These Highlights Do Not Include All The Information Needed To Use Spinosad Topical Suspension Safely And Effectively. See Full Prescribing Information For Spinosad Topical Suspension.
8e87bd3b-db25-4e97-bf57-96c875f67ce1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 04/2021 Dosage and Administration ( 2 ) 04/2021 Warnings and Precautions ( 5 ) 04/2021
Indications and Usage
Spinosad Topical Suspension is a pediculicide indicated for the topical treatment of head lice infestations in adult and pediatric patients 6 months of age and older. ( 1.1 ) Spinosad Topical Suspension is a scabicide indicated for the topical treatment of scabies infestations in adult and pediatric patients 4 years of age and older, ( 1.2 )
Dosage and Administration
For topical use only. Not for oral, ophthalmic, or intravaginal use. ( 2 ) Treatment of head lice infestations ( 2.2 ): Shake bottle well Apply a sufficient amount to cover dry scalp, then apply to dry hair Rinse off with warm water after 10 minutes Repeat treatment only if live lice are seen 7 days after first treatment Treatment of scabies infestations ( 2.3 ): Shake bottle well Apply product to skin by rubbing it in to completely cover the body from the neck down to the soles of the feet Patients with balding scalp should also apply product to the scalp, hairline, temples, and forehead Allow to absorb in the skin and dry for 10 minutes before getting dressed Leave on the skin for at least 6 hours before showering or bathing
Warnings and Precautions
Benzyl Alcohol Toxicity: Not recommended in infants below the age of 6 months; potential for increased systemic absorption. ( 5.1 )
Contraindications
None.
Adverse Reactions
Most common adverse events for lice treatment (>1%) were application site erythema and ocular erythema. ( 6.1 ) Most common adverse reactions for scabies treatment (>1%) were application site irritation (pain and burning) and dry skin. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Allegis Pharmaceuticals, LLC at 1-866-633-9033 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Medication Information
Warnings and Precautions
Benzyl Alcohol Toxicity: Not recommended in infants below the age of 6 months; potential for increased systemic absorption. ( 5.1 )
Indications and Usage
Spinosad Topical Suspension is a pediculicide indicated for the topical treatment of head lice infestations in adult and pediatric patients 6 months of age and older. ( 1.1 ) Spinosad Topical Suspension is a scabicide indicated for the topical treatment of scabies infestations in adult and pediatric patients 4 years of age and older, ( 1.2 )
Dosage and Administration
For topical use only. Not for oral, ophthalmic, or intravaginal use. ( 2 ) Treatment of head lice infestations ( 2.2 ): Shake bottle well Apply a sufficient amount to cover dry scalp, then apply to dry hair Rinse off with warm water after 10 minutes Repeat treatment only if live lice are seen 7 days after first treatment Treatment of scabies infestations ( 2.3 ): Shake bottle well Apply product to skin by rubbing it in to completely cover the body from the neck down to the soles of the feet Patients with balding scalp should also apply product to the scalp, hairline, temples, and forehead Allow to absorb in the skin and dry for 10 minutes before getting dressed Leave on the skin for at least 6 hours before showering or bathing
Contraindications
None.
Adverse Reactions
Most common adverse events for lice treatment (>1%) were application site erythema and ocular erythema. ( 6.1 ) Most common adverse reactions for scabies treatment (>1%) were application site irritation (pain and burning) and dry skin. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Allegis Pharmaceuticals, LLC at 1-866-633-9033 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Description
Indications and Usage ( 1 ) 04/2021 Dosage and Administration ( 2 ) 04/2021 Warnings and Precautions ( 5 ) 04/2021
Section 42229-5
Adjunctive Measures for Head Lice Infestations
Spinosad Topical Suspension should be used in the context of an overall lice management program:
- Wash in hot water or dry-clean all recently worn clothing, hats, used bedding and towels.
- Wash personal care items such as combs, brushes and hair clips in hot water.
- A fine-tooth comb or special nit comb may be used to remove dead lice and nits.
Section 42230-3
| Patient Information for Head Lice Treatment
Spinosad Topical Suspension |
|---|
| Important: For use on scalp hair and scalp only. Do not get Spinosad Topical Suspension in your eyes, mouth, or vagina. |
| What is Spinosad Topical Suspension? |
| Spinosad Topical Suspension is a prescription medicine used to treat head lice on the scalp and hair of adults and children 6 months of age and older.
