These Highlights Do Not Include All The Information Needed To Use Livdelzi Safely And Effectively. See Full Prescribing Information For Livdelzi.
89f79d98-135c-40b2-a585-6996c04e599c
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
LIVDELZI is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA. This indication is approved under accelerated approval based on a reduction of alkaline phosphatase (ALP) [see Clinical Studies (14) ] . Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
Indications and Usage
LIVDELZI is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA. This indication is approved under accelerated approval based on a reduction of alkaline phosphatase (ALP) [see Clinical Studies (14) ] . Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
Dosage and Administration
The recommended dosage of LIVDELZI is 10 mg orally once daily. Administer LIVDELZI with or without food. ( 2.1 )
Warnings and Precautions
Fractures : Consider the risk of fracture in patients treated with LIVDELZI. Monitor bone health according to current standards of care. ( 5.1 ) Liver Test Abnormalities : Obtain baseline clinical and laboratory liver assessments prior to starting LIVDELZI and monitor during treatment. Interrupt or discontinue LIVDELZI if the liver tests worsen. ( 5.2 ) Biliary Obstruction : Avoid use in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt LIVDELZI and treat as clinically indicated. ( 5.3 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in labeling: Fractures [see Warnings and Precautions (5.1) ] Liver Test Abnormalities [see Warnings and Precautions (5.2) ]
Drug Interactions
Probenecid : Avoid concomitant use. ( 7.1 ) Strong CYP2C9 Inhibitors : Monitor for adverse effects. ( 7.1 ) Dual Moderate CYP2C9 and Moderate to Strong CYP3A4 Inhibitors : Monitor for adverse effects. ( 7.1 ) CYP2C9 Poor Metabolizers using Moderate to Strong CYP3A4 Inhibitors : Monitor for adverse effects. ( 7.1 ) Dual or Multiple Clinical Inhibitors of Drug Transporters OATP1B1, OATP1B3, and BCRP : Monitor for adverse effects. ( 7.1 ) Rifampin : Monitor biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin. ( 7.1 ) Bile Acid Sequestrants : Administer at least 4 hours before or 4 hours after taking a bile acid sequestrant, or at as great an interval as possible. ( 7.1 )
Medication Information
Warnings and Precautions
Fractures : Consider the risk of fracture in patients treated with LIVDELZI. Monitor bone health according to current standards of care. ( 5.1 ) Liver Test Abnormalities : Obtain baseline clinical and laboratory liver assessments prior to starting LIVDELZI and monitor during treatment. Interrupt or discontinue LIVDELZI if the liver tests worsen. ( 5.2 ) Biliary Obstruction : Avoid use in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt LIVDELZI and treat as clinically indicated. ( 5.3 )
Indications and Usage
LIVDELZI is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA. This indication is approved under accelerated approval based on a reduction of alkaline phosphatase (ALP) [see Clinical Studies (14) ] . Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
Dosage and Administration
The recommended dosage of LIVDELZI is 10 mg orally once daily. Administer LIVDELZI with or without food. ( 2.1 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in labeling: Fractures [see Warnings and Precautions (5.1) ] Liver Test Abnormalities [see Warnings and Precautions (5.2) ]
Drug Interactions
Probenecid : Avoid concomitant use. ( 7.1 ) Strong CYP2C9 Inhibitors : Monitor for adverse effects. ( 7.1 ) Dual Moderate CYP2C9 and Moderate to Strong CYP3A4 Inhibitors : Monitor for adverse effects. ( 7.1 ) CYP2C9 Poor Metabolizers using Moderate to Strong CYP3A4 Inhibitors : Monitor for adverse effects. ( 7.1 ) Dual or Multiple Clinical Inhibitors of Drug Transporters OATP1B1, OATP1B3, and BCRP : Monitor for adverse effects. ( 7.1 ) Rifampin : Monitor biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin. ( 7.1 ) Bile Acid Sequestrants : Administer at least 4 hours before or 4 hours after taking a bile acid sequestrant, or at as great an interval as possible. ( 7.1 )
Description
LIVDELZI is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA. This indication is approved under accelerated approval based on a reduction of alkaline phosphatase (ALP) [see Clinical Studies (14) ] . Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
Section 42229-5
Limitations of Use
Use of LIVDELZI is not recommended in patients who have or develop decompensated cirrhosis (e.g., ascites, variceal bleeding, hepatic encephalopathy) [see Use in Specific Populations (8.7)].
