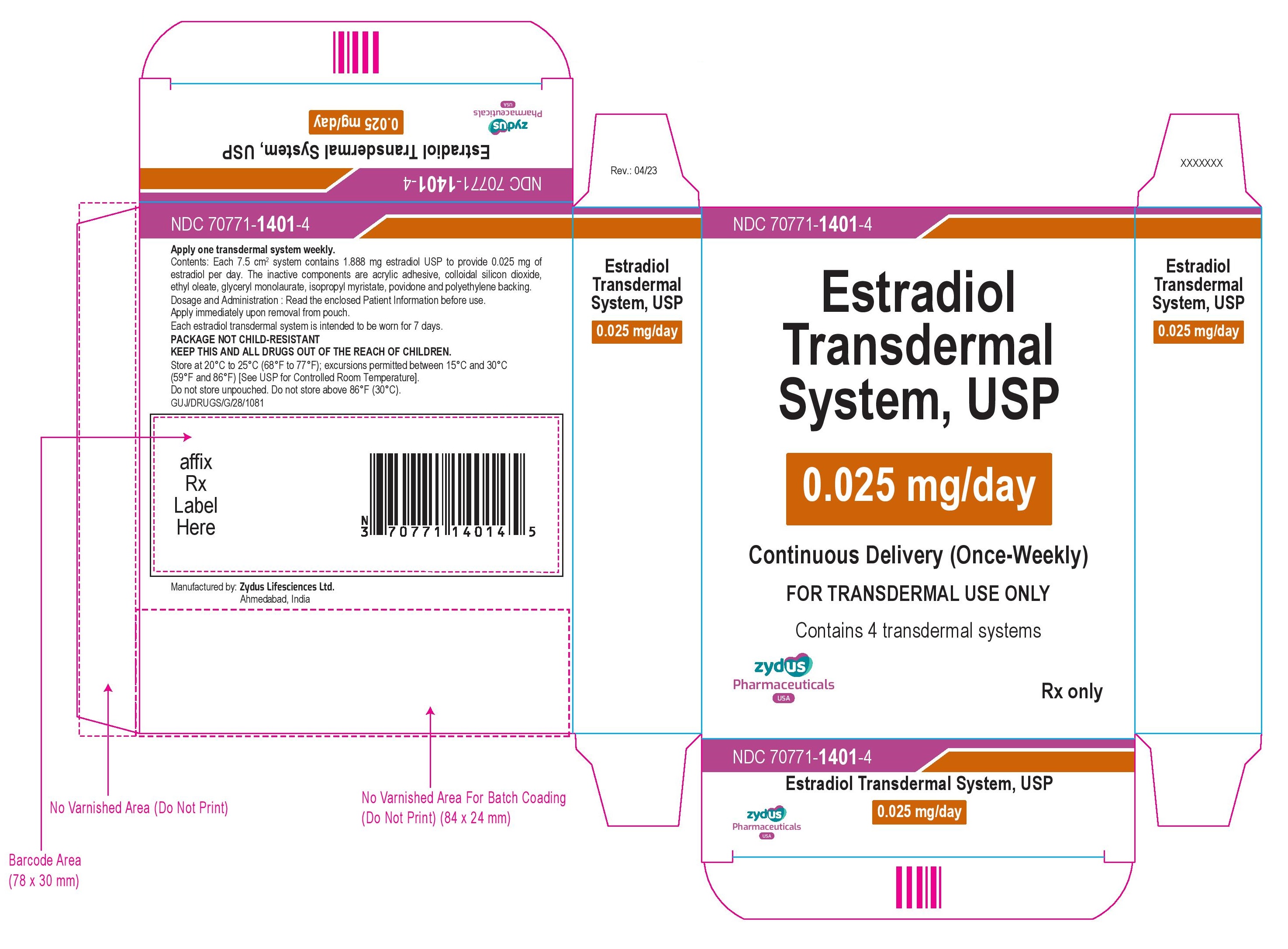
[{"name": "ESTRADIOL", "unii": "4TI98Z838E", "classCode": "ACTIB", "strengthNumerator": "0.025", "strengthDenominator": "1", "strengthNumeratorUnit": "mg", "strengthDenominatorUnit": "d"}, {"name": "2-ETHYLHEXYL ACRYLATE", "unii": "HR49R9S6XG", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "2-HYDROXYETHYL ACRYLATE", "unii": "25GT92NY0C", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ETHYL OLEATE", "unii": "Z2Z439864Y", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERYL LAURATE", "unii": "Y98611C087", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCIDYL METHACRYLATE", "unii": "R8WN29J8VF", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ISOPROPYL MYRISTATE", "unii": "0RE8K4LNJS", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "POVIDONE", "unii": "FZ989GH94E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SILICON DIOXIDE", "unii": "ETJ7Z6XBU4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "VINYL ACETATE", "unii": "L9MK238N77", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ESTRADIOL", "unii": "4TI98Z838E", "classCode": "ACTIB", "strengthNumerator": "0.0375", "strengthDenominator": "1", "strengthNumeratorUnit": "mg", "strengthDenominatorUnit": "d"}, {"name": "2-ETHYLHEXYL ACRYLATE", "unii": "HR49R9S6XG", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "2-HYDROXYETHYL ACRYLATE", "unii": "25GT92NY0C", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ETHYL OLEATE", "unii": "Z2Z439864Y", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERYL LAURATE", "unii": "Y98611C087", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCIDYL METHACRYLATE", "unii": "R8WN29J8VF", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ISOPROPYL MYRISTATE", "unii": "0RE8K4LNJS", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "POVIDONE", "unii": "FZ989GH94E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SILICON DIOXIDE", "unii": "ETJ7Z6XBU4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "VINYL ACETATE", "unii": "L9MK238N77", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ESTRADIOL", "unii": "4TI98Z838E", "classCode": "ACTIB", "strengthNumerator": "0.05", "strengthDenominator": "1", "strengthNumeratorUnit": "mg", "strengthDenominatorUnit": "d"}, {"name": "2-ETHYLHEXYL ACRYLATE", "unii": "HR49R9S6XG", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "2-HYDROXYETHYL ACRYLATE", "unii": "25GT92NY0C", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ETHYL OLEATE", "unii": "Z2Z439864Y", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERYL LAURATE", "unii": "Y98611C087", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCIDYL METHACRYLATE", "unii": "R8WN29J8VF", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ISOPROPYL MYRISTATE", "unii": "0RE8K4LNJS", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "POVIDONE", "unii": "FZ989GH94E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SILICON DIOXIDE", "unii": "ETJ7Z6XBU4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "VINYL