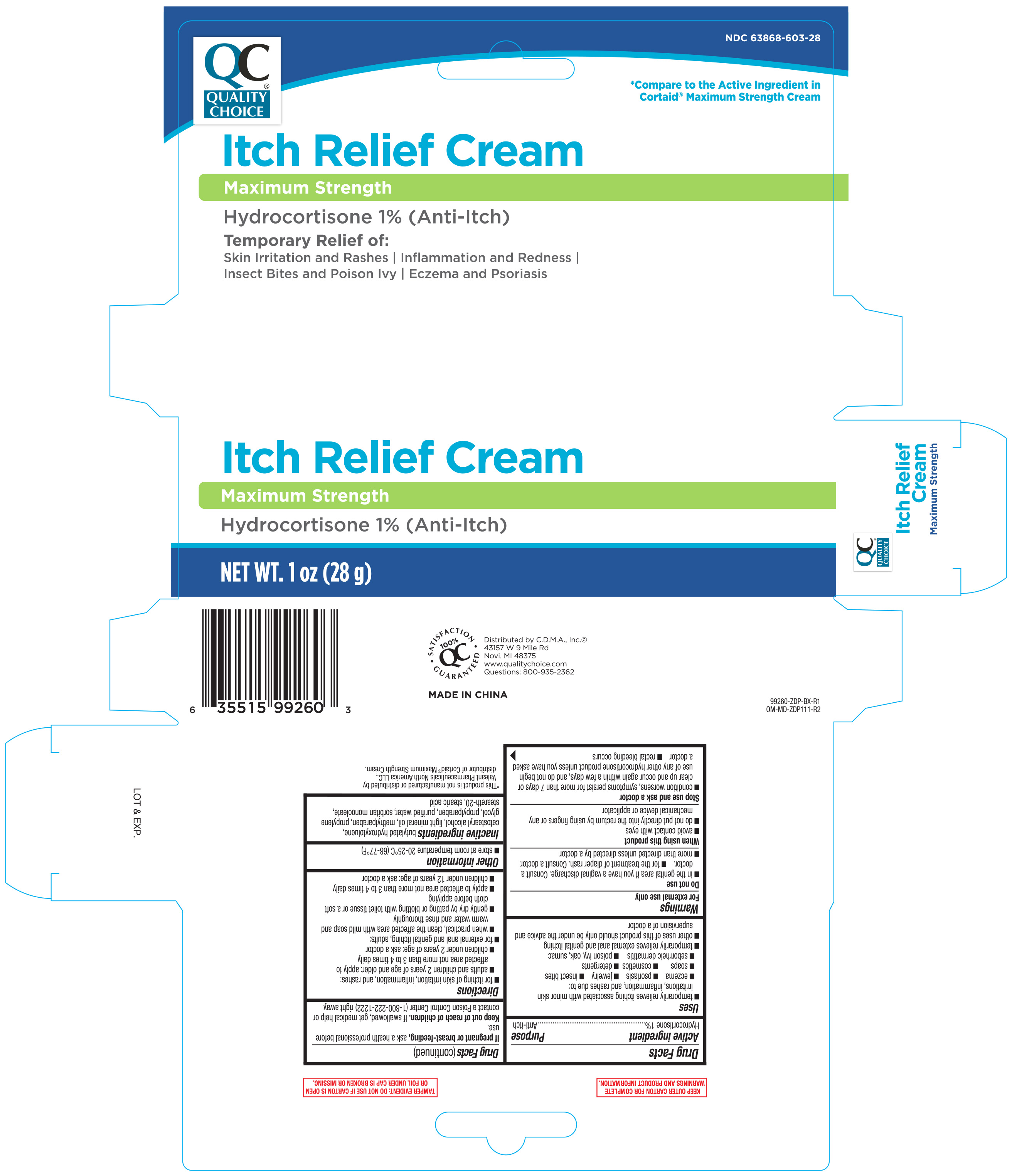
Qc 1% Hydrocortisone Cream Max. Strength 1 Oz 99260 (2019)
8dc1ac1e-de7f-44e1-e053-2a95a90a182e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Purpose Hydrocortisone 1%..................................................Anti-itch
Medication Information
Warnings and Precautions
Warnings
For external use only
Indications and Usage
Directions
- for itching of skin irritation, inflammation, and rashes:
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
- for external anal and genital itching, adults:
- when practical, clean the affected area with mild soap and warm water and rinse thoroughly
- gently dry by patting or blotting with toilet tissue or a soft cloth before applying
- apply to affected area not more than 3 to 4 times daily
- children under 12 years of age: ask a doctor
Dosage and Administration
Distributed by:
C.D.M.A., Inc.
