These Highlights Do Not Include All The Information Needed To Use Isentress Safely And Effectively. See Full Prescribing Information For Isentress.
89a5ec53-d956-4329-8004-0f40f51c88a3
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Adult Patients: ISENTRESS and ISENTRESS HD are human immunodeficiency virus integrase strand transfer inhibitors (HIV-1 INSTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in adult patients ( 1 ). Pediatric Patients: ISENTRESS is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 2 kg ( 1 ). ISENTRESS HD is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 40 kg ( 1 ).
Indications and Usage
Adult Patients: ISENTRESS and ISENTRESS HD are human immunodeficiency virus integrase strand transfer inhibitors (HIV-1 INSTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in adult patients ( 1 ). Pediatric Patients: ISENTRESS is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 2 kg ( 1 ). ISENTRESS HD is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 40 kg ( 1 ).
Dosage and Administration
ISENTRESS and ISENTRESS HD can be administered with or without food ( 2.1 ). Do not substitute ISENTRESS chewable tablets or ISENTRESS for oral suspension for the ISENTRESS 400 mg or 600 mg film-coated tablet. See specific dosing guidance for chewable tablets and the formulation for oral suspension ( 2.1 ). Adults Treatment-naïve patients or patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily: 1200 mg (2 × 600 mg) film-coated tablet orally, once daily or 400 mg film-coated tablet orally, twice daily ( 2.2 ). Treatment-experienced patients: 400 mg film-coated tablet orally, twice daily ( 2.2 ). During coadministration with rifampin in adults, 800 mg (2 × 400 mg) twice daily ( 2.2 ). Pediatrics If weighing at least 40 kg, and either treatment-naïve patients or patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily: 1200 mg (2 × 600 mg) film-coated tablet orally, once daily or 400 mg film-coated tablet orally, twice daily or 300 mg (3 × 100 mg) chewable tablets, twice daily ( 2.3 ). If weighing at least 25 kg: One 400 mg film-coated tablet orally, twice daily. If unable to swallow a tablet, consider the chewable tablet, as specified in Table 2 ( 2.3 ). If weighing at least 3 kg to less than 25 kg: Weight-based dosing using the chewable tablet or oral suspension, as specified in Table 4 ( 2.3 ). For neonates (birth to 4 weeks [28 days] of age): Weight-based dosing of the oral suspension as specified in Table 5 ( 2.3 ).
Warnings and Precautions
Severe, potentially life-threatening and fatal skin reactions have been reported. This includes cases of Stevens-Johnson syndrome, hypersensitivity reaction and toxic epidermal necrolysis. Immediately discontinue treatment with ISENTRESS or ISENTRESS HD and other suspect agents if severe hypersensitivity, severe rash, or rash with systemic symptoms or liver aminotransferase elevations develops and monitor clinical status, including liver aminotransferases closely ( 5.1 ). Monitor for Immune Reconstitution Syndrome ( 5.2 ). Inform patients with phenylketonuria that the 100 mg and 25 mg chewable tablets contain phenylalanine ( 5.3 ).
Contraindications
None
Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Drug Interactions
Coadministration of ISENTRESS or ISENTRESS HD and other drugs may alter the plasma concentration of raltegravir. The potential for drug-drug interactions must be considered prior to and during therapy ( 7 ). Coadministration of ISENTRESS or ISENTRESS HD with drugs that are strong inducers of UGT1A1, such as rifampin, may result in reduced plasma concentrations of raltegravir ( 2.1 , 7.1 ).
Storage and Handling
ISENTRESS tablets 400 mg are pink, oval-shaped, film-coated tablets with "227" on one side. They are supplied as follows: NDC 0006-0227-61 unit-of-use bottles of 60. ISENTRESS HD tablets 600 mg are yellow, oval-shaped, film-coated tablets with corporate logo and "242" on one side and plain on the other side. They are supplied as follows: NDC 0006-3080-01 unit-of-use bottles of 60. ISENTRESS tablets 100 mg are pale orange, oval-shaped, orange-banana flavored, chewable tablets scored on both sides and imprinted on one face with the corporate logo and "477" on opposite sides of the score. They are supplied as follows: NDC 0006-0477-61 unit-of-use bottles of 60. ISENTRESS tablets 25 mg are pale yellow, round, orange-banana flavored, chewable tablets with the corporate logo on one side and "473" on the other side. They are supplied as follows: NDC 0006-0473-61 unit-of-use bottles of 60. ISENTRESS for oral suspension 100 mg is a white to off-white granular powder that may contain yellow or beige to tan particles, in child resistant single-use foil packets, packaged as A kit with two 1 mL syringes, two 3 mL syringes, two 10 mL syringes and two mixing cups. It is supplied as follows: NDC 0006-3603-61 unit of use carton with 60 packets. NDC 0006-3603-01 individual packet.
How Supplied
ISENTRESS tablets 400 mg are pink, oval-shaped, film-coated tablets with "227" on one side. They are supplied as follows: NDC 0006-0227-61 unit-of-use bottles of 60. ISENTRESS HD tablets 600 mg are yellow, oval-shaped, film-coated tablets with corporate logo and "242" on one side and plain on the other side. They are supplied as follows: NDC 0006-3080-01 unit-of-use bottles of 60. ISENTRESS tablets 100 mg are pale orange, oval-shaped, orange-banana flavored, chewable tablets scored on both sides and imprinted on one face with the corporate logo and "477" on opposite sides of the score. They are supplied as follows: NDC 0006-0477-61 unit-of-use bottles of 60. ISENTRESS tablets 25 mg are pale yellow, round, orange-banana flavored, chewable tablets with the corporate logo on one side and "473" on the other side. They are supplied as follows: NDC 0006-0473-61 unit-of-use bottles of 60. ISENTRESS for oral suspension 100 mg is a white to off-white granular powder that may contain yellow or beige to tan particles, in child resistant single-use foil packets, packaged as A kit with two 1 mL syringes, two 3 mL syringes, two 10 mL syringes and two mixing cups. It is supplied as follows: NDC 0006-3603-61 unit of use carton with 60 packets. NDC 0006-3603-01 individual packet.
Medication Information
Warnings and Precautions
Severe, potentially life-threatening and fatal skin reactions have been reported. This includes cases of Stevens-Johnson syndrome, hypersensitivity reaction and toxic epidermal necrolysis. Immediately discontinue treatment with ISENTRESS or ISENTRESS HD and other suspect agents if severe hypersensitivity, severe rash, or rash with systemic symptoms or liver aminotransferase elevations develops and monitor clinical status, including liver aminotransferases closely ( 5.1 ). Monitor for Immune Reconstitution Syndrome ( 5.2 ). Inform patients with phenylketonuria that the 100 mg and 25 mg chewable tablets contain phenylalanine ( 5.3 ).
Indications and Usage
Adult Patients: ISENTRESS and ISENTRESS HD are human immunodeficiency virus integrase strand transfer inhibitors (HIV-1 INSTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in adult patients ( 1 ). Pediatric Patients: ISENTRESS is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 2 kg ( 1 ). ISENTRESS HD is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 40 kg ( 1 ).
Dosage and Administration
ISENTRESS and ISENTRESS HD can be administered with or without food ( 2.1 ). Do not substitute ISENTRESS chewable tablets or ISENTRESS for oral suspension for the ISENTRESS 400 mg or 600 mg film-coated tablet. See specific dosing guidance for chewable tablets and the formulation for oral suspension ( 2.1 ). Adults Treatment-naïve patients or patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily: 1200 mg (2 × 600 mg) film-coated tablet orally, once daily or 400 mg film-coated tablet orally, twice daily ( 2.2 ). Treatment-experienced patients: 400 mg film-coated tablet orally, twice daily ( 2.2 ). During coadministration with rifampin in adults, 800 mg (2 × 400 mg) twice daily ( 2.2 ). Pediatrics If weighing at least 40 kg, and either treatment-naïve patients or patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily: 1200 mg (2 × 600 mg) film-coated tablet orally, once daily or 400 mg film-coated tablet orally, twice daily or 300 mg (3 × 100 mg) chewable tablets, twice daily ( 2.3 ). If weighing at least 25 kg: One 400 mg film-coated tablet orally, twice daily. If unable to swallow a tablet, consider the chewable tablet, as specified in Table 2 ( 2.3 ). If weighing at least 3 kg to less than 25 kg: Weight-based dosing using the chewable tablet or oral suspension, as specified in Table 4 ( 2.3 ). For neonates (birth to 4 weeks [28 days] of age): Weight-based dosing of the oral suspension as specified in Table 5 ( 2.3 ).
Contraindications
None
Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Drug Interactions
Coadministration of ISENTRESS or ISENTRESS HD and other drugs may alter the plasma concentration of raltegravir. The potential for drug-drug interactions must be considered prior to and during therapy ( 7 ). Coadministration of ISENTRESS or ISENTRESS HD with drugs that are strong inducers of UGT1A1, such as rifampin, may result in reduced plasma concentrations of raltegravir ( 2.1 , 7.1 ).
Storage and Handling
ISENTRESS tablets 400 mg are pink, oval-shaped, film-coated tablets with "227" on one side. They are supplied as follows: NDC 0006-0227-61 unit-of-use bottles of 60. ISENTRESS HD tablets 600 mg are yellow, oval-shaped, film-coated tablets with corporate logo and "242" on one side and plain on the other side. They are supplied as follows: NDC 0006-3080-01 unit-of-use bottles of 60. ISENTRESS tablets 100 mg are pale orange, oval-shaped, orange-banana flavored, chewable tablets scored on both sides and imprinted on one face with the corporate logo and "477" on opposite sides of the score. They are supplied as follows: NDC 0006-0477-61 unit-of-use bottles of 60. ISENTRESS tablets 25 mg are pale yellow, round, orange-banana flavored, chewable tablets with the corporate logo on one side and "473" on the other side. They are supplied as follows: NDC 0006-0473-61 unit-of-use bottles of 60. ISENTRESS for oral suspension 100 mg is a white to off-white granular powder that may contain yellow or beige to tan particles, in child resistant single-use foil packets, packaged as A kit with two 1 mL syringes, two 3 mL syringes, two 10 mL syringes and two mixing cups. It is supplied as follows: NDC 0006-3603-61 unit of use carton with 60 packets. NDC 0006-3603-01 individual packet.
