These Highlights Do Not Include All The Information Needed To Use Inlyta Safely And Effectively. See Full Prescribing Information For Inlyta.
8a903e31-936e-4ed7-8a59-59f32374f338
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
INLYTA is a kinase inhibitor indicated: • in combination with avelumab, for the first-line treatment of patients with advanced renal cell carcinoma (RCC). ( 1.1 ) • in combination with pembrolizumab, for the first-line treatment of patients with advanced RCC. ( 1.1 ) • as a single agent, for the treatment of advanced renal cell carcinoma (RCC) after failure of one prior systemic therapy. ( 1.2 )
Indications and Usage
INLYTA is a kinase inhibitor indicated: • in combination with avelumab, for the first-line treatment of patients with advanced renal cell carcinoma (RCC). ( 1.1 ) • in combination with pembrolizumab, for the first-line treatment of patients with advanced RCC. ( 1.1 ) • as a single agent, for the treatment of advanced renal cell carcinoma (RCC) after failure of one prior systemic therapy. ( 1.2 )
Dosage and Administration
• INLYTA 5 mg orally twice daily with avelumab 800 mg every 2 weeks. ( 2.1 ) • INLYTA 5 mg orally twice daily with pembrolizumab 200 mg every 3 weeks or 400 mg every 6 weeks. ( 2.1 ) • INLYTA as a single agent the starting dose is 5 mg orally twice daily. ( 2.1 ) • Dose adjustments can be made based on individual safety and tolerability. ( 2.2 ) • Administer INLYTA dose approximately 12 hours apart with or without food. ( 2.1 ) • INLYTA should be swallowed whole with a glass of water. ( 2.1 ) • See Full Prescribing Information for dosage modifications for adverse reactions. ( 2.2 ) • If a strong CYP3A4/5 inhibitor is required, decrease the INLYTA dose by approximately half. ( 2.2 ) • For patients with moderate hepatic impairment, decrease the starting dose by approximately half. ( 2.2 )
Warnings and Precautions
• Hypertension: Hypertension including hypertensive crisis has been observed. Blood pressure should be well-controlled prior to initiating INLYTA. Monitor for hypertension and treat as needed. Withhold and then dose reduce INLYTA or permanently discontinue based on severity of hypertension. ( 5.1 ) • Arterial and Venous Thromboembolic Events: Arterial and venous thrombotic events have been observed and can be fatal. Use with caution in patients who are at increased risk for these events. Permanently discontinue INLYTA if an arterial thromboembolic event occurs during treatment. Withhold INLYTA and then resume at same dose or permanently discontinue based on severity of VTE. ( 5.2 , 5.3 ) • Hemorrhage: Hemorrhagic events, including fatal events, have been reported. INLYTA has not been studied in patients with evidence of untreated brain metastasis or recent active gastrointestinal bleeding and should not be used in those patients. Withhold and then dose reduce INLYTA or discontinue based on severity and persistence of hemorrhage. ( 5.4 ) • Cardiac Failure: Cardiac failure has been observed and can be fatal. Monitor for signs or symptoms of cardiac failure throughout treatment with INLYTA. Management of cardiac failure may require dose reduction, dose interruption or permanent discontinuation of INLYTA. ( 5.5 ) • Gastrointestinal Perforation and Fistula Formation: Gastrointestinal perforation and fistula, including death, have occurred. Use with caution in patients at risk for gastrointestinal perforation or fistula. ( 5.6 ) • Hypothyroidism: Hypothyroidism requiring thyroid hormone replacement has been reported. Monitor thyroid function before initiation of, and periodically throughout, treatment with INLYTA. ( 5.7 ) • Impaired Wound Healing: Withhold INLYTA for at least 2 days prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. Resume INLYTA at a reduced dose or discontinue based on severity and persistence of the impaired wound healing. The safety of resumption of INLYTA after resolution of wound healing complications has not been established. ( 5.8 ) • Reversible Posterior Leukoencephalopathy Syndrome (RPLS): RPLS has been observed. Permanently discontinue INLYTA if signs or symptoms of RPLS occur. ( 5.9 ) • Proteinuria: Monitor for proteinuria before initiation of, and periodically throughout, treatment with INLYTA. For moderate to severe proteinuria, withhold and then dose reduce INLYTA. ( 5.10 ) • Hepatotoxicity: Liver enzyme elevation has occurred during treatment with INLYTA as a single agent. Monitor ALT, AST and bilirubin before initiation of, and periodically throughout, treatment with INLYTA. When used in combination with avelumab or pembrolizumab, higher frequencies of Grade 3 and 4 ALT and AST elevation may occur. Consider more frequent monitoring of liver enzymes. Withhold INLYTA and avelumab or pembrolizumab, initiate corticosteroid therapy as needed, and/or permanently discontinue the combination for severe or life-threatening hepatotoxicity. ( 5.11 ) • Use in Patients with Hepatic Impairment: Decrease the starting dose of INLYTA if used in patients with moderate hepatic impairment. INLYTA has not been studied in patients with severe hepatic impairment. ( 2.2 , 5.12 ) • Major adverse cardiovascular events (INLYTA in combination with avelumab): Optimize management of cardiovascular risk factors. Permanently discontinue INLYTA in combination with avelumab for Grade 3–4 events. ( 5.13 ) • Embryo-Fetal Toxicity: INLYTA can cause fetal harm. Advise patients of the potential risk to the fetus and to use effective contraception. ( 5.14 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are discussed elsewhere in the labeling [see Warnings and Precautions (5) ] : • Hypertension [see Warnings and Precautions (5.1) ] • Arterial thromboembolic events [see Warnings and Precautions (5.2) ] • Venous thromboembolic events [see Warnings and Precautions (5.3) ] • Hemorrhage [see Warnings and Precautions (5.4) ] • Cardiac failure [see Warnings and Precautions (5.5) ] • Gastrointestinal perforation and fistula formation [see Warnings and Precautions (5.6) ] • Thyroid dysfunction [see Warnings and Precautions (5.7) ] • Reversible posterior leukoencephalopathy syndrome [see Warnings and Precautions (5.9) ] • Proteinuria [see Warnings and Precautions (5.10) ] • Hepatotoxicity [see Warnings and Precautions (5.11) ] • Hepatic impairment [see Warnings and Precautions (5.12) ]
Drug Interactions
Storage and Handling
INLYTA tablets are supplied as follows: • 1 mg tablets are red film-coated, oval tablets debossed with "Pfizer" on one side and "1 XNB" on the other; available in bottles of 180: NDC 0069-0145-01. • 5 mg tablets are red film-coated, triangular tablets debossed with "Pfizer" on one side and "5 XNB" on the other; available in bottles of 60: NDC 0069-0151-11. • Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
How Supplied
INLYTA tablets are supplied as follows: • 1 mg tablets are red film-coated, oval tablets debossed with "Pfizer" on one side and "1 XNB" on the other; available in bottles of 180: NDC 0069-0145-01. • 5 mg tablets are red film-coated, triangular tablets debossed with "Pfizer" on one side and "5 XNB" on the other; available in bottles of 60: NDC 0069-0151-11. • Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Medication Information
Warnings and Precautions
• Hypertension: Hypertension including hypertensive crisis has been observed. Blood pressure should be well-controlled prior to initiating INLYTA. Monitor for hypertension and treat as needed. Withhold and then dose reduce INLYTA or permanently discontinue based on severity of hypertension. ( 5.1 ) • Arterial and Venous Thromboembolic Events: Arterial and venous thrombotic events have been observed and can be fatal. Use with caution in patients who are at increased risk for these events. Permanently discontinue INLYTA if an arterial thromboembolic event occurs during treatment. Withhold INLYTA and then resume at same dose or permanently discontinue based on severity of VTE. ( 5.2 , 5.3 ) • Hemorrhage: Hemorrhagic events, including fatal events, have been reported. INLYTA has not been studied in patients with evidence of untreated brain metastasis or recent active gastrointestinal bleeding and should not be used in those patients. Withhold and then dose reduce INLYTA or discontinue based on severity and persistence of hemorrhage. ( 5.4 ) • Cardiac Failure: Cardiac failure has been observed and can be fatal. Monitor for signs or symptoms of cardiac failure throughout treatment with INLYTA. Management of cardiac failure may require dose reduction, dose interruption or permanent discontinuation of INLYTA. ( 5.5 ) • Gastrointestinal Perforation and Fistula Formation: Gastrointestinal perforation and fistula, including death, have occurred. Use with caution in patients at risk for gastrointestinal perforation or fistula. ( 5.6 ) • Hypothyroidism: Hypothyroidism requiring thyroid hormone replacement has been reported. Monitor thyroid function before initiation of, and periodically throughout, treatment with INLYTA. ( 5.7 ) • Impaired Wound Healing: Withhold INLYTA for at least 2 days prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. Resume INLYTA at a reduced dose or discontinue based on severity and persistence of the impaired wound healing. The safety of resumption of INLYTA after resolution of wound healing complications has not been established. ( 5.8 ) • Reversible Posterior Leukoencephalopathy Syndrome (RPLS): RPLS has been observed. Permanently discontinue INLYTA if signs or symptoms of RPLS occur. ( 5.9 ) • Proteinuria: Monitor for proteinuria before initiation of, and periodically throughout, treatment with INLYTA. For moderate to severe proteinuria, withhold and then dose reduce INLYTA. ( 5.10 ) • Hepatotoxicity: Liver enzyme elevation has occurred during treatment with INLYTA as a single agent. Monitor ALT, AST and bilirubin before initiation of, and periodically throughout, treatment with INLYTA. When used in combination with avelumab or pembrolizumab, higher frequencies of Grade 3 and 4 ALT and AST elevation may occur. Consider more frequent monitoring of liver enzymes. Withhold INLYTA and avelumab or pembrolizumab, initiate corticosteroid therapy as needed, and/or permanently discontinue the combination for severe or life-threatening hepatotoxicity. ( 5.11 ) • Use in Patients with Hepatic Impairment: Decrease the starting dose of INLYTA if used in patients with moderate hepatic impairment. INLYTA has not been studied in patients with severe hepatic impairment. ( 2.2 , 5.12 ) • Major adverse cardiovascular events (INLYTA in combination with avelumab): Optimize management of cardiovascular risk factors. Permanently discontinue INLYTA in combination with avelumab for Grade 3–4 events. ( 5.13 ) • Embryo-Fetal Toxicity: INLYTA can cause fetal harm. Advise patients of the potential risk to the fetus and to use effective contraception. ( 5.14 , 8.1 , 8.3 )
