8c2acd62-2f81-4dde-b258-870cd9469f6e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
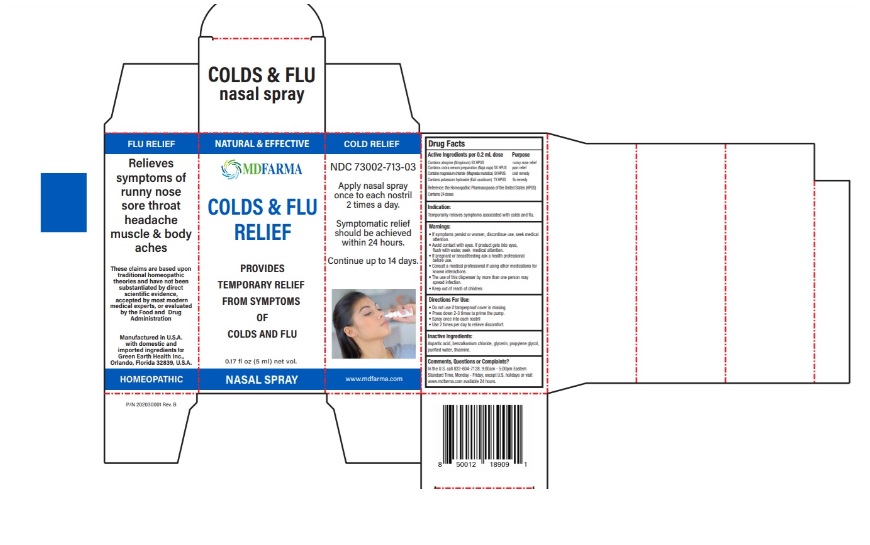
Active ingredient Purpose Contains atropine (Atropinum) 5X HPUS …………………………………....... runny nose relief Contains cobra venom preparation (Naja naja) 5X HPUS ………..... pain relief Contains magnesium chloride (Magnesia muriatica) 5X HPUS….... cold remedy Contains potassium hydroxide (Kali causticum) 7X HPUS ………. .... flu remedy Reference: the Homeopathic Pharmacopoeia of the United States (HPUS) Contains 24 doses
Medication Information
Description
Active ingredient Purpose Contains atropine (Atropinum) 5X HPUS …………………………………....... runny nose relief Contains cobra venom preparation (Naja naja) 5X HPUS ………..... pain relief Contains magnesium chloride (Magnesia muriatica) 5X HPUS….... cold remedy Contains potassium hydroxide (Kali causticum) 7X HPUS ………. .... flu remedy Reference: the Homeopathic Pharmacopoeia of the United States (HPUS) Contains 24 doses
Use
Temporarily relieves symptoms associated with colds and flu.
Section 50565-1
Keep out of reach of children.
Section 55105-1
Warnings
If symptoms persist or worsen, discontinue use, seek medical attention.
• Avoid contact with eyes. If product gets into eyes, flush with water, seek medical attention.
• If pregnant or breastfeeding ask a health professional before use.
• Consult a medical professional if using other medications for known interactions.
• The use of this dispenser by more than one person may spread infection.
Directions
- Do not use if tamperproof cover is missing.
- Press down 2-3 times to prime the pump.
- Spray once into each nostril
- Use 2 times per day to relieve discomfort.
Product Label
Active Ingredient
Active ingredient Purpose
Contains atropine (Atropinum) 5X HPUS …………………………………....... runny nose relief
Contains cobra venom preparation (Naja naja) 5X HPUS ………..... pain relief
Contains magnesium chloride (Magnesia muriatica) 5X HPUS….... cold remedy
Contains potassium hydroxide (Kali causticum) 7X HPUS ………. .... flu remedy
Reference: the Homeopathic Pharmacopoeia of the United States (HPUS)
Contains 24 doses
Inactive Ingredients
Aspartic acid, benzalkonium chloride, glycerin, propylene glycol, purified water, thiamine.
Comments, Questions or Complaints?
In the U.S. call 833-604-7128, 9:00am - 5:00pm Eastern Standard Time, Monday - Friday, except U.S. holidays or visit www.mdfarma.com available 24 hours.
Structured Label Content
Use
Temporarily relieves symptoms associated with colds and flu.
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 55105-1 (55105-1)
Warnings
If symptoms persist or worsen, discontinue use, seek medical attention.
• Avoid contact with eyes. If product gets into eyes, flush with water, seek medical attention.
• If pregnant or breastfeeding ask a health professional before use.
• Consult a medical professional if using other medications for known interactions.
• The use of this dispenser by more than one person may spread infection.
Directions
- Do not use if tamperproof cover is missing.
- Press down 2-3 times to prime the pump.
- Spray once into each nostril
- Use 2 times per day to relieve discomfort.
Product Label (Product label)
Active Ingredient
Active ingredient Purpose
Contains atropine (Atropinum) 5X HPUS …………………………………....... runny nose relief
Contains cobra venom preparation (Naja naja) 5X HPUS ………..... pain relief
Contains magnesium chloride (Magnesia muriatica) 5X HPUS….... cold remedy
Contains potassium hydroxide (Kali causticum) 7X HPUS ………. .... flu remedy
Reference: the Homeopathic Pharmacopoeia of the United States (HPUS)
Contains 24 doses
Inactive Ingredients (Inactive ingredients)
Aspartic acid, benzalkonium chloride, glycerin, propylene glycol, purified water, thiamine.
Comments, Questions or Complaints?
In the U.S. call 833-604-7128, 9:00am - 5:00pm Eastern Standard Time, Monday - Friday, except U.S. holidays or visit www.mdfarma.com available 24 hours.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:35.837287 · Updated: 2026-03-14T23:09:28.158875