How Supplied
ISENTRESS tablets 400 mg are pink, oval-shaped, film-coated tablets with "227" on one side. They are supplied as follows: NDC 0006-0227-61 unit-of-use bottles of 60. ISENTRESS HD tablets 600 mg are yellow, oval-shaped, film-coated tablets with corporate logo and "242" on one side and plain on the other side. They are supplied as follows: NDC 0006-3080-01 unit-of-use bottles of 60. ISENTRESS tablets 100 mg are pale orange, oval-shaped, orange-banana flavored, chewable tablets scored on both sides and imprinted on one face with the corporate logo and "477" on opposite sides of the score. They are supplied as follows: NDC 0006-0477-61 unit-of-use bottles of 60. ISENTRESS tablets 25 mg are pale yellow, round, orange-banana flavored, chewable tablets with the corporate logo on one side and "473" on the other side. They are supplied as follows: NDC 0006-0473-61 unit-of-use bottles of 60. ISENTRESS for oral suspension 100 mg is a white to off-white granular powder that may contain yellow or beige to tan particles, in child resistant single-use foil packets, packaged as A kit with two 1 mL syringes, two 3 mL syringes, two 10 mL syringes and two mixing cups. It is supplied as follows: NDC 0006-3603-61 unit of use carton with 60 packets. NDC 0006-3603-01 individual packet.
Description
Adult Patients: ISENTRESS and ISENTRESS HD are human immunodeficiency virus integrase strand transfer inhibitors (HIV-1 INSTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in adult patients ( 1 ). Pediatric Patients: ISENTRESS is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 2 kg ( 1 ). ISENTRESS HD is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 40 kg ( 1 ).
Section 42229-5
Adult Patients:
ISENTRESS® and ISENTRESS® HD are indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) infection in adult patients.
Section 42230-3
| PATIENT INFORMATION | ||||
|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised May 2022 | |||
| ISENTRESS® (eye sen tris) (raltegravir) film-coated tablets |
ISENTRESS® (eye sen tris) (raltegravir) chewable tablets |
|||
| ISENTRESS® HD (eye sen tris HD) (raltegravir) film-coated tablets |
ISENTRESS® (eye sen tris) (raltegravir) for oral suspension |
|||
| What are ISENTRESS and ISENTRESS HD? | ||||
| ISENTRESS is a prescription medicine used with other HIV-1 medicines to treat Human Immunodeficiency Virus-1 (HIV-1) infection in adults, and in children weighing at least 4.4 pounds (2 kg). HIV is the virus that causes AIDS (Acquired Immune Deficiency Syndrome). | ||||
| ISENTRESS HD is a prescription medicine used with other HIV-1 medicines to treat HIV-1 infection in adults, and in children weighing at least 88 pounds (40 kg). ISENTRESS should not be used in children who weigh less than 4.4 pounds (2 kg). |
||||
Before you take ISENTRESS or ISENTRESS HD, tell your doctor about all of your medical conditions, including if you:
|
||||
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some medicines interact with ISENTRESS and ISENTRESS HD.
|
||||
How should I take ISENTRESS or ISENTRESS HD?
If ISENTRESS for oral suspension is prescribed for your child, be sure to read the following information:
|
||||
| What are the possible side effects of ISENTRESS or ISENTRESS HD? | ||||
ISENTRESS and ISENTRESS HD can cause serious side effects including:
|
||||
|
|
|||
| Sometimes allergic reactions can affect body organs, such as your liver. Call your doctor right away if you have any of the following signs or symptoms of liver problems: | ||||
|
|
|||
|
||||
| The most common side effects of ISENTRESS and ISENTRESS HD include: | ||||
|
|
|||
| Less common side effects of ISENTRESS and ISENTRESS HD include: | ||||
|
|
|||
| Tell your doctor right away if you get unexplained muscle pain, tenderness, or weakness during treatment with ISENTRESS or ISENTRESS HD. These may be signs of a rare serious muscle problem that can lead to kidney problems. | ||||
| These are not all the possible side effects of ISENTRESS and ISENTRESS HD. | ||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
| How should I store ISENTRESS and ISENTRESS HD? | ||||
ISENTRESS and ISENTRESS HD film-coated tablets:
|
||||
ISENTRESS chewable tablets:
|
||||
ISENTRESS for oral suspension:
|
||||
| Keep ISENTRESS and all medicines out of the reach of children. | ||||
| General information about the safe and effective use of ISENTRESS and ISENTRESS HD | ||||
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. | ||||
| Do not use ISENTRESS or ISENTRESS HD for a condition for which it was not prescribed. Do not give ISENTRESS or ISENTRESS HD to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about ISENTRESS or ISENTRESS HD that is written for health professionals. | ||||
| What are the ingredients in ISENTRESS and ISENTRESS HD? | ||||
| ISENTRESS 400 mg film-coated tablets: | ||||
| Active ingredient: raltegravir | ||||
| Inactive ingredients: calcium phosphate dibasic anhydrous, hypromellose 2208, lactose monohydrate, magnesium stearate, microcrystalline cellulose, poloxamer 407 (contains 0.01% butylated hydroxytoluene as antioxidant), sodium stearyl fumarate. | ||||
| The film coating contains: black iron oxide, polyethylene glycol 3350, polyvinyl alcohol, red iron oxide, talc and titanium dioxide. | ||||
| ISENTRESS HD 600 mg film-coated tablets: | ||||
| Active ingredient: raltegravir | ||||
| Inactive ingredients: croscarmellose sodium, hypromellose 2910, magnesium stearate, microcrystalline cellulose. | ||||
| The film coating contains: ferrosoferric oxide, hypromellose 2910, iron oxide yellow, lactose monohydrate, triacetin and titanium dioxide. | ||||
| The tablet may also contain trace amount of carnauba wax. | ||||
| ISENTRESS chewable tablets: | ||||
| Active ingredient: raltegravir | ||||
| Inactive ingredients: ammonium hydroxide, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, magnesium stearate, mannitol, medium chain triglycerides, monoammonium glycyrrhizinate, natural and artificial flavors (orange, banana, and masking that contains aspartame), oleic acid, PEG 400, saccharin sodium, sodium citrate dihydrate, sodium stearyl fumarate, sorbitol, sucralose and yellow iron oxide. The 100 mg chewable tablet also contains red iron oxide. | ||||
| ISENTRESS for oral suspension: | ||||
| Active ingredient: raltegravir | ||||
| Inactive ingredients: ammonium hydroxide, banana with other natural flavors, carboxymethylcellulose sodium, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, macrogol/PEG 400, magnesium stearate, maltodextrin, mannitol, medium chain triglycerides, microcrystalline cellulose, monoammonium glycyrrhizinate, oleic acid, sorbitol, sucralose and sucrose. | ||||
| Distributed by: Merck Sharp & Dohme LLC Rahway, NJ 07065, USA |
||||
| For patent information: www.msd.com/research/patent | ||||
| Copyright © 2007-2022 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved. | ||||
| usppi-mk0518-mf-2205r030 For more information go to www.ISENTRESS.com or call 1-800-622-4477. |
Section 44425-7
Storage and Handling
Section 51945-4
PRINCIPAL DISPLAY PANEL - 100 mg Packet Carton
NDC 0006-3603-61
Isentress
®
(raltegravir)
For Oral Suspension
100 mg
For Pediatric Use
For Oral Administration Only
Each packet contains 108.6 mg raltegravir potassium, equivalent to 100 mg raltegravir.
Rx only
60 packets
Multi-Dose Kit
DIRECTIONS FOR USE:
See instructions for use
booklet and package insert
for additional information.
Section 59845-8
Bring this booklet to your child's appointments.
ISENTRESS (raltegravir)
for oral suspension
Instructions for Use
for babies and toddlers
Before You Start
| Note: Make sure your doctor shows you how to prepare and give ISENTRESS for oral suspension. |
- Be sure you understand these instructions before you start. Call your doctor if you have any questions.
- It is very important that you measure the water and ISENTRESS carefully using the correct syringe.
- Before you give ISENTRESS to your child, check the expiration date. The expiration date is printed on the box and the ISENTRESS packets.
- Do not open the ISENTRESS packets until you are ready to mix a dose.
- The amount of ISENTRESS depends on your child's age and weight, so it will change over time.
Your doctor will tell you the right dose at each check-up after weighing your child.
Be sure to keep your doctor's appointments so you get new dosing information as your child grows.
During your child's first week of life, you will give ISENTRESS 1 time a day. After the first week of life, you will give it 2 times a day. - This booklet tells you how to:
- Mix ISENTRESS into a liquid form
- Measure the right dose using a syringe
- Give mixed ISENTRESS to your child
- Clean up
Kit Contents
|
|
||
|
|
| 2 blue (10mL) syringes | 2 green (3mL) syringes | 2 white (1mL) syringes |
The kit has an extra cup and set of syringes in case one is lost or damaged.
Do not use any damaged cups or syringes.
Step 1. Get ready
- Put your child in a safe place. You will need both hands to prepare ISENTRESS.
- Wash your hands with soap and water.
- Take out what you need from the kit as shown below to make 1 dose and place on a clean surface:
| 1 mixing cup (Using the tab on the mixing cup, pull open the lid) |
1 packet of ISENTRESS | a clean glass | 3 syringes (Have one of each size ready, but you will only need 1 or 2, depending on the prescribed dose) |
Step 2. Fill a clean glass with water
| Fill a clean glass with room-temperature drinking water from your sink or use bottled water. |
Step 3. Fill the blue syringe with water
| Push the plunger of the blue syringe into the syringe as far as it goes. | |
| Put the tip of the syringe into the glass of water. Pull back the plunger to draw up water into the syringe. Stop when you get to the 10mL mark. |
Step 4. Check for air bubbles
| Hold the syringe with tip up. Tap it with your finger to move any air bubbles up towards the tip. Slowly push up on the plunger to make the air come out. You may see some drops of water come out. |
|
|
Re-check the amount of water in the syringe. If it is less than 10mL, put the tip back into the water and pull back on the plunger until you get to the 10mL mark. |
Step 5. Add the 10mL of water to the mixing cup
Add the 10 mL of water from the syringe to the mixing cup by pushing all the way down on the plunger.