It is not known if Spinosad Topical Suspension is safe and effective for children under 6 months of age. See " How do I stop the spread of lice?" at the end of this leaflet for additional information on ways to stop the spread of lice. |
Before you use Spinosad Topical Suspension, tell your healthcare provider about all of your medical conditions, including if you or your child:
|
| How should I use Spinosad Topical Suspension? |
|
| What are the possible side effects of Spinosad Topical Suspension? |
| The most common side effects of Spinosad Topical Suspension include redness where Spinosad Topical Suspension is applied and redness to eyes. |
| These are not all the possible side effects of Spinosad Topical Suspension. |
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
| How should I store Spinosad Topical Suspension? |
|
| Keep Spinosad Topical Suspension and all medicines out of the reach of children. |
| General information about the safe and effective use of Spinosad Topical Suspension. |
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Spinosad Topical Suspension for a condition for which it was not prescribed. Do not give Spinosad Topical Suspension to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Spinosad Topical Suspension that is written for health professionals. |
| What are the ingredients in Spinosad Topical Suspension? |
| Active ingredient: spinosad |
| Inactive ingredients: Benzyl Alcohol, Butylated Hydroxytoluene, Ceteareth-20, Cetearyl Alcohol, FD&C Yellow #6, Hexylene Glycol, Hydroxyethyl Cellulose, Isopropyl Alcohol, Propylene Glycol, Stearalkonium Chloride, Water, Hydrochloric acid (HCl) as pH adjuster. |
Section 43683-2
Section 44425-7
Storage and Handling
Store at 25° C (77° F); excursions permitted to 15° to 30° C (59° to 86° F).
10 Overdosage
No specific antidotes for spinosad overdosage are known. If oral ingestion occurs, contact Poison Control (1-800-222-1222) for latest recommendations and seek medical attention immediately.
11 Description
Spinosad Topical Suspension is a slightly opaque, light orange-colored, viscous topical suspension.
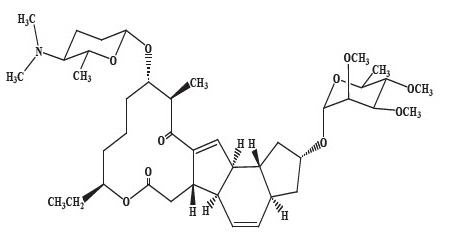
Spinosad Topical Suspension is a pediculicide and scabicide. Spinosad, the active ingredient, is derived from the fermentation of a soil actinomycete bacterium, Saccharopolyspora spinosa.
Spinosad is a mixture of spinosyn A and spinosyn D in a ratio of approximately 5 to 1 (spinosyn A to spinosyn D).
8.5 Geriatric Use
Clinical studies of Spinosad Topical Suspension did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
4 Contraindications
None.
6 Adverse Reactions
Most common adverse events for lice treatment (>1%) were application site erythema and ocular erythema. ( 6.1)
Most common adverse reactions for scabies treatment (>1%) were application site irritation (pain and burning) and dry skin. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Allegis Pharmaceuticals, LLC at 1-866-633-9033 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Instructions for Use
Before you use Spinosad Topical Suspension, it is important that you read this Instructions for Use. Be sure that you read, understand, and follow this Instructions for Use so that you use Spinosad Topical Suspension the right way. Ask your healthcare provider or pharmacist if you have questions about the right way to use Spinosad Topical Suspension.
Important information:
- Your hair and scalp must be dry before applying Spinosad Topical Suspension.
- For very thick, medium length hair or long hair, an entire bottle (120mL) of Spinosad Topical Suspension may be needed to cover the scalp and hair. Less Spinosad Topical Suspension may be needed for shorter, thinner hair.
How to apply Spinosad Topical Suspension to your scalp and hair:
Step 1
- Shake Spinosad Topical Suspension bottle well right before use.
Step 2
- Cover your face and eyes with a towel and keep your eyes closed tightly.
- Apply Spinosad Topical Suspension directly to dry hair and scalp.
- Completely cover the scalp and hair closest to the scalp first, and then apply outwards towards the ends of the hair.
- It is important to apply enough Spinosad Topical Suspension to cover your entire scalp and hair so that all lice and eggs are exposed to Spinosad Topical Suspension.
Step 3
- Allow Spinosad Topical Suspension to stay on your scalp and hair for 10 minutes. Use a timer or clock and start timing after you have completely covered your hair and scalp with Spinosad Topical Suspension.
- Continue to keep eyes covered to prevent dripping into your eyes.
- After 10 minutes, completely rinse Spinosad Topical Suspension from your hair and scalp with warm water.
