenitram is formulated as the diolamine salt of treprostinil, a tricyclic benzindene analogue of prostacyclin. The chemical name is Acetic acid, 2-[[(1R,2R,3aS,9aS)-2,3,3a,4,9,9a-hexahydro-2-hydroxy-1-[(3S)-3-hydroxyoctyl]-1H-benz[f]inden-5-yl]oxy]-, complexed with 2,2'-iminobis[ethanol] (1:1). The molecular formula is C23H34O5∙C4H11NO2, the molecular weight is 495.65, and it has the following structural formula:
Orenitram tablets are formulated in five strengths, which contain 0.125 mg of treprostinil (equivalent to 0.159 mg treprostinil diolamine), 0.25 mg of treprostinil (equivalent to 0.317 mg treprostinil diolamine), 1 mg of treprostinil (equivalent to 1.27 mg treprostinil diolamine), 2.5 mg of treprostinil (equivalent to 3.17 mg treprostinil diolamine), or 5 mg of treprostinil (equivalent to 6.35 mg treprostinil diolamine). The formulations also contain xylitol, maltodextrin, sodium lauryl sulfate, magnesium stearate, cellulose acetate, triethyl citrate, polyvinyl alcohol, titanium dioxide, polyethylene glycol, and talc. In addition, tablets may contain colorants FD&C Blue #2, iron oxide yellow, and iron oxide red. The imprint ink contains shellac glaze, ethanol, isopropyl alcohol USP, iron oxide black, n-butyl alcohol, and propylene glycol.
Orenitram is designed to release treprostinil at a near zero-order rate using an osmotic tablet technology. The tablet core is coated with a semi-permeable membrane and has a laser-drilled aperture through the membrane. Upon contact with water (e.g., after ingestion), the core tablet absorbs water through the semi-permeable membrane. The water dissolves the water-soluble treprostinil diolamine and the water-soluble osmotic excipients, which creates hydrostatic pressure within the membrane, eventually forcing the drug across the membrane at a controlled rate.
16.1 How Supplied
Orenitram is an 8-mm round biconvex tablet with strength identified by color and print and supplied as follows:
| Strength | Color | Printing on Tablets | NDC # 100-Count Bottle |
NDC # 10-Count Bottle |
NDC # 10-Count Carton (Unit-Dose Blister) |
|---|---|---|---|---|---|
| 0.125 mg | White | UT 0.125 | 66302-300-01 | 66302-300-10 | 66302-300-02 |
| 0.25 mg | Green | UT 0.25 | 66302-302-01 | 66302-302-10 | 66302-302-02 |
| 1 mg | Yellow | UT 1 | 66302-310-01 | 66302-310-10 | 66302-310-02 |
| 2.5 mg | Pink | UT 2.5 | 66302-325-01 | 66302-325-10 | 66302-325-02 |
| 5 mg | Red | UT 5 | 66302-350-01 | 66302-350-10 | 66302-350-02 |
Titration Kits for Orenitram are supplied in the following configurations:
| Titration Kit | Blister Configurations | NDC # |
|---|---|---|
| Month 1 containing four weekly cartons | Seven daily wallet blister packs in each weekly carton containing the following total number of tablets:
|
66302-361-28 |
| Month 2 containing four weekly cartons | Seven daily wallet blister packs in each weekly carton containing the following total number of tablets:
|
66302-362-56 |
| Month 3 containing four weekly cartons | Seven daily wallet blister packs in each weekly carton containing the following total number of tablets:
|
66302-363-84 |
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Use of Orenitram in patients aged 65 years and over demonstrated slightly higher absolute and relative adverse event rates compared to younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications (4 CONTRAINDICATIONS)
Severe hepatic impairment (Child Pugh Class C) [see Use In Specific Populations (8.6) and Clinical Pharmacology (12.3)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (incidence >10%) reported in clinical studies in patients treated with Orenitram compared with placebo are headache, diarrhea, nausea, vomiting, jaw pain, and flushing. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact United Therapeutics Corp. at 1-866-458-6479 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
12.2 Pharmacodynamics
In a clinical trial of 240 healthy adult volunteers, single doses of inhaled treprostinil 54 µg (the target clinical dose) and 84 µg (supratherapeutic inhalation dose) prolonged the corrected QTc interval by approximately 10 msec. The QTc effect dissipated rapidly as the concentration of treprostinil decreased. Orenitram has not been evaluated in a thorough QTc study.