Step 6. Add ISENTRESS to the mixing cup
|
Note before adding ISENTRESS:
Make sure you and your child are ready! After mixing ISENTRESS, use it within 30 minutes. Throw away any leftover ISENTRESS after you have given the dose to your child. |
|
|
Take 1 packet of ISENTRESS and shake the powder to the bottom of the packet. |
| Tear or cut open the packet at the dotted line and add all of the powder to the water in the mixing cup. Make sure the packet is completely empty. |
Step 7. Mix ISENTRESS and water
| Snap the lid of the mixing cup shut. Gently swirl the mixing cup for 45 seconds in a circular motion to mix the powder and water. Use a clock or timer to time for 45 seconds. Do not shake the mixture. |
|
| Check to make sure the powder is mixed. If it is not mixed, swirl it some more. The mixture should look cloudy. |
Step 8. Check your prescription
Find the dose amount in 'mL' prescribed by your doctor. This is written on the prescription label on the box from your pharmacy.
Remember that the dose may change each time you go to the doctor, so make sure you check the prescribed dose each time. Be sure to go to all of your doctor's appointments so your child gets the right dose!
Step 9. Choose the syringe you need
Your doctor will prescribe the dose in milliliters (mL). Choose the right syringe for your child's dose:
|
WHITE
(1mL) for 1mL or less |
GREEN
(3mL) for 1.5mL to 3mL |
BLUE
(10mL) for 3.5mL to 10mL |
Then find the mL mark on the syringe that matches your child's dose.
Step 10. Measure ISENTRESS
| Push the plunger into the barrel of the syringe as far as it goes. | |
| Put the tip of the syringe into the cup of the mixed ISENTRESS and pull back on the plunger. Stop when you get to the line that matches your child's prescribed dose. |
IMPORTANT:
|
Step 11. Check for air bubbles
| Hold the syringe with tip up. Tap it with your finger to move any air bubbles up towards the tip. Slowly push the plunger to make the air come out. You may see some drops of medicine come out. |
|
| Re-check the amount of ISENTRESS in the syringe. If it is less than the prescribed dose, put the tip back into the cup of mixed ISENTRESS and pull back on the plunger until you get to the right dose mark. |
Step 12. Give ISENTRESS to your child
| Place the tip of the syringe inside your child's mouth so that it touches either the right or left cheek. |
Slowly push in the plunger to give the dose of mixed ISENTRESS to your child. If your child fusses, take the tip of the syringe out of the mouth and try again. It is important that your child takes all of the prescribed dose (a little left in the syringe tip is OK).
|
IMPORTANT:
If your child does not take all of the prescribed dose or spits some of it out, call your doctor to find out what to do. |
Step 13. Clean up
Pour the leftover mixed ISENTRESS into the trash.
Do not pour it into the sink.
Pull the plungers out of any syringes you used.
Hand wash the syringes, plungers, and mixing cup with warm water and dish soap. Do not wash in the dishwasher.
| Rinse with water and let air dry. Put everything in a clean, dry place. |
|
How should I store ISENTRESS?
Store the ISENTRESS for oral suspension kit at room temperature between 68°F to 77°F (20°C to 25°C).
Store in the original container.
Keep ISENTRESS and all medicines out of the reach of children.
| Be sure to keep your doctor's appointments so you always know how much ISENTRESS to give to your child. |
For more information, go to www.ISENTRESS.com or call 1-800-622-4477.
2.2 Adults
The recommended adult dosage of ISENTRESS film-coated tablets is displayed in Table 1. ISENTRESS and ISENTRESS HD should be taken by mouth and may be taken with or without food [see Clinical Pharmacology (12.3)].
| Population | Recommended Dose |
|---|---|
| Treatment-naïve patients or patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily | 1200 mg (2 × 600 mg) once daily or 400 mg twice daily |
| Treatment-experienced | 400 mg twice daily |
| Treatment-naïve or treatment-experienced when coadministered with rifampin [see Drug Interactions (7.1)] | 800 mg (2 × 400 mg) twice daily |
10 Overdosage
In the event of an overdose, it is reasonable to employ the standard supportive measures, e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring (including obtaining an electrocardiogram), and institute supportive therapy if required. The extent to which ISENTRESS may be dialyzable is unknown.
11 Description
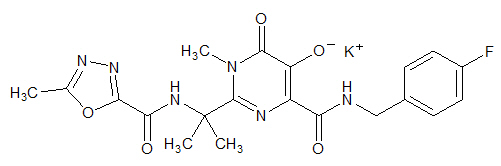
ISENTRESS contains raltegravir potassium, a human immunodeficiency virus integrase strand transfer inhibitor. The chemical name for raltegravir potassium is N-[(4-Fluorophenyl) methyl]-1,6-dihydro-5-hydroxy-1-methyl-2-[1-methyl-1-[[(5-methyl-1,3,4-oxadiazol-2-yl)carbonyl]amino]ethyl]-6-oxo-4-pyrimidinecarboxamide monopotassium salt.
The empirical formula is C20H20FKN6O5 and the molecular weight is 482.51. The structural formula is:
Raltegravir potassium is a white to off-white powder. It is soluble in water, slightly soluble in methanol, very slightly soluble in ethanol and acetonitrile and insoluble in isopropanol.
Each 400 mg film-coated tablet of ISENTRESS for oral administration contains 434.4 mg of raltegravir (as potassium salt), equivalent to 400 mg of raltegravir free phenol and the following inactive ingredients: calcium phosphate dibasic anhydrous, hypromellose 2208, lactose monohydrate, magnesium stearate, microcrystalline cellulose, poloxamer 407 (contains 0.01% butylated hydroxytoluene as antioxidant), sodium stearyl fumarate. In addition, the film coating contains the following inactive ingredients: black iron oxide, polyethylene glycol 3350, polyvinyl alcohol, red iron oxide, talc and titanium dioxide.
Each 600 mg film-coated tablet of ISENTRESS HD for oral administration contains 651.6 mg of raltegravir (as potassium salt), equivalent to 600 mg of raltegravir free phenol and the following inactive ingredients: croscarmellose sodium, hypromellose 2910, magnesium stearate, microcrystalline cellulose. The film coating contains the following inactive ingredients: ferrosoferric oxide, hypromellose 2910, iron oxide yellow, lactose monohydrate, triacetin and titanium dioxide. The tablet may also contain trace amount of carnauba wax.
Each 100 mg chewable tablet of ISENTRESS for oral administration contains 108.6 mg of raltegravir (as potassium salt), equivalent to 100 mg of raltegravir free phenol and the following inactive ingredients: ammonium hydroxide, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, magnesium stearate, mannitol, medium chain triglycerides, monoammonium glycyrrhizinate, natural and artificial flavors (orange, banana, and masking that contains aspartame), oleic acid, PEG 400, red iron oxide, saccharin sodium, sodium citrate dihydrate, sodium stearyl fumarate, sorbitol, sucralose and yellow iron oxide.
Each 25 mg chewable tablet of ISENTRESS for oral administration contains 27.16 mg of raltegravir (as potassium salt), equivalent to 25 mg of raltegravir free phenol and the following inactive ingredients: ammonium hydroxide, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, magnesium stearate, mannitol, medium chain triglycerides, monoammonium glycyrrhizinate, natural and artificial flavors (orange, banana, and masking that contains aspartame), oleic acid, PEG 400, saccharin sodium, sodium citrate dihydrate, sodium stearyl fumarate, sorbitol, sucralose and yellow iron oxide.
Each packet of ISENTRESS for oral suspension 100 mg, contains 108.6 mg of raltegravir (as potassium salt), equivalent to 100 mg of raltegravir free phenol and the following inactive ingredients: ammonium hydroxide, banana with other natural flavors, carboxymethylcellulose sodium, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, macrogol/PEG 400, magnesium stearate, maltodextrin, mannitol, medium chain triglycerides, microcrystalline cellulose, monoammonium glycyrrhizinate, oleic acid, sorbitol, sucralose and sucrose.
2.3 Pediatrics
The recommended pediatric dosage of ISENTRESS is displayed in Table 2. ISENTRESS film-coated tablets, chewable tablets and for oral suspension should be taken by mouth and may be taken with or without food [see Clinical Pharmacology (12.3)].
| Recommended Pediatric Dosage and Formulation | ||||
|---|---|---|---|---|
| Population/Weight | Film-Coated Tablets 400 mg | Film-Coated Tablets 600 mg | Chewable Tablets 100 mg and 25 mg | For Oral Suspension 100 mg |
If at least 40 kg and either:
|
400 mg twice daily | 1200 mg (2 × 600 mg) once daily | 300 mg twice daily (see Table 3) | NA |
| If at least 25 kg | 400 mg twice daily If able to swallow a tablet
|
NA | Weight-based dosing twice daily (see Table 3) | NA |
| If at least 4 weeks of age and weighing 3 kg to less than 25 kg | NA | NA | Weight-based dosing twice daily (see Table 4) | Weight-based dosing twice daily up to 20 kg (see Table 4) |
| From birth to 4 weeks (28 days) weighing at least 2 kg | NA | NA | NA | Weight-based dosing once daily or twice daily (see Table 5) |
| Body Weight (kg) |
Dose | Number of 100 mg Chewable Tablets |
|---|---|---|
| 25 to less than 28 | 150 mg twice daily | 1.5 × 100 mg The 100 mg chewable tablet can be divided into equal halves. twice daily |
| 28 to less than 40 | 200 mg twice daily | 2 × 100 mg twice daily |
| At least 40 | 300 mg twice daily | 3 × 100 mg twice daily |
| Body Weight (kg) |
Volume (Dose) of Suspension to be Administered | Number of Chewable Tablets The chewable tablets are available as 25 mg and 100 mg tablets.
|
|---|---|---|
| 3 to less than 4 | 2.5 mL (25 mg) twice daily | 1 × 25 mg twice daily May be administered as a crushed tablet(s); see
General Dosing Recommendations (2.1)
for guidance.
|
| 4 to less than 6 | 3 mL (30 mg) twice daily | |
| 6 to less than 8 | 4 mL (40 mg) twice daily | 2 × 25 mg twice daily |
| 8 to less than 10 | 6 mL (60 mg) twice daily | |
| 10 to less than 14 | 8 mL (80 mg) twice daily | 3 × 25 mg twice daily |
| 14 to less than 20 | 10 mL (100 mg) twice daily | 1 × 100 mg twice daily |
| 20 to less than 25 | Not applicable | 1.5 × 100 mg The 100 mg chewable tablet can be divided into equal halves. twice daily |
- For full-term neonates (birth to 4 weeks [28 days] of age): Weight-based dosing of the oral suspension as specified in Table 5.