Indications and Usage
INLYTA is a kinase inhibitor indicated: • in combination with avelumab, for the first-line treatment of patients with advanced renal cell carcinoma (RCC). ( 1.1 ) • in combination with pembrolizumab, for the first-line treatment of patients with advanced RCC. ( 1.1 ) • as a single agent, for the treatment of advanced renal cell carcinoma (RCC) after failure of one prior systemic therapy. ( 1.2 )
Dosage and Administration
• INLYTA 5 mg orally twice daily with avelumab 800 mg every 2 weeks. ( 2.1 ) • INLYTA 5 mg orally twice daily with pembrolizumab 200 mg every 3 weeks or 400 mg every 6 weeks. ( 2.1 ) • INLYTA as a single agent the starting dose is 5 mg orally twice daily. ( 2.1 ) • Dose adjustments can be made based on individual safety and tolerability. ( 2.2 ) • Administer INLYTA dose approximately 12 hours apart with or without food. ( 2.1 ) • INLYTA should be swallowed whole with a glass of water. ( 2.1 ) • See Full Prescribing Information for dosage modifications for adverse reactions. ( 2.2 ) • If a strong CYP3A4/5 inhibitor is required, decrease the INLYTA dose by approximately half. ( 2.2 ) • For patients with moderate hepatic impairment, decrease the starting dose by approximately half. ( 2.2 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are discussed elsewhere in the labeling [see Warnings and Precautions (5) ] : • Hypertension [see Warnings and Precautions (5.1) ] • Arterial thromboembolic events [see Warnings and Precautions (5.2) ] • Venous thromboembolic events [see Warnings and Precautions (5.3) ] • Hemorrhage [see Warnings and Precautions (5.4) ] • Cardiac failure [see Warnings and Precautions (5.5) ] • Gastrointestinal perforation and fistula formation [see Warnings and Precautions (5.6) ] • Thyroid dysfunction [see Warnings and Precautions (5.7) ] • Reversible posterior leukoencephalopathy syndrome [see Warnings and Precautions (5.9) ] • Proteinuria [see Warnings and Precautions (5.10) ] • Hepatotoxicity [see Warnings and Precautions (5.11) ] • Hepatic impairment [see Warnings and Precautions (5.12) ]
Drug Interactions
Storage and Handling
INLYTA tablets are supplied as follows: • 1 mg tablets are red film-coated, oval tablets debossed with "Pfizer" on one side and "1 XNB" on the other; available in bottles of 180: NDC 0069-0145-01. • 5 mg tablets are red film-coated, triangular tablets debossed with "Pfizer" on one side and "5 XNB" on the other; available in bottles of 60: NDC 0069-0151-11. • Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
How Supplied
INLYTA tablets are supplied as follows: • 1 mg tablets are red film-coated, oval tablets debossed with "Pfizer" on one side and "1 XNB" on the other; available in bottles of 180: NDC 0069-0145-01. • 5 mg tablets are red film-coated, triangular tablets debossed with "Pfizer" on one side and "5 XNB" on the other; available in bottles of 60: NDC 0069-0151-11. • Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Description
INLYTA is a kinase inhibitor indicated: • in combination with avelumab, for the first-line treatment of patients with advanced renal cell carcinoma (RCC). ( 1.1 ) • in combination with pembrolizumab, for the first-line treatment of patients with advanced RCC. ( 1.1 ) • as a single agent, for the treatment of advanced renal cell carcinoma (RCC) after failure of one prior systemic therapy. ( 1.2 )
Section 42229-5
First-Line Advanced RCC
Section 42230-3
|
PATIENT INFORMATION
|
|||
|
Important information: If your healthcare provider prescribes INLYTA for you to be taken with avelumab or pembrolizumab, also read the Medication Guide for avelumab or pembrolizumab. |
|||
|
What is INLYTA?
It is not known if INLYTA is safe and effective in children. |
|||
|
Before taking INLYTA, tell your healthcare provider about all of your medical conditions, including if you:
For females, tell your healthcare provider if you:
For males with female partners who are able to become pregnant:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. INLYTA and certain other medicines can affect each other causing serious side effects. |
|||
|
How should I take INLYTA?
|
|||
|
What should I avoid while taking INLYTA?
|
|||
|
What are the possible side effects of INLYTA?
|
|||
|
|
||
|
|||
|
|||
|
|
||
|
|||
|
|||
|
|
||
|
|||
|
|||
|
|
||
|
|||
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
||
|
The most common side effects of INLYTA with avelumab include: |
|||
|
|
||
|
The most common side effects of INLYTA with pembrolizumab include: |
|||
|
|
||
|
The most common side effects of INLYTA when used alone include: |
|||
|
|
||
|
INLYTA may cause fertility problems in males and females, which may affect your ability to have a child. Talk to your healthcare provider if this is a concern for you. |
|||
|
How should I store INLYTA? Store INLYTA at room temperature between 68°F to 77°F (20°C to 25°C). Keep INLYTA and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of INLYTA.
|
|||
|
What are the ingredients in INLYTA?
|
|||
|
LAB-0439-8.0 |
|||
|
For more information, go to www.inlyta.com or call 877-0744-5675 |
This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 07/2024
10 Overdosage
There is no specific treatment for INLYTA overdose.
In a controlled clinical study with INLYTA for the treatment of patients with RCC, 1 patient inadvertently received a dose of 20 mg twice daily for 4 days and experienced dizziness (Grade 1).
In a clinical dose finding study with INLYTA, subjects who received starting doses of 10 mg twice daily or 20 mg twice daily experienced adverse reactions which included hypertension, seizures associated with hypertension, and fatal hemoptysis.
In cases of suspected overdose, INLYTA should be withheld and supportive care instituted.
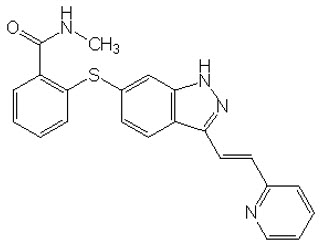
11 Description
INLYTA (axitinib) is a kinase inhibitor. Axitinib has the chemical name N-methyl-2-[3-((E)-2-pyridin-2-yl-vinyl)-1H-indazol-6-ylsulfanyl]-benzamide. The molecular formula is C22H18N4OS and the molecular weight is 386.47 Daltons. The chemical structure is:
Axitinib is a white to light-yellow powder with a pKa of 4.8. The solubility of axitinib in aqueous media over the range pH 1.1 to pH 7.8 is in excess of 0.2 µg/mL. The partition coefficient (n-octanol/water) is 3.5.
INLYTA is supplied as red, film-coated tablets containing either 1 mg or 5 mg of axitinib together with microcrystalline cellulose, lactose monohydrate, croscarmellose sodium, magnesium stearate, and Opadry® II red 32K15441 as inactive ingredients. The Opadry II red 32K15441 film coating contains lactose monohydrate, HPMC 2910/Hypromellose 15cP, titanium dioxide, triacetin (glycerol triacetate), and red iron oxide.
5.4 Hemorrhage
In a controlled clinical study with INLYTA for the treatment of patients with RCC, hemorrhagic events were reported in 58/359 patients (16%) receiving INLYTA and 64/355 patients (18%) receiving sorafenib. Grade 3/4 hemorrhagic events were reported in 5/359 (1%) patients receiving INLYTA (including cerebral hemorrhage, hematuria, hemoptysis, lower gastrointestinal hemorrhage, and melena) and 11/355 (3%) patients receiving sorafenib. Fatal hemorrhage was reported in 1/359 patients (<1%) receiving INLYTA (gastric hemorrhage) and 3/355 patients (1%) receiving sorafenib.
INLYTA has not been studied in patients who have evidence of untreated brain metastasis or recent active gastrointestinal bleeding and should not be used in those patients. Withhold and then dose reduce INLYTA or discontinue based on severity and persistence of hemorrhage.
5.1 Hypertension
In a controlled clinical study with INLYTA for the treatment of patients with RCC, hypertension was reported in 145/359 patients (40%) receiving INLYTA and 103/355 patients (29%) receiving sorafenib. Grade 3/4 hypertension was observed in 56/359 patients (16%) receiving INLYTA and 39/355 patients (11%) receiving sorafenib. Hypertensive crisis was reported in 2/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib. The median onset time for hypertension (systolic blood pressure >150 mmHg or diastolic blood pressure >100 mmHg) was within the first month of the start of INLYTA treatment and blood pressure increases have been observed as early as 4 days after starting INLYTA. Hypertension was managed with standard anti-hypertensive therapy. Discontinuation of INLYTA treatment due to hypertension occurred in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib [see Adverse Reactions (6.1)].
Ensure that blood pressure is well-controlled prior to initiating INLYTA. Monitor patients for hypertension and treat as needed with standard anti-hypertensive therapy. Withhold and then dose reduce INLYTA or permanently discontinue based on severity of hypertension [see Dosage and Administration (2.2)].
5.10 Proteinuria
In a controlled clinical study with INLYTA for the treatment of patients with RCC, proteinuria was reported in 39/359 patients (11%) receiving INLYTA and 26/355 patients (7%) receiving sorafenib. Grade 3 proteinuria was reported in 11/359 patients (3%) receiving INLYTA and 6/355 patients (2%) receiving sorafenib [see Adverse Reactions (6.1)].
Monitoring for proteinuria before initiation of, and periodically throughout, treatment with INLYTA is recommended. For patients who develop moderate to severe proteinuria, withhold and then dose reduce INLYTA [see Dosage and Administration (2.2)].
8.4 Pediatric Use
The safety and effectiveness of INLYTA in pediatric patients have not been established.
The safety and effectiveness of INLYTA were assessed, but not established, in two open label studies: a dose finding study of INLYTA as a single agent in 17 pediatric patients aged 5 to <17 years with recurrent or refractory solid tumors (ADVL1315, NCT02164838) and a randomized study of INLYTA as a single agent or in combination in 7 pediatric patients aged 7 to <17 years (AREN1721, NCT03595124).
No new safety signals were observed with INLYTA in pediatric patients across these studies.
Exposure in pediatric patients who received INLYTA at the maximum tolerated dosage were lower than those previously observed in adults who received the approved recommended starting dosage.
8.5 Geriatric Use
In a controlled clinical study with INLYTA for the treatment of patients with RCC, 123/359 patients (34%) treated with INLYTA were ≥65 years of age. Although greater sensitivity in some older individuals cannot be ruled out, no overall differences were observed in the safety and effectiveness of INLYTA between patients who were ≥65 years of age and younger.
Of the 434 patients randomized to INLYTA 5 mg twice daily administered in combination with avelumab 10 mg/kg in the JAVELIN Renal 101 trial, 38% were 65 years or older and 8% were 75 years or older. No overall difference in safety or efficacy was reported between patients who were ≥65 years of age and younger.