- You may use a fine-tooth comb to remove treated lice and nits from the hair and scalp, but combing is not required.
- Wash your hands after applying Spinosad Topical Suspension.
- It is okay to shampoo your hair any time after the treatment.
If you see live lice on your scalp or hair one week (7 days), after your first treatment, repeat the steps above.
How do I stop the spread of lice?
To help prevent the spread of lice from one person to another, here are some steps you can take:
- Avoid direct head-to-head contact with anyone known to have live, crawling lice.
- Do not share combs, brushes, hats, scarves, bandannas, ribbons, barrettes, hair bands, towels, helmets, or other hair-related personal items with anyone else, whether they have lice or not.
- Avoid sleepovers and slumber parties during lice outbreaks. Lice can live in bedding, pillows, and carpets that have recently been used by someone with lice.
- After finishing treatment with lice medicine, check everyone in your family for lice after one week. Be sure to talk to your healthcare provider about treatments for those who have lice.
- Machine-wash or dry-clean any bedding, towels and clothing used by anyone having lice. Machine-wash at high temperatures (150°F) and tumble in a hot dryer for 20 minutes.
- Wash personal items such as combs, brushes, and hair clips in hot water.
Distributed by: Allegis Pharmaceuticals LLC, Canton, MS 39046
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Revised: 04/2021
12.2 Pharmacodynamics
The pharmacodynamics of Spinosad Topical Suspension has not been studied.
1 Indications and Usage
Spinosad Topical Suspension is a pediculicide indicated for the topical treatment of head lice infestations in adult and pediatric patients 6 months of age and older. ( 1.1)
Spinosad Topical Suspension is a scabicide indicated for the topical treatment of scabies infestations in adult and pediatric patients 4 years of age and older, ( 1.2)
1.2 Scabies Infestations
Spinosad Topical Suspension is indicated for the topical treatment of scabies infestations in adult and pediatric patients 4 years of age and older.
12.1 Mechanism of Action
Spinosad causes neuronal excitation in insects. After periods of hyperexcitation, lice and mites become paralyzed and die.
14.2 Scabies Infestations
Two multicenter, randomized, double-blind, vehicle-controlled trials (Trial 1 [NCT02485717] and Trial 2 [NCT02485704]) were conducted in subjects from 206 households in which at least one household member 4 years of age or older had an active scabies infestation. An active scabies infestation was defined as the presence of clinical signs and symptoms (evidence of burrows or presence of inflammatory/ noninflammatory lesions and pruritus) as well as microscopic evidence from a skin scraping or dermatoscopy to demonstrate the presence of mites, eggs, and/or scybala. All members of the household were treated with the same randomized treatment (Spinosad Topical Suspension or vehicle), whether or not the household member had an active scabies infestation. Subjects applied a single application of Spinosad Topical Suspension or vehicle on Day 1 and returned for evaluation on Day 28.
The two studies enrolled 533 adult and pediatric subjects 4 years of age and older. A total of 286 subjects (212 adults and 74 pediatrics) were treated with Spinosad Topical Suspension and 247 subjects (176 adults and 71 pediatrics) were treated with vehicle. For the evaluation of efficacy, the youngest subject from each household with an active scabies infestation was considered to be the primary subject of the household, and other members in the household were enrolled in the trial as secondary subjects and evaluated for all safety parameters. In Trial 1, 43 primary subjects were randomized to Spinosad Topical Suspension, and 43 primary subjects were randomized to vehicle. In Trial 2, 62 and 58 primary subjects were randomized to Spinosad Topical Suspension and vehicle, respectively.
Efficacy was assessed as the proportion of primary subjects who achieved complete cure 28 days after treatment. Complete cure is defined as a demonstration of clinical cure (all signs and symptoms have completely resolved, including burrows, inflammatory/non-inflammatory lesions and pruritus) and microscopic or dermatoscopic cure demonstrating the absence of mites, eggs, and/or scybala, and negative dermatoscopy for burrows. Table 4 contains the proportion of primary subjects who achieved complete cure in each of the two trials.
| Trial 1 | Trial 2 | ||||
|---|---|---|---|---|---|
| Spinosad
N=43 |
Vehicle
N=43 |
Estimated Difference
95% CI |
Spinosad
N=62 |
Vehicle
N=58 |
Estimated Difference
95% CI |
| CI= Confidence Interval | |||||
| 30 (69.8%) | 20 (46.5%) | 22.7%
(1.8%, 43.5%) |
52 (83.9%) | 20 (34.5%) | 49.7%
(36.0%, 63.5%) |
1.1 Head Lice Infestations
Spinosad Topical Suspension is indicated for the topical treatment of head lice infestations in adult and pediatric patients 6 months of age and older.