- No data are available in pre-term neonates. The use of ISENTRESS is not recommended in pre-term neonates.
| Body Weight (kg) |
Volume (Dose) of Suspension to be Administered |
|---|---|
| Note: If the mother has taken ISENTRESS or ISENTRESS HD 2-24 hours before delivery, the neonate's first dose should be given between 24-48 hours after birth. | |
|
Birth to 1 Week - Once daily dosing The dosing recommendations are based on approximately 1.5 mg/kg/dose.
|
|
| 2 to less than 3 | 0.4 mL (4 mg) once daily |
| 3 to less than 4 | 0.5 mL (5 mg) once daily |
| 4 to less than 5 | 0.7 mL (7 mg) once daily |
|
1 to 4 Weeks - Twice daily dosing The dosing recommendations are based on approximately 3 mg/kg/dose.
|
|
| 2 to less than 3 | 0.8 mL (8 mg) twice daily |
| 3 to less than 4 | 1 mL (10 mg) twice daily |
| 4 to less than 5 | 1.5 mL (15 mg) twice daily |
8.5 Geriatric Use
Clinical studies of ISENTRESS/ISENTRESS HD did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications
None
6 Adverse Reactions
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7 Drug Interactions
- Coadministration of ISENTRESS or ISENTRESS HD and other drugs may alter the plasma concentration of raltegravir. The potential for drug-drug interactions must be considered prior to and during therapy (7).
- Coadministration of ISENTRESS or ISENTRESS HD with drugs that are strong inducers of UGT1A1, such as rifampin, may result in reduced plasma concentrations of raltegravir (2.1, 7.1).
5.3 Phenylketonurics
ISENTRESS Chewable Tablets contain phenylalanine, a component of aspartame. Each 25 mg ISENTRESS Chewable Tablet contains approximately 0.05 mg phenylalanine. Each 100 mg ISENTRESS Chewable Tablet contains approximately 0.10 mg phenylalanine. Phenylalanine can be harmful to patients with phenylketonuria.
12.2 Pharmacodynamics
In a monotherapy study raltegravir (400 mg twice daily) demonstrated rapid antiviral activity with mean viral load reduction of 1.66 log10 copies/mL by day 10.
1 Indications and Usage
Adult Patients:
ISENTRESS and ISENTRESS HD are human immunodeficiency virus integrase strand transfer inhibitors (HIV-1 INSTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in adult patients (1).
Pediatric Patients:
ISENTRESS is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 2 kg (1).
ISENTRESS HD is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 40 kg (1).
12.1 Mechanism of Action
Raltegravir is an HIV-1 antiviral drug [see Microbiology (12.4)].
5 Warnings and Precautions
- Severe, potentially life-threatening and fatal skin reactions have been reported. This includes cases of Stevens-Johnson syndrome, hypersensitivity reaction and toxic epidermal necrolysis. Immediately discontinue treatment with ISENTRESS or ISENTRESS HD and other suspect agents if severe hypersensitivity, severe rash, or rash with systemic symptoms or liver aminotransferase elevations develops and monitor clinical status, including liver aminotransferases closely (5.1).
- Monitor for Immune Reconstitution Syndrome (5.2).
- Inform patients with phenylketonuria that the 100 mg and 25 mg chewable tablets contain phenylalanine (5.3).
2 Dosage and Administration
ISENTRESS and ISENTRESS HD can be administered with or without food (2.1).
Do not substitute ISENTRESS chewable tablets or ISENTRESS for oral suspension for the ISENTRESS 400 mg or 600 mg film-coated tablet.
See specific dosing guidance for chewable tablets and the formulation for oral suspension (2.1).
Adults
- Treatment-naïve patients or patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily:
- 1200 mg (2 × 600 mg) film-coated tablet orally, once daily or
- 400 mg film-coated tablet orally, twice daily (2.2).
- Treatment-experienced patients:
- 400 mg film-coated tablet orally, twice daily (2.2).
- During coadministration with rifampin in adults, 800 mg (2 × 400 mg) twice daily (2.2).
Pediatrics
- If weighing at least 40 kg, and either
- treatment-naïve patients or
- patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily:
- 1200 mg (2 × 600 mg) film-coated tablet orally, once daily or
- 400 mg film-coated tablet orally, twice daily or
- 300 mg (3 × 100 mg) chewable tablets, twice daily (2.3).
- If weighing at least 25 kg: One 400 mg film-coated tablet orally, twice daily. If unable to swallow a tablet, consider the chewable tablet, as specified in Table 2 (2.3).
- If weighing at least 3 kg to less than 25 kg: Weight-based dosing using the chewable tablet or oral suspension, as specified in Table 4 (2.3).
- For neonates (birth to 4 weeks [28 days] of age): Weight-based dosing of the oral suspension as specified in Table 5 (2.3).
3 Dosage Forms and Strengths
- Film-coated Tablets
- 400 mg pink, oval-shaped, film-coated tablets with "227" on one side (ISENTRESS).
- 600 mg yellow, oval-shaped, film-coated tablets with corporate logo and "242" on one side and plain on the other side (ISENTRESS HD).
- Chewable Tablets
- 100 mg pale orange, oval-shaped, orange-banana flavored, chewable tablets scored on both sides and imprinted on one face with the corporate logo and "477" on opposite sides of the score.
- 25 mg pale yellow, round, orange-banana flavored, chewable tablets with the corporate logo on one side and "473" on the other side.
- For Oral Suspension
- 100 mg white to off-white, banana flavored, granular powder that may contain yellow or beige to tan particles in a child resistant single-use foil packet.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ISENTRESS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: thrombocytopenia
Gastrointestinal Disorders: diarrhea
Hepatobiliary Disorders: hepatic failure (with and without associated hypersensitivity) in patients with underlying liver disease and/or concomitant medications
Musculoskeletal and Connective Tissue Disorders: rhabdomyolysis
Nervous System Disorders: cerebellar ataxia
Psychiatric Disorders: anxiety, paranoia
8 Use in Specific Populations
Lactation: Women infected with HIV should be instructed not to breastfeed due to the potential for HIV transmission (8.2).
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
2.1 General Dosing Recommendations
- Because the formulations have different pharmacokinetic profiles, do not substitute ISENTRESS chewable tablets or ISENTRESS for oral suspension for the ISENTRESS 400 mg film-coated tablet or the ISENTRESS HD 600 mg film-coated tablet. See specific dosing guidance for chewable tablets and the formulation for oral suspension.
- Because the extent to which ISENTRESS may be dialyzable is unknown, dosing before a dialysis session should be avoided [see Clinical Pharmacology (12.3)].
- ISENTRESS film-coated tablets must be swallowed whole.
- ISENTRESS chewable tablets may be chewed or swallowed whole. Maximum daily dose is 300 mg taken by mouth twice daily.
- For children who have difficulty chewing the 25 mg chewable tablet, the tablet may be crushed.
- Preparation of the crushed 25 mg chewable tablet:
- Place the tablet(s) in a small, clean cup. For each tablet, add a teaspoonful (~5 mL) of liquid (for example, water, juice, or breast milk).
- Within 2 minutes, the tablet(s) will absorb the liquid and fall apart.
- Using a spoon, crush any remaining pieces of the tablet(s). Immediately administer the entire dose orally.
- If any portion of the dose is left in the cup, add another teaspoonful (~5 mL) of liquid, swirl and administer immediately.
- Preparation of the crushed 25 mg chewable tablet:
- ISENTRESS for oral suspension:
- See Instructions for Use for details on preparation and administration of ISENTRESS for oral suspension.
- Using the provided mixing cup, combine 10 mL of water and the entire contents of one packet of ISENTRESS for oral suspension and mix. Each single-use packet for oral suspension contains 100 mg of raltegravir which is suspended in 10 mL of water giving a final concentration of 10 mg per mL. Maximum daily dose is 100 mg taken by mouth twice daily.
- Gently swirl the mixing cup for 45 seconds in a circular motion to mix the powder into a uniform suspension. Do not shake.
- Once mixed, measure the prescribed dose volume of suspension with a syringe and administer the dose orally. The dose should be administered orally within 30 minutes of mixing.
- Discard any remaining suspension into the trash.