Section 42230-3
| PATIENT INFORMATION LIVDELZI (liv del' zee) (seladelpar) capsules, for oral use |
||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 01/2026 | |
| What is LIVDELZI? | ||
| LIVDELZI is a prescription medicine used to treat primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have not responded well to UDCA or used alone in patients unable to tolerate UDCA. | ||
| LIVDELZI is not recommended for use in people who have advanced liver disease (decompensated cirrhosis). Symptoms of advanced liver disease may include confusion, having fluid in the stomach-area (abdomen), black, tarry, or bloody stools, coughing up or vomiting blood, or having vomit that looks like "coffee grounds". | ||
| It is not known if taking LIVDELZI will improve your chance of survival or prevent liver decompensation. | ||
| It is not known if LIVDELZI is safe and effective in children. | ||
Before taking LIVDELZI, tell your healthcare provider about all your medical conditions, including if you:
|
||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Certain other medicines may affect the way LIVDELZI works. | ||
How should I take LIVDELZI?
|
||
| What are the possible side effects of LIVDELZI? | ||
LIVDELZI can cause serious side effects, including:
|
||
|
|
|
| The most common side effects of LIVDELZI include: | ||
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or does not go away. | ||
| These are not all the possible side effects of LIVDELZI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
How should I store LIVDELZI?
|
||
| General information about the safe and effective use of LIVDELZI. | ||
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use LIVDELZI for a condition for which it was not prescribed. Do not give LIVDELZI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about LIVDELZI that is written for health professionals. | ||
| What are the ingredients in LIVDELZI? | ||
| Active ingredient: seladelpar lysine | ||
| Inactive ingredients: butylated hydroxytoluene, colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, and hard gelatin shells. | ||
| The light gray opaque (body) and dark blue opaque (cap) capsule shells contain gelatin, titanium dioxide, black iron oxide, yellow iron oxide, red iron oxide and the colorant FD&C Blue #2. | ||
| Distributed by: Gilead Sciences, Inc., Foster City, CA 94404. | ||
| LIVDELZI is a trademark of Gilead Sciences, Inc., or its related companies. | ||
| © 2026 Gilead Sciences, Inc. All rights reserved. 217899-GS-002 | ||
| For more information, call 1-800-445-3235 or go to www.LIVDELZI.com. |
Section 44425-7
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Dispense only in original container to protect from light.
10 Overdosage
PBC patients who received 5-times the recommended dosage or 20-times the recommended dosage of LIVDELZI experienced an increase in liver transaminases, muscle pain, and/or elevations in creatine phosphokinase, which resolved upon LIVDELZI discontinuation [see Warnings and Precautions (5.2)].
There is no specific treatment for overdose with LIVDELZI. General supportive care of the patient is indicated, as appropriate. If indicated, elimination of unabsorbed drug should be achieved by emesis or gastric lavage; usual precautions should be observed to maintain the airway. Because seladelpar is highly bound to plasma proteins, hemodialysis should not be considered.
5.1 Fractures
Fractures occurred in 4% of LIVDELZI-treated patients compared to no placebo-treated patients [see Adverse Reactions (6.1)].
Consider the risk of fracture in the care of patients treated with LIVDELZI and monitor bone health according to current standards of care.
11 Description
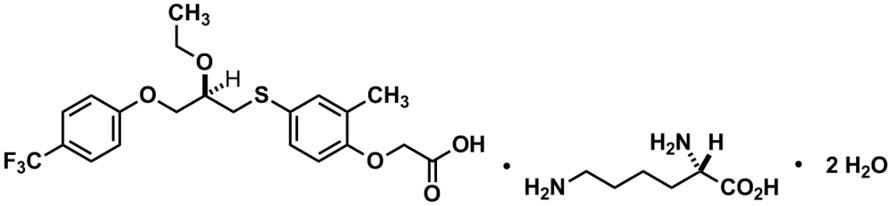
LIVDELZI capsules contain seladelpar lysine, a peroxisome proliferator-activated receptor (PPAR)-delta (δ) agonist. Seladelpar is a single enantiomer of the R-configuration and is present as a lysine dihydrate salt. Seladelpar lysine dihydrate is a white to off-white powder with a molecular formula of C21H23F3O5S ∙C6H14N2O2 ∙2H2O and a molecular weight of 626.7 g/mol. Its solubility in water is pH dependent. It is slightly soluble at low pH and very soluble at high pH. The chemical name for seladelpar lysine dihydrate is 2-[4-[[(2R)-2-ethoxy-3-[4-(trifluoromethyl)phenoxy]propyl]thio]-2-methylphenoxy]acetic acid, lysine dihydrate, and the chemical structure is:
LIVDELZI (seladelpar) capsules are supplied in a 10 mg strength for oral administration. Each capsule contains 14.1 mg of seladelpar lysine and the following inactive ingredients: butylated hydroxytoluene, colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, and hard gelatin shells.
The light gray opaque (body)/dark blue opaque (cap) capsule shells contain gelatin, titanium dioxide, black iron oxide, yellow iron oxide, red iron oxide and the colorant FD&C Blue #2.
8.4 Pediatric Use
The safety and effectiveness of LIVDELZI in pediatric patients have not been established.
8.5 Geriatric Use
Of the 128 LIVDELZI-treated patients in Trial 1, 29 (23%) patients were 65 years of age and older and 2 (2%) were 75 years of age and older. No overall differences in safety or effectiveness were observed between patients 65 to 75 years of age and younger adult patients. No dosage adjustment for patients 65 years of age and older is necessary.
