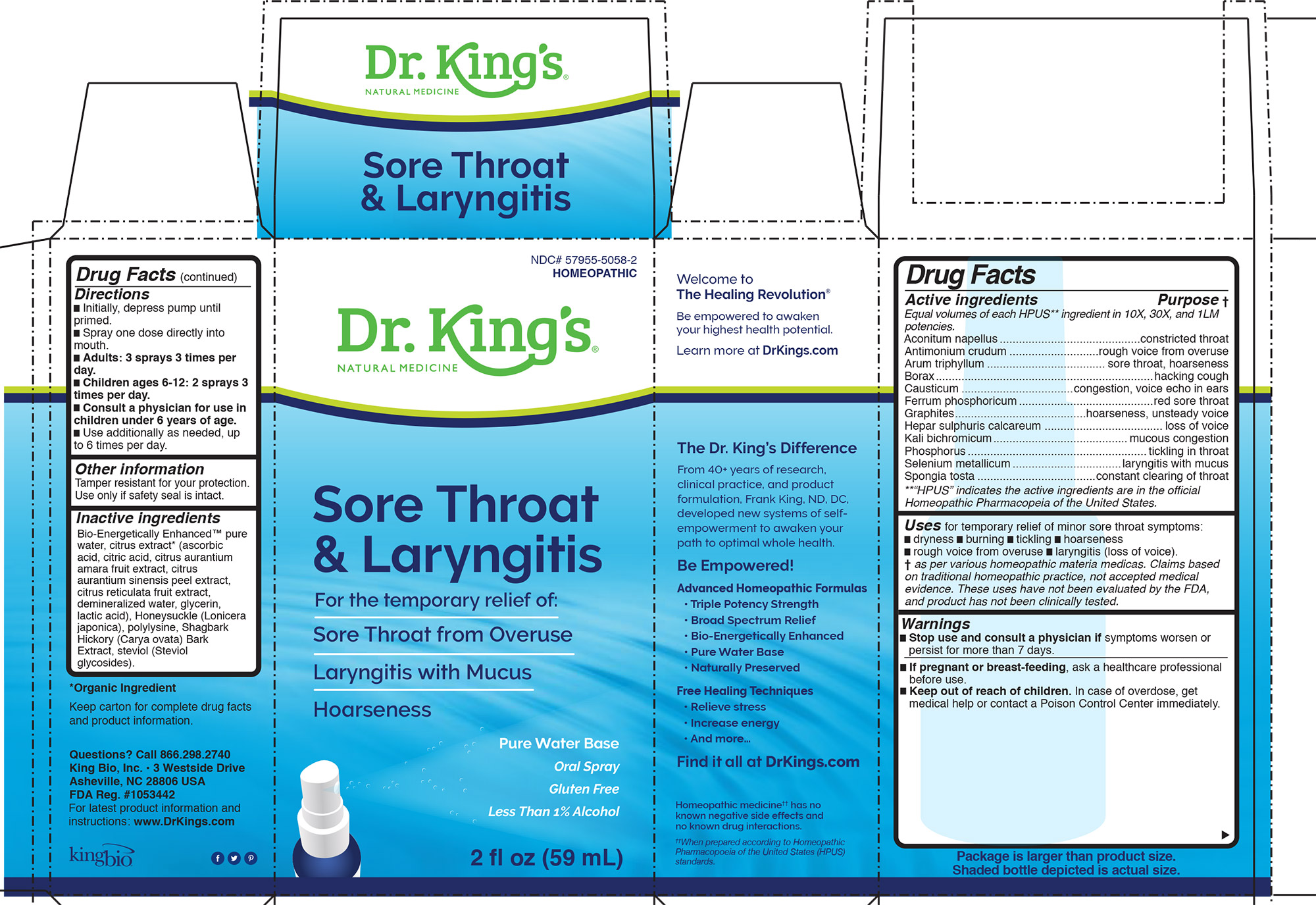
Sore Throat And Laryngitis
8e6cca4f-f27c-41c0-9113-879e25a3298b
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
HPUS active ingredients Equal volumes of each ingredient in 10X, 30X, and 1LM potencies. Aconitum napellus, Antimonium crudum, Arum triphyllum, Borax, Causticum, Ferrum phosphoricum, Graphites, Hepar sulphuris calcareum, Kali bichromicum, Phosphorus, Selenium metallicum, Spongia tosta
Medication Information
Warnings and Precautions
Warnings
Stop use and consult a physician if symptoms worsen or
persist for more than 7 days.
Indications and Usage
Uses for temporary relief of minor sore throat symptoms:
- dryness
- burning
- tickling
- hoarseness
- rough voice from overuse
- laryngitis (loss of voice).
† as per various homeopathic materia medicas. Claims based
on traditional homeopathic practice, not accepted medical
evidence. These uses have not been evaluated by the FDA,
and product has not been clinically tested.
Dosage and Administration
Directions:
- Initially, depress pump until primed.
- Spray one dose directly into mouth.
- Adults: 3 sprays 3 times per day.
- Children ages 6-12: 2 sprays 3 times per day.
- Consult a physician for use in children under 6 years of age.
- Use additionally as needed, up to 6 times per day.
Description
HPUS active ingredients Equal volumes of each ingredient in 10X, 30X, and 1LM potencies. Aconitum napellus, Antimonium crudum, Arum triphyllum, Borax, Causticum, Ferrum phosphoricum, Graphites, Hepar sulphuris calcareum, Kali bichromicum, Phosphorus, Selenium metallicum, Spongia tosta
Section 44425-7
Other Information:
Tamper resistant for your protection. Use only if safety seal is intact.