5.2 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including ISENTRESS. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jiroveci pneumonia, tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
14.1 Description of Clinical Studies
The evidence of durable efficacy of ISENTRESS 400 mg twice daily is based on the analyses of 240-week data from a randomized, double-blind, active-controlled trial, STARTMRK evaluating ISENTRESS 400 mg twice daily in antiretroviral treatment-naïve HIV-1 infected adult subjects, the analysis of 96-week data from a randomized, double-blind, active-control trial, ONCEMRK evaluating ISENTRESS HD 1200 mg (2 × 600 mg) once daily in treatment-naïve adult subjects, and 96-week data from 2 randomized, double-blind, placebo-controlled studies, BENCHMRK 1 and BENCHMRK 2, evaluating ISENTRESS 400 mg twice daily in antiretroviral treatment-experienced HIV-1 infected adult subjects. See Table 18.
| Trial | Study Type | Population | Study Arms (N) | Dose/Formulation | Timepoint |
|---|---|---|---|---|---|
| STARTMRK | Randomized, double-blind, active-controlled | Treatment-Naïve Adults | ISENTRESS 400 mg Twice Daily (281) Efavirenz 600 mg At Bedtime (282) Both in combination with emtricitabine (+) tenofovir disoproxil fumarate |
400 mg film-coated tablet | Week 240 |
| ONCEMRK | Randomized, double-blind, active-controlled | Treatment-Naïve Adults | ISENTRESS HD 1200 mg Once Daily (531) | 600 mg film-coated tablet | Week 96 |
| ISENTRESS 400 mg Twice Daily (266) Both in combination with emtricitabine (+) tenofovir disoproxil fumarate |
400 mg film-coated tablet | ||||
| BENCHMRK 1 | Randomized, double-blind, placebo-controlled | Treatment-Experienced Adults | ISENTRESS 400 mg Twice Daily (232) Placebo (118) Both in combination with optimized background therapy |
400 mg film-coated tablet | Week 240 (Week 156 on double-blind plus Week 84 on open-label) |
| BENCHMRK 2 | Randomized, double-blind, placebo-controlled | Treatment-Experienced Adults | ISENTRESS 400 mg Twice Daily (230) Placebo (119) Both in combination with optimized background therapy |
400 mg film-coated tablet | Week 240 (Week 156 on double-blind plus Week 84 on open-label) |
| IMPAACT P1066 | Open-label, non-comparative | Pediatric Patients – 4 weeks to 18 years of age (Treatment-Experienced or Failed Prior PMTCT) |
ISENTRESS 400 mg tablet Twice Daily – 12 to 18 years or 6 to <12 years and ≥25 kg (87) ISENTRESS chewable tablet- Weight-Based Dose to Approximate 6 mg/kg Twice Daily – 2 to <12 years (39) ISENTRESS for oral suspension- Weight-Based Dose to Approximate 6 mg/kg Twice Daily – 4 weeks to <2 years (26) In combination with optimized background therapy |
400 mg film-coated tablet 25 mg and 100 mg chewable tablet 100 mg sachet for oral suspension |
Week 240 |
16 How Supplied/storage and Handling
ISENTRESS tablets 400 mg are pink, oval-shaped, film-coated tablets with "227" on one side. They are supplied as follows:
-
NDC 0006-0227-61 unit-of-use bottles of 60.
ISENTRESS HD tablets 600 mg are yellow, oval-shaped, film-coated tablets with corporate logo and "242" on one side and plain on the other side. They are supplied as follows:
-
NDC 0006-3080-01 unit-of-use bottles of 60.
ISENTRESS tablets 100 mg are pale orange, oval-shaped, orange-banana flavored, chewable tablets scored on both sides and imprinted on one face with the corporate logo and "477" on opposite sides of the score. They are supplied as follows:
-
NDC 0006-0477-61 unit-of-use bottles of 60.
ISENTRESS tablets 25 mg are pale yellow, round, orange-banana flavored, chewable tablets with the corporate logo on one side and "473" on the other side. They are supplied as follows:
-
NDC 0006-0473-61 unit-of-use bottles of 60.
ISENTRESS for oral suspension 100 mg is a white to off-white granular powder that may contain yellow or beige to tan particles, in child resistant single-use foil packets, packaged as
A kit with two 1 mL syringes, two 3 mL syringes, two 10 mL syringes and two mixing cups. It is supplied as follows:
-
NDC 0006-3603-61 unit of use carton with 60 packets.
-
NDC 0006-3603-01 individual packet.
14.3 Treatment Experienced Adult Subjects
BENCHMRK 1 and BENCHMRK 2 are Phase 3 studies to evaluate the safety and antiretroviral activity of ISENTRESS 400 mg twice daily in combination with an optimized background therapy (OBT), versus OBT alone, in HIV-1-infected subjects, 16 years or older, with documented resistance to at least 1 drug in each of 3 classes (NNRTIs, NRTIs, PIs) of antiretroviral therapies. Randomization was stratified by degree of resistance to PI (1PI vs. >1PI) and the use of enfuvirtide in the OBT. Prior to randomization, OBT was selected by the investigator based on genotypic/phenotypic resistance testing and prior ART history.
Table 20 shows the demographic characteristics of subjects in the group receiving ISENTRESS 400 mg twice daily and subjects in the placebo group.
| Randomized Studies BENCHMRK 1 and BENCHMRK 2 |
ISENTRESS 400 mg Twice Daily + OBT (N = 462) |
Placebo + OBT (N = 237) |
|---|---|---|
| Gender | ||
| Male | 88% | 89% |
| Female | 12% | 11% |
| Race | ||
| White | 65% | 73% |
| Black | 14% | 11% |
| Asian | 3% | 3% |
| Hispanic | 11% | 8% |
| Others | 6% | 5% |
| Age (years) | ||
| Median (min, max) | 45 (16 to 74) | 45 (17 to 70) |
| CD4+ Cell Count | ||
| Median (min, max), cells/mm3 | 119 (1 to 792) | 123 (0 to 759) |
| ≤50 cells/mm3 | 32% | 33% |
| >50 and ≤200 cells/mm3 | 37% | 36% |
| Plasma HIV-1 RNA | ||
| Median (min, max), log10 copies/mL | 4.8 (2 to 6) | 4.7 (2 to 6) |
| >100,000 copies/mL | 36% | 33% |
| History of AIDS | ||
| Yes | 92% | 91% |
| Prior Use of ART, Median (1st Quartile, 3rd Quartile) | ||
| Years of ART Use | 10 (7 to 12) | 10 (8 to 12) |
| Number of ART | 12 (9 to 15) | 12 (9 to 14) |
|
Hepatitis Co-infection Hepatitis B virus surface antigen positive or hepatitis C virus antibody positive.
|
||
| No Hepatitis B or C virus | 83% | 84% |
| Hepatitis B virus only | 8% | 3% |
| Hepatitis C virus only | 8% | 12% |
| Co-infection of Hepatitis B and C virus | 1% | 1% |
| Stratum | ||
| Enfuvirtide in OBT | 38% | 38% |
| Resistant to ≥2 PI | 97% | 95% |
Table 21 compares the characteristics of optimized background therapy at baseline in the group receiving ISENTRESS 400 mg twice daily and subjects in the control group.
| Randomized Studies BENCHMRK 1 and BENCHMRK 2 |
ISENTRESS 400 mg Twice Daily + OBT (N = 462) |
Placebo + OBT (N = 237) |
|---|---|---|
| Number of ARTs in OBT | ||
| Median (min, max) | 4 (1 to 7) | 4 (2 to 7) |
|
Number of Active PI in OBT by Phenotypic Resistance Test Darunavir use in OBT in darunavir-naïve subjects was counted as one active PI.
|
||
| 0 | 36% | 41% |
| 1 or more | 60% | 58% |
|
Phenotypic Sensitivity Score (PSS) The Phenotypic Sensitivity Score (PSS) and the Genotypic Sensitivity Score (GSS) were defined as the total oral ARTs in OBT to which a subject's viral isolate showed phenotypic sensitivity and genotypic sensitivity, respectively, based upon phenotypic and genotypic resistance tests. Enfuvirtide use in OBT in enfuvirtide-naïve subjects was counted as one active drug in OBT in the GSS and PSS. Similarly, darunavir use in OBT in darunavir-naïve subjects was counted as one active drug in OBT.
|
||
| 0 | 15% | 18% |
| 1 | 31% | 30% |
| 2 | 31% | 28% |
| 3 or more | 18% | 20% |
| Genotypic Sensitivity Score (GSS) | ||
| 0 | 25% | 27% |
| 1 | 38% | 40% |
| 2 | 24% | 21% |
| 3 or more | 11% | 10% |
Week 96 outcomes for the 699 subjects randomized and treated with the recommended dose of ISENTRESS 400 mg twice daily or placebo in the pooled BENCHMRK 1 and 2 studies are shown in Table 22.
| ISENTRESS 400 mg Twice Daily + OBT (N = 462) |
Placebo + OBT (N = 237) |
|
|---|---|---|
| Subjects with HIV-1 RNA less than 50 copies/mL | 55% | 27% |
|
Virologic Failure Includes subjects who switched to open-label raltegravir after Week 16 due to the protocol-defined virologic failure, subjects who discontinued prior to Week 96 for lack of efficacy, subjects changed OBT due to lack of efficacy prior to Week 96, or subjects who were ≥50 copies in the 96 week window.
|
35% | 66% |
| No virologic data at Week 96 Window | ||
| Reasons | ||
| Discontinued study due to AE or death Includes subjects who discontinued due to AE or death at any time point from day 1 through the Week 96 window if this resulted in no virologic data on treatment during the Week 96 window.
|
3% | 3% |
| Discontinued study for other reasons Other includes: withdrew consent, loss to follow-up, moved etc., if the viral load at the time of discontinuation was <50 copies/mL.
|
4% | 4% |
| Missing data during window but on study | 4% | <1% |
The mean changes in CD4 count from baseline were 118 cells/mm3 in the group receiving ISENTRESS 400 mg twice daily and 47 cells/mm3 for the control group.
Treatment-emergent CDC Category C events occurred in 4% of the group receiving ISENTRESS 400 mg twice daily and 5% of the control group.