5 Warnings and Precautions
Benzyl Alcohol Toxicity: Not recommended in infants below the age of 6 months; potential for increased systemic absorption. ( 5.1)
14.1 Head Lice Infestations
Two multicenter, randomized, investigator-blind, active-controlled trials were conducted in 1038 subjects 6 months of age and older with head lice infestation. A total of 552 subjects were treated with Spinosad Topical Suspension. For the evaluation of efficacy, the youngest subject from each household was considered to be the primary subject of the household, and other members in the household were enrolled in the study as secondary subjects and evaluated for all safety parameters.
In Study 1, 91 primary subjects were randomized to Spinosad Topical Suspension, and 89 primary subjects were randomized to permethrin 1%. In Study 2, 83 and 84 primary subjects were randomized to Spinosad Topical Suspension and permethrin 1%, respectively.
In both trials, all subjects who were treated on Day 0 returned for efficacy evaluation at Day 7. Subjects with live lice present at Day 7 received a second treatment. Subjects who were lice free on Day 7 were to return on Day 14 for evaluation. Subjects with live lice and who received a second treatment were to return on Days 14 and 21.
Efficacy was assessed as the proportion of primary subjects who were free of live lice 14 days after the final treatment. Table 3 contains the proportion of primary subjects who were free of live lice in each of the two trials.
| Study 1 | Study 2 | ||
|---|---|---|---|
| Spinosad
(N=91) |
Permethrin 1%
(N=89) |
Spinosad
(N=83) |
Permethrin 1%
(N=84) |
| 77 (84.6%) | 40 (44.9%) | 72 (86.7%) | 36 (42.9%) |
2 Dosage and Administration
- For topical use only. Not for oral, ophthalmic, or intravaginal use. ( 2)
- Treatment of head lice infestations (
2.2):
- Shake bottle well
- Apply a sufficient amount to cover dry scalp, then apply to dry hair
- Rinse off with warm water after 10 minutes
- Repeat treatment only if live lice are seen 7 days after first treatment
- Treatment of scabies infestations (
2.3):
- Shake bottle well
- Apply product to skin by rubbing it in to completely cover the body from the neck down to the soles of the feet
- Patients with balding scalp should also apply product to the scalp, hairline, temples, and forehead
- Allow to absorb in the skin and dry for 10 minutes before getting dressed
- Leave on the skin for at least 6 hours before showering or bathing
5.1 Benzyl Alcohol Toxicity
Spinosad Topical Suspension contains benzyl alcohol and is not approved for use in neonates and infants below the age of 6 months. Systemic exposure to benzyl alcohol has been associated with serious adverse reactions and death in neonates and low birth-weight infants when administered intravenously [See Use in Specific Populations (8.4)] .
3 Dosage Forms and Strengths
Topical suspension: 0.9% w/w; each gram contains 9 mg of spinosad in a viscous, slightly opaque (white soft particles may be visible), light orange-colored suspension in 120 mL bottles.
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Patient Information)
2.3 Treatment of Scabies Infestations
- Shake bottle well.
- Apply a sufficient amount of Spinosad Topical Suspension to skin to completely cover the body from the neck to the toes (including the soles of the feet).
- For patients with balding scalp, also apply product to the scalp, hairline, temples, and forehead.
- Allow to absorb into the skin and dry for 10 minutes before getting dressed.
- Leave on the skin for at least 6 hours before showering or bathing.
- Apply Spinosad Topical Suspension on pediatric patient only under direct supervision of an adult .
2.2 Treatment of Head Lice Infestations
- Shake bottle well.
- Apply a sufficient amount of Spinosad Topical Suspension to cover dry scalp, then apply to dry hair. Depending on hair length, apply up to 120 mL (one bottle) to adequately cover scalp and hair.
- Leave on for 10 minutes, then thoroughly rinse off with warm water.
- Wash hands after use.
- If live lice are seen 7 days after the first treatment, a second treatment should be applied.
- Apply Spinosad Topical Suspension on pediatric patient only under direct supervision of an adult [see Warnings and Precautions (5.1)].
2.1 Important Administration Instructions
- For topical use only. Spinosad Topical Suspension is not for oral, ophthalmic, or intravaginal use.
- Avoid contact with eyes. If Spinosad Topical Suspension gets in or near the eyes, rinse thoroughly with water.