Of the 432 patients randomized to INLYTA 5 mg twice daily administered in combination with pembrolizumab 200 mg in the KEYNOTE-426 trial, 40% were 65 years or older. No overall difference in safety or efficacy was reported between patients who were ≥65 years of age and younger.
No dosage adjustment is required in elderly patients [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
4 Contraindications
None.
5.5 Cardiac Failure
In a controlled clinical study with INLYTA for the treatment of patients with RCC, cardiac failure was reported in 6/359 patients (2%) receiving INLYTA and 3/355 patients (1%) receiving sorafenib. Grade 3/4 cardiac failure was observed in 2/359 patients (1%) receiving INLYTA and 1/355 patients (<1%) receiving sorafenib. Fatal cardiac failure was reported in 2/359 patients (1%) receiving INLYTA and 1/355 patients (<1%) receiving sorafenib. Monitor for signs or symptoms of cardiac failure throughout treatment with INLYTA. Management of cardiac failure may require dose reduction, dose interruption or permanent discontinuation of INLYTA [see Dosage and Administration (2.2)].
6 Adverse Reactions
The following clinically significant adverse reactions are discussed elsewhere in the labeling [see Warnings and Precautions (5)]:
-
•Hypertension [see Warnings and Precautions (5.1)]
-
•Arterial thromboembolic events [see Warnings and Precautions (5.2)]
-
•Venous thromboembolic events [see Warnings and Precautions (5.3)]
-
•Hemorrhage [see Warnings and Precautions (5.4)]
-
•Cardiac failure [see Warnings and Precautions (5.5)]
-
•Gastrointestinal perforation and fistula formation [see Warnings and Precautions (5.6)]
-
•Thyroid dysfunction [see Warnings and Precautions (5.7)]
-
•Reversible posterior leukoencephalopathy syndrome [see Warnings and Precautions (5.9)]
-
•Proteinuria [see Warnings and Precautions (5.10)]
-
•Hepatotoxicity [see Warnings and Precautions (5.11)]
-
•Hepatic impairment [see Warnings and Precautions (5.12)]
7 Drug Interactions
8.7 Renal Impairment
No dedicated renal impairment trial for axitinib has been conducted. Based on the population pharmacokinetic analyses, no significant difference in axitinib clearance was observed in patients with pre-existing mild to severe renal impairment (15 mL/min ≤creatinine clearance [CLcr] <89 mL/min) [see Clinical Pharmacology (12.3)]. No starting dose adjustment is needed for patients with pre-existing mild to severe renal impairment. Caution should be used in patients with end-stage renal disease (CLcr <15 mL/min).
12.2 Pharmacodynamics
The effect of a single oral dose of INLYTA (5 mg) in the absence and presence of 400 mg ketoconazole on the QTc interval was evaluated in a randomized, single-blinded, two-way crossover study in 35 healthy subjects. No large changes in mean QTc interval (i.e., >20 ms) from placebo were detected up to 3 hours post-dose. However, small increases in mean QTc interval (i.e., <10 ms) cannot be ruled out.
12.3 Pharmacokinetics
The population pharmacokinetic analysis pooled data from 17 trials in healthy subjects and patients with cancer. A two-compartment disposition model with first-order absorption and lag-time adequately describes the axitinib concentration-time profile.
7.2 Cyp3a4/5 Inducers
Co-administration of rifampin, a strong inducer of CYP3A4/5, reduced the plasma exposure of axitinib in healthy volunteers. Co-administration of INLYTA with strong CYP3A4/5 inducers (e.g., rifampin, dexamethasone, phenytoin, carbamazepine, rifabutin, rifapentine, phenobarbital, and St. John's wort) should be avoided. Selection of concomitant medication with no or minimal CYP3A4/5 induction potential is recommended [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)]. Moderate CYP3A4/5 inducers (e.g., bosentan, efavirenz, etravirine, modafinil, and nafcillin) may also reduce the plasma exposure of axitinib and should be avoided if possible.
8.6 Hepatic Impairment
In a dedicated hepatic impairment trial, compared to subjects with normal hepatic function, systemic exposure following a single dose of INLYTA was similar in subjects with baseline mild hepatic impairment (Child-Pugh class A) and higher in subjects with baseline moderate hepatic impairment (Child-Pugh class B).
No starting dose adjustment is required when administering INLYTA to patients with mild hepatic impairment (Child-Pugh class A). A starting dose decrease is recommended when administering INLYTA to patients with moderate hepatic impairment (Child-Pugh class B) [see Dosage and Administration (2.2), Warnings and Precautions (5.12), Clinical Pharmacology (12.3)].
INLYTA has not been studied in subjects with severe hepatic impairment (Child-Pugh class C).
1 Indications and Usage
INLYTA is a kinase inhibitor indicated:
-
•in combination with avelumab, for the first-line treatment of patients with advanced renal cell carcinoma (RCC). (1.1)
-
•in combination with pembrolizumab, for the first-line treatment of patients with advanced RCC. (1.1)
-
•as a single agent, for the treatment of advanced renal cell carcinoma (RCC) after failure of one prior systemic therapy. (1.2)
5.7 Thyroid Dysfunction
In a controlled clinical study with INLYTA for the treatment of patients with RCC, hypothyroidism was reported in 69/359 patients (19%) receiving INLYTA and 29/355 patients (8%) receiving sorafenib. Hyperthyroidism was reported in 4/359 patients (1%) receiving INLYTA and 4/355 patients (1%) receiving sorafenib. In patients who had thyroid stimulating hormone (TSH) <5 μU/mL before treatment, elevations of TSH to ≥10 μU/mL occurred in 79/245 patients (32%) receiving INLYTA and 25/232 patients (11%) receiving sorafenib [see Adverse Reactions (6.1)].
Monitor thyroid function before initiation of, and periodically throughout, treatment with INLYTA. Treat hypothyroidism and hyperthyroidism according to standard medical practice to maintain euthyroid state.
7.1 Cyp3a4/5 Inhibitors
Co-administration of ketoconazole, a strong inhibitor of CYP3A4/5, increased the plasma exposure of axitinib in healthy volunteers. Co-administration of INLYTA with strong CYP3A4/5 inhibitors should be avoided. Grapefruit or grapefruit juice may also increase axitinib plasma concentrations and should be avoided. Selection of concomitant medication with no or minimal CYP3A4/5 inhibition potential is recommended. If a strong CYP3A4/5 inhibitor must be co-administered, the INLYTA dose should be reduced [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
12.1 Mechanism of Action
Axitinib has been shown to inhibit receptor tyrosine kinases including vascular endothelial growth factor receptors (VEGFR)-1, VEGFR-2, and VEGFR-3 at therapeutic plasma concentrations. These receptors are implicated in pathologic angiogenesis, tumor growth, and cancer progression. VEGF-mediated endothelial cell proliferation and survival were inhibited by axitinib in vitro and in mouse models. Axitinib was shown to inhibit tumor growth and phosphorylation of VEGFR-2 in tumor xenograft mouse models.
5 Warnings and Precautions
-
•Hypertension: Hypertension including hypertensive crisis has been observed. Blood pressure should be well-controlled prior to initiating INLYTA. Monitor for hypertension and treat as needed. Withhold and then dose reduce INLYTA or permanently discontinue based on severity of hypertension. (5.1)
-
•Arterial and Venous Thromboembolic Events: Arterial and venous thrombotic events have been observed and can be fatal. Use with caution in patients who are at increased risk for these events. Permanently discontinue INLYTA if an arterial thromboembolic event occurs during treatment. Withhold INLYTA and then resume at same dose or permanently discontinue based on severity of VTE. (5.2, 5.3)
-
•Hemorrhage: Hemorrhagic events, including fatal events, have been reported. INLYTA has not been studied in patients with evidence of untreated brain metastasis or recent active gastrointestinal bleeding and should not be used in those patients. Withhold and then dose reduce INLYTA or discontinue based on severity and persistence of hemorrhage. (5.4)
-
•Cardiac Failure: Cardiac failure has been observed and can be fatal. Monitor for signs or symptoms of cardiac failure throughout treatment with INLYTA. Management of cardiac failure may require dose reduction, dose interruption or permanent discontinuation of INLYTA. (5.5)
-
•Gastrointestinal Perforation and Fistula Formation: Gastrointestinal perforation and fistula, including death, have occurred. Use with caution in patients at risk for gastrointestinal perforation or fistula. (5.6)
-
•Hypothyroidism: Hypothyroidism requiring thyroid hormone replacement has been reported. Monitor thyroid function before initiation of, and periodically throughout, treatment with INLYTA. (5.7)
-
•Impaired Wound Healing: Withhold INLYTA for at least 2 days prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. Resume INLYTA at a reduced dose or discontinue based on severity and persistence of the impaired wound healing. The safety of resumption of INLYTA after resolution of wound healing complications has not been established. (5.8)
-
•Reversible Posterior Leukoencephalopathy Syndrome (RPLS): RPLS has been observed. Permanently discontinue INLYTA if signs or symptoms of RPLS occur. (5.9)
-
•Proteinuria: Monitor for proteinuria before initiation of, and periodically throughout, treatment with INLYTA. For moderate to severe proteinuria, withhold and then dose reduce INLYTA. (5.10)
-
•Hepatotoxicity: Liver enzyme elevation has occurred during treatment with INLYTA as a single agent. Monitor ALT, AST and bilirubin before initiation of, and periodically throughout, treatment with INLYTA. When used in combination with avelumab or pembrolizumab, higher frequencies of Grade 3 and 4 ALT and AST elevation may occur. Consider more frequent monitoring of liver enzymes. Withhold INLYTA and avelumab or pembrolizumab, initiate corticosteroid therapy as needed, and/or permanently discontinue the combination for severe or life-threatening hepatotoxicity. (5.11)
-
•Use in Patients with Hepatic Impairment: Decrease the starting dose of INLYTA if used in patients with moderate hepatic impairment. INLYTA has not been studied in patients with severe hepatic impairment. (2.2, 5.12)
-
•Major adverse cardiovascular events (INLYTA in combination with avelumab): Optimize management of cardiovascular risk factors. Permanently discontinue INLYTA in combination with avelumab for Grade 3–4 events. (5.13)
-
•Embryo-Fetal Toxicity: INLYTA can cause fetal harm. Advise patients of the potential risk to the fetus and to use effective contraception. (5.14, 8.1, 8.3)
5.14 Embryo Fetal Toxicity
Based on its mechanism of action and findings from animal studies, INLYTA can cause fetal harm when administered to a pregnant woman. There are no available human data to inform the drug-associated risk. In developmental toxicity studies in mice, axitinib was teratogenic, embryotoxic and fetotoxic at maternal exposures that were lower than human exposures at the recommended clinical dose. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception during treatment with INLYTA and for 1 week after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with INLYTA and for 1 week after the last dose [see Use in Specific Populations (8.1, 8.3), Clinical Pharmacology (12.1)].