12.3 Pharmacokinetics
In patients with PAH, pharmacokinetics of treprostinil is dose-proportional for systemic exposure (AUC0-t) over the dose range from 0.5 to 15 mg BID. Upon repeat administration with a BID regimen, the accumulation in the systemic exposures to treprostinil is minimal and results in a peak-to-trough ratio of approximately 7. However, a TID regimen will reduce the peak-to-trough fluctuations to approximately 2.5 for the same total daily dose.
2.1 Recommended Dosing
Take Orenitram with food. Swallow Orenitram tablets whole; do not crush, split, or chew.
The recommended starting dose of Orenitram is 0.125 mg three times daily (TID) with food, taken approximately 8 hours apart or 0.25 mg twice daily (BID) with food, taken approximately 12 hours apart.
Titrate by 0.125 mg TID or 0.25 or 0.5 mg BID not more frequently than every 3 to 4 days. Increase the dose to the highest tolerated dose. The recommended maximum daily dose is 120 mg.
If dose increments are not tolerated, consider titrating slower. If intolerable pharmacologic effects occur, decrease the dose in increments of 0.125 mg TID or 0.25 mg BID. Avoid abrupt discontinuation [see Warnings and Precautions (5.1)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Orenitram is a prostacyclin mimetic indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1):
- To delay disease progression and to improve exercise capacity. The studies that established effectiveness included predominately patients with WHO functional class II-III symptoms and etiologies of idiopathic or heritable PAH (66%) or PAH associated with connective tissue disease (26%). (1.1)
12.1 Mechanism of Action
The major pharmacologic actions of treprostinil are direct vasodilation of pulmonary and systemic arterial vascular beds, inhibition of platelet aggregation, and inhibition of smooth muscle cell proliferation.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Give with food. Swallow tablets whole; use only intact tablets. (2.1)
- Starting dose: 0.125 mg TID or 0.25 mg BID. (2.1)
- Titrate by 0.125 mg TID or by 0.25 mg or 0.5 mg BID, not more frequently than every 3 to 4 days as tolerated. The maximum daily dose is 120 mg. (2.1)
- If transitioning from intravenous (IV) or subcutaneous (SC) Remodulin®, the Orenitram dose should be increased while simultaneously decreasing the IV/SC infusion rate. (2.2)
- Mild hepatic impairment (Child Pugh Class A): Initiate at 0.125 mg BID. Increment at 0.125 mg BID not more frequently than every 3 to 4 days. (2.3)
- Avoid use in patients with moderate hepatic impairment. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Orenitram (treprostinil) extended-release tablets are available in the following five strengths:
-
-0.125 mg [White tablet imprinted with UT 0.125]
-
-0.25 mg [Green tablet imprinted with UT 0.25]
-
-1 mg [Yellow tablet imprinted with UT 1]
-
-2.5 mg [Pink tablet imprinted with UT 2.5]
-
-5 mg [Red tablet imprinted with UT 5]
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
The following adverse reactions have been identified during postapproval use of Orenitram: dizziness, dyspepsia, vomiting, myalgia, and arthralgia. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In a 12-week, placebo-controlled, monotherapy study (Study 1; WHO Group 1; functional class II-III), and an event-driven, placebo-controlled, combination therapy study (Study 4; WHO Group 1; functional class I-III), the most commonly reported adverse reactions that occurred in patients receiving Orenitram included: headache, diarrhea, nausea, and flushing.
