Cycloserine Capsules Usp, 250 Mg
8e7e2665-7a3d-3f54-9f92-5fe845f02ef9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
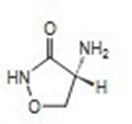
D -Cycloserine, (R)-4-amino-3-isoxazolidinone, is a broad-spectrum antibiotic that is produced by a strain of Streptomyces orchidaceus and has also been synthesized. Cycloserine is a white to off-white powder that is soluble in water and stable in alkaline solution. It is rapidly destroyed at a neutral or acid pH. Cycloserine has a pH between 5.5 and 6.5 in a solution containing 100 mg/mL. The molecular weight of cycloserine is 102.09, and it has an empirical formula of C 3H6N2O 2 . The structural formula of cycloserine is as follows: Each capsule contains cycloserine, 250 mg (2.45 mmol); D & C Yellow No. 10, F D & C Blue No. 1, F D & C Red No. 3, F D & C Yellow No. 6, gelatin, iron oxide, talc, and titanium dioxide.
Indications and Usage
Cycloserine is indicated in the treatment of active pulmonary and extrapulmonary tuberculosis (including renal disease) when the causative organisms are susceptible to this drug and when treatment with the primary medications (streptomycin, isoniazid, rifampin, and ethambutol) has proved inadequate. Like all antituberculosis drugs, cycloserine should be administered in conjunction with other effective chemotherapy and not as the sole therapeutic agent. Cycloserine may be effective in the treatment of acute urinary tract infections caused by susceptible strains of gram-positive and gram- negative bacteria. Use of cycloserine in these infections should be considered only when more conventional therapy has failed and when the organism has been demonstrated to be susceptible to the drug
Dosage and Administration
Cycloserine is effective orally and is currently administered only by this route. The usual dosage is 500 mg to 1 g daily in divided doses monitored by blood levels. 1 The initial adult dosage most frequently given is 250 mg twice daily at 12-hour intervals for the first 2 weeks. A daily dosage of 1 g should not be exceeded.
Contraindications
Administration is contraindicated in patients with any of the following: • Hypersensitivity to cycloserine • Epilepsy • Depression, severe anxiety, or psychosis • Severe renal insufficiency • Excessive concurrent use of alcoho
Adverse Reactions
Most adverse reactions occurring during therapy with cycloserine involve the nervous system or are manifestations of drug hypersensitivity. The following side effects have been observed in patients receiving cycloserine: Nervous system symptoms (which appear to be related to higher dosages of the drug, i.e., more than 500 mg daily) • Convulsions • Drowsiness and somnolence • Headache • Tremor • Dysarthria • Vertigo • Confusion and disorientation with loss of memory • Psychoses, possibly with suicidal tendencies • Character changes • Hyperirritability • Aggression • Paresis • Hyperreflexia • Paresthesia • Major & minor (localized) clonic seizures • Coma Cardiovascular: Sudden development of congestive heart failure in patients receiving 1 to 1.5 g of cycloserine daily has been reported. Allergy (apparently not related to dosage) Skin rash Miscellaneous: Elevated serum transaminase, especially in patients with preexisting liver disease To report SUSPECTED ADVERSE REACTIONS, contact Cerovene, Inc. at 1-833-304-9569 or FDA at 1‑800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
Cycloserine is available as a 250 mg capsule with an opaque red cap and opaque gray body imprinted with “PGC” and “F04” in edible black ink on both the cap and the body. Cycloserine Capsules, USP are supplied in cartons of 3 cards. Each card is a ten-capsules blister card as follows: NDC 43598-235-31: carton with 3 cards. NDC 43598-235-10 (ten 250mg capsules per card). Store at controlled room temperature, 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Medication Information
Indications and Usage
Cycloserine is indicated in the treatment
of active pulmonary and extrapulmonary
tuberculosis (including renal disease) when
the causative organisms are susceptible to
this drug and when treatment with the primary
medications (streptomycin, isoniazid, rifampin,
and ethambutol) has proved inadequate. Like
all antituberculosis drugs, cycloserine should be
administered in conjunction with other effective
chemotherapy and not as the sole therapeutic
agent.
