1019-qch-2024-0812
8d985ad7-71b9-493f-9b9a-d6ef280f72dd
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Nighttime sleep-aid
Medication Information
Purpose
Nighttime sleep-aid
Description
Drug Facts
Use
helps to reduce difficulty falling asleep
Section 42229-5
Drug Facts
Section 50566-9
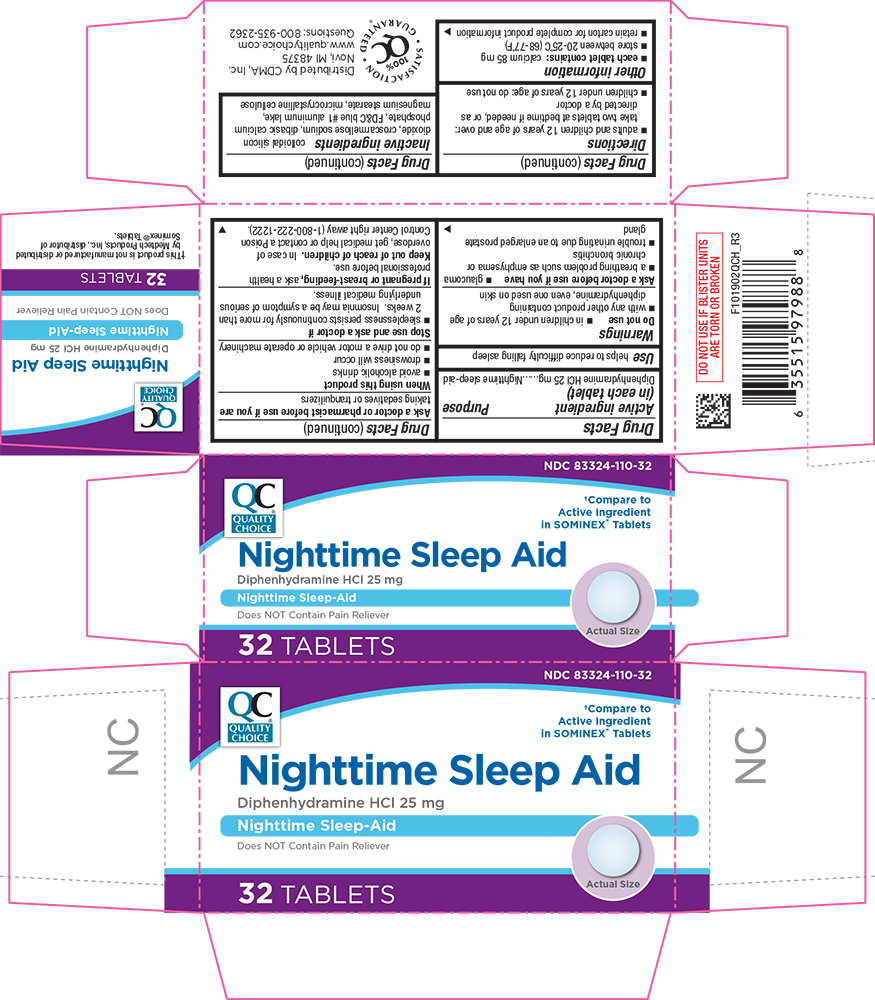
Stop use and ask a doctor if
- sleeplessness persists continuously for more than 2 weeks.
Insomnia may be a symptom of serious underlying medical illness.
Section 50567-7
When using this product
- avoid alcoholic drinks
- drowsiness will occur
- do not drive a motor vehicle or operate machinery
Section 50568-5
Ask a doctor or pharmacist before use if you aretaking sedatives or tranquilizers
Section 53414-9
If pregnant or breast-feeding,ask a health professional before use.
Directions
- adults and children 12 years of age and over: take two tablets at bedtime if needed, or as directed by a doctor
- children under 12 years of age: do not use
Do Not Use
- in children under 12 years of age
- with any other product containing diphenhydramine, even one used on skin
Other Information
- each tablet contains:calcium 85 mg
- store between 20-25°C (68-77°F)
- retain carton for complete product information
Inactive Ingredients
colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate, FD&C blue #1 aluminum lake, magnesium stearate, microcrystalline cellulose
Principal Display Panel
QUALITY CHOICE®
NDC 83324-110-32
†Compare to Active Ingredient in SOMINEX® Tablets
Nighttime Sleep Aid
Diphenhydramine HCl 25 mg
Nighttime Sleep-Aid
Does NOT Contain Pain Reliever
32 TABLETS
Actual Size
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Active Ingredient (in Each Tablet)
Diphenhydramine HCl 25 mg
Ask A Doctor Before Use If You Have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Structured Label Content
Use
helps to reduce difficulty falling asleep
Section 42229-5 (42229-5)
Drug Facts
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- sleeplessness persists continuously for more than 2 weeks.
Insomnia may be a symptom of serious underlying medical illness.
Section 50567-7 (50567-7)
When using this product
- avoid alcoholic drinks
- drowsiness will occur
- do not drive a motor vehicle or operate machinery
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you aretaking sedatives or tranquilizers
Section 53414-9 (53414-9)
If pregnant or breast-feeding,ask a health professional before use.
Purpose
Nighttime sleep-aid
Directions
- adults and children 12 years of age and over: take two tablets at bedtime if needed, or as directed by a doctor
- children under 12 years of age: do not use
Do Not Use (Do not use)
- in children under 12 years of age
- with any other product containing diphenhydramine, even one used on skin
Other Information (Other information)
- each tablet contains:calcium 85 mg
- store between 20-25°C (68-77°F)
- retain carton for complete product information
Inactive Ingredients (Inactive ingredients)
colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate, FD&C blue #1 aluminum lake, magnesium stearate, microcrystalline cellulose
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
QUALITY CHOICE®
NDC 83324-110-32
†Compare to Active Ingredient in SOMINEX® Tablets
Nighttime Sleep Aid
Diphenhydramine HCl 25 mg
Nighttime Sleep-Aid
Does NOT Contain Pain Reliever
32 TABLETS
Actual Size
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Diphenhydramine HCl 25 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- trouble urinating due to an enlarged prostate gland
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:24.869604 · Updated: 2026-03-14T23:03:28.306097