Clinical studies of LIVDELZI did not include sufficient numbers of patients 75 years of age and older to determine whether they respond differently from younger adult patients. Because of limited clinical experience with LIVDELZI in patients older than 75 years old, closer monitoring of adverse events in patients older than 75 years is recommended [see Clinical Pharmacology (12.3)].
14 Clinical Studies
The efficacy of LIVDELZI was evaluated in Trial 1 (NCT04620733), a 12-month, randomized, double-blind, placebo-controlled trial. The trial included 193 adult patients with PBC with an inadequate response or intolerance to UDCA. Patients were included in the trial if their ALP was greater than or equal to 1.67-times the ULN and total bilirubin (TB) was less than or equal to 2-times the ULN. Patients were excluded from the trial if they had other chronic liver diseases, clinically important hepatic decompensation including portal hypertension with complications, or cirrhosis with complications (e.g., Model for End Stage Liver Disease [MELD] score of 12 or greater, known esophageal varices or history of variceal bleeds, history of hepatorenal syndrome).
Patients were randomized to receive LIVDELZI 10 mg (N=128) or placebo (N=65) once daily for 12 months. LIVDELZI or placebo was administered in combination with UDCA in 181 (94%) patients during the trial, or as a monotherapy in 12 (6%) patients who were unable to tolerate UDCA.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in labeling:
- Fractures [see Warnings and Precautions (5.1)]
- Liver Test Abnormalities [see Warnings and Precautions (5.2)]
7 Drug Interactions
- Probenecid: Avoid concomitant use. (7.1)
- Strong CYP2C9 Inhibitors: Monitor for adverse effects. (7.1)
- Dual Moderate CYP2C9 and Moderate to Strong CYP3A4 Inhibitors: Monitor for adverse effects. (7.1)
- CYP2C9 Poor Metabolizers using Moderate to Strong CYP3A4 Inhibitors: Monitor for adverse effects. (7.1)
- Dual or Multiple Clinical Inhibitors of Drug Transporters OATP1B1, OATP1B3, and BCRP: Monitor for adverse effects. (7.1)
- Rifampin: Monitor biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin. (7.1)
- Bile Acid Sequestrants: Administer at least 4 hours before or 4 hours after taking a bile acid sequestrant, or at as great an interval as possible. (7.1)
8.6 Renal Impairment
The recommended dosage in patients with mild, moderate, or severe renal impairment is the same as in patients with normal renal function. Patients with end-stage renal disease on dialysis have not been studied [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
Following a single dose administration, seladelpar systemic exposure increased dose-proportionally from 2 mg (0.2 times the recommended dosage) to 15 mg (1.5 times the recommended dosage) and greater than dose proportionally at higher doses. For a dose increase from 10 mg to 200 mg (20 times the recommended dosage), mean Cmax and mean AUC for seladelpar increased 70-fold and 27-fold, respectively.
Following once daily dosing, seladelpar steady-state was achieved by day 4 and AUC increase was less than 30%. In PBC patients, median (CV) Cmax and AUC for seladelpar was 90.5 (42.5%) ng/mL and 817 (44%) ng*h/mL, respectively at steady state following once daily dosing of 10 mg.
12.5 Pharmacogenomics
CYP2C9 activity is decreased in individuals with genetic variants such as CYP2C9*2 and CYP2C9*3. Compared to CYP2C9 normal metabolizers (*1/*1, n=84) after a single dose of seladelpar 1 mg to 15 mg, dose-normalized AUC0–inf was 48% higher in CYP2C9 poor metabolizers (*2/*3, n=2) and 24% higher in CYP2C9 intermediate metabolizers (*1/*2, *1/*8, *1/*3, *2/*2, n=28). Dose-normalized Cmax was similar for CYP2C9 normal, intermediate, and poor metabolizers. Seladelpar pharmacokinetics was not evaluated in patients who are CYP2C9 poor metabolizers with two no function alleles (e.g., *3/*3). CYP2C9 poor metabolizers may have increased AUC when seladelpar is used concomitantly with a moderate to strong CYP3A4 inhibitor [see Drug Interactions (7.1), Use in Specific Populations (8.8)].
The prevalence of CYP2C9 poor metabolizers is approximately 2 to 3% in White populations, 0.5 to 4% in Asian populations, and <1% in African American populations. Additional decreased or nonfunctional alleles (e.g., *5, *6, *11) are more prevalent in African American populations.
8.7 Hepatic Impairment
No dosage adjustment is recommended for PBC patients with mild hepatic impairment (Child-Pugh A) [see Clinical Pharmacology (12.3)].
The safety and efficacy of LIVDELZI in patients with decompensated cirrhosis have not been established. Use of LIVDELZI is not recommended in patients who have or develop decompensated cirrhosis (e.g., ascites, variceal bleeding, hepatic encephalopathy).
Monitor patients with cirrhosis for evidence of decompensation. Consider discontinuing LIVDELZI if the patient progresses to moderate or severe hepatic impairment (Child-Pugh B or C).