Cycloserine may be effective in the treatment
of acute urinary tract infections caused by
susceptible strains of gram-positive and gram-
negative bacteria. Use of cycloserine in these
infections should be considered only when
more conventional therapy has failed and when
the organism has been demonstrated to be
susceptible to the drug
Dosage and Administration
Cycloserine is effective orally and is currently
administered only by this route. The usual
dosage is 500 mg to 1 g daily in divided doses
monitored by blood levels.1 The initial adult
dosage most frequently given is 250 mg twice
daily at 12-hour intervals for the first 2 weeks.
A daily dosage of 1 g should not be exceeded.
Contraindications
Administration is contraindicated in patients with
any of the following:
• Hypersensitivity to cycloserine
• Epilepsy
• Depression, severe anxiety, or psychosis
• Severe renal insufficiency
• Excessive concurrent use of alcoho
Adverse Reactions
Most adverse reactions occurring during therapy
with cycloserine involve the nervous system or
are manifestations of drug hypersensitivity. The
following side effects have been observed in
patients receiving cycloserine:
Nervous system symptoms (which appear to
be related to higher dosages of the drug, i.e.,
more than 500 mg daily)
• Convulsions
• Drowsiness and somnolence
• Headache
• Tremor
• Dysarthria
• Vertigo
• Confusion and disorientation with loss of
memory
• Psychoses, possibly with suicidal tendencies
• Character changes
• Hyperirritability
• Aggression
• Paresis
• Hyperreflexia
• Paresthesia
• Major & minor (localized) clonic seizures
• Coma
Cardiovascular: Sudden development of
congestive heart failure in patients receiving
1 to 1.5 g of cycloserine daily has been
reported.
Allergy (apparently not related to dosage)
Skin rash
Miscellaneous: Elevated serum transaminase,
especially in patients with preexisting liver
disease
To report SUSPECTED ADVERSE REACTIONS, contact Cerovene, Inc. at 1-833-304-9569 or FDA at 1‑800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
Cycloserine is available as a 250 mg capsule
with an opaque red cap and opaque gray body
imprinted with “PGC” and “F04” in edible black
ink on both the cap and the body.
Cycloserine Capsules, USP are supplied in cartons of 3 cards. Each card is a ten-capsules blister card as follows:
NDC 43598-235-31: carton with 3 cards.
NDC 43598-235-10 (ten 250mg capsules per card).
Store at controlled room temperature,
20° to 25°C (68° to 77°F) [see USP Controlled
Room Temperature].
Description
D -Cycloserine, (R)-4-amino-3-isoxazolidinone,
is a broad-spectrum antibiotic that is produced
by a strain of Streptomyces orchidaceus and has
also been synthesized. Cycloserine is a white
to off-white powder that is soluble in water and
stable in alkaline solution. It is rapidly destroyed
at a neutral or acid pH.
Cycloserine has a pH between 5.5 and 6.5 in a
solution containing 100 mg/mL. The molecular
weight of cycloserine is 102.09, and it has an
empirical formula of C 3H6N2O 2 . The structural
formula of cycloserine is as follows:
Each capsule contains cycloserine, 250 mg
(2.45 mmol); D & C Yellow No. 10, F D & C Blue
No. 1, F D & C Red No. 3, F D & C Yellow No.
6, gelatin, iron oxide, talc, and titanium dioxide.
Section 42229-5
Distributed By:
Dr. Reddy’s Laboratories, Inc.
Princeton, NJ 08540
Rev 02/23
Warnings
Administration of cycloserine should be
discontinued or the dosage reduced if
the patient develops allergic dermatitis or
symptoms of CNS toxicity, such as convulsions,
psychosis, somnolence, depression, confusion,
hyperreflexia, headache, tremor, vertigo
paresis, or dysarthria.
The toxicity of cycloserine is closely related to
excessive blood levels (above 30 mcg/mL), as
determined by high dosage or inadequate renal
clearance. The ratio of toxic dose to effective
dose in tuberculosis is small.
The risk of convulsions is increased in chronic
alcoholics.
Patients should be monitored by hematologic,
renal excretion, blood level, and liver function
studies.