Principal Display Panel 120 Ml Bottle Carton
NDC 28595-570-04
Rx Only
Spinosad
Topical Suspension
0.9% w/w
For topical use only
120 mL
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In an oral (diet) mouse carcinogenicity study, spinosad was administered to CD-1 mice at doses of 0.0025, 0.008, and 0.036% in the diet (approximately 3.4, 11.4, and 50.9 mg/kg/day for males and 4.2, 13.8, and 67.0 mg/kg/day for females) for 18 months. No treatment-related tumors were noted in the mouse carcinogenicity study up to the highest doses evaluated in this study of 50.9 mg/kg/day in male mice and 13.8 mg/kg/day in female mice. Female mice treated with a dose of 67.0 mg/kg/day were not evaluated in this study due to high mortality.
In an oral (diet) rat carcinogenicity study, spinosad was administered to Fischer 344 rats at doses of 0.005, 0.02, 0.05, and 0.1% in the diet (approximately 2.4, 9.5, 24.1 and 49.4 mg/kg/day for males and 3.0, 12.0, 30.1 and 62.8 mg/kg/day for females) for 24 months. No treatment-related tumors were noted in the rat carcinogenicity study in male or female rats up to the highest doses evaluated in this study of 24.1 mg/kg/day in male rats and 30.1 mg/kg/day in female rats. Rats in the highest dose group in this study were not evaluated due to high mortality.
Spinosad demonstrated no evidence of mutagenic or clastogenic potential based on the results of four in vitro genotoxicity tests (Ames assay, mouse lymphoma L5178Y assay, Chinese hamster ovary cell chromosome aberration assay, and rat hepatocyte unscheduled DNA synthesis assay) and one in vivo genotoxicity test (mouse bone marrow micronucleus assay).
Oral administration of spinosad (in diet) to rats, throughout mating, gestation, parturition and lactation, demonstrated no effects on growth, fertility or reproduction, at doses up to 10 mg/kg/day [see Use In Specific Populations (8.1)] .
Structured Label Content
Section 42229-5 (42229-5)
Adjunctive Measures for Head Lice Infestations
Spinosad Topical Suspension should be used in the context of an overall lice management program:
- Wash in hot water or dry-clean all recently worn clothing, hats, used bedding and towels.
- Wash personal care items such as combs, brushes and hair clips in hot water.
- A fine-tooth comb or special nit comb may be used to remove dead lice and nits.
Section 42230-3 (42230-3)
| Patient Information for Head Lice Treatment
Spinosad Topical Suspension |
|---|
| Important: For use on scalp hair and scalp only. Do not get Spinosad Topical Suspension in your eyes, mouth, or vagina. |
| What is Spinosad Topical Suspension? |
| Spinosad Topical Suspension is a prescription medicine used to treat head lice on the scalp and hair of adults and children 6 months of age and older.
It is not known if Spinosad Topical Suspension is safe and effective for children under 6 months of age. See " How do I stop the spread of lice?" at the end of this leaflet for additional information on ways to stop the spread of lice. |
Before you use Spinosad Topical Suspension, tell your healthcare provider about all of your medical conditions, including if you or your child:
|
| How should I use Spinosad Topical Suspension? |
|
| What are the possible side effects of Spinosad Topical Suspension? |
| The most common side effects of Spinosad Topical Suspension include redness where Spinosad Topical Suspension is applied and redness to eyes. |
| These are not all the possible side effects of Spinosad Topical Suspension. |
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
| How should I store Spinosad Topical Suspension? |
|
| Keep Spinosad Topical Suspension and all medicines out of the reach of children. |
| General information about the safe and effective use of Spinosad Topical Suspension. |
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use Spinosad Topical Suspension for a condition for which it was not prescribed. Do not give Spinosad Topical Suspension to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about Spinosad Topical Suspension that is written for health professionals. |
| What are the ingredients in Spinosad Topical Suspension? |
| Active ingredient: spinosad |
| Inactive ingredients: Benzyl Alcohol, Butylated Hydroxytoluene, Ceteareth-20, Cetearyl Alcohol, FD&C Yellow #6, Hexylene Glycol, Hydroxyethyl Cellulose, Isopropyl Alcohol, Propylene Glycol, Stearalkonium Chloride, Water, Hydrochloric acid (HCl) as pH adjuster. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling
Store at 25° C (77° F); excursions permitted to 15° to 30° C (59° to 86° F).