1 Indications and Usage
LIVDELZI is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA.
This indication is approved under accelerated approval based on a reduction of alkaline phosphatase (ALP) [see Clinical Studies (14)]. Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
5.3 Biliary Obstruction
Avoid use of LIVDELZI in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt LIVDELZI and treat as clinically indicated.
12.1 Mechanism of Action
Seladelpar is a peroxisome proliferator-activated receptor (PPAR)-delta (δ) agonist. However, the mechanism by which seladelpar exerts its therapeutic effects in patients with PBC is not well understood. Pharmacological activity that is potentially relevant to therapeutic effects includes inhibition of bile acid synthesis through activation of PPARδ, which is a nuclear receptor expressed in most tissues, including the liver. Published studies show that PPARδ activation by seladelpar reduces bile acid synthesis through Fibroblast Growth Factor 21 (FGF21)-dependent downregulation of CYP7A1, the key enzyme for the synthesis of bile acids from cholesterol.
5 Warnings and Precautions
- Fractures: Consider the risk of fracture in patients treated with LIVDELZI. Monitor bone health according to current standards of care. (5.1)
- Liver Test Abnormalities: Obtain baseline clinical and laboratory liver assessments prior to starting LIVDELZI and monitor during treatment. Interrupt or discontinue LIVDELZI if the liver tests worsen. (5.2)
- Biliary Obstruction: Avoid use in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt LIVDELZI and treat as clinically indicated. (5.3)
2 Dosage and Administration
The recommended dosage of LIVDELZI is 10 mg orally once daily. Administer LIVDELZI with or without food. (2.1)
3 Dosage Forms and Strengths
Capsules: 10 mg, opaque, hard gelatin capsules, size 1, with light gray opaque body and a dark blue opaque cap, printed with "CBAY" on the cap and "10" on the body.
5.2 Liver Test Abnormalities
LIVDELZI has been associated with dose-related increases in serum transaminase (aspartate aminotransferase [AST] and alanine aminotransferase [ALT]) levels greater than 3-times upper limit of normal (ULN) in PBC patients receiving 50 mg once daily (5-times higher than the recommended dosage) and 200 mg (20-times higher than the recommended dosage) once daily. Transaminase levels returned to pretreatment levels upon LIVDELZI discontinuation. LIVDELZI 10 mg once daily did not show a similar pattern for increases in transaminase levels [see Overdosage (10)].
Obtain baseline clinical and laboratory assessments at treatment initiation with LIVDELZI and monitor thereafter according to routine patient management. Interrupt LIVDELZI treatment if the liver tests (ALT, AST, total bilirubin [TB], and/or alkaline phosphatase [ALP]) worsen, or the patient develops signs and symptoms consistent with clinical hepatitis (e.g., jaundice, right upper quadrant pain, eosinophilia). Consider permanent discontinuation if liver tests worsen after restarting LIVDELZI.
8.8 Cyp2c9 Poor Metabolizers
Monitor CYP2C9 poor metabolizers who receive a concomitant moderate to strong CYP3A4 inhibitor more frequently for adverse reactions.
Seladelpar is a CYP2C9 and CYP3A4 substrate. Increased seladelpar AUC is expected in patients who are CYP2C9 poor metabolizers with concomitant use of a moderate to strong CYP3A4 inhibitor [see Drug Interactions (7.1), Clinical Pharmacology (12.5)].
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In Trial 1, 193 patients were randomized to receive either LIVDELZI 10 mg (N=128) or placebo (N=65) once daily for 12 months [see Clinical Studies (14)]. LIVDELZI or placebo was administered in combination with UDCA in 94% of patients and as monotherapy in 6% of patients who were unable to tolerate UDCA.
8 Use in Specific Populations
Hepatic Impairment: Monitor patients with cirrhosis for evidence of decompensation. Consider discontinuation if patient progresses to moderate or severe hepatic impairment (Child-Pugh B or C). (8.7)
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
7.1 Effect of Other Drugs On Livdelzi
Table 2 includes clinically significant drug interactions affecting LIVDELZI.
| Concomitant Drug or Class | Potential Effect on Seladelpar Exposure ↑ = Increase, ↓ = Decrease.