Orenitram patients in Study 1 (N=151) had access to 0.25 mg tablets at randomization. Approximately 91% of such patients in Study 1 experienced an adverse reaction, but only 4% discontinued therapy for an adverse reaction (compared to 3% receiving placebo). Study 4 enrolled a total of 690 patients, 346 received Orenitram and 344 received placebo. Overall, 19% of patients treated with Orenitram discontinued treatment in Study 4 due to an adverse event (compared to 4% of patients receiving placebo). The exposure to Orenitram in Study 4 was up to 5.1 years with a median duration of exposure of 1.2 years. Table 1 summarizes adverse events with rates at least 5% higher on Orenitram therapy than on placebo that were reported in either Study 1 or 4.
| Reaction | Study 1 N=228 Includes all subjects in the Primary Analysis Population
|
Study 4 N=690 |
||
|---|---|---|---|---|
| Orenitram n=151 |
Placebo n=77 |
Orenitram n=346 |
Placebo n=344 |
|
| Headache | 63% | 19% | 75% | 35% |
| Diarrhea | 30% | 16% | 69% | 29% |
| Nausea | 30% | 18% | 40% | 23% |
| Vomiting | 17% | 16% | 36% | 10% |
| Flushing | 15% | 6% | 45% | 8% |
| Pain in jaw | 11% | 4% | 18% | 3% |
| Pain in extremity | 14% | 8% | 18% | 9% |
| Hypokalemia | 9% | 3% | 4% | 3% |
| Abdominal discomfort | 6% | 0% | 8% | 4% |
| Upper abdominal pain | 5% | 3% | 12% | 5% |
Orenitram was studied in a long-term, open-label, extension study in which 824 patients were dosed for a mean duration of approximately 2 years. About 70% of patients continued treatment with Orenitram for at least a year. The mean dose was 4.2 mg BID at one year. The adverse reactions were similar to those observed in the placebo-controlled trials.
The safety of Orenitram was also evaluated in an open-label study transitioning patients from Remodulin. The safety profile during this study was similar to that observed in the three pivotal studies.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA-approved patient labeling (Patient Information).
Tell patients:
- Abrupt discontinuation of therapy could result in worsening of PAH symptoms.
- Take Orenitram with food.
- Swallow Orenitram tablets whole. Do not split, chew, crush, or break. Do not take a tablet that is damaged or broken.
- The biologically inert components of the tablet remain intact during gastrointestinal transit and are eliminated in the feces as an insoluble shell.
8.7 Patients With Renal Impairment (8.7 Patients with Renal Impairment)
No dose adjustments are required in patients with renal impairment. Orenitram is not removed by dialysis [see Clinical Pharmacology (12.3)].
1.1 Pulmonary Arterial Hypertension
Orenitram is indicated for the treatment of pulmonary arterial hypertension (PAH) (WHO Group 1) to delay disease progression and to improve exercise capacity.
The studies that established effectiveness included predominately patients with WHO functional class II-III symptoms and etiologies of idiopathic or heritable PAH (66%) or PAH associated with connective tissue disease (26%).
8.6 Patients With Hepatic Impairment (8.6 Patients with Hepatic Impairment)
There is a marked increase in the systemic exposure to treprostinil in hepatically impaired patients [see Dosage and Administration (2.3), Contraindications (4), and Clinical Pharmacology (12.3)].
2.5 Interruptions and Discontinuation
If a dose of medication is missed, the patient should take the missed dose as soon as possible, with food. If a patient misses two or more doses, restart at a lower dose and re-titrate.
In the event of a planned short-term treatment interruption for patients unable to take oral medications, consider a temporary infusion of subcutaneous or intravenous treprostinil. To calculate the total daily dose (mg) of treprostinil for the parenteral route use the following equation:
| Remodulin (ng/kg/min) = | 139 × Orenitram total daily dose (mg) |
| weight (kg) |
When discontinuing Orenitram, reduce the dose in steps of 0.5 to 1 mg per day [see Warnings and Precautions (5.1)].