When INLYTA is used in combination with avelumab or pembrolizumab, refer to the full prescribing information of avelumab or pembrolizumab for pregnancy and contraception information.
5.8 Impaired Wound Healing
Impaired wound healing can occur in patients who receive drugs that inhibit the vascular endothelial growth factor (VEGF) signaling pathway. Therefore, INLYTA has the potential to adversely affect wound healing.
Withhold INLYTA for at least 2 days prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. Resume INLYTA at a reduced dose or discontinue based on severity and persistence of the impaired wound healing. The safety of resumption of INLYTA after resolution of wound healing complications has not been established [see Dosage and Administration (2.2)].
2 Dosage and Administration
-
•INLYTA 5 mg orally twice daily with avelumab 800 mg every 2 weeks. (2.1)
-
•INLYTA 5 mg orally twice daily with pembrolizumab 200 mg every 3 weeks or 400 mg every 6 weeks. (2.1)
-
•INLYTA as a single agent the starting dose is 5 mg orally twice daily. (2.1)
-
•Dose adjustments can be made based on individual safety and tolerability. (2.2)
-
•Administer INLYTA dose approximately 12 hours apart with or without food. (2.1)
-
•INLYTA should be swallowed whole with a glass of water. (2.1)
-
•See Full Prescribing Information for dosage modifications for adverse reactions. (2.2)
-
•If a strong CYP3A4/5 inhibitor is required, decrease the INLYTA dose by approximately half. (2.2)
-
•For patients with moderate hepatic impairment, decrease the starting dose by approximately half. (2.2)
3 Dosage Forms and Strengths
-
•1 mg tablets of INLYTA: red, film-coated, oval tablets, debossed with "Pfizer" on one side and "1 XNB" on the other side.
-
•5 mg tablets of INLYTA: red, film-coated, triangular tablets, debossed with "Pfizer" on one side and "5 XNB" on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of INLYTA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Vascular disorders: arterial (including aortic), aneurysms, dissections, and rupture.
14.2 Second Line Advanced Rcc
The safety and efficacy of INLYTA were evaluated in a randomized, open-label, multicenter Phase 3 study. Patients (N=723) with advanced RCC whose disease had progressed on or after treatment with 1 prior systemic therapy, including sunitinib-, bevacizumab-, temsirolimus-, or cytokine-containing regimens were randomized (1:1) to receive INLYTA (N=361) or sorafenib (N=362). Progression-free survival (PFS) was assessed by a blinded independent central review committee. Other endpoints included objective response rate (ORR) and overall survival (OS).
Of the patients enrolled in this study, 389 patients (54%) had received 1 prior sunitinib-based therapy, 251 patients (35%) had received 1 prior cytokine-based therapy (interleukin-2 or interferon-alfa), 59 patients (8%) had received 1 prior bevacizumab-based therapy, and 24 patients (3%) had received 1 prior temsirolimus-based therapy. The baseline demographic and disease characteristics were similar between the INLYTA and sorafenib groups with regard to age (median 61 years), gender (72% male), race (75% white, 21% Asian), Eastern Cooperative Oncology Group (ECOG) performance status (55% 0, 45% 1), and histology (99% clear cell).
There was a statistically significant advantage for INLYTA over sorafenib for the endpoint of PFS (see Table 12 and Figure 4). There was no statistically significant difference between the arms in OS.
| Endpoint/Study Population | INLYTA | Sorafenib | HR (95% CI) | P-value |
|---|---|---|---|---|
| CI: Confidence interval; HR: Hazard ratio (INLYTA/sorafenib); ITT: Intent-to-treat; ORR: Objective response rate; NS: Not significant; OS: Overall survival; PFS: Progression-free survival | ||||
|
Overall ITT |
N= 361 |
N = 362 |
||
|
Median PFS Time from randomization to progression or death due to any cause, whichever occurs first.
,
Assessed by independent radiology review according to RECIST. in months (95% CI)
|
6.7 (6.3, 8.6) |
4.7 (4.6, 5.6) |
0.67 (0.54, 0.81) |
<0.0001 One-sided p-value from a log-rank test of treatment stratified by ECOG performance status and prior therapy (comparison is considered statistically significant if the one-sided p-value is <0.023).
|
|
Median OS in months |
20.1 (16.7, 23.4) |
19.2 (17.5, 22.3) |
0.97 (0.80, 1.17) |
NS |
|
ORR % (95% CI) |
19.4 (15.4, 23.9) |
9.4 (6.6, 12.9) |
2.06 Risk ratio is used for ORR. A risk ratio >1 indicated a higher likelihood of responding in the axitinib arm; a risk ratio <1 indicated a higher likelihood of responding in the sorafenib arm. (1.41, 3.00)
|
- P-value not included since it was not adjusted for multiple testing.
|
|
PFS by prior treatment |
||||
|
Sunitinib-refractory subgroup |
N=194 |
N=195 |
||
|
Median, months (95% CI) |
4.8 (4.5, 6.4) |
3.4 (2.8, 4.7) |
0.74 (0.57, 0.96) |
- |
|
Cytokine-refractory subgroup |
N=126 |
N=125 |
||
|
Median, months (95% CI) |
12.1 (10.1, 13.9) |
6.5 (6.3, 8.3) |
0.46 (0.32, 0.68) |
- |
Figure 4. Kaplan-Meier Curve for Progression-Free Survival by Independent Assessment (Intent-to-Treat Population)
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of INLYTA has been evaluated in combination with avelumab in JAVELIN Renal 101 and pembrolizumab in KEYNOTE-426 for the first-line treatment of patients with advanced RCC [see Clinical Studies (14.1)]. The data described [see Adverse Reactions (6.1)] reflect exposure to INLYTA in combination with avelumab in 434 patients and pembrolizumab in 429 patients [see Clinical Studies (14.1)].
The safety of INLYTA has been evaluated in 715 patients in second-line monotherapy studies, which included 537 patients with advanced RCC. The data described [see Adverse Reactions (6.1)] reflect exposure to INLYTA in 359 patients with advanced RCC who participated in a randomized clinical study versus sorafenib [see Clinical Studies (14.2)].
2.2 Dose Modification Guidelines
Dose increase or reduction is recommended based on individual safety and tolerability.
Recommended INLYTA dosage increases and reductions are provided in Table 1.
Over the course of treatment, patients who tolerate INLYTA for at least two consecutive weeks with no adverse reactions Grade >2 (according to the Common Toxicity Criteria for Adverse Events [CTCAE]), are normotensive, and are not receiving anti-hypertension medication, may have their dose increased.
| Dose Modification | Dose Regimen |
|---|---|
|
Recommended starting dosage |
5 mg twice daily |
|
Dosage increase |
|
|
First dose increase |
7 mg twice daily |
|
Second dose increase |
10 mg twice daily |
|
Dosage reduction for management of adverse drug reactions
|
|
|
First dose reduction from 5 mg twice daily
|
3 mg twice daily |
|
Second dose reduction |
2 mg twice daily |
Recommended dosage modifications for adverse reactions for INLYTA are provided in Table 2.
| Adverse Reaction | Severity | Dosage Modifications for INLYTA |
|---|---|---|
|
Hypertension [see Warnings and Precautions (5.1)] |
SBP >150 mmHg or DBP >100 mmHg despite antihypertensive treatment |
|
|
SBP >160 mmHg or DBP >105 mmHg |
|
|
|
Grade 4 or hypertensive crisis |
|
|
|
Hemorrhage [see Warnings and Precautions (5.4)] |
Grade 3 or 4 |
|
|
Cardiac failure [see Warnings and Precautions (5.5)] |
Asymptomatic cardiomyopathy (left ventricular ejection fraction greater than 20% but less than 50% below baseline or below the lower limit of normal if baseline was not obtained) |
|
|
Clinically manifested congestive heart failure |
|
|
|
Impaired wound healing [see Warnings and Precautions (5.8)] |
Any Grade |
|
|
Reversible Posterior Leukoencephalopathy Syndrome [see Warnings and Precautions (5.9)] |
Any Grade |
|
|
Proteinuria [see Warnings and Precautions (5.10) ] |
2 or more grams proteinuria per 24 hours |
|
|
Other Adverse Reactions |
Grade 3 |
|
|
Grade 4 |
|
Table 3 represents additional recommended dosage modifications for adverse reactions when INLYTA is administered in combination with avelumab or pembrolizumab.
See the Full Prescribing Information for additional dosage information for avelumab or pembrolizumab including dose modifications for immune-mediated adverse reactions.
| Treatment | Adverse Reaction |
Severity
Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.0.
|
Dosage Modifications for INLYTA |
|---|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal | |||
|
INLYTA in combination with avelumab OR pembrolizumab |
Liver enzyme elevations Consider corticosteroid therapy
|
ALT or AST at least 3 times ULN but less than 10 times ULN without concurrent total bilirubin at least 2 times ULN |
|
|
ALT or AST increases to more than 3 times ULN with concurrent total bilirubin at least 2 times ULN or ALT or AST at least 10 times ULN |
|
||
|
Diarrhea |
Grade 1–2 |
|
|
|
Grade 3 |
|
||
|
Grade 4 |
|
||
|
INLYTA in combination with avelumab |
Major Adverse Cardiovascular Events (MACE) |
Grade 3 or 4 |
|
5.3 Venous Thromboembolic Events
In clinical trials, venous thromboembolic events have been reported, including deaths. In a controlled clinical study with INLYTA for the treatment of patients with RCC, venous thromboembolic events were reported in 11/359 patients (3%) receiving INLYTA and 2/355 patients (1%) receiving sorafenib. Grade 3/4 venous thromboembolic events were reported in 9/359 patients (3%) receiving INLYTA (including pulmonary embolism, deep vein thrombosis, retinal vein occlusion and retinal vein thrombosis) and 2/355 patients (1%) receiving sorafenib. Fatal pulmonary embolism was reported in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib.
INLYTA has not been studied in patients who had a venous thromboembolic event within the previous 6 months. In clinical trials with INLYTA, venous thromboembolic events were reported in 22/715 patients (3%), with two deaths secondary to pulmonary embolism.
Monitor for signs and symptoms of VTE and PE. Withhold INLYTA and then resume at same dose or permanently discontinue based on severity of VTE.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
5.2 Arterial Thromboembolic Events
In clinical trials, arterial thromboembolic events have been reported, including deaths. In a controlled clinical study with INLYTA for the treatment of patients with RCC, Grade 3/4 arterial thromboembolic events were reported in 4/359 patients (1%) receiving INLYTA and 4/355 patients (1%) receiving sorafenib. Fatal cerebrovascular accident was reported in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib [see Adverse Reactions (6.1)].
