75112-009 Rilakkuma
8b7b661d-918e-4403-86e5-d16c297fbc7c
34390-5
HUMAN OTC DRUG LABEL
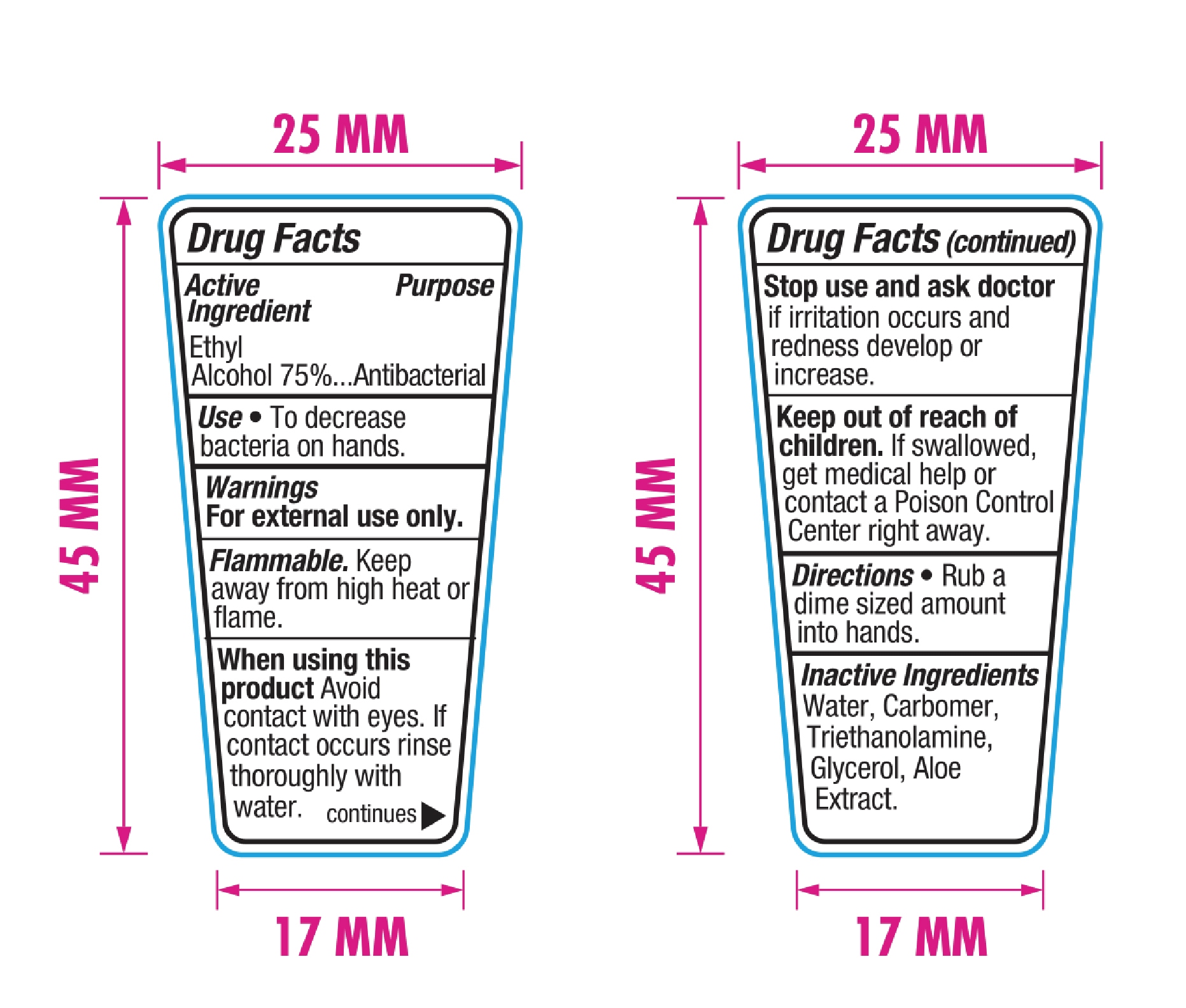
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl Alcohol 75% v/v
Purpose
Antibacterial
Medication Information
Purpose
Antibacterial
Description
Ethyl Alcohol 75% v/v
Use
To decrease bacterial on hands
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if irritation occurs and redness develops or increases.
Section 50567-7
When using this product, avoid contact with eyes. If contact occurs, rinse thoroughly with water.
Section 51945-4
Warnings
For external use only. Flammable. Keep away from heat or flame.
Directions
Rub a dime sized amount into hands
Active Ingredient(s)
Ethyl Alcohol 75% v/v
Inactive Ingredients
Water, carbomer, triethanolamine, glycerol, aloe extract
Structured Label Content
Use
To decrease bacterial on hands
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if irritation occurs and redness develops or increases.
Section 50567-7 (50567-7)
When using this product, avoid contact with eyes. If contact occurs, rinse thoroughly with water.
Section 51945-4 (51945-4)
Purpose
Antibacterial
Warnings
For external use only. Flammable. Keep away from heat or flame.
Directions
Rub a dime sized amount into hands
Active Ingredient(s)
Ethyl Alcohol 75% v/v
Inactive Ingredients (Inactive ingredients)
Water, carbomer, triethanolamine, glycerol, aloe extract
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:19.080080 · Updated: 2026-03-14T23:00:41.107943