Section 50565-1
Keep out of reach of children. In case of overdose, get
medical help or contact a Poison Control Center immediately.
Section 51727-6
Inactive Ingredients:
Bio-Energetically Enhanced™ pure
water, citrus extract* (ascorbic
acid, citric acid, citrus aurantium
amara fruit extract, citrus
aurantium sinensis peel extract,
citrus reticulata fruit extract,
demineralized water, glycerin,
lactic acid), Honeysuckle (Lonicera
japonica), polylysine, Shagbark
Hickory (Carya ovata) Bark
Extract, steviol (Steviol
glycosides).
Section 51945-4
Section 53413-1
Questions? Call 866.298.2740
King Bio, Inc. • 3 Westside Drive
Asheville, NC 28806 USA
FDA Reg. #1053442
For latest product information and
instructions: www.DrKings.com
Section 53414-9
If pregnant or breast-feeding, ask a healthcare professional
before use.
Section 55105-1
HPUS active ingredients Purpose
Equal volumes of each HPUS** ingredient in 10X, 30X, and 1LM
potencies.
Aconitum napellus............................................constricted throat
Antimonium crudum ............................rough voice from overuse
Arum triphyllum ..................................... sore throat, hoarseness
Borax....................................................................hacking cough
Causticum ...................................congestion, voice echo in ears
Ferrum phosphoricum..........................................red sore throat
Graphites.........................................hoarseness, unsteady voice
Hepar sulphuris calcareum ..................................... loss of voice
Kali bichromicum..........................................mucous congestion
Phosphorus........................................................tickling in throat
Selenium metallicum..................................laryngitis with mucus
Spongia tosta .....................................constant clearing of throat
**“HPUS” indicates the active ingredients are in the official
Homeopathic Pharmacopeia of the United States.
Section 55106-9
HPUS active ingredients
Equal volumes of each ingredient in 10X, 30X, and 1LM potencies.
Aconitum napellus, Antimonium crudum, Arum triphyllum, Borax, Causticum, Ferrum phosphoricum, Graphites, Hepar sulphuris calcareum, Kali bichromicum, Phosphorus, Selenium metallicum, Spongia tosta
Structured Label Content
Indications and Usage (34067-9)
Uses for temporary relief of minor sore throat symptoms:
- dryness
- burning
- tickling
- hoarseness
- rough voice from overuse
- laryngitis (loss of voice).
† as per various homeopathic materia medicas. Claims based
on traditional homeopathic practice, not accepted medical
evidence. These uses have not been evaluated by the FDA,
and product has not been clinically tested.
Dosage and Administration (34068-7)
Directions:
- Initially, depress pump until primed.
- Spray one dose directly into mouth.
- Adults: 3 sprays 3 times per day.
- Children ages 6-12: 2 sprays 3 times per day.
- Consult a physician for use in children under 6 years of age.
- Use additionally as needed, up to 6 times per day.
Warnings and Precautions (34071-1)
Warnings
Stop use and consult a physician if symptoms worsen or
persist for more than 7 days.
Section 44425-7 (44425-7)
Other Information:
Tamper resistant for your protection. Use only if safety seal is intact.
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get
medical help or contact a Poison Control Center immediately.
Section 51727-6 (51727-6)
Inactive Ingredients:
Bio-Energetically Enhanced™ pure
water, citrus extract* (ascorbic
acid, citric acid, citrus aurantium
amara fruit extract, citrus
aurantium sinensis peel extract,
citrus reticulata fruit extract,
demineralized water, glycerin,
lactic acid), Honeysuckle (Lonicera
japonica), polylysine, Shagbark
Hickory (Carya ovata) Bark
Extract, steviol (Steviol
glycosides).
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
Questions? Call 866.298.2740
King Bio, Inc. • 3 Westside Drive
Asheville, NC 28806 USA
FDA Reg. #1053442
For latest product information and
instructions: www.DrKings.com
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a healthcare professional
before use.
Section 55105-1 (55105-1)
HPUS active ingredients Purpose
Equal volumes of each HPUS** ingredient in 10X, 30X, and 1LM
potencies.
Aconitum napellus............................................constricted throat
Antimonium crudum ............................rough voice from overuse
Arum triphyllum ..................................... sore throat, hoarseness
Borax....................................................................hacking cough
Causticum ...................................congestion, voice echo in ears
Ferrum phosphoricum..........................................red sore throat
Graphites.........................................hoarseness, unsteady voice
Hepar sulphuris calcareum ..................................... loss of voice
Kali bichromicum..........................................mucous congestion
Phosphorus........................................................tickling in throat
Selenium metallicum..................................laryngitis with mucus
Spongia tosta .....................................constant clearing of throat
**“HPUS” indicates the active ingredients are in the official
Homeopathic Pharmacopeia of the United States.
Section 55106-9 (55106-9)
HPUS active ingredients
Equal volumes of each ingredient in 10X, 30X, and 1LM potencies.
Aconitum napellus, Antimonium crudum, Arum triphyllum, Borax, Causticum, Ferrum phosphoricum, Graphites, Hepar sulphuris calcareum, Kali bichromicum, Phosphorus, Selenium metallicum, Spongia tosta
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:55.655109 · Updated: 2026-03-14T23:14:57.671574