INLYTA has not been studied in patients who had an arterial thromboembolic event within the previous 12 months. In clinical trials with INLYTA, arterial thromboembolic events (including transient ischemic attack, cerebrovascular accident, myocardial infarction, and retinal artery occlusion) were reported in 17/715 patients (2%), with two deaths secondary to cerebrovascular accident.
Permanently discontinue INLYTA if an arterial thromboembolic event occurs during treatment.
16 How Supplied/storage and Handling
INLYTA tablets are supplied as follows:
-
•1 mg tablets are red film-coated, oval tablets debossed with "Pfizer" on one side and "1 XNB" on the other; available in bottles of 180: NDC 0069-0145-01.
-
•5 mg tablets are red film-coated, triangular tablets debossed with "Pfizer" on one side and "5 XNB" on the other; available in bottles of 60: NDC 0069-0151-11.
-
•Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
1.1 First Line Advanced Renal Cell Carcinoma
INLYTA in combination with avelumab is indicated for the first-line treatment of patients with advanced renal cell carcinoma (RCC).
INLYTA in combination with pembrolizumab is indicated for the first-line treatment of patients with advanced RCC.
5.12 Use in Patients With Hepatic Impairment
The systemic exposure to axitinib was higher in subjects with moderate hepatic impairment (Child-Pugh class B) compared to subjects with normal hepatic function. A dose decrease is recommended when administering INLYTA to patients with moderate hepatic impairment (Child-Pugh class B). INLYTA has not been studied in patients with severe hepatic impairment (Child-Pugh class C) [see Dosage and Administration (2.2), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
1.2 Second Line Advanced Renal Cell Carcinoma
INLYTA as a single agent is indicated for the treatment of advanced RCC after failure of one prior systemic therapy.
2.4 Dosage Modification for Hepatic Impairment
No starting dose adjustment is required when administering INLYTA to patients with mild hepatic impairment (Child-Pugh class A). Based on the pharmacokinetic data, the INLYTA starting dose should be reduced by approximately half in patients with baseline moderate hepatic impairment (Child-Pugh class B). The subsequent doses can be increased or decreased based on individual safety and tolerability. INLYTA has not been studied in patients with severe hepatic impairment (Child-Pugh class C) [see Warnings and Precautions (5.12), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
5.13 Major Adverse Cardiovascular Events (mace)
INLYTA in combination with avelumab can cause severe and fatal cardiovascular events. Consider baseline and periodic evaluations of left ventricular ejection fraction. Monitor for signs and symptoms of cardiovascular events. Optimize management of cardiovascular risk factors, such as hypertension, diabetes, or dyslipidemia. Permanently discontinue INLYTA and avelumab for Grade 3–4 cardiovascular events.
MACE occurred in 7% of patients with advanced RCC treated with INLYTA in combination with avelumab compared to 3.4% treated with sunitinib in a randomized trial, JAVELIN Renal 101. These events included death due to cardiac events (1.4%), Grade 3–4 myocardial infarction (2.8%), and Grade 3–4 congestive heart failure (1.8%). Median time to onset of MACE was 4.2 months (range: 2 days to 24.5 months).
8.3 Females and Males of Reproductive Potential
Based on findings in animal studies, INLYTA can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)]. When INLYTA is used in combination with avelumab or pembrolizumab, refer to the full prescribing information of avelumab or pembrolizumab for contraception information.
Principal Display Panel 1 Mg Tablet Bottle Label
PROFESSIONAL SAMPLE – NOT FOR SALE
Pfizer
NDC 63539-026-01
Inlyta
®
(axitinib) tablets
1 mg
90 Tablets
Rx only
Principal Display Panel 5 Mg Tablet Bottle Label
PROFESSIONAL SAMPLE – NOT FOR SALE
Pfizer
NDC 63539-044-02
Inlyta
®
(axitinib) tablets
5 mg
30 Tablets
Rx only
5.9 Reversible Posterior Leukoencephalopathy Syndrome
In a controlled clinical study with INLYTA for the treatment of patients with RCC, reversible posterior leukoencephalopathy syndrome (RPLS) was reported in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib [see Adverse Reactions (6.1)]. There were two additional reports of RPLS in other clinical trials with INLYTA.
RPLS is a neurological disorder which can present with headache, seizure, lethargy, confusion, blindness and other visual and neurologic disturbances. Mild to severe hypertension may be present. Magnetic resonance imaging is necessary to confirm the diagnosis of RPLS. Permanently discontinue INLYTA in patients developing RPLS. The safety of reinitiating INLYTA therapy in patients previously experiencing RPLS is not known [see Dosage and Administration (2.2)].
5.6 Gastrointestinal Perforation and Fistula Formation
In a controlled clinical study with INLYTA for the treatment of patients with RCC, gastrointestinal perforation was reported in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib. In clinical trials with INLYTA, gastrointestinal perforation was reported in 5/715 patients (1%), including one death. In addition to cases of gastrointestinal perforation, fistulas were reported in 4/715 patients (1%).
Monitor for symptoms of gastrointestinal perforation or fistula periodically throughout treatment with INLYTA.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with axitinib.
Axitinib was not mutagenic in an in vitro bacterial reverse mutation (Ames) assay and was not clastogenic in the in vitro human lymphocyte chromosome aberration assay. Axitinib was genotoxic in the in vivo mouse bone marrow micronucleus assay.
INLYTA has the potential to impair reproductive function and fertility in humans. In repeat-dose toxicology studies, findings in the male reproductive tract were observed in the testes/epididymis (decreased organ weight, atrophy or degeneration, decreased numbers of germinal cells, hypospermia or abnormal sperm forms, reduced sperm density and count) at ≥15 mg/kg/dose administered orally twice daily in mice (approximately 7 times the systemic exposure (AUC) in patients at the recommended starting dose) and ≥1.5 mg/kg/dose administered orally twice daily in dogs (approximately 0.1 times the AUC in patients at the recommended starting dose). Findings in the female reproductive tract in mice and dogs included signs of delayed sexual maturity, reduced or absent corpora lutea, decreased uterine weights and uterine atrophy at ≥5 mg/kg/dose (approximately 1.5 or 0.3 times the AUC in patients at the recommended starting dose compared to mice and dogs, respectively).
In a fertility study in mice, axitinib did not affect mating or fertility rate when administered orally twice daily to males at any dose tested up to 50 mg/kg/dose following at least 70 days of administration (approximately 57 times the AUC in patients at the recommended starting dose). In female mice, reduced fertility and embryonic viability were observed at all doses tested (≥15 mg/kg/dose administered orally twice daily) following at least 15 days of treatment with axitinib (approximately 10 times the AUC in patients at the recommended starting dose).
Structured Label Content
Section 42229-5 (42229-5)
First-Line Advanced RCC
Section 42230-3 (42230-3)
|
PATIENT INFORMATION
|
|||
|
Important information: If your healthcare provider prescribes INLYTA for you to be taken with avelumab or pembrolizumab, also read the Medication Guide for avelumab or pembrolizumab. |
|||
|
What is INLYTA?
It is not known if INLYTA is safe and effective in children. |
|||
|
Before taking INLYTA, tell your healthcare provider about all of your medical conditions, including if you:
For females, tell your healthcare provider if you:
For males with female partners who are able to become pregnant:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. INLYTA and certain other medicines can affect each other causing serious side effects. |
|||
|
How should I take INLYTA?
|
|||
|
What should I avoid while taking INLYTA?
|
|||
|
What are the possible side effects of INLYTA?
|
|||
|
|
||
|
|||
|
|||
|
|
||
|
|||
|
|||
|
|
||
|
|||
|
|||
|
|
||
|
|||
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
||
|
The most common side effects of INLYTA with avelumab include: |
|||
|
|
||
|
The most common side effects of INLYTA with pembrolizumab include: |
|||
|
|
||
|
The most common side effects of INLYTA when used alone include: |
|||
|
|
||
|
INLYTA may cause fertility problems in males and females, which may affect your ability to have a child. Talk to your healthcare provider if this is a concern for you. |
|||
|
How should I store INLYTA? Store INLYTA at room temperature between 68°F to 77°F (20°C to 25°C). Keep INLYTA and all medicines out of the reach of children. |
|||
|
General information about the safe and effective use of INLYTA.
|
|||
|
What are the ingredients in INLYTA?
|
|||
|
LAB-0439-8.0 |
|||
|
For more information, go to www.inlyta.com or call 877-0744-5675 |
This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 07/2024
10 Overdosage (10 OVERDOSAGE)
There is no specific treatment for INLYTA overdose.
In a controlled clinical study with INLYTA for the treatment of patients with RCC, 1 patient inadvertently received a dose of 20 mg twice daily for 4 days and experienced dizziness (Grade 1).
In a clinical dose finding study with INLYTA, subjects who received starting doses of 10 mg twice daily or 20 mg twice daily experienced adverse reactions which included hypertension, seizures associated with hypertension, and fatal hemoptysis.
In cases of suspected overdose, INLYTA should be withheld and supportive care instituted.
11 Description (11 DESCRIPTION)
INLYTA (axitinib) is a kinase inhibitor. Axitinib has the chemical name N-methyl-2-[3-((E)-2-pyridin-2-yl-vinyl)-1H-indazol-6-ylsulfanyl]-benzamide. The molecular formula is C22H18N4OS and the molecular weight is 386.47 Daltons. The chemical structure is:
Axitinib is a white to light-yellow powder with a pKa of 4.8. The solubility of axitinib in aqueous media over the range pH 1.1 to pH 7.8 is in excess of 0.2 µg/mL. The partition coefficient (n-octanol/water) is 3.5.
INLYTA is supplied as red, film-coated tablets containing either 1 mg or 5 mg of axitinib together with microcrystalline cellulose, lactose monohydrate, croscarmellose sodium, magnesium stearate, and Opadry® II red 32K15441 as inactive ingredients. The Opadry II red 32K15441 film coating contains lactose monohydrate, HPMC 2910/Hypromellose 15cP, titanium dioxide, triacetin (glycerol triacetate), and red iron oxide.
5.4 Hemorrhage
In a controlled clinical study with INLYTA for the treatment of patients with RCC, hemorrhagic events were reported in 58/359 patients (16%) receiving INLYTA and 64/355 patients (18%) receiving sorafenib. Grade 3/4 hemorrhagic events were reported in 5/359 (1%) patients receiving INLYTA (including cerebral hemorrhage, hematuria, hemoptysis, lower gastrointestinal hemorrhage, and melena) and 11/355 (3%) patients receiving sorafenib. Fatal hemorrhage was reported in 1/359 patients (<1%) receiving INLYTA (gastric hemorrhage) and 3/355 patients (1%) receiving sorafenib.