Virologic responses at Week 96 by baseline genotypic and phenotypic sensitivity score are shown in Table 23.
| Percent with HIV-1 RNA <50 copies/mL At Week 96 | ||||
|---|---|---|---|---|
| n | ISENTRESS 400 mg Twice Daily + OBT (N = 462) |
n | Placebo + OBT (N = 237) |
|
|
Phenotypic Sensitivity Score (PSS) The Phenotypic Sensitivity Score (PSS) and the Genotypic Sensitivity Score (GSS) were defined as the total oral ARTs in OBT to which a subject's viral isolate showed phenotypic sensitivity and genotypic sensitivity, respectively, based upon phenotypic and genotypic resistance tests. Enfuvirtide use in OBT in enfuvirtide-naïve subjects was counted as one active drug in OBT in the GSS and PSS. Similarly, darunavir use in OBT in darunavir-naïve subjects was counted as one active drug in OBT.
|
||||
| 0 | 67 | 43 | 43 | 5 |
| 1 | 144 | 58 | 71 | 23 |
| 2 | 142 | 61 | 66 | 32 |
| 3 or more | 85 | 48 | 48 | 42 |
| Genotypic Sensitivity Score (GSS) | ||||
| 0 | 116 | 39 | 65 | 5 |
| 1 | 177 | 62 | 95 | 26 |
| 2 | 111 | 61 | 49 | 53 |
| 3 or more | 51 | 49 | 23 | 35 |
8.7 Use in Patients With Renal Impairment
No dosage adjustment of ISENTRESS or ISENTRESS HD is necessary in patients with any degree of renal impairment [see Clinical Pharmacology (12.3)]. The extent to which ISENTRESS may be dialyzable is unknown; therefore, dosing before a dialysis session should be avoided.
8.6 Use in Patients With Hepatic Impairment
No dosage adjustment of ISENTRESS is necessary for patients with mild to moderate (Child-Pugh A and B) hepatic impairment. No hepatic impairment study has been conducted with ISENTRESS HD and therefore administration in subjects with hepatic impairment is not recommended. The effect of severe hepatic impairment on the pharmacokinetics of raltegravir has not been studied [see Clinical Pharmacology (12.3)].
Principal Display Panel 600 Mg Bottle Label
NDC 0006-3080-01
Isentress
®
HD
(raltegravir) tablets
600 mg
RECOMMENDED DOSAGE: Two tablets once daily.
See Package Insert.
Each tablet contains 651.6 mg raltegravir
potassium, equivalent to
600 mg raltegravir.
Rx only
60 Tablets
5.1 Severe Skin and Hypersensitivity Reactions
Severe, potentially life-threatening, and fatal skin reactions have been reported. These include cases of Stevens-Johnson syndrome and toxic epidermal necrolysis. Hypersensitivity reactions have also been reported and were characterized by rash, constitutional findings, and sometimes, organ dysfunction, including hepatic failure. Discontinue ISENTRESS or ISENTRESS HD and other suspect agents immediately if signs or symptoms of severe skin reactions or hypersensitivity reactions develop (including, but not limited to, severe rash or rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, hepatitis, eosinophilia, angioedema). Clinical status including liver aminotransferases should be monitored and appropriate therapy initiated. Delay in stopping ISENTRESS or ISENTRESS HD treatment or other suspect agents after the onset of severe rash may result in a life-threatening reaction.
Principal Display Panel 25 Mg Tablet Bottle Label
NDC 0006-0473-61
Isentress®
(raltegravir) CHEWABLE Tablets
25 mg
For Pediatric Use
Phenylketonurics:
contains phenylalanine (a component
of aspartame) 0.05 mg per 25 mg chewable tablet.
Rx only
60 Tablets
Principal Display Panel 100 Mg Tablet Bottle Label
NDC 0006-0477-61
Isentress®
(raltegravir) CHEWABLE Tablets
100 mg
For Pediatric Use
Phenylketonurics:
contains phenylalanine (a component
of aspartame) 0.10 mg per 100 mg chewable tablet.
Rx only
60 Tablets
Principal Display Panel 400 Mg Tablet Bottle Label
NDC 0006-0227-61
Isentress®
(raltegravir) tablets
400 mg
Each tablet contains 434.4 mg raltegravir
potassium, equivalent to 400 mg raltegravir.
USUAL DOSAGE: See Package Insert.
Rx only
60 Tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies of raltegravir in mice did not show any carcinogenic potential. At the highest dose levels, 400 mg/kg/day in females and 250 mg/kg/day in males, systemic exposure was 1.8-fold (females) or 1.2-fold (males) greater than the AUC (54 µM∙hr) at the 400-mg twice daily human dose. Treatment-related squamous cell carcinoma of nose/nasopharynx was observed in female rats dosed with 600 mg/kg/day raltegravir for 104 weeks. These tumors were possibly the result of local irritation and inflammation due to local deposition and/or aspiration of drug in the mucosa of the nose/nasopharynx during dosing. No tumors of the nose/nasopharynx were observed in rats dosed with 150 mg/kg/day (males) and 50 mg/kg/day (females) and the systemic exposure in rats was 1.7-fold (males) to 1.4-fold (females) greater than the AUC (54 µM∙hr) at the 400-mg twice daily human dose.
No evidence of mutagenicity or genotoxicity was observed in in vitro microbial mutagenesis (Ames) tests, in vitro alkaline elution assays for DNA breakage, and in vitro and in vivo chromosomal aberration studies.
No effect on fertility was seen in male and female rats at doses up to 600 mg/kg/day which resulted in a 3-fold exposure above the exposure at the recommended human dose.
7.1 Effect of Other Agents On the Pharmacokinetics of Raltegravir
Raltegravir is not a substrate of cytochrome P450 (CYP) enzymes. Based on in vivo and in vitro studies, raltegravir is eliminated mainly by metabolism via a UGT1A1-mediated glucuronidation pathway. Coadministration of ISENTRESS with drugs that inhibit UGT1A1 may increase plasma levels of raltegravir and coadministration of ISENTRESS with drugs that induce UGT1A1, such as rifampin, may reduce plasma levels of raltegravir (see Table 11).
Selected drug interactions are presented in Table 11 [see Clinical Pharmacology (12.3)]. In some cases, recommendations differ for ISENTRESS versus ISENTRESS HD.
| Concomitant Drug Class: Drug Name |
Effect on Concentration of Raltegravir | Clinical Comment for ISENTRESS | Clinical Comment for ISENTRESS HD |
|---|---|---|---|
| Metal-Containing Antacids | |||
| Aluminum and/or magnesium-containing antacids | ↓ | Coadministration or staggered administration is not recommended. | |
| Calcium carbonate antacid | ↓ | No dose adjustment | Co-administration is not recommended |
| Other Agents | |||
| Rifampin | ↓ | The recommended dosage is 800 mg twice daily during coadministration with rifampin. There are no data to guide co-administration of ISENTRESS with rifampin in patients below 18 years of age [see Dosage and Administration (2.1)]. | Coadministration is not recommended. |
| Tipranavir/ritonavir | No dose adjustment | Coadministration is not recommended | |
| Etravirine | ↓ | No dose adjustment | Coadministration is not recommended. |
| Strong inducers of drug metabolizing enzymes not mentioned above e.g., Carbamazepine Phenobarbital Phenytoin |
↓↔ | The impact of other strong inducers of drug metabolizing enzymes on raltegravir is unknown. Coadministration is not recommended. |
Structured Label Content
Section 42229-5 (42229-5)
Adult Patients:
ISENTRESS® and ISENTRESS® HD are indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) infection in adult patients.
Section 42230-3 (42230-3)
| PATIENT INFORMATION | ||||
|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised May 2022 | |||
| ISENTRESS® (eye sen tris) (raltegravir) film-coated tablets |
ISENTRESS® (eye sen tris) (raltegravir) chewable tablets |
|||
| ISENTRESS® HD (eye sen tris HD) (raltegravir) film-coated tablets |
ISENTRESS® (eye sen tris) (raltegravir) for oral suspension |
|||
| What are ISENTRESS and ISENTRESS HD? | ||||
| ISENTRESS is a prescription medicine used with other HIV-1 medicines to treat Human Immunodeficiency Virus-1 (HIV-1) infection in adults, and in children weighing at least 4.4 pounds (2 kg). HIV is the virus that causes AIDS (Acquired Immune Deficiency Syndrome). | ||||
| ISENTRESS HD is a prescription medicine used with other HIV-1 medicines to treat HIV-1 infection in adults, and in children weighing at least 88 pounds (40 kg). ISENTRESS should not be used in children who weigh less than 4.4 pounds (2 kg). |
||||
Before you take ISENTRESS or ISENTRESS HD, tell your doctor about all of your medical conditions, including if you:
|
||||
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Some medicines interact with ISENTRESS and ISENTRESS HD.
|
||||
How should I take ISENTRESS or ISENTRESS HD?