5.2 Use in Patients With Blind End Pouches (5.2 Use in Patients with Blind-end Pouches)
The tablet shell does not dissolve. In patients with diverticulosis, Orenitram tablets can lodge in a diverticulum.
7.1 Effect of Cyp2c8 Inhibitors On Treprostinil (7.1 Effect of CYP2C8 Inhibitors on Treprostinil)
Co-administration of Orenitram and the CYP2C8 enzyme inhibitor gemfibrozil in healthy adult volunteers increases exposure to treprostinil. Reduce the starting dose of Orenitram to 0.125 mg BID and use 0.125 mg BID increments not more frequently than every 3 to 4 days [see Dosage and Administration (2.4) and Clinical Pharmacology (12.3)].
5.1 Worsening Pah Symptoms Upon Abrupt Withdrawal (5.1 Worsening PAH Symptoms upon Abrupt Withdrawal)
Abrupt discontinuation or sudden large reductions in dosage of Orenitram may result in worsening of PAH symptoms.
2.4 Dose Adjustment for Use With Cyp2c8 Inhibitors (2.4 Dose Adjustment for Use with CYP2C8 Inhibitors)
When co-administered with strong CYP2C8 inhibitors (e.g., gemfibrozil) the initial dose is 0.125 mg BID with 0.125 mg BID dose increments not more frequently than every 3 to 4 days.
Principal Display Panel 1 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Label)
NDC 66302-310-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
1 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
Principal Display Panel 5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label)
NDC 66302-350-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
5 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
Principal Display Panel 2.5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 2.5 mg Tablet Bottle Label)
NDC 66302-325-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
2.5 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
Principal Display Panel 0.25 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 0.25 mg Tablet Bottle Label)
NDC 66302-302-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
0.25 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
Principal Display Panel 0.125 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 0.125 mg Tablet Bottle Label)
NDC 66302-300-01
100 Tablets
Orenitram®
(treprostinil)
Extended-Release
Tablets
0.125 mg
Swallow whole.
Do not split, chew, crush,
or break tablets.
14.1 Clinical Trials in Pulmonary Arterial Hypertension
Four multicenter, randomized, double-blind studies were conducted and compared Orenitram to placebo in a total of 349 (Study 1), 350 (Study 2), 310 (Study 3), and 690 (Study 4) patients with PAH.
2.3 Dose Adjustment in Patients With Hepatic Impairment (2.3 Dose Adjustment in Patients with Hepatic Impairment)
In patients with mild hepatic impairment (Child Pugh Class A) start at 0.125 mg BID with 0.125 mg BID dose increments not more frequently than every 3 to 4 days. Avoid use of Orenitram in patients with moderate hepatic impairment (Child Pugh Class B). Orenitram is contraindicated in patients with severe hepatic impairment (Child Pugh Class C) due to increases in systemic exposure [see Contraindications (4), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Treprostinil diolamine did not demonstrate any carcinogenic effects in mouse or rat carcinogenicity studies. Oral administration of treprostinil diolamine to Tg.rasH2 mice at 0, 5, 10, and 20 mg/kg/day in males and 0, 3, 7.5, and 15 mg/kg/day in females daily for 26 weeks did not significantly increase the incidence of tumors. Oral administration of treprostinil diolamine to Sprague Dawley rats at 0, 1, 3, and 10 mg/kg/day daily for 104 weeks did not significantly increase the incidence of tumors. The exposures obtained at the highest dose levels used in males and females are about 13- and 18-fold, respectively, the human exposure at the dose of 3.5 mg BID on an AUC basis.
In vitro genotoxicity studies with high doses of treprostinil did not demonstrate any mutagenic or clastogenic effects. Treprostinil diolamine was tested in vivo in a rat micronucleus assay and did not induce an increased incidence of micronucleated polychromatic erythrocytes.