|
Clinical Intervention |
|---|---|---|
| Probenecid | ↑ seladelpar | Avoid concomitant administration of LIVDELZI with probenecid. |
| Strong CYP2C9 Inhibitors | ↑ seladelpar | Monitor patients for adverse effects during concomitant use of LIVDELZI with strong CYP2C9 inhibitors. |
| Dual Moderate CYP2C9 and Moderate or Strong CYP3A4 Inhibitors (e.g., fluconazole) | ↑ seladelpar | Monitor patients for adverse effects during concomitant use of LIVDELZI with drugs that are dual moderate CYP2C9 and moderate or strong CYP3A4 inhibitors. |
| CYP2C9 Poor Metabolizers Using Moderate or Strong CYP3A4 Inhibitors | ↑ seladelpar | Monitor patients who are CYP2C9 poor metabolizers for adverse effects during concomitant use of LIVDELZI with moderate or strong CYP3A4 inhibitor. |
| Dual or Multiple Clinical Inhibitors of Drug Transporters OATP1B1, OATP1B3, and BCRP (e.g, cyclosporine) | ↑ seladelpar | Monitor patients for adverse effects during concomitant use of LIVDELZI with dual or multiple clinical inhibitors of drug transporters OATP1B1, OATP1B3, and BCRP. |
| Rifampin | ↓ seladelpar | Concomitant use of LIVDELZI with rifampin, an inducer of metabolizing enzymes, may result in delayed or suboptimal LIVDELZI biochemical response. Monitor the biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin during LIVDELZI treatment. |
| Bile Acid Sequestrants | ↓ seladelpar | Bile acid sequestrants may interfere with the action of LIVDELZI by reducing its absorption and systemic exposure, which may reduce LIVDELZI efficacy. Administer LIVDELZI at least 4 hours before or 4 hours after taking a bile acid sequestrant, or at as great an interval as possible [see Dosage and Administration (2.2)]. |
2.1 Recommended Dosage and Administration
The recommended dosage of LIVDELZI is 10 mg orally once daily. Administer LIVDELZI with or without food [see Clinical Pharmacology (12.3)].
13.2 Animal Toxicology And/or Pharmacology
In a 2-year study in CD-1 mice, seladelpar produced an increased incidence of lens cataracts at 5 mg/kg/day in both sexes (6-times and 19-times the recommended dose in male and female mice, respectively, based on AUC). In a 2-year study in Sprague-Dawley rats, seladelpar produced an increased incidence of cornea inflammation at 10 mg/kg/day (14-times the recommended dose based on AUC) and cornea mineralization at 30 mg/kg/day (79-times the recommended dose based on AUC), with both effects observed in males only. The incidence of cornea inflammation was not increased in male rats at 3 mg/kg/day (5-times the recommended dose based on AUC).
Principal Display Panel 10 Mg Capsule Bottle Label
NDC 61958-3301-1
Livdelzi®
seladelpar
10 mg
For Oral Use Only
30 capsules
Rx only
GILEAD
2.2 Administration Modification for Bile Acid Sequestrants
Administer LIVDELZI at least 4 hours before or 4 hours after taking bile acid sequestrants, or at as great an interval as possible [see Drug Interactions (7.1)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
Use of LIVDELZI is not recommended in patients who have or develop decompensated cirrhosis (e.g., ascites, variceal bleeding, hepatic encephalopathy) [see Use in Specific Populations (8.7)].
Section 42230-3 (42230-3)
| PATIENT INFORMATION LIVDELZI (liv del' zee) (seladelpar) capsules, for oral use |
||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 01/2026 | |
| What is LIVDELZI? | ||
| LIVDELZI is a prescription medicine used to treat primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have not responded well to UDCA or used alone in patients unable to tolerate UDCA. | ||
| LIVDELZI is not recommended for use in people who have advanced liver disease (decompensated cirrhosis). Symptoms of advanced liver disease may include confusion, having fluid in the stomach-area (abdomen), black, tarry, or bloody stools, coughing up or vomiting blood, or having vomit that looks like "coffee grounds". | ||
| It is not known if taking LIVDELZI will improve your chance of survival or prevent liver decompensation. | ||
| It is not known if LIVDELZI is safe and effective in children. | ||
Before taking LIVDELZI, tell your healthcare provider about all your medical conditions, including if you:
|
||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Certain other medicines may affect the way LIVDELZI works. | ||
How should I take LIVDELZI?
|
||
| What are the possible side effects of LIVDELZI? | ||
LIVDELZI can cause serious side effects, including:
|
||
|
|
|
| The most common side effects of LIVDELZI include: | ||
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or does not go away. | ||
| These are not all the possible side effects of LIVDELZI. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
How should I store LIVDELZI?
|
||
| General information about the safe and effective use of LIVDELZI. | ||
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use LIVDELZI for a condition for which it was not prescribed. Do not give LIVDELZI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about LIVDELZI that is written for health professionals. | ||
| What are the ingredients in LIVDELZI? | ||
| Active ingredient: seladelpar lysine | ||
| Inactive ingredients: butylated hydroxytoluene, colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, and hard gelatin shells. | ||
| The light gray opaque (body) and dark blue opaque (cap) capsule shells contain gelatin, titanium dioxide, black iron oxide, yellow iron oxide, red iron oxide and the colorant FD&C Blue #2. | ||
| Distributed by: Gilead Sciences, Inc., Foster City, CA 94404. | ||
| LIVDELZI is a trademark of Gilead Sciences, Inc., or its related companies. | ||
| © 2026 Gilead Sciences, Inc. All rights reserved. 217899-GS-002 | ||
| For more information, call 1-800-445-3235 or go to www.LIVDELZI.com. |
Section 44425-7 (44425-7)
Storage and Handling
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Dispense only in original container to protect from light.