43157 W 9 Mile Rd
Novi, MI 48375
www.qualitychoice.com
Made in China
Description
Active ingredient Purpose Hydrocortisone 1%..................................................Anti-itch
Section 44425-7
Other information
- store at room temperature 20-25°C (68-77°F)
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9
Stop use and ask a doctor
- condition worsens, symptoms persist for more than 7 days or clear up and occur again within a few days, and do not begin use of any other hydrocortisone product unless you have asked a doctor
- rectal bleeding occurs
Section 50567-7
When using this product
- avoid contact with eyes
- do not put directly into the rectum by using fingers or any mechanical device or applicator
Section 50570-1
Do not use
- in the genital area if you have a vaginal discharge. Consult a doctor.
- for the treatment of diaper rash. Consult a doctor.
- more than directed unless directed by a doctor
Section 51727-6
Inactive ingredients
butylated hydroxytoluene, cetostearyl alcohol, light mineral oil, methylparaben, propylene glycol, propylparaben, purified water, sorbitan monooleate, steareth-20, stearic acid
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Uses
- temporarily relieves itching associated with minor skin irritation, inflammation, and rashed due to
- eczema
- psoriasis
- jewelry
- insect bites
- soaps
- cosmetics
- detergents
- seborrheic dermatitis
- poison ivy, oak sumac
Section 55106-9
Active ingredient Purpose
Hydrocortisone 1%..................................................Anti-itch
Structured Label Content
Indications and Usage (34067-9)
Directions
- for itching of skin irritation, inflammation, and rashes:
- adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
- children under 2 years of age: ask a doctor
- for external anal and genital itching, adults:
- when practical, clean the affected area with mild soap and warm water and rinse thoroughly
- gently dry by patting or blotting with toilet tissue or a soft cloth before applying
- apply to affected area not more than 3 to 4 times daily
- children under 12 years of age: ask a doctor
Dosage and Administration (34068-7)
Distributed by:
C.D.M.A., Inc.
43157 W 9 Mile Rd
Novi, MI 48375
www.qualitychoice.com
Made in China
Warnings and Precautions (34071-1)
Warnings
For external use only
Section 44425-7 (44425-7)
Other information
- store at room temperature 20-25°C (68-77°F)
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor
- condition worsens, symptoms persist for more than 7 days or clear up and occur again within a few days, and do not begin use of any other hydrocortisone product unless you have asked a doctor
- rectal bleeding occurs
Section 50567-7 (50567-7)
When using this product
- avoid contact with eyes
- do not put directly into the rectum by using fingers or any mechanical device or applicator
Section 50570-1 (50570-1)
Do not use
- in the genital area if you have a vaginal discharge. Consult a doctor.
- for the treatment of diaper rash. Consult a doctor.
- more than directed unless directed by a doctor
Section 51727-6 (51727-6)
Inactive ingredients
butylated hydroxytoluene, cetostearyl alcohol, light mineral oil, methylparaben, propylene glycol, propylparaben, purified water, sorbitan monooleate, steareth-20, stearic acid
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Uses
- temporarily relieves itching associated with minor skin irritation, inflammation, and rashed due to
- eczema
- psoriasis
- jewelry
- insect bites
- soaps
- cosmetics
- detergents
- seborrheic dermatitis
- poison ivy, oak sumac
Section 55106-9 (55106-9)
Active ingredient Purpose
Hydrocortisone 1%..................................................Anti-itch
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:57.307633 · Updated: 2026-03-14T22:55:46.109144