Overdosage
Signs and Symptoms: Acute toxicity from
cycloserine can occur if more than 1 g is
ingested by an adult. Chronic toxicity from
cycloserine is dose related and can occur if
more than 500 mg is administered daily. The
central nervous system is the most common
organ system involved with toxicity. Toxic effects
may include headache, vertigo, confusion,
drowsiness, hyperirritability, paresthesias,
dysarthria, psychosis paresis, convulsions, and
coma.
Treatment: In adults, many of the neurotoxic
effects of cycloserine can be both treated and
prevented with the administration of 200 to 300 mg
of pyridoxine daily.
Hemodialysis has been shown to remove
cycloserine from the bloodstream. This
procedure should be reserved for patients with
life threatening toxicity that is unresponsive to
less invasive therapy.
References
1. Jones LR: Colorimetric determination of
cycloserine, a new antibiotic. Anal Chem
1956;28:39.
Precautions
General: Before treatment with cycloserine
is initiated, cultures should be taken and the
organism’s susceptibility to the drug should
be established. In tuberculous infections,
the organism’s susceptibility to the other
antituberculosis agents in the regimen should
also be demonstrated.
Anticonvulsant drugs or sedatives may be
effective in controlling symptoms of CNS
toxicity, such as convulsions, anxiety, and
tremor. Patients receiving more than 500 mg
of cycloserine daily should be closely observed
for such symptoms. The value of pyridoxine in
preventing CNS toxicity from cycloserine has
not been proved.
Administration of cycloserine and other
antituberculosis drugs has been associated in
a few instances with vitamin B 12 and/or folic-
acid deficiency, megaloblastic anemia, and
sideroblastic anemia. If evidence of anemia
develops during treatment, appropriate studies
and therapy should be instituted.
Laboratory Tests: Blood levels should be
determined at least weekly for patients with
reduced renal function, for individuals receiving
a daily dosage of more than 500 mg, and for
those showing signs and symptoms suggestive
of toxicity. The dosage should be adjusted to
keep the blood level below 30 mcg/mL.
Drug Interactions: Concurrent administration
of ethionamide has been reported to potentiate
neurotoxic side effects.
Alcohol and cycloserine are incompatible,
especially during a regimen calling for large
doses of the latter. Alcohol increases the
possibility and risk of epileptic episodes.
Concurrent administration of isoniazid may
result in increased incidence of CNS effects,
such as dizziness or drowsiness. Dosage
adjustments may be necessary and patients
should be monitored closely for signs of CNS
toxicity.
Carcinogenesis, Mutagenicity, and
Impairment of Fertility: Studies have not
been performed to determine potential for
carcinogenicity. The Ames test and unscheduled
DNA repair test were negative. A study in 2
generations of rats showed no impairment of
fertility relative to controls for the first mating but
somewhat lower fertility in the second mating.
Pregnancy Category C: There are no
adequate and well-controlled studies with
the use of Cycloserine in pregnant women.
A study in 2 generations of rats given doses
up to 100 mg/kg/day (approximately equivalent
to the maximum recommended human dose
on a body surface area basis) demonstrated
no teratogenic effect in offspring. Cycloserine
should be used during pregnancy only if the
potential benefit justifies the potential risk to the
fetus.
Nursing Mothers: Because of the potential
for serious adverse reactions in nursing infants
from cycloserine, a decision should be made
whether to discontinue nursing or to discontinue
the drug, taking into account the importance of
the drug to the mother.
Usage in Pediatric Patients: Safety and
effectiveness in pediatric patients have not been
established.
Geriatric Use: Clinical studies of cycloserine
did not include sufficient numbers of subjects
aged 65 and over to determine whether they
responded differently from younger subjects.
Other reported clinical experience has not
identified differences in responses between
the elderly and younger patients. In general,
dose selection for an elderly patient should be
cautious, usually starting at the low end of the
dosing range, reflecting the greater frequency
of decreased hepatic, renal, or cardiac function,
and of concomitant disease or other drug
therapy.
This drug is known to be substantially excreted
by the kidney, and the risk of toxic reactions
to this drug may be greater in patients with
impaired renal function. Because elderly
patients are more likely to have decreased renal
function, care should be taken in dose selection,
and it may be useful to monitor renal function.