If ISENTRESS for oral suspension is prescribed for your child, be sure to read the following information:
|
||||
| What are the possible side effects of ISENTRESS or ISENTRESS HD? | ||||
ISENTRESS and ISENTRESS HD can cause serious side effects including:
|
||||
|
|
|||
| Sometimes allergic reactions can affect body organs, such as your liver. Call your doctor right away if you have any of the following signs or symptoms of liver problems: | ||||
|
|
|||
|
||||
| The most common side effects of ISENTRESS and ISENTRESS HD include: | ||||
|
|
|||
| Less common side effects of ISENTRESS and ISENTRESS HD include: | ||||
|
|
|||
| Tell your doctor right away if you get unexplained muscle pain, tenderness, or weakness during treatment with ISENTRESS or ISENTRESS HD. These may be signs of a rare serious muscle problem that can lead to kidney problems. | ||||
| These are not all the possible side effects of ISENTRESS and ISENTRESS HD. | ||||
| Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||||
| How should I store ISENTRESS and ISENTRESS HD? | ||||
ISENTRESS and ISENTRESS HD film-coated tablets:
|
||||
ISENTRESS chewable tablets:
|
||||
ISENTRESS for oral suspension:
|
||||
| Keep ISENTRESS and all medicines out of the reach of children. | ||||
| General information about the safe and effective use of ISENTRESS and ISENTRESS HD | ||||
| Medicines are sometimes prescribed for purposes other than those listed in a Patient Information Leaflet. | ||||
| Do not use ISENTRESS or ISENTRESS HD for a condition for which it was not prescribed. Do not give ISENTRESS or ISENTRESS HD to other people, even if they have the same symptoms that you have. It may harm them. You can ask your doctor or pharmacist for information about ISENTRESS or ISENTRESS HD that is written for health professionals. | ||||
| What are the ingredients in ISENTRESS and ISENTRESS HD? | ||||
| ISENTRESS 400 mg film-coated tablets: | ||||
| Active ingredient: raltegravir | ||||
| Inactive ingredients: calcium phosphate dibasic anhydrous, hypromellose 2208, lactose monohydrate, magnesium stearate, microcrystalline cellulose, poloxamer 407 (contains 0.01% butylated hydroxytoluene as antioxidant), sodium stearyl fumarate. | ||||
| The film coating contains: black iron oxide, polyethylene glycol 3350, polyvinyl alcohol, red iron oxide, talc and titanium dioxide. | ||||
| ISENTRESS HD 600 mg film-coated tablets: | ||||
| Active ingredient: raltegravir | ||||
| Inactive ingredients: croscarmellose sodium, hypromellose 2910, magnesium stearate, microcrystalline cellulose. | ||||
| The film coating contains: ferrosoferric oxide, hypromellose 2910, iron oxide yellow, lactose monohydrate, triacetin and titanium dioxide. | ||||
| The tablet may also contain trace amount of carnauba wax. | ||||
| ISENTRESS chewable tablets: | ||||
| Active ingredient: raltegravir | ||||
| Inactive ingredients: ammonium hydroxide, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, magnesium stearate, mannitol, medium chain triglycerides, monoammonium glycyrrhizinate, natural and artificial flavors (orange, banana, and masking that contains aspartame), oleic acid, PEG 400, saccharin sodium, sodium citrate dihydrate, sodium stearyl fumarate, sorbitol, sucralose and yellow iron oxide. The 100 mg chewable tablet also contains red iron oxide. | ||||
| ISENTRESS for oral suspension: | ||||
| Active ingredient: raltegravir | ||||
| Inactive ingredients: ammonium hydroxide, banana with other natural flavors, carboxymethylcellulose sodium, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, macrogol/PEG 400, magnesium stearate, maltodextrin, mannitol, medium chain triglycerides, microcrystalline cellulose, monoammonium glycyrrhizinate, oleic acid, sorbitol, sucralose and sucrose. | ||||
| Distributed by: Merck Sharp & Dohme LLC Rahway, NJ 07065, USA |
||||
| For patent information: www.msd.com/research/patent | ||||
| Copyright © 2007-2022 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved. | ||||
| usppi-mk0518-mf-2205r030 For more information go to www.ISENTRESS.com or call 1-800-622-4477. |
Section 44425-7 (44425-7)
Storage and Handling
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL - 100 mg Packet Carton
NDC 0006-3603-61
Isentress
®
(raltegravir)
For Oral Suspension
100 mg
For Pediatric Use
For Oral Administration Only
Each packet contains 108.6 mg raltegravir potassium, equivalent to 100 mg raltegravir.
Rx only
60 packets
Multi-Dose Kit
DIRECTIONS FOR USE:
See instructions for use
booklet and package insert
for additional information.
Section 59845-8 (59845-8)
Bring this booklet to your child's appointments.
ISENTRESS (raltegravir)
for oral suspension
Instructions for Use
for babies and toddlers
Before You Start
| Note: Make sure your doctor shows you how to prepare and give ISENTRESS for oral suspension. |
- Be sure you understand these instructions before you start. Call your doctor if you have any questions.
- It is very important that you measure the water and ISENTRESS carefully using the correct syringe.
- Before you give ISENTRESS to your child, check the expiration date. The expiration date is printed on the box and the ISENTRESS packets.
- Do not open the ISENTRESS packets until you are ready to mix a dose.
- The amount of ISENTRESS depends on your child's age and weight, so it will change over time.
Your doctor will tell you the right dose at each check-up after weighing your child.
Be sure to keep your doctor's appointments so you get new dosing information as your child grows.
During your child's first week of life, you will give ISENTRESS 1 time a day. After the first week of life, you will give it 2 times a day. - This booklet tells you how to:
- Mix ISENTRESS into a liquid form
- Measure the right dose using a syringe
- Give mixed ISENTRESS to your child
- Clean up
Kit Contents
|
|
||
|
|
| 2 blue (10mL) syringes | 2 green (3mL) syringes | 2 white (1mL) syringes |
The kit has an extra cup and set of syringes in case one is lost or damaged.
Do not use any damaged cups or syringes.
Step 1. Get ready
- Put your child in a safe place. You will need both hands to prepare ISENTRESS.
- Wash your hands with soap and water.
- Take out what you need from the kit as shown below to make 1 dose and place on a clean surface:
| 1 mixing cup (Using the tab on the mixing cup, pull open the lid) |
1 packet of ISENTRESS | a clean glass | 3 syringes (Have one of each size ready, but you will only need 1 or 2, depending on the prescribed dose) |
Step 2. Fill a clean glass with water
| Fill a clean glass with room-temperature drinking water from your sink or use bottled water. |
Step 3. Fill the blue syringe with water
| Push the plunger of the blue syringe into the syringe as far as it goes. | |
| Put the tip of the syringe into the glass of water. Pull back the plunger to draw up water into the syringe. Stop when you get to the 10mL mark. |
Step 4. Check for air bubbles
| Hold the syringe with tip up. Tap it with your finger to move any air bubbles up towards the tip. Slowly push up on the plunger to make the air come out. You may see some drops of water come out. |
|
|
Re-check the amount of water in the syringe. If it is less than 10mL, put the tip back into the water and pull back on the plunger until you get to the 10mL mark. |
Step 5. Add the 10mL of water to the mixing cup
Add the 10 mL of water from the syringe to the mixing cup by pushing all the way down on the plunger.
Step 6. Add ISENTRESS to the mixing cup
|
Note before adding ISENTRESS:
Make sure you and your child are ready! After mixing ISENTRESS, use it within 30 minutes. Throw away any leftover ISENTRESS after you have given the dose to your child. |
|
|
Take 1 packet of ISENTRESS and shake the powder to the bottom of the packet. |
| Tear or cut open the packet at the dotted line and add all of the powder to the water in the mixing cup. Make sure the packet is completely empty. |
Step 7. Mix ISENTRESS and water
| Snap the lid of the mixing cup shut. Gently swirl the mixing cup for 45 seconds in a circular motion to mix the powder and water. Use a clock or timer to time for 45 seconds. Do not shake the mixture. |
|
| Check to make sure the powder is mixed. If it is not mixed, swirl it some more. The mixture should look cloudy. |
Step 8. Check your prescription
Find the dose amount in 'mL' prescribed by your doctor. This is written on the prescription label on the box from your pharmacy.
Remember that the dose may change each time you go to the doctor, so make sure you check the prescribed dose each time. Be sure to go to all of your doctor's appointments so your child gets the right dose!
Step 9. Choose the syringe you need
Your doctor will prescribe the dose in milliliters (mL). Choose the right syringe for your child's dose:
|
WHITE
(1mL) for 1mL or less |
GREEN
(3mL) for 1.5mL to 3mL |
BLUE
(10mL) for 3.5mL to 10mL |
Then find the mL mark on the syringe that matches your child's dose.
Step 10. Measure ISENTRESS
| Push the plunger into the barrel of the syringe as far as it goes. | |
| Put the tip of the syringe into the cup of the mixed ISENTRESS and pull back on the plunger. Stop when you get to the line that matches your child's prescribed dose. |
IMPORTANT:
|
Step 11. Check for air bubbles
| Hold the syringe with tip up. Tap it with your finger to move any air bubbles up towards the tip. Slowly push the plunger to make the air come out. You may see some drops of medicine come out. |
|
| Re-check the amount of ISENTRESS in the syringe. If it is less than the prescribed dose, put the tip back into the cup of mixed ISENTRESS and pull back on the plunger until you get to the right dose mark. |
Step 12. Give ISENTRESS to your child
| Place the tip of the syringe inside your child's mouth so that it touches either the right or left cheek. |
Slowly push in the plunger to give the dose of mixed ISENTRESS to your child. If your child fusses, take the tip of the syringe out of the mouth and try again. It is important that your child takes all of the prescribed dose (a little left in the syringe tip is OK).
|
IMPORTANT:
If your child does not take all of the prescribed dose or spits some of it out, call your doctor to find out what to do. |
Step 13. Clean up
Pour the leftover mixed ISENTRESS into the trash.
Do not pour it into the sink.
Pull the plungers out of any syringes you used.
Hand wash the syringes, plungers, and mixing cup with warm water and dish soap. Do not wash in the dishwasher.
| Rinse with water and let air dry. Put everything in a clean, dry place. |
|
How should I store ISENTRESS?
Store the ISENTRESS for oral suspension kit at room temperature between 68°F to 77°F (20°C to 25°C).
Store in the original container.
Keep ISENTRESS and all medicines out of the reach of children.
| Be sure to keep your doctor's appointments so you always know how much ISENTRESS to give to your child. |
For more information, go to www.ISENTRESS.com or call 1-800-622-4477.
2.2 Adults
The recommended adult dosage of ISENTRESS film-coated tablets is displayed in Table 1. ISENTRESS and ISENTRESS HD should be taken by mouth and may be taken with or without food [see Clinical Pharmacology (12.3)].
| Population | Recommended Dose |
|---|---|
| Treatment-naïve patients or patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily | 1200 mg (2 × 600 mg) once daily or 400 mg twice daily |
| Treatment-experienced | 400 mg twice daily |
| Treatment-naïve or treatment-experienced when coadministered with rifampin [see Drug Interactions (7.1)] | 800 mg (2 × 400 mg) twice daily |
10 Overdosage (10 OVERDOSAGE)
In the event of an overdose, it is reasonable to employ the standard supportive measures, e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring (including obtaining an electrocardiogram), and institute supportive therapy if required. The extent to which ISENTRESS may be dialyzable is unknown.