INLYTA has not been studied in patients who have evidence of untreated brain metastasis or recent active gastrointestinal bleeding and should not be used in those patients. Withhold and then dose reduce INLYTA or discontinue based on severity and persistence of hemorrhage.
5.1 Hypertension
In a controlled clinical study with INLYTA for the treatment of patients with RCC, hypertension was reported in 145/359 patients (40%) receiving INLYTA and 103/355 patients (29%) receiving sorafenib. Grade 3/4 hypertension was observed in 56/359 patients (16%) receiving INLYTA and 39/355 patients (11%) receiving sorafenib. Hypertensive crisis was reported in 2/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib. The median onset time for hypertension (systolic blood pressure >150 mmHg or diastolic blood pressure >100 mmHg) was within the first month of the start of INLYTA treatment and blood pressure increases have been observed as early as 4 days after starting INLYTA. Hypertension was managed with standard anti-hypertensive therapy. Discontinuation of INLYTA treatment due to hypertension occurred in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib [see Adverse Reactions (6.1)].
Ensure that blood pressure is well-controlled prior to initiating INLYTA. Monitor patients for hypertension and treat as needed with standard anti-hypertensive therapy. Withhold and then dose reduce INLYTA or permanently discontinue based on severity of hypertension [see Dosage and Administration (2.2)].
5.10 Proteinuria
In a controlled clinical study with INLYTA for the treatment of patients with RCC, proteinuria was reported in 39/359 patients (11%) receiving INLYTA and 26/355 patients (7%) receiving sorafenib. Grade 3 proteinuria was reported in 11/359 patients (3%) receiving INLYTA and 6/355 patients (2%) receiving sorafenib [see Adverse Reactions (6.1)].
Monitoring for proteinuria before initiation of, and periodically throughout, treatment with INLYTA is recommended. For patients who develop moderate to severe proteinuria, withhold and then dose reduce INLYTA [see Dosage and Administration (2.2)].
8.4 Pediatric Use
The safety and effectiveness of INLYTA in pediatric patients have not been established.
The safety and effectiveness of INLYTA were assessed, but not established, in two open label studies: a dose finding study of INLYTA as a single agent in 17 pediatric patients aged 5 to <17 years with recurrent or refractory solid tumors (ADVL1315, NCT02164838) and a randomized study of INLYTA as a single agent or in combination in 7 pediatric patients aged 7 to <17 years (AREN1721, NCT03595124).
No new safety signals were observed with INLYTA in pediatric patients across these studies.
Exposure in pediatric patients who received INLYTA at the maximum tolerated dosage were lower than those previously observed in adults who received the approved recommended starting dosage.
8.5 Geriatric Use
In a controlled clinical study with INLYTA for the treatment of patients with RCC, 123/359 patients (34%) treated with INLYTA were ≥65 years of age. Although greater sensitivity in some older individuals cannot be ruled out, no overall differences were observed in the safety and effectiveness of INLYTA between patients who were ≥65 years of age and younger.
Of the 434 patients randomized to INLYTA 5 mg twice daily administered in combination with avelumab 10 mg/kg in the JAVELIN Renal 101 trial, 38% were 65 years or older and 8% were 75 years or older. No overall difference in safety or efficacy was reported between patients who were ≥65 years of age and younger.
Of the 432 patients randomized to INLYTA 5 mg twice daily administered in combination with pembrolizumab 200 mg in the KEYNOTE-426 trial, 40% were 65 years or older. No overall difference in safety or efficacy was reported between patients who were ≥65 years of age and younger.
No dosage adjustment is required in elderly patients [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
None.
5.5 Cardiac Failure
In a controlled clinical study with INLYTA for the treatment of patients with RCC, cardiac failure was reported in 6/359 patients (2%) receiving INLYTA and 3/355 patients (1%) receiving sorafenib. Grade 3/4 cardiac failure was observed in 2/359 patients (1%) receiving INLYTA and 1/355 patients (<1%) receiving sorafenib. Fatal cardiac failure was reported in 2/359 patients (1%) receiving INLYTA and 1/355 patients (<1%) receiving sorafenib. Monitor for signs or symptoms of cardiac failure throughout treatment with INLYTA. Management of cardiac failure may require dose reduction, dose interruption or permanent discontinuation of INLYTA [see Dosage and Administration (2.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed elsewhere in the labeling [see Warnings and Precautions (5)]:
-
•Hypertension [see Warnings and Precautions (5.1)]
-
•Arterial thromboembolic events [see Warnings and Precautions (5.2)]
-
•Venous thromboembolic events [see Warnings and Precautions (5.3)]
-
•Hemorrhage [see Warnings and Precautions (5.4)]
-
•Cardiac failure [see Warnings and Precautions (5.5)]
-
•Gastrointestinal perforation and fistula formation [see Warnings and Precautions (5.6)]
-
•Thyroid dysfunction [see Warnings and Precautions (5.7)]
-
•Reversible posterior leukoencephalopathy syndrome [see Warnings and Precautions (5.9)]
-
•Proteinuria [see Warnings and Precautions (5.10)]
-
•Hepatotoxicity [see Warnings and Precautions (5.11)]
-
•Hepatic impairment [see Warnings and Precautions (5.12)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.7 Renal Impairment
No dedicated renal impairment trial for axitinib has been conducted. Based on the population pharmacokinetic analyses, no significant difference in axitinib clearance was observed in patients with pre-existing mild to severe renal impairment (15 mL/min ≤creatinine clearance [CLcr] <89 mL/min) [see Clinical Pharmacology (12.3)]. No starting dose adjustment is needed for patients with pre-existing mild to severe renal impairment. Caution should be used in patients with end-stage renal disease (CLcr <15 mL/min).
12.2 Pharmacodynamics
The effect of a single oral dose of INLYTA (5 mg) in the absence and presence of 400 mg ketoconazole on the QTc interval was evaluated in a randomized, single-blinded, two-way crossover study in 35 healthy subjects. No large changes in mean QTc interval (i.e., >20 ms) from placebo were detected up to 3 hours post-dose. However, small increases in mean QTc interval (i.e., <10 ms) cannot be ruled out.
12.3 Pharmacokinetics
The population pharmacokinetic analysis pooled data from 17 trials in healthy subjects and patients with cancer. A two-compartment disposition model with first-order absorption and lag-time adequately describes the axitinib concentration-time profile.
7.2 Cyp3a4/5 Inducers (7.2 CYP3A4/5 Inducers)
Co-administration of rifampin, a strong inducer of CYP3A4/5, reduced the plasma exposure of axitinib in healthy volunteers. Co-administration of INLYTA with strong CYP3A4/5 inducers (e.g., rifampin, dexamethasone, phenytoin, carbamazepine, rifabutin, rifapentine, phenobarbital, and St. John's wort) should be avoided. Selection of concomitant medication with no or minimal CYP3A4/5 induction potential is recommended [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)]. Moderate CYP3A4/5 inducers (e.g., bosentan, efavirenz, etravirine, modafinil, and nafcillin) may also reduce the plasma exposure of axitinib and should be avoided if possible.
8.6 Hepatic Impairment
In a dedicated hepatic impairment trial, compared to subjects with normal hepatic function, systemic exposure following a single dose of INLYTA was similar in subjects with baseline mild hepatic impairment (Child-Pugh class A) and higher in subjects with baseline moderate hepatic impairment (Child-Pugh class B).
No starting dose adjustment is required when administering INLYTA to patients with mild hepatic impairment (Child-Pugh class A). A starting dose decrease is recommended when administering INLYTA to patients with moderate hepatic impairment (Child-Pugh class B) [see Dosage and Administration (2.2), Warnings and Precautions (5.12), Clinical Pharmacology (12.3)].
INLYTA has not been studied in subjects with severe hepatic impairment (Child-Pugh class C).
1 Indications and Usage (1 INDICATIONS AND USAGE)
INLYTA is a kinase inhibitor indicated:
-
•in combination with avelumab, for the first-line treatment of patients with advanced renal cell carcinoma (RCC). (1.1)
-
•in combination with pembrolizumab, for the first-line treatment of patients with advanced RCC. (1.1)
-
•as a single agent, for the treatment of advanced renal cell carcinoma (RCC) after failure of one prior systemic therapy. (1.2)
5.7 Thyroid Dysfunction
In a controlled clinical study with INLYTA for the treatment of patients with RCC, hypothyroidism was reported in 69/359 patients (19%) receiving INLYTA and 29/355 patients (8%) receiving sorafenib. Hyperthyroidism was reported in 4/359 patients (1%) receiving INLYTA and 4/355 patients (1%) receiving sorafenib. In patients who had thyroid stimulating hormone (TSH) <5 μU/mL before treatment, elevations of TSH to ≥10 μU/mL occurred in 79/245 patients (32%) receiving INLYTA and 25/232 patients (11%) receiving sorafenib [see Adverse Reactions (6.1)].
Monitor thyroid function before initiation of, and periodically throughout, treatment with INLYTA. Treat hypothyroidism and hyperthyroidism according to standard medical practice to maintain euthyroid state.