The toxicity of cycloserine is closely related
to excessive blood levels (above 30 mcg/mL)
as determined by high dosage or inadequate
renal clearance (see WARNINGS). Blood
levels should be determined at least weekly
for patients with reduced renal function, for
individuals receiving a daily dosage of more
than 500 mg, and for those showing signs
and symptoms suggestive of toxicity. The
dosage should be adjusted to keep the blood
level below 30 mcg/mL (see PRECAUTIONS,
Laboratory Tests).
Clinical Pharmacology
After oral administration, cycloserine is
readily absorbed from the gastrointestinal
tract, with peak blood levels occurring in 4
to 8 hours. Blood levels of 25 to 30 mcg/mL
can generally be maintained with the usual
dosage of 250 mg twice a day, although the
relationship of plasma levels to dosage is
not always consistent. Concentrations in the
cerebrospinal fluid, pleural fluid, fetal blood,
and mother’s milk approach those found in
the serum. Detectable amounts are found
in ascitic fluid, file, sputum, amniotic fluid,
and lung and lymph tissues. Approximately
65% of a single dose of cycloserine can
be recovered in the urine within 72 hours
after oral administration. The remaining
35% is apparently metabolized to unknown
substances. The maximum excretion rate
occurs 2 to 6 hours after administration, with
50% of the drug eliminated in 12 hours.
Mechanism of Action: The antibacterial activity
of Cycloserine results from inhibition of cell-wall
synthesis in susceptible strains of gram-positive
and gram-negative bacteria.
Antibacterial Activity: Cycloserine has been
shown to be active against most isolates of the
following microorganism, both in vitro and in
clinical infections [see Indications and Usage]:
Mycobacterium tuberculosis.
Package Label Display
Cycloserine Capsules USP, 250 mg - Carton Label
Structured Label Content
Section 42229-5 (42229-5)
Distributed By:
Dr. Reddy’s Laboratories, Inc.
Princeton, NJ 08540
Rev 02/23
Warnings (WARNINGS)
Administration of cycloserine should be
discontinued or the dosage reduced if
the patient develops allergic dermatitis or
symptoms of CNS toxicity, such as convulsions,
psychosis, somnolence, depression, confusion,
hyperreflexia, headache, tremor, vertigo
paresis, or dysarthria.
The toxicity of cycloserine is closely related to
excessive blood levels (above 30 mcg/mL), as
determined by high dosage or inadequate renal
clearance. The ratio of toxic dose to effective
dose in tuberculosis is small.
The risk of convulsions is increased in chronic
alcoholics.
Patients should be monitored by hematologic,
renal excretion, blood level, and liver function
studies.
Overdosage (OVERDOSAGE)
Signs and Symptoms: Acute toxicity from
cycloserine can occur if more than 1 g is
ingested by an adult. Chronic toxicity from
cycloserine is dose related and can occur if
more than 500 mg is administered daily. The
central nervous system is the most common
organ system involved with toxicity. Toxic effects
may include headache, vertigo, confusion,
drowsiness, hyperirritability, paresthesias,
dysarthria, psychosis paresis, convulsions, and
coma.
Treatment: In adults, many of the neurotoxic
effects of cycloserine can be both treated and
prevented with the administration of 200 to 300 mg
of pyridoxine daily.
Hemodialysis has been shown to remove
cycloserine from the bloodstream. This
procedure should be reserved for patients with
life threatening toxicity that is unresponsive to
less invasive therapy.
References (REFERENCES)
1. Jones LR: Colorimetric determination of
cycloserine, a new antibiotic. Anal Chem
1956;28:39.
Description (DESCRIPTION)
D -Cycloserine, (R)-4-amino-3-isoxazolidinone,
is a broad-spectrum antibiotic that is produced
by a strain of Streptomyces orchidaceus and has
also been synthesized. Cycloserine is a white
to off-white powder that is soluble in water and
stable in alkaline solution. It is rapidly destroyed
at a neutral or acid pH.
Cycloserine has a pH between 5.5 and 6.5 in a
solution containing 100 mg/mL. The molecular
weight of cycloserine is 102.09, and it has an
empirical formula of C 3H6N2O 2 . The structural
formula of cycloserine is as follows:
Each capsule contains cycloserine, 250 mg
(2.45 mmol); D & C Yellow No. 10, F D & C Blue
No. 1, F D & C Red No. 3, F D & C Yellow No.
6, gelatin, iron oxide, talc, and titanium dioxide.