In rats, treatment with treprostinil diolamine had no effect on reproductive performance or sperm motility at doses up to 10 mg/kg/day. The exposures at this dose level are about 6- (male) to 11- (female) fold the human exposure at the dose of 3.5 mg BID on an AUC basis.
Principal Display Panel 1 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 1 mg Tablet Blister Pack Carton)
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
1 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-310-02
Principal Display Panel 5 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 5 mg Tablet Blister Pack Carton)
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
5 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-350-02
Principal Display Panel 2.5 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 2.5 mg Tablet Blister Pack Carton)
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
2.5 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-325-02
Principal Display Panel 0.25 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 0.25 mg Tablet Blister Pack Carton)
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
0.25 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-302-02
Principal Display Panel 0.125 Mg Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 0.125 mg Tablet Blister Pack Carton)
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
0.125 mg
per tablet
Swallow whole.
Do not split, chew,
crush, or break tablets.
10 Tablets
(1 x 10 tablet blister pack)
Pharmacist:
Dispense the accompanying Patient
Information Leaflet to each patient.
Rx Only
NDC 66302-300-02
Principal Display Panel 0.125 Mg 0.25 Mg Titration Kit Carton Month 1 (PRINCIPAL DISPLAY PANEL - 0.125 mg 0.25 mg Titration Kit Carton - Month 1)
Rx Only
NDC 66302-361-28
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
Titration Kit
Month 1
0.125 mg
per tablet
0.25 mg
per tablet
Swallow whole.
Do not split, chew, crush, or break tablets.
Kit Contains:
168 tablets (126 x 0.125 mg per tablet and
42 x 0.25 mg per tablet).
Monthly carton contains 4 individual weekly cartons
each with 7 daily wallet blister packs.
Principal Display Panel 0.125 Mg 0.25 Mg Titration Kit Carton Month 2 (PRINCIPAL DISPLAY PANEL - 0.125 mg 0.25 mg Titration Kit Carton - Month 2)
Rx Only
NDC 66302-362-56
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
Titration Kit
Month 2
0.125 mg
per tablet
0.25 mg
per tablet
Swallow whole.
Do not split, chew, crush, or break tablets.
Kit Contains:
336 tablets (126 x 0.125 mg per tablet and
210 x 0.25 mg per tablet).
Monthly carton contains 4 individual weekly cartons
each with 7 daily wallet blister packs.
Principal Display Panel 0.125 Mg 0.25 Mg 1 Mg Titration Kit Carton Month 3 (PRINCIPAL DISPLAY PANEL - 0.125 mg 0.25 mg 1 mg Titration Kit Carton - Month 3)
Rx Only
NDC 66302-363-84
Orenitram®
(treprostinil)
EXTENDED-RELEASE TABLETS
Titration Kit
Month 3
0.125 mg
per tablet
0.25 mg
per tablet
1 mg
per tablet
Swallow whole.
Do not split, chew, crush, or break tablets.
Kit Contains:
252 tablets (126 x 0.125 mg per tablet,
42 x 0.25 mg per tablet, and 84 × 1 mg per tablet).
Monthly carton contains 4 individual weekly cartons
each with 7 daily wallet blister packs.
2.2 Transitioning From Subcutaneous Or Intravenous Routes of Administration of Treprostinil (2.2 Transitioning from Subcutaneous or Intravenous Routes of Administration of Treprostinil)
Decrease the dose of Remodulin while simultaneously increasing the dose of Orenitram. The dose of Remodulin can be reduced up to 30 ng/kg/min per day and the dose of Orenitram simultaneously increased up to 6 mg per day (2 mg TID) if tolerated. The following equation can be used to estimate a target total daily dose of Orenitram in mg using a patient's dose of intravenous (IV)/subcutaneous (SC) treprostinil (in ng/kg/min) and weight (in kg).
Orenitram total daily dose (mg) = 0.0072 × Remodulin dose (ng/kg/min) × weight (kg)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:01.885655 · Updated: 2026-03-14T22:18:26.158923