7.1 Cyp3a4/5 Inhibitors (7.1 CYP3A4/5 Inhibitors)
Co-administration of ketoconazole, a strong inhibitor of CYP3A4/5, increased the plasma exposure of axitinib in healthy volunteers. Co-administration of INLYTA with strong CYP3A4/5 inhibitors should be avoided. Grapefruit or grapefruit juice may also increase axitinib plasma concentrations and should be avoided. Selection of concomitant medication with no or minimal CYP3A4/5 inhibition potential is recommended. If a strong CYP3A4/5 inhibitor must be co-administered, the INLYTA dose should be reduced [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
12.1 Mechanism of Action
Axitinib has been shown to inhibit receptor tyrosine kinases including vascular endothelial growth factor receptors (VEGFR)-1, VEGFR-2, and VEGFR-3 at therapeutic plasma concentrations. These receptors are implicated in pathologic angiogenesis, tumor growth, and cancer progression. VEGF-mediated endothelial cell proliferation and survival were inhibited by axitinib in vitro and in mouse models. Axitinib was shown to inhibit tumor growth and phosphorylation of VEGFR-2 in tumor xenograft mouse models.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Hypertension: Hypertension including hypertensive crisis has been observed. Blood pressure should be well-controlled prior to initiating INLYTA. Monitor for hypertension and treat as needed. Withhold and then dose reduce INLYTA or permanently discontinue based on severity of hypertension. (5.1)
-
•Arterial and Venous Thromboembolic Events: Arterial and venous thrombotic events have been observed and can be fatal. Use with caution in patients who are at increased risk for these events. Permanently discontinue INLYTA if an arterial thromboembolic event occurs during treatment. Withhold INLYTA and then resume at same dose or permanently discontinue based on severity of VTE. (5.2, 5.3)
-
•Hemorrhage: Hemorrhagic events, including fatal events, have been reported. INLYTA has not been studied in patients with evidence of untreated brain metastasis or recent active gastrointestinal bleeding and should not be used in those patients. Withhold and then dose reduce INLYTA or discontinue based on severity and persistence of hemorrhage. (5.4)
-
•Cardiac Failure: Cardiac failure has been observed and can be fatal. Monitor for signs or symptoms of cardiac failure throughout treatment with INLYTA. Management of cardiac failure may require dose reduction, dose interruption or permanent discontinuation of INLYTA. (5.5)
-
•Gastrointestinal Perforation and Fistula Formation: Gastrointestinal perforation and fistula, including death, have occurred. Use with caution in patients at risk for gastrointestinal perforation or fistula. (5.6)
-
•Hypothyroidism: Hypothyroidism requiring thyroid hormone replacement has been reported. Monitor thyroid function before initiation of, and periodically throughout, treatment with INLYTA. (5.7)
-
•Impaired Wound Healing: Withhold INLYTA for at least 2 days prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. Resume INLYTA at a reduced dose or discontinue based on severity and persistence of the impaired wound healing. The safety of resumption of INLYTA after resolution of wound healing complications has not been established. (5.8)
-
•Reversible Posterior Leukoencephalopathy Syndrome (RPLS): RPLS has been observed. Permanently discontinue INLYTA if signs or symptoms of RPLS occur. (5.9)
-
•Proteinuria: Monitor for proteinuria before initiation of, and periodically throughout, treatment with INLYTA. For moderate to severe proteinuria, withhold and then dose reduce INLYTA. (5.10)
-
•Hepatotoxicity: Liver enzyme elevation has occurred during treatment with INLYTA as a single agent. Monitor ALT, AST and bilirubin before initiation of, and periodically throughout, treatment with INLYTA. When used in combination with avelumab or pembrolizumab, higher frequencies of Grade 3 and 4 ALT and AST elevation may occur. Consider more frequent monitoring of liver enzymes. Withhold INLYTA and avelumab or pembrolizumab, initiate corticosteroid therapy as needed, and/or permanently discontinue the combination for severe or life-threatening hepatotoxicity. (5.11)
-
•Use in Patients with Hepatic Impairment: Decrease the starting dose of INLYTA if used in patients with moderate hepatic impairment. INLYTA has not been studied in patients with severe hepatic impairment. (2.2, 5.12)
-
•Major adverse cardiovascular events (INLYTA in combination with avelumab): Optimize management of cardiovascular risk factors. Permanently discontinue INLYTA in combination with avelumab for Grade 3–4 events. (5.13)
-
•Embryo-Fetal Toxicity: INLYTA can cause fetal harm. Advise patients of the potential risk to the fetus and to use effective contraception. (5.14, 8.1, 8.3)
5.14 Embryo Fetal Toxicity (5.14 Embryo-Fetal Toxicity)
Based on its mechanism of action and findings from animal studies, INLYTA can cause fetal harm when administered to a pregnant woman. There are no available human data to inform the drug-associated risk. In developmental toxicity studies in mice, axitinib was teratogenic, embryotoxic and fetotoxic at maternal exposures that were lower than human exposures at the recommended clinical dose. Advise females of reproductive potential of the potential risk to the fetus and to use effective contraception during treatment with INLYTA and for 1 week after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with INLYTA and for 1 week after the last dose [see Use in Specific Populations (8.1, 8.3), Clinical Pharmacology (12.1)].
When INLYTA is used in combination with avelumab or pembrolizumab, refer to the full prescribing information of avelumab or pembrolizumab for pregnancy and contraception information.
5.8 Impaired Wound Healing
Impaired wound healing can occur in patients who receive drugs that inhibit the vascular endothelial growth factor (VEGF) signaling pathway. Therefore, INLYTA has the potential to adversely affect wound healing.
Withhold INLYTA for at least 2 days prior to elective surgery. Do not administer for at least 2 weeks following major surgery and until adequate wound healing. Resume INLYTA at a reduced dose or discontinue based on severity and persistence of the impaired wound healing. The safety of resumption of INLYTA after resolution of wound healing complications has not been established [see Dosage and Administration (2.2)].
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•INLYTA 5 mg orally twice daily with avelumab 800 mg every 2 weeks. (2.1)
-
•INLYTA 5 mg orally twice daily with pembrolizumab 200 mg every 3 weeks or 400 mg every 6 weeks. (2.1)
-
•INLYTA as a single agent the starting dose is 5 mg orally twice daily. (2.1)
-
•Dose adjustments can be made based on individual safety and tolerability. (2.2)
-
•Administer INLYTA dose approximately 12 hours apart with or without food. (2.1)
-
•INLYTA should be swallowed whole with a glass of water. (2.1)
-
•See Full Prescribing Information for dosage modifications for adverse reactions. (2.2)
-
•If a strong CYP3A4/5 inhibitor is required, decrease the INLYTA dose by approximately half. (2.2)
-
•For patients with moderate hepatic impairment, decrease the starting dose by approximately half. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
-
•1 mg tablets of INLYTA: red, film-coated, oval tablets, debossed with "Pfizer" on one side and "1 XNB" on the other side.
-
•5 mg tablets of INLYTA: red, film-coated, triangular tablets, debossed with "Pfizer" on one side and "5 XNB" on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of INLYTA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Vascular disorders: arterial (including aortic), aneurysms, dissections, and rupture.
14.2 Second Line Advanced Rcc (14.2 Second-Line Advanced RCC)
The safety and efficacy of INLYTA were evaluated in a randomized, open-label, multicenter Phase 3 study. Patients (N=723) with advanced RCC whose disease had progressed on or after treatment with 1 prior systemic therapy, including sunitinib-, bevacizumab-, temsirolimus-, or cytokine-containing regimens were randomized (1:1) to receive INLYTA (N=361) or sorafenib (N=362). Progression-free survival (PFS) was assessed by a blinded independent central review committee. Other endpoints included objective response rate (ORR) and overall survival (OS).
Of the patients enrolled in this study, 389 patients (54%) had received 1 prior sunitinib-based therapy, 251 patients (35%) had received 1 prior cytokine-based therapy (interleukin-2 or interferon-alfa), 59 patients (8%) had received 1 prior bevacizumab-based therapy, and 24 patients (3%) had received 1 prior temsirolimus-based therapy. The baseline demographic and disease characteristics were similar between the INLYTA and sorafenib groups with regard to age (median 61 years), gender (72% male), race (75% white, 21% Asian), Eastern Cooperative Oncology Group (ECOG) performance status (55% 0, 45% 1), and histology (99% clear cell).
There was a statistically significant advantage for INLYTA over sorafenib for the endpoint of PFS (see Table 12 and Figure 4). There was no statistically significant difference between the arms in OS.
| Endpoint/Study Population | INLYTA | Sorafenib | HR (95% CI) | P-value |
|---|---|---|---|---|
| CI: Confidence interval; HR: Hazard ratio (INLYTA/sorafenib); ITT: Intent-to-treat; ORR: Objective response rate; NS: Not significant; OS: Overall survival; PFS: Progression-free survival | ||||
|
Overall ITT |
N= 361 |
N = 362 |
||
|
Median PFS Time from randomization to progression or death due to any cause, whichever occurs first.
,
Assessed by independent radiology review according to RECIST. in months (95% CI)
|
6.7 (6.3, 8.6) |
4.7 (4.6, 5.6) |
0.67 (0.54, 0.81) |
<0.0001 One-sided p-value from a log-rank test of treatment stratified by ECOG performance status and prior therapy (comparison is considered statistically significant if the one-sided p-value is <0.023).
|
|
Median OS in months |
20.1 (16.7, 23.4) |
19.2 (17.5, 22.3) |
0.97 (0.80, 1.17) |
NS |
|
ORR % (95% CI) |
19.4 (15.4, 23.9) |
9.4 (6.6, 12.9) |
2.06 Risk ratio is used for ORR. A risk ratio >1 indicated a higher likelihood of responding in the axitinib arm; a risk ratio <1 indicated a higher likelihood of responding in the sorafenib arm. (1.41, 3.00)
|
- P-value not included since it was not adjusted for multiple testing.
|
|
PFS by prior treatment |
||||
|
Sunitinib-refractory subgroup |
N=194 |
N=195 |
||
|
Median, months (95% CI) |
4.8 (4.5, 6.4) |
3.4 (2.8, 4.7) |
0.74 (0.57, 0.96) |
- |
|
Cytokine-refractory subgroup |
N=126 |
N=125 |
||
|
Median, months (95% CI) |
12.1 (10.1, 13.9) |
6.5 (6.3, 8.3) |
0.46 (0.32, 0.68) |
- |
Figure 4. Kaplan-Meier Curve for Progression-Free Survival by Independent Assessment (Intent-to-Treat Population)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of INLYTA has been evaluated in combination with avelumab in JAVELIN Renal 101 and pembrolizumab in KEYNOTE-426 for the first-line treatment of patients with advanced RCC [see Clinical Studies (14.1)]. The data described [see Adverse Reactions (6.1)] reflect exposure to INLYTA in combination with avelumab in 434 patients and pembrolizumab in 429 patients [see Clinical Studies (14.1)].
The safety of INLYTA has been evaluated in 715 patients in second-line monotherapy studies, which included 537 patients with advanced RCC. The data described [see Adverse Reactions (6.1)] reflect exposure to INLYTA in 359 patients with advanced RCC who participated in a randomized clinical study versus sorafenib [see Clinical Studies (14.2)].
2.2 Dose Modification Guidelines
Dose increase or reduction is recommended based on individual safety and tolerability.
Recommended INLYTA dosage increases and reductions are provided in Table 1.