10 Overdosage (10 OVERDOSAGE)
PBC patients who received 5-times the recommended dosage or 20-times the recommended dosage of LIVDELZI experienced an increase in liver transaminases, muscle pain, and/or elevations in creatine phosphokinase, which resolved upon LIVDELZI discontinuation [see Warnings and Precautions (5.2)].
There is no specific treatment for overdose with LIVDELZI. General supportive care of the patient is indicated, as appropriate. If indicated, elimination of unabsorbed drug should be achieved by emesis or gastric lavage; usual precautions should be observed to maintain the airway. Because seladelpar is highly bound to plasma proteins, hemodialysis should not be considered.
5.1 Fractures
Fractures occurred in 4% of LIVDELZI-treated patients compared to no placebo-treated patients [see Adverse Reactions (6.1)].
Consider the risk of fracture in the care of patients treated with LIVDELZI and monitor bone health according to current standards of care.
11 Description (11 DESCRIPTION)
LIVDELZI capsules contain seladelpar lysine, a peroxisome proliferator-activated receptor (PPAR)-delta (δ) agonist. Seladelpar is a single enantiomer of the R-configuration and is present as a lysine dihydrate salt. Seladelpar lysine dihydrate is a white to off-white powder with a molecular formula of C21H23F3O5S ∙C6H14N2O2 ∙2H2O and a molecular weight of 626.7 g/mol. Its solubility in water is pH dependent. It is slightly soluble at low pH and very soluble at high pH. The chemical name for seladelpar lysine dihydrate is 2-[4-[[(2R)-2-ethoxy-3-[4-(trifluoromethyl)phenoxy]propyl]thio]-2-methylphenoxy]acetic acid, lysine dihydrate, and the chemical structure is:
LIVDELZI (seladelpar) capsules are supplied in a 10 mg strength for oral administration. Each capsule contains 14.1 mg of seladelpar lysine and the following inactive ingredients: butylated hydroxytoluene, colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, mannitol, microcrystalline cellulose, and hard gelatin shells.
The light gray opaque (body)/dark blue opaque (cap) capsule shells contain gelatin, titanium dioxide, black iron oxide, yellow iron oxide, red iron oxide and the colorant FD&C Blue #2.
8.4 Pediatric Use
The safety and effectiveness of LIVDELZI in pediatric patients have not been established.
8.5 Geriatric Use
Of the 128 LIVDELZI-treated patients in Trial 1, 29 (23%) patients were 65 years of age and older and 2 (2%) were 75 years of age and older. No overall differences in safety or effectiveness were observed between patients 65 to 75 years of age and younger adult patients. No dosage adjustment for patients 65 years of age and older is necessary.
Clinical studies of LIVDELZI did not include sufficient numbers of patients 75 years of age and older to determine whether they respond differently from younger adult patients. Because of limited clinical experience with LIVDELZI in patients older than 75 years old, closer monitoring of adverse events in patients older than 75 years is recommended [see Clinical Pharmacology (12.3)].
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of LIVDELZI was evaluated in Trial 1 (NCT04620733), a 12-month, randomized, double-blind, placebo-controlled trial. The trial included 193 adult patients with PBC with an inadequate response or intolerance to UDCA. Patients were included in the trial if their ALP was greater than or equal to 1.67-times the ULN and total bilirubin (TB) was less than or equal to 2-times the ULN. Patients were excluded from the trial if they had other chronic liver diseases, clinically important hepatic decompensation including portal hypertension with complications, or cirrhosis with complications (e.g., Model for End Stage Liver Disease [MELD] score of 12 or greater, known esophageal varices or history of variceal bleeds, history of hepatorenal syndrome).
Patients were randomized to receive LIVDELZI 10 mg (N=128) or placebo (N=65) once daily for 12 months. LIVDELZI or placebo was administered in combination with UDCA in 181 (94%) patients during the trial, or as a monotherapy in 12 (6%) patients who were unable to tolerate UDCA.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in labeling:
- Fractures [see Warnings and Precautions (5.1)]
- Liver Test Abnormalities [see Warnings and Precautions (5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Probenecid: Avoid concomitant use. (7.1)
- Strong CYP2C9 Inhibitors: Monitor for adverse effects. (7.1)
- Dual Moderate CYP2C9 and Moderate to Strong CYP3A4 Inhibitors: Monitor for adverse effects. (7.1)
- CYP2C9 Poor Metabolizers using Moderate to Strong CYP3A4 Inhibitors: Monitor for adverse effects. (7.1)
- Dual or Multiple Clinical Inhibitors of Drug Transporters OATP1B1, OATP1B3, and BCRP: Monitor for adverse effects. (7.1)
- Rifampin: Monitor biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin. (7.1)
- Bile Acid Sequestrants: Administer at least 4 hours before or 4 hours after taking a bile acid sequestrant, or at as great an interval as possible. (7.1)
8.6 Renal Impairment
The recommended dosage in patients with mild, moderate, or severe renal impairment is the same as in patients with normal renal function. Patients with end-stage renal disease on dialysis have not been studied [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
Following a single dose administration, seladelpar systemic exposure increased dose-proportionally from 2 mg (0.2 times the recommended dosage) to 15 mg (1.5 times the recommended dosage) and greater than dose proportionally at higher doses. For a dose increase from 10 mg to 200 mg (20 times the recommended dosage), mean Cmax and mean AUC for seladelpar increased 70-fold and 27-fold, respectively.