Precautions (PRECAUTIONS)
General: Before treatment with cycloserine
is initiated, cultures should be taken and the
organism’s susceptibility to the drug should
be established. In tuberculous infections,
the organism’s susceptibility to the other
antituberculosis agents in the regimen should
also be demonstrated.
Anticonvulsant drugs or sedatives may be
effective in controlling symptoms of CNS
toxicity, such as convulsions, anxiety, and
tremor. Patients receiving more than 500 mg
of cycloserine daily should be closely observed
for such symptoms. The value of pyridoxine in
preventing CNS toxicity from cycloserine has
not been proved.
Administration of cycloserine and other
antituberculosis drugs has been associated in
a few instances with vitamin B 12 and/or folic-
acid deficiency, megaloblastic anemia, and
sideroblastic anemia. If evidence of anemia
develops during treatment, appropriate studies
and therapy should be instituted.
Laboratory Tests: Blood levels should be
determined at least weekly for patients with
reduced renal function, for individuals receiving
a daily dosage of more than 500 mg, and for
those showing signs and symptoms suggestive
of toxicity. The dosage should be adjusted to
keep the blood level below 30 mcg/mL.
Drug Interactions: Concurrent administration
of ethionamide has been reported to potentiate
neurotoxic side effects.
Alcohol and cycloserine are incompatible,
especially during a regimen calling for large
doses of the latter. Alcohol increases the
possibility and risk of epileptic episodes.
Concurrent administration of isoniazid may
result in increased incidence of CNS effects,
such as dizziness or drowsiness. Dosage
adjustments may be necessary and patients
should be monitored closely for signs of CNS
toxicity.
Carcinogenesis, Mutagenicity, and
Impairment of Fertility: Studies have not
been performed to determine potential for
carcinogenicity. The Ames test and unscheduled
DNA repair test were negative. A study in 2
generations of rats showed no impairment of
fertility relative to controls for the first mating but
somewhat lower fertility in the second mating.
Pregnancy Category C: There are no
adequate and well-controlled studies with
the use of Cycloserine in pregnant women.
A study in 2 generations of rats given doses
up to 100 mg/kg/day (approximately equivalent
to the maximum recommended human dose
on a body surface area basis) demonstrated
no teratogenic effect in offspring. Cycloserine
should be used during pregnancy only if the
potential benefit justifies the potential risk to the
fetus.
Nursing Mothers: Because of the potential
for serious adverse reactions in nursing infants
from cycloserine, a decision should be made
whether to discontinue nursing or to discontinue
the drug, taking into account the importance of
the drug to the mother.
Usage in Pediatric Patients: Safety and
effectiveness in pediatric patients have not been
established.
Geriatric Use: Clinical studies of cycloserine
did not include sufficient numbers of subjects
aged 65 and over to determine whether they
responded differently from younger subjects.
Other reported clinical experience has not
identified differences in responses between
the elderly and younger patients. In general,
dose selection for an elderly patient should be
cautious, usually starting at the low end of the
dosing range, reflecting the greater frequency
of decreased hepatic, renal, or cardiac function,
and of concomitant disease or other drug
therapy.
This drug is known to be substantially excreted
by the kidney, and the risk of toxic reactions
to this drug may be greater in patients with
impaired renal function. Because elderly
patients are more likely to have decreased renal
function, care should be taken in dose selection,
and it may be useful to monitor renal function.
The toxicity of cycloserine is closely related
to excessive blood levels (above 30 mcg/mL)
as determined by high dosage or inadequate
renal clearance (see WARNINGS). Blood
levels should be determined at least weekly
for patients with reduced renal function, for
individuals receiving a daily dosage of more
than 500 mg, and for those showing signs
and symptoms suggestive of toxicity. The
dosage should be adjusted to keep the blood
level below 30 mcg/mL (see PRECAUTIONS,
Laboratory Tests).
How Supplied (HOW SUPPLIED)
Cycloserine is available as a 250 mg capsule
with an opaque red cap and opaque gray body
imprinted with “PGC” and “F04” in edible black
ink on both the cap and the body.
Cycloserine Capsules, USP are supplied in cartons of 3 cards. Each card is a ten-capsules blister card as follows:
NDC 43598-235-31: carton with 3 cards.