ACETATE", "unii": "L9MK238N77", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ESTRADIOL", "unii": "4TI98Z838E", "classCode": "ACTIB", "strengthNumerator": "0.06", "strengthDenominator": "1", "strengthNumeratorUnit": "mg", "strengthDenominatorUnit": "d"}, {"name": "2-ETHYLHEXYL ACRYLATE", "unii": "HR49R9S6XG", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "2-HYDROXYETHYL ACRYLATE", "unii": "25GT92NY0C", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ETHYL OLEATE", "unii": "Z2Z439864Y", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERYL LAURATE", "unii": "Y98611C087", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCIDYL METHACRYLATE", "unii": "R8WN29J8VF", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ISOPROPYL MYRISTATE", "unii": "0RE8K4LNJS", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "POVIDONE", "unii": "FZ989GH94E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SILICON DIOXIDE", "unii": "ETJ7Z6XBU4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "VINYL ACETATE", "unii": "L9MK238N77", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ESTRADIOL", "unii": "4TI98Z838E", "classCode": "ACTIB", "strengthNumerator": "0.075", "strengthDenominator": "1", "strengthNumeratorUnit": "mg", "strengthDenominatorUnit": "d"}, {"name": "2-ETHYLHEXYL ACRYLATE", "unii": "HR49R9S6XG", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "2-HYDROXYETHYL ACRYLATE", "unii": "25GT92NY0C", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ETHYL OLEATE", "unii": "Z2Z439864Y", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERYL LAURATE", "unii": "Y98611C087", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCIDYL METHACRYLATE", "unii": "R8WN29J8VF", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ISOPROPYL MYRISTATE", "unii": "0RE8K4LNJS", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "POVIDONE", "unii": "FZ989GH94E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SILICON DIOXIDE", "unii": "ETJ7Z6XBU4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "VINYL ACETATE", "unii": "L9MK238N77", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ESTRADIOL", "unii": "4TI98Z838E", "classCode": "ACTIB", "strengthNumerator": "0.1", "strengthDenominator": "1", "strengthNumeratorUnit": "mg", "strengthDenominatorUnit": "d"}, {"name": "2-ETHYLHEXYL ACRYLATE", "unii": "HR49R9S6XG", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "2-HYDROXYETHYL ACRYLATE", "unii": "25GT92NY0C", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ETHYL OLEATE", "unii": "Z2Z439864Y", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERYL LAURATE", "unii": "Y98611C087", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCIDYL METHACRYLATE", "unii": "R8WN29J8VF", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ISOPROPYL MYRISTATE", "unii": "0RE8K4LNJS", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "POVIDONE", "unii": "FZ989GH94E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SILICON DIOXIDE", "unii": "ETJ7Z6XBU4", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "VINYL ACETATE", "unii": "L9MK238N77", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}]