10 Overdosage (10 OVERDOSAGE)
No specific antidotes for spinosad overdosage are known. If oral ingestion occurs, contact Poison Control (1-800-222-1222) for latest recommendations and seek medical attention immediately.
11 Description (11 DESCRIPTION)
Spinosad Topical Suspension is a slightly opaque, light orange-colored, viscous topical suspension.
Spinosad Topical Suspension is a pediculicide and scabicide. Spinosad, the active ingredient, is derived from the fermentation of a soil actinomycete bacterium, Saccharopolyspora spinosa.
Spinosad is a mixture of spinosyn A and spinosyn D in a ratio of approximately 5 to 1 (spinosyn A to spinosyn D).
8.5 Geriatric Use
Clinical studies of Spinosad Topical Suspension did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse events for lice treatment (>1%) were application site erythema and ocular erythema. ( 6.1)
Most common adverse reactions for scabies treatment (>1%) were application site irritation (pain and burning) and dry skin. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Allegis Pharmaceuticals, LLC at 1-866-633-9033 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Instructions for Use
Before you use Spinosad Topical Suspension, it is important that you read this Instructions for Use. Be sure that you read, understand, and follow this Instructions for Use so that you use Spinosad Topical Suspension the right way. Ask your healthcare provider or pharmacist if you have questions about the right way to use Spinosad Topical Suspension.
Important information:
- Your hair and scalp must be dry before applying Spinosad Topical Suspension.
- For very thick, medium length hair or long hair, an entire bottle (120mL) of Spinosad Topical Suspension may be needed to cover the scalp and hair. Less Spinosad Topical Suspension may be needed for shorter, thinner hair.
How to apply Spinosad Topical Suspension to your scalp and hair:
Step 1
- Shake Spinosad Topical Suspension bottle well right before use.
Step 2
- Cover your face and eyes with a towel and keep your eyes closed tightly.
- Apply Spinosad Topical Suspension directly to dry hair and scalp.
- Completely cover the scalp and hair closest to the scalp first, and then apply outwards towards the ends of the hair.
- It is important to apply enough Spinosad Topical Suspension to cover your entire scalp and hair so that all lice and eggs are exposed to Spinosad Topical Suspension.
Step 3
- Allow Spinosad Topical Suspension to stay on your scalp and hair for 10 minutes. Use a timer or clock and start timing after you have completely covered your hair and scalp with Spinosad Topical Suspension.
- Continue to keep eyes covered to prevent dripping into your eyes.
- After 10 minutes, completely rinse Spinosad Topical Suspension from your hair and scalp with warm water.
- You may use a fine-tooth comb to remove treated lice and nits from the hair and scalp, but combing is not required.
- Wash your hands after applying Spinosad Topical Suspension.
- It is okay to shampoo your hair any time after the treatment.
If you see live lice on your scalp or hair one week (7 days), after your first treatment, repeat the steps above.
How do I stop the spread of lice?
To help prevent the spread of lice from one person to another, here are some steps you can take:
- Avoid direct head-to-head contact with anyone known to have live, crawling lice.
- Do not share combs, brushes, hats, scarves, bandannas, ribbons, barrettes, hair bands, towels, helmets, or other hair-related personal items with anyone else, whether they have lice or not.
- Avoid sleepovers and slumber parties during lice outbreaks. Lice can live in bedding, pillows, and carpets that have recently been used by someone with lice.
- After finishing treatment with lice medicine, check everyone in your family for lice after one week. Be sure to talk to your healthcare provider about treatments for those who have lice.
- Machine-wash or dry-clean any bedding, towels and clothing used by anyone having lice. Machine-wash at high temperatures (150°F) and tumble in a hot dryer for 20 minutes.
- Wash personal items such as combs, brushes, and hair clips in hot water.
Distributed by: Allegis Pharmaceuticals LLC, Canton, MS 39046
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Revised: 04/2021
12.2 Pharmacodynamics
The pharmacodynamics of Spinosad Topical Suspension has not been studied.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Spinosad Topical Suspension is a pediculicide indicated for the topical treatment of head lice infestations in adult and pediatric patients 6 months of age and older. ( 1.1)
Spinosad Topical Suspension is a scabicide indicated for the topical treatment of scabies infestations in adult and pediatric patients 4 years of age and older, ( 1.2)
1.2 Scabies Infestations
Spinosad Topical Suspension is indicated for the topical treatment of scabies infestations in adult and pediatric patients 4 years of age and older.
12.1 Mechanism of Action
Spinosad causes neuronal excitation in insects. After periods of hyperexcitation, lice and mites become paralyzed and die.