Following once daily dosing, seladelpar steady-state was achieved by day 4 and AUC increase was less than 30%. In PBC patients, median (CV) Cmax and AUC for seladelpar was 90.5 (42.5%) ng/mL and 817 (44%) ng*h/mL, respectively at steady state following once daily dosing of 10 mg.
12.5 Pharmacogenomics
CYP2C9 activity is decreased in individuals with genetic variants such as CYP2C9*2 and CYP2C9*3. Compared to CYP2C9 normal metabolizers (*1/*1, n=84) after a single dose of seladelpar 1 mg to 15 mg, dose-normalized AUC0–inf was 48% higher in CYP2C9 poor metabolizers (*2/*3, n=2) and 24% higher in CYP2C9 intermediate metabolizers (*1/*2, *1/*8, *1/*3, *2/*2, n=28). Dose-normalized Cmax was similar for CYP2C9 normal, intermediate, and poor metabolizers. Seladelpar pharmacokinetics was not evaluated in patients who are CYP2C9 poor metabolizers with two no function alleles (e.g., *3/*3). CYP2C9 poor metabolizers may have increased AUC when seladelpar is used concomitantly with a moderate to strong CYP3A4 inhibitor [see Drug Interactions (7.1), Use in Specific Populations (8.8)].
The prevalence of CYP2C9 poor metabolizers is approximately 2 to 3% in White populations, 0.5 to 4% in Asian populations, and <1% in African American populations. Additional decreased or nonfunctional alleles (e.g., *5, *6, *11) are more prevalent in African American populations.
8.7 Hepatic Impairment
No dosage adjustment is recommended for PBC patients with mild hepatic impairment (Child-Pugh A) [see Clinical Pharmacology (12.3)].
The safety and efficacy of LIVDELZI in patients with decompensated cirrhosis have not been established. Use of LIVDELZI is not recommended in patients who have or develop decompensated cirrhosis (e.g., ascites, variceal bleeding, hepatic encephalopathy).
Monitor patients with cirrhosis for evidence of decompensation. Consider discontinuing LIVDELZI if the patient progresses to moderate or severe hepatic impairment (Child-Pugh B or C).
1 Indications and Usage (1 INDICATIONS AND USAGE)
LIVDELZI is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA.
This indication is approved under accelerated approval based on a reduction of alkaline phosphatase (ALP) [see Clinical Studies (14)]. Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
5.3 Biliary Obstruction
Avoid use of LIVDELZI in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt LIVDELZI and treat as clinically indicated.
12.1 Mechanism of Action
Seladelpar is a peroxisome proliferator-activated receptor (PPAR)-delta (δ) agonist. However, the mechanism by which seladelpar exerts its therapeutic effects in patients with PBC is not well understood. Pharmacological activity that is potentially relevant to therapeutic effects includes inhibition of bile acid synthesis through activation of PPARδ, which is a nuclear receptor expressed in most tissues, including the liver. Published studies show that PPARδ activation by seladelpar reduces bile acid synthesis through Fibroblast Growth Factor 21 (FGF21)-dependent downregulation of CYP7A1, the key enzyme for the synthesis of bile acids from cholesterol.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Fractures: Consider the risk of fracture in patients treated with LIVDELZI. Monitor bone health according to current standards of care. (5.1)
- Liver Test Abnormalities: Obtain baseline clinical and laboratory liver assessments prior to starting LIVDELZI and monitor during treatment. Interrupt or discontinue LIVDELZI if the liver tests worsen. (5.2)
- Biliary Obstruction: Avoid use in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt LIVDELZI and treat as clinically indicated. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dosage of LIVDELZI is 10 mg orally once daily. Administer LIVDELZI with or without food. (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsules: 10 mg, opaque, hard gelatin capsules, size 1, with light gray opaque body and a dark blue opaque cap, printed with "CBAY" on the cap and "10" on the body.
5.2 Liver Test Abnormalities
LIVDELZI has been associated with dose-related increases in serum transaminase (aspartate aminotransferase [AST] and alanine aminotransferase [ALT]) levels greater than 3-times upper limit of normal (ULN) in PBC patients receiving 50 mg once daily (5-times higher than the recommended dosage) and 200 mg (20-times higher than the recommended dosage) once daily. Transaminase levels returned to pretreatment levels upon LIVDELZI discontinuation. LIVDELZI 10 mg once daily did not show a similar pattern for increases in transaminase levels [see Overdosage (10)].