Over the course of treatment, patients who tolerate INLYTA for at least two consecutive weeks with no adverse reactions Grade >2 (according to the Common Toxicity Criteria for Adverse Events [CTCAE]), are normotensive, and are not receiving anti-hypertension medication, may have their dose increased.
| Dose Modification | Dose Regimen |
|---|---|
|
Recommended starting dosage |
5 mg twice daily |
|
Dosage increase |
|
|
First dose increase |
7 mg twice daily |
|
Second dose increase |
10 mg twice daily |
|
Dosage reduction for management of adverse drug reactions
|
|
|
First dose reduction from 5 mg twice daily
|
3 mg twice daily |
|
Second dose reduction |
2 mg twice daily |
Recommended dosage modifications for adverse reactions for INLYTA are provided in Table 2.
| Adverse Reaction | Severity | Dosage Modifications for INLYTA |
|---|---|---|
|
Hypertension [see Warnings and Precautions (5.1)] |
SBP >150 mmHg or DBP >100 mmHg despite antihypertensive treatment |
|
|
SBP >160 mmHg or DBP >105 mmHg |
|
|
|
Grade 4 or hypertensive crisis |
|
|
|
Hemorrhage [see Warnings and Precautions (5.4)] |
Grade 3 or 4 |
|
|
Cardiac failure [see Warnings and Precautions (5.5)] |
Asymptomatic cardiomyopathy (left ventricular ejection fraction greater than 20% but less than 50% below baseline or below the lower limit of normal if baseline was not obtained) |
|
|
Clinically manifested congestive heart failure |
|
|
|
Impaired wound healing [see Warnings and Precautions (5.8)] |
Any Grade |
|
|
Reversible Posterior Leukoencephalopathy Syndrome [see Warnings and Precautions (5.9)] |
Any Grade |
|
|
Proteinuria [see Warnings and Precautions (5.10) ] |
2 or more grams proteinuria per 24 hours |
|
|
Other Adverse Reactions |
Grade 3 |
|
|
Grade 4 |
|
Table 3 represents additional recommended dosage modifications for adverse reactions when INLYTA is administered in combination with avelumab or pembrolizumab.
See the Full Prescribing Information for additional dosage information for avelumab or pembrolizumab including dose modifications for immune-mediated adverse reactions.
| Treatment | Adverse Reaction |
Severity
Based on Common Terminology Criteria for Adverse Events (CTCAE), version 4.0.
|
Dosage Modifications for INLYTA |
|---|---|---|---|
| ALT = alanine aminotransferase, AST = aspartate aminotransferase, ULN = upper limit normal | |||
|
INLYTA in combination with avelumab OR pembrolizumab |
Liver enzyme elevations Consider corticosteroid therapy
|
ALT or AST at least 3 times ULN but less than 10 times ULN without concurrent total bilirubin at least 2 times ULN |
|
|
ALT or AST increases to more than 3 times ULN with concurrent total bilirubin at least 2 times ULN or ALT or AST at least 10 times ULN |
|
||
|
Diarrhea |
Grade 1–2 |
|
|
|
Grade 3 |
|
||
|
Grade 4 |
|
||
|
INLYTA in combination with avelumab |
Major Adverse Cardiovascular Events (MACE) |
Grade 3 or 4 |
|
5.3 Venous Thromboembolic Events
In clinical trials, venous thromboembolic events have been reported, including deaths. In a controlled clinical study with INLYTA for the treatment of patients with RCC, venous thromboembolic events were reported in 11/359 patients (3%) receiving INLYTA and 2/355 patients (1%) receiving sorafenib. Grade 3/4 venous thromboembolic events were reported in 9/359 patients (3%) receiving INLYTA (including pulmonary embolism, deep vein thrombosis, retinal vein occlusion and retinal vein thrombosis) and 2/355 patients (1%) receiving sorafenib. Fatal pulmonary embolism was reported in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib.
INLYTA has not been studied in patients who had a venous thromboembolic event within the previous 6 months. In clinical trials with INLYTA, venous thromboembolic events were reported in 22/715 patients (3%), with two deaths secondary to pulmonary embolism.
Monitor for signs and symptoms of VTE and PE. Withhold INLYTA and then resume at same dose or permanently discontinue based on severity of VTE.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
5.2 Arterial Thromboembolic Events
In clinical trials, arterial thromboembolic events have been reported, including deaths. In a controlled clinical study with INLYTA for the treatment of patients with RCC, Grade 3/4 arterial thromboembolic events were reported in 4/359 patients (1%) receiving INLYTA and 4/355 patients (1%) receiving sorafenib. Fatal cerebrovascular accident was reported in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib [see Adverse Reactions (6.1)].
INLYTA has not been studied in patients who had an arterial thromboembolic event within the previous 12 months. In clinical trials with INLYTA, arterial thromboembolic events (including transient ischemic attack, cerebrovascular accident, myocardial infarction, and retinal artery occlusion) were reported in 17/715 patients (2%), with two deaths secondary to cerebrovascular accident.
Permanently discontinue INLYTA if an arterial thromboembolic event occurs during treatment.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
INLYTA tablets are supplied as follows:
-
•1 mg tablets are red film-coated, oval tablets debossed with "Pfizer" on one side and "1 XNB" on the other; available in bottles of 180: NDC 0069-0145-01.
-
•5 mg tablets are red film-coated, triangular tablets debossed with "Pfizer" on one side and "5 XNB" on the other; available in bottles of 60: NDC 0069-0151-11.
-
•Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
1.1 First Line Advanced Renal Cell Carcinoma (1.1 First-Line Advanced Renal Cell Carcinoma)
INLYTA in combination with avelumab is indicated for the first-line treatment of patients with advanced renal cell carcinoma (RCC).
INLYTA in combination with pembrolizumab is indicated for the first-line treatment of patients with advanced RCC.
5.12 Use in Patients With Hepatic Impairment (5.12 Use in Patients with Hepatic Impairment)
The systemic exposure to axitinib was higher in subjects with moderate hepatic impairment (Child-Pugh class B) compared to subjects with normal hepatic function. A dose decrease is recommended when administering INLYTA to patients with moderate hepatic impairment (Child-Pugh class B). INLYTA has not been studied in patients with severe hepatic impairment (Child-Pugh class C) [see Dosage and Administration (2.2), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
1.2 Second Line Advanced Renal Cell Carcinoma (1.2 Second-Line Advanced Renal Cell Carcinoma)
INLYTA as a single agent is indicated for the treatment of advanced RCC after failure of one prior systemic therapy.
2.4 Dosage Modification for Hepatic Impairment
No starting dose adjustment is required when administering INLYTA to patients with mild hepatic impairment (Child-Pugh class A). Based on the pharmacokinetic data, the INLYTA starting dose should be reduced by approximately half in patients with baseline moderate hepatic impairment (Child-Pugh class B). The subsequent doses can be increased or decreased based on individual safety and tolerability. INLYTA has not been studied in patients with severe hepatic impairment (Child-Pugh class C) [see Warnings and Precautions (5.12), Use in Specific Populations (8.6), and Clinical Pharmacology (12.3)].
5.13 Major Adverse Cardiovascular Events (mace) (5.13 Major Adverse Cardiovascular Events (MACE))
INLYTA in combination with avelumab can cause severe and fatal cardiovascular events. Consider baseline and periodic evaluations of left ventricular ejection fraction. Monitor for signs and symptoms of cardiovascular events. Optimize management of cardiovascular risk factors, such as hypertension, diabetes, or dyslipidemia. Permanently discontinue INLYTA and avelumab for Grade 3–4 cardiovascular events.
MACE occurred in 7% of patients with advanced RCC treated with INLYTA in combination with avelumab compared to 3.4% treated with sunitinib in a randomized trial, JAVELIN Renal 101. These events included death due to cardiac events (1.4%), Grade 3–4 myocardial infarction (2.8%), and Grade 3–4 congestive heart failure (1.8%). Median time to onset of MACE was 4.2 months (range: 2 days to 24.5 months).
8.3 Females and Males of Reproductive Potential
Based on findings in animal studies, INLYTA can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1)]. When INLYTA is used in combination with avelumab or pembrolizumab, refer to the full prescribing information of avelumab or pembrolizumab for contraception information.
Principal Display Panel 1 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 1 mg Tablet Bottle Label)
PROFESSIONAL SAMPLE – NOT FOR SALE
Pfizer
NDC 63539-026-01
Inlyta
®
(axitinib) tablets
1 mg
90 Tablets
Rx only
Principal Display Panel 5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 5 mg Tablet Bottle Label)
PROFESSIONAL SAMPLE – NOT FOR SALE
Pfizer
NDC 63539-044-02
Inlyta
®
(axitinib) tablets
5 mg
30 Tablets
Rx only
5.9 Reversible Posterior Leukoencephalopathy Syndrome
In a controlled clinical study with INLYTA for the treatment of patients with RCC, reversible posterior leukoencephalopathy syndrome (RPLS) was reported in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib [see Adverse Reactions (6.1)]. There were two additional reports of RPLS in other clinical trials with INLYTA.
RPLS is a neurological disorder which can present with headache, seizure, lethargy, confusion, blindness and other visual and neurologic disturbances. Mild to severe hypertension may be present. Magnetic resonance imaging is necessary to confirm the diagnosis of RPLS. Permanently discontinue INLYTA in patients developing RPLS. The safety of reinitiating INLYTA therapy in patients previously experiencing RPLS is not known [see Dosage and Administration (2.2)].
5.6 Gastrointestinal Perforation and Fistula Formation
In a controlled clinical study with INLYTA for the treatment of patients with RCC, gastrointestinal perforation was reported in 1/359 patients (<1%) receiving INLYTA and none of the patients receiving sorafenib. In clinical trials with INLYTA, gastrointestinal perforation was reported in 5/715 patients (1%), including one death. In addition to cases of gastrointestinal perforation, fistulas were reported in 4/715 patients (1%).
Monitor for symptoms of gastrointestinal perforation or fistula periodically throughout treatment with INLYTA.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with axitinib.
Axitinib was not mutagenic in an in vitro bacterial reverse mutation (Ames) assay and was not clastogenic in the in vitro human lymphocyte chromosome aberration assay. Axitinib was genotoxic in the in vivo mouse bone marrow micronucleus assay.
INLYTA has the potential to impair reproductive function and fertility in humans. In repeat-dose toxicology studies, findings in the male reproductive tract were observed in the testes/epididymis (decreased organ weight, atrophy or degeneration, decreased numbers of germinal cells, hypospermia or abnormal sperm forms, reduced sperm density and count) at ≥15 mg/kg/dose administered orally twice daily in mice (approximately 7 times the systemic exposure (AUC) in patients at the recommended starting dose) and ≥1.5 mg/kg/dose administered orally twice daily in dogs (approximately 0.1 times the AUC in patients at the recommended starting dose). Findings in the female reproductive tract in mice and dogs included signs of delayed sexual maturity, reduced or absent corpora lutea, decreased uterine weights and uterine atrophy at ≥5 mg/kg/dose (approximately 1.5 or 0.3 times the AUC in patients at the recommended starting dose compared to mice and dogs, respectively).
In a fertility study in mice, axitinib did not affect mating or fertility rate when administered orally twice daily to males at any dose tested up to 50 mg/kg/dose following at least 70 days of administration (approximately 57 times the AUC in patients at the recommended starting dose). In female mice, reduced fertility and embryonic viability were observed at all doses tested (≥15 mg/kg/dose administered orally twice daily) following at least 15 days of treatment with axitinib (approximately 10 times the AUC in patients at the recommended starting dose).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:44:29.416192 · Updated: 2026-03-14T22:20:22.923169