14.2 Scabies Infestations
Two multicenter, randomized, double-blind, vehicle-controlled trials (Trial 1 [NCT02485717] and Trial 2 [NCT02485704]) were conducted in subjects from 206 households in which at least one household member 4 years of age or older had an active scabies infestation. An active scabies infestation was defined as the presence of clinical signs and symptoms (evidence of burrows or presence of inflammatory/ noninflammatory lesions and pruritus) as well as microscopic evidence from a skin scraping or dermatoscopy to demonstrate the presence of mites, eggs, and/or scybala. All members of the household were treated with the same randomized treatment (Spinosad Topical Suspension or vehicle), whether or not the household member had an active scabies infestation. Subjects applied a single application of Spinosad Topical Suspension or vehicle on Day 1 and returned for evaluation on Day 28.
The two studies enrolled 533 adult and pediatric subjects 4 years of age and older. A total of 286 subjects (212 adults and 74 pediatrics) were treated with Spinosad Topical Suspension and 247 subjects (176 adults and 71 pediatrics) were treated with vehicle. For the evaluation of efficacy, the youngest subject from each household with an active scabies infestation was considered to be the primary subject of the household, and other members in the household were enrolled in the trial as secondary subjects and evaluated for all safety parameters. In Trial 1, 43 primary subjects were randomized to Spinosad Topical Suspension, and 43 primary subjects were randomized to vehicle. In Trial 2, 62 and 58 primary subjects were randomized to Spinosad Topical Suspension and vehicle, respectively.
Efficacy was assessed as the proportion of primary subjects who achieved complete cure 28 days after treatment. Complete cure is defined as a demonstration of clinical cure (all signs and symptoms have completely resolved, including burrows, inflammatory/non-inflammatory lesions and pruritus) and microscopic or dermatoscopic cure demonstrating the absence of mites, eggs, and/or scybala, and negative dermatoscopy for burrows. Table 4 contains the proportion of primary subjects who achieved complete cure in each of the two trials.
| Trial 1 | Trial 2 | ||||
|---|---|---|---|---|---|
| Spinosad
N=43 |
Vehicle
N=43 |
Estimated Difference
95% CI |
Spinosad
N=62 |
Vehicle
N=58 |
Estimated Difference
95% CI |
| CI= Confidence Interval | |||||
| 30 (69.8%) | 20 (46.5%) | 22.7%
(1.8%, 43.5%) |
52 (83.9%) | 20 (34.5%) | 49.7%
(36.0%, 63.5%) |
1.1 Head Lice Infestations
Spinosad Topical Suspension is indicated for the topical treatment of head lice infestations in adult and pediatric patients 6 months of age and older.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Benzyl Alcohol Toxicity: Not recommended in infants below the age of 6 months; potential for increased systemic absorption. ( 5.1)
14.1 Head Lice Infestations
Two multicenter, randomized, investigator-blind, active-controlled trials were conducted in 1038 subjects 6 months of age and older with head lice infestation. A total of 552 subjects were treated with Spinosad Topical Suspension. For the evaluation of efficacy, the youngest subject from each household was considered to be the primary subject of the household, and other members in the household were enrolled in the study as secondary subjects and evaluated for all safety parameters.
In Study 1, 91 primary subjects were randomized to Spinosad Topical Suspension, and 89 primary subjects were randomized to permethrin 1%. In Study 2, 83 and 84 primary subjects were randomized to Spinosad Topical Suspension and permethrin 1%, respectively.
In both trials, all subjects who were treated on Day 0 returned for efficacy evaluation at Day 7. Subjects with live lice present at Day 7 received a second treatment. Subjects who were lice free on Day 7 were to return on Day 14 for evaluation. Subjects with live lice and who received a second treatment were to return on Days 14 and 21.
Efficacy was assessed as the proportion of primary subjects who were free of live lice 14 days after the final treatment. Table 3 contains the proportion of primary subjects who were free of live lice in each of the two trials.