Obtain baseline clinical and laboratory assessments at treatment initiation with LIVDELZI and monitor thereafter according to routine patient management. Interrupt LIVDELZI treatment if the liver tests (ALT, AST, total bilirubin [TB], and/or alkaline phosphatase [ALP]) worsen, or the patient develops signs and symptoms consistent with clinical hepatitis (e.g., jaundice, right upper quadrant pain, eosinophilia). Consider permanent discontinuation if liver tests worsen after restarting LIVDELZI.
8.8 Cyp2c9 Poor Metabolizers (8.8 CYP2C9 Poor Metabolizers)
Monitor CYP2C9 poor metabolizers who receive a concomitant moderate to strong CYP3A4 inhibitor more frequently for adverse reactions.
Seladelpar is a CYP2C9 and CYP3A4 substrate. Increased seladelpar AUC is expected in patients who are CYP2C9 poor metabolizers with concomitant use of a moderate to strong CYP3A4 inhibitor [see Drug Interactions (7.1), Clinical Pharmacology (12.5)].
6.1 Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In Trial 1, 193 patients were randomized to receive either LIVDELZI 10 mg (N=128) or placebo (N=65) once daily for 12 months [see Clinical Studies (14)]. LIVDELZI or placebo was administered in combination with UDCA in 94% of patients and as monotherapy in 6% of patients who were unable to tolerate UDCA.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Hepatic Impairment: Monitor patients with cirrhosis for evidence of decompensation. Consider discontinuation if patient progresses to moderate or severe hepatic impairment (Child-Pugh B or C). (8.7)
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
7.1 Effect of Other Drugs On Livdelzi (7.1 Effect of Other Drugs on LIVDELZI)
Table 2 includes clinically significant drug interactions affecting LIVDELZI.
| Concomitant Drug or Class | Potential Effect on Seladelpar Exposure ↑ = Increase, ↓ = Decrease.
|
Clinical Intervention |
|---|---|---|
| Probenecid | ↑ seladelpar | Avoid concomitant administration of LIVDELZI with probenecid. |
| Strong CYP2C9 Inhibitors | ↑ seladelpar | Monitor patients for adverse effects during concomitant use of LIVDELZI with strong CYP2C9 inhibitors. |
| Dual Moderate CYP2C9 and Moderate or Strong CYP3A4 Inhibitors (e.g., fluconazole) | ↑ seladelpar | Monitor patients for adverse effects during concomitant use of LIVDELZI with drugs that are dual moderate CYP2C9 and moderate or strong CYP3A4 inhibitors. |
| CYP2C9 Poor Metabolizers Using Moderate or Strong CYP3A4 Inhibitors | ↑ seladelpar | Monitor patients who are CYP2C9 poor metabolizers for adverse effects during concomitant use of LIVDELZI with moderate or strong CYP3A4 inhibitor. |
| Dual or Multiple Clinical Inhibitors of Drug Transporters OATP1B1, OATP1B3, and BCRP (e.g, cyclosporine) | ↑ seladelpar | Monitor patients for adverse effects during concomitant use of LIVDELZI with dual or multiple clinical inhibitors of drug transporters OATP1B1, OATP1B3, and BCRP. |
| Rifampin | ↓ seladelpar | Concomitant use of LIVDELZI with rifampin, an inducer of metabolizing enzymes, may result in delayed or suboptimal LIVDELZI biochemical response. Monitor the biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin during LIVDELZI treatment. |
| Bile Acid Sequestrants | ↓ seladelpar | Bile acid sequestrants may interfere with the action of LIVDELZI by reducing its absorption and systemic exposure, which may reduce LIVDELZI efficacy. Administer LIVDELZI at least 4 hours before or 4 hours after taking a bile acid sequestrant, or at as great an interval as possible [see Dosage and Administration (2.2)]. |
2.1 Recommended Dosage and Administration
The recommended dosage of LIVDELZI is 10 mg orally once daily. Administer LIVDELZI with or without food [see Clinical Pharmacology (12.3)].
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
In a 2-year study in CD-1 mice, seladelpar produced an increased incidence of lens cataracts at 5 mg/kg/day in both sexes (6-times and 19-times the recommended dose in male and female mice, respectively, based on AUC). In a 2-year study in Sprague-Dawley rats, seladelpar produced an increased incidence of cornea inflammation at 10 mg/kg/day (14-times the recommended dose based on AUC) and cornea mineralization at 30 mg/kg/day (79-times the recommended dose based on AUC), with both effects observed in males only. The incidence of cornea inflammation was not increased in male rats at 3 mg/kg/day (5-times the recommended dose based on AUC).
Principal Display Panel 10 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Capsule Bottle Label)
NDC 61958-3301-1
Livdelzi®
seladelpar
10 mg
For Oral Use Only
30 capsules
Rx only
GILEAD
2.2 Administration Modification for Bile Acid Sequestrants
Administer LIVDELZI at least 4 hours before or 4 hours after taking bile acid sequestrants, or at as great an interval as possible [see Drug Interactions (7.1)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:42:28.236666 · Updated: 2026-03-14T22:46:15.425828