NDC 43598-235-10 (ten 250mg capsules per card).
Store at controlled room temperature,
20° to 25°C (68° to 77°F) [see USP Controlled
Room Temperature].
Adverse Reactions (ADVERSE REACTIONS)
Most adverse reactions occurring during therapy
with cycloserine involve the nervous system or
are manifestations of drug hypersensitivity. The
following side effects have been observed in
patients receiving cycloserine:
Nervous system symptoms (which appear to
be related to higher dosages of the drug, i.e.,
more than 500 mg daily)
• Convulsions
• Drowsiness and somnolence
• Headache
• Tremor
• Dysarthria
• Vertigo
• Confusion and disorientation with loss of
memory
• Psychoses, possibly with suicidal tendencies
• Character changes
• Hyperirritability
• Aggression
• Paresis
• Hyperreflexia
• Paresthesia
• Major & minor (localized) clonic seizures
• Coma
Cardiovascular: Sudden development of
congestive heart failure in patients receiving
1 to 1.5 g of cycloserine daily has been
reported.
Allergy (apparently not related to dosage)
Skin rash
Miscellaneous: Elevated serum transaminase,
especially in patients with preexisting liver
disease
To report SUSPECTED ADVERSE REACTIONS, contact Cerovene, Inc. at 1-833-304-9569 or FDA at 1‑800-FDA-1088 or www.fda.gov/medwatch.
Contraindications (CONTRAINDICATIONS)
Administration is contraindicated in patients with
any of the following:
• Hypersensitivity to cycloserine
• Epilepsy
• Depression, severe anxiety, or psychosis
• Severe renal insufficiency
• Excessive concurrent use of alcoho
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
After oral administration, cycloserine is
readily absorbed from the gastrointestinal
tract, with peak blood levels occurring in 4
to 8 hours. Blood levels of 25 to 30 mcg/mL
can generally be maintained with the usual
dosage of 250 mg twice a day, although the
relationship of plasma levels to dosage is
not always consistent. Concentrations in the
cerebrospinal fluid, pleural fluid, fetal blood,
and mother’s milk approach those found in
the serum. Detectable amounts are found
in ascitic fluid, file, sputum, amniotic fluid,
and lung and lymph tissues. Approximately
65% of a single dose of cycloserine can
be recovered in the urine within 72 hours
after oral administration. The remaining
35% is apparently metabolized to unknown
substances. The maximum excretion rate
occurs 2 to 6 hours after administration, with
50% of the drug eliminated in 12 hours.
Mechanism of Action: The antibacterial activity
of Cycloserine results from inhibition of cell-wall
synthesis in susceptible strains of gram-positive
and gram-negative bacteria.
Antibacterial Activity: Cycloserine has been
shown to be active against most isolates of the
following microorganism, both in vitro and in
clinical infections [see Indications and Usage]:
Mycobacterium tuberculosis.
Indications and Usage (INDICATIONS AND USAGE)
Cycloserine is indicated in the treatment
of active pulmonary and extrapulmonary
tuberculosis (including renal disease) when
the causative organisms are susceptible to
this drug and when treatment with the primary
medications (streptomycin, isoniazid, rifampin,
and ethambutol) has proved inadequate. Like
all antituberculosis drugs, cycloserine should be
administered in conjunction with other effective
chemotherapy and not as the sole therapeutic
agent.
Cycloserine may be effective in the treatment
of acute urinary tract infections caused by
susceptible strains of gram-positive and gram-
negative bacteria. Use of cycloserine in these
infections should be considered only when
more conventional therapy has failed and when
the organism has been demonstrated to be
susceptible to the drug
Package Label Display (PACKAGE LABEL DISPLAY)
Cycloserine Capsules USP, 250 mg - Carton Label
Dosage and Administration (DOSAGE AND ADMINISTRATION)
Cycloserine is effective orally and is currently
administered only by this route. The usual
dosage is 500 mg to 1 g daily in divided doses
monitored by blood levels.1 The initial adult
dosage most frequently given is 250 mg twice
daily at 12-hour intervals for the first 2 weeks.
A daily dosage of 1 g should not be exceeded.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:42:10.774822 · Updated: 2026-03-14T22:08:06.283890