| Study 1 | Study 2 | ||
|---|---|---|---|
| Spinosad
(N=91) |
Permethrin 1%
(N=89) |
Spinosad
(N=83) |
Permethrin 1%
(N=84) |
| 77 (84.6%) | 40 (44.9%) | 72 (86.7%) | 36 (42.9%) |
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For topical use only. Not for oral, ophthalmic, or intravaginal use. ( 2)
- Treatment of head lice infestations (
2.2):
- Shake bottle well
- Apply a sufficient amount to cover dry scalp, then apply to dry hair
- Rinse off with warm water after 10 minutes
- Repeat treatment only if live lice are seen 7 days after first treatment
- Treatment of scabies infestations (
2.3):
- Shake bottle well
- Apply product to skin by rubbing it in to completely cover the body from the neck down to the soles of the feet
- Patients with balding scalp should also apply product to the scalp, hairline, temples, and forehead
- Allow to absorb in the skin and dry for 10 minutes before getting dressed
- Leave on the skin for at least 6 hours before showering or bathing
5.1 Benzyl Alcohol Toxicity
Spinosad Topical Suspension contains benzyl alcohol and is not approved for use in neonates and infants below the age of 6 months. Systemic exposure to benzyl alcohol has been associated with serious adverse reactions and death in neonates and low birth-weight infants when administered intravenously [See Use in Specific Populations (8.4)] .
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Topical suspension: 0.9% w/w; each gram contains 9 mg of spinosad in a viscous, slightly opaque (white soft particles may be visible), light orange-colored suspension in 120 mL bottles.
6.1 Clinical Studies Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Patient Information)
2.3 Treatment of Scabies Infestations
- Shake bottle well.
- Apply a sufficient amount of Spinosad Topical Suspension to skin to completely cover the body from the neck to the toes (including the soles of the feet).
- For patients with balding scalp, also apply product to the scalp, hairline, temples, and forehead.
- Allow to absorb into the skin and dry for 10 minutes before getting dressed.
- Leave on the skin for at least 6 hours before showering or bathing.
- Apply Spinosad Topical Suspension on pediatric patient only under direct supervision of an adult .
2.2 Treatment of Head Lice Infestations
- Shake bottle well.
- Apply a sufficient amount of Spinosad Topical Suspension to cover dry scalp, then apply to dry hair. Depending on hair length, apply up to 120 mL (one bottle) to adequately cover scalp and hair.
- Leave on for 10 minutes, then thoroughly rinse off with warm water.
- Wash hands after use.
- If live lice are seen 7 days after the first treatment, a second treatment should be applied.
- Apply Spinosad Topical Suspension on pediatric patient only under direct supervision of an adult [see Warnings and Precautions (5.1)].
2.1 Important Administration Instructions
- For topical use only. Spinosad Topical Suspension is not for oral, ophthalmic, or intravaginal use.
- Avoid contact with eyes. If Spinosad Topical Suspension gets in or near the eyes, rinse thoroughly with water.
Principal Display Panel 120 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 120 mL Bottle Carton)
NDC 28595-570-04
Rx Only
Spinosad
Topical Suspension
0.9% w/w
For topical use only
120 mL
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In an oral (diet) mouse carcinogenicity study, spinosad was administered to CD-1 mice at doses of 0.0025, 0.008, and 0.036% in the diet (approximately 3.4, 11.4, and 50.9 mg/kg/day for males and 4.2, 13.8, and 67.0 mg/kg/day for females) for 18 months. No treatment-related tumors were noted in the mouse carcinogenicity study up to the highest doses evaluated in this study of 50.9 mg/kg/day in male mice and 13.8 mg/kg/day in female mice. Female mice treated with a dose of 67.0 mg/kg/day were not evaluated in this study due to high mortality.
In an oral (diet) rat carcinogenicity study, spinosad was administered to Fischer 344 rats at doses of 0.005, 0.02, 0.05, and 0.1% in the diet (approximately 2.4, 9.5, 24.1 and 49.4 mg/kg/day for males and 3.0, 12.0, 30.1 and 62.8 mg/kg/day for females) for 24 months. No treatment-related tumors were noted in the rat carcinogenicity study in male or female rats up to the highest doses evaluated in this study of 24.1 mg/kg/day in male rats and 30.1 mg/kg/day in female rats. Rats in the highest dose group in this study were not evaluated due to high mortality.
Spinosad demonstrated no evidence of mutagenic or clastogenic potential based on the results of four in vitro genotoxicity tests (Ames assay, mouse lymphoma L5178Y assay, Chinese hamster ovary cell chromosome aberration assay, and rat hepatocyte unscheduled DNA synthesis assay) and one in vivo genotoxicity test (mouse bone marrow micronucleus assay).
Oral administration of spinosad (in diet) to rats, throughout mating, gestation, parturition and lactation, demonstrated no effects on growth, fertility or reproduction, at doses up to 10 mg/kg/day [see Use In Specific Populations (8.1)] .
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:22.667810 · Updated: 2026-03-14T22:40:12.922880