11 Description (11 DESCRIPTION)
ISENTRESS contains raltegravir potassium, a human immunodeficiency virus integrase strand transfer inhibitor. The chemical name for raltegravir potassium is N-[(4-Fluorophenyl) methyl]-1,6-dihydro-5-hydroxy-1-methyl-2-[1-methyl-1-[[(5-methyl-1,3,4-oxadiazol-2-yl)carbonyl]amino]ethyl]-6-oxo-4-pyrimidinecarboxamide monopotassium salt.
The empirical formula is C20H20FKN6O5 and the molecular weight is 482.51. The structural formula is:
Raltegravir potassium is a white to off-white powder. It is soluble in water, slightly soluble in methanol, very slightly soluble in ethanol and acetonitrile and insoluble in isopropanol.
Each 400 mg film-coated tablet of ISENTRESS for oral administration contains 434.4 mg of raltegravir (as potassium salt), equivalent to 400 mg of raltegravir free phenol and the following inactive ingredients: calcium phosphate dibasic anhydrous, hypromellose 2208, lactose monohydrate, magnesium stearate, microcrystalline cellulose, poloxamer 407 (contains 0.01% butylated hydroxytoluene as antioxidant), sodium stearyl fumarate. In addition, the film coating contains the following inactive ingredients: black iron oxide, polyethylene glycol 3350, polyvinyl alcohol, red iron oxide, talc and titanium dioxide.
Each 600 mg film-coated tablet of ISENTRESS HD for oral administration contains 651.6 mg of raltegravir (as potassium salt), equivalent to 600 mg of raltegravir free phenol and the following inactive ingredients: croscarmellose sodium, hypromellose 2910, magnesium stearate, microcrystalline cellulose. The film coating contains the following inactive ingredients: ferrosoferric oxide, hypromellose 2910, iron oxide yellow, lactose monohydrate, triacetin and titanium dioxide. The tablet may also contain trace amount of carnauba wax.
Each 100 mg chewable tablet of ISENTRESS for oral administration contains 108.6 mg of raltegravir (as potassium salt), equivalent to 100 mg of raltegravir free phenol and the following inactive ingredients: ammonium hydroxide, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, magnesium stearate, mannitol, medium chain triglycerides, monoammonium glycyrrhizinate, natural and artificial flavors (orange, banana, and masking that contains aspartame), oleic acid, PEG 400, red iron oxide, saccharin sodium, sodium citrate dihydrate, sodium stearyl fumarate, sorbitol, sucralose and yellow iron oxide.
Each 25 mg chewable tablet of ISENTRESS for oral administration contains 27.16 mg of raltegravir (as potassium salt), equivalent to 25 mg of raltegravir free phenol and the following inactive ingredients: ammonium hydroxide, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, magnesium stearate, mannitol, medium chain triglycerides, monoammonium glycyrrhizinate, natural and artificial flavors (orange, banana, and masking that contains aspartame), oleic acid, PEG 400, saccharin sodium, sodium citrate dihydrate, sodium stearyl fumarate, sorbitol, sucralose and yellow iron oxide.
Each packet of ISENTRESS for oral suspension 100 mg, contains 108.6 mg of raltegravir (as potassium salt), equivalent to 100 mg of raltegravir free phenol and the following inactive ingredients: ammonium hydroxide, banana with other natural flavors, carboxymethylcellulose sodium, crospovidone, ethylcellulose 20 cP, fructose, hydroxypropyl cellulose, hypromellose 2910/6cP, macrogol/PEG 400, magnesium stearate, maltodextrin, mannitol, medium chain triglycerides, microcrystalline cellulose, monoammonium glycyrrhizinate, oleic acid, sorbitol, sucralose and sucrose.
2.3 Pediatrics
The recommended pediatric dosage of ISENTRESS is displayed in Table 2. ISENTRESS film-coated tablets, chewable tablets and for oral suspension should be taken by mouth and may be taken with or without food [see Clinical Pharmacology (12.3)].
| Recommended Pediatric Dosage and Formulation | ||||
|---|---|---|---|---|
| Population/Weight | Film-Coated Tablets 400 mg | Film-Coated Tablets 600 mg | Chewable Tablets 100 mg and 25 mg | For Oral Suspension 100 mg |
If at least 40 kg and either:
|
400 mg twice daily | 1200 mg (2 × 600 mg) once daily | 300 mg twice daily (see Table 3) | NA |
| If at least 25 kg | 400 mg twice daily If able to swallow a tablet
|
NA | Weight-based dosing twice daily (see Table 3) | NA |
| If at least 4 weeks of age and weighing 3 kg to less than 25 kg | NA | NA | Weight-based dosing twice daily (see Table 4) | Weight-based dosing twice daily up to 20 kg (see Table 4) |
| From birth to 4 weeks (28 days) weighing at least 2 kg | NA | NA | NA | Weight-based dosing once daily or twice daily (see Table 5) |
| Body Weight (kg) |
Dose | Number of 100 mg Chewable Tablets |
|---|---|---|
| 25 to less than 28 | 150 mg twice daily | 1.5 × 100 mg The 100 mg chewable tablet can be divided into equal halves. twice daily |
| 28 to less than 40 | 200 mg twice daily | 2 × 100 mg twice daily |
| At least 40 | 300 mg twice daily | 3 × 100 mg twice daily |
| Body Weight (kg) |
Volume (Dose) of Suspension to be Administered | Number of Chewable Tablets The chewable tablets are available as 25 mg and 100 mg tablets.
|
|---|---|---|
| 3 to less than 4 | 2.5 mL (25 mg) twice daily | 1 × 25 mg twice daily May be administered as a crushed tablet(s); see
General Dosing Recommendations (2.1)
for guidance.
|
| 4 to less than 6 | 3 mL (30 mg) twice daily | |
| 6 to less than 8 | 4 mL (40 mg) twice daily | 2 × 25 mg twice daily |
| 8 to less than 10 | 6 mL (60 mg) twice daily | |
| 10 to less than 14 | 8 mL (80 mg) twice daily | 3 × 25 mg twice daily |
| 14 to less than 20 | 10 mL (100 mg) twice daily | 1 × 100 mg twice daily |
| 20 to less than 25 | Not applicable | 1.5 × 100 mg The 100 mg chewable tablet can be divided into equal halves. twice daily |
- For full-term neonates (birth to 4 weeks [28 days] of age): Weight-based dosing of the oral suspension as specified in Table 5.
- No data are available in pre-term neonates. The use of ISENTRESS is not recommended in pre-term neonates.
| Body Weight (kg) |
Volume (Dose) of Suspension to be Administered |
|---|---|
| Note: If the mother has taken ISENTRESS or ISENTRESS HD 2-24 hours before delivery, the neonate's first dose should be given between 24-48 hours after birth. | |
|
Birth to 1 Week - Once daily dosing The dosing recommendations are based on approximately 1.5 mg/kg/dose.
|
|
| 2 to less than 3 | 0.4 mL (4 mg) once daily |
| 3 to less than 4 | 0.5 mL (5 mg) once daily |
| 4 to less than 5 | 0.7 mL (7 mg) once daily |
|
1 to 4 Weeks - Twice daily dosing The dosing recommendations are based on approximately 3 mg/kg/dose.
|
|
| 2 to less than 3 | 0.8 mL (8 mg) twice daily |
| 3 to less than 4 | 1 mL (10 mg) twice daily |
| 4 to less than 5 | 1.5 mL (15 mg) twice daily |
8.5 Geriatric Use
Clinical studies of ISENTRESS/ISENTRESS HD did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
4 Contraindications (4 CONTRAINDICATIONS)
None
6 Adverse Reactions (6 ADVERSE REACTIONS)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
7 Drug Interactions (7 DRUG INTERACTIONS)
- Coadministration of ISENTRESS or ISENTRESS HD and other drugs may alter the plasma concentration of raltegravir. The potential for drug-drug interactions must be considered prior to and during therapy (7).
- Coadministration of ISENTRESS or ISENTRESS HD with drugs that are strong inducers of UGT1A1, such as rifampin, may result in reduced plasma concentrations of raltegravir (2.1, 7.1).
5.3 Phenylketonurics
ISENTRESS Chewable Tablets contain phenylalanine, a component of aspartame. Each 25 mg ISENTRESS Chewable Tablet contains approximately 0.05 mg phenylalanine. Each 100 mg ISENTRESS Chewable Tablet contains approximately 0.10 mg phenylalanine. Phenylalanine can be harmful to patients with phenylketonuria.
12.2 Pharmacodynamics
In a monotherapy study raltegravir (400 mg twice daily) demonstrated rapid antiviral activity with mean viral load reduction of 1.66 log10 copies/mL by day 10.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Adult Patients:
ISENTRESS and ISENTRESS HD are human immunodeficiency virus integrase strand transfer inhibitors (HIV-1 INSTI) indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in adult patients (1).
Pediatric Patients:
ISENTRESS is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 2 kg (1).
ISENTRESS HD is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection in pediatric patients weighing at least 40 kg (1).
12.1 Mechanism of Action
Raltegravir is an HIV-1 antiviral drug [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Severe, potentially life-threatening and fatal skin reactions have been reported. This includes cases of Stevens-Johnson syndrome, hypersensitivity reaction and toxic epidermal necrolysis. Immediately discontinue treatment with ISENTRESS or ISENTRESS HD and other suspect agents if severe hypersensitivity, severe rash, or rash with systemic symptoms or liver aminotransferase elevations develops and monitor clinical status, including liver aminotransferases closely (5.1).
- Monitor for Immune Reconstitution Syndrome (5.2).
- Inform patients with phenylketonuria that the 100 mg and 25 mg chewable tablets contain phenylalanine (5.3).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
ISENTRESS and ISENTRESS HD can be administered with or without food (2.1).
Do not substitute ISENTRESS chewable tablets or ISENTRESS for oral suspension for the ISENTRESS 400 mg or 600 mg film-coated tablet.
See specific dosing guidance for chewable tablets and the formulation for oral suspension (2.1).
Adults
- Treatment-naïve patients or patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily:
- 1200 mg (2 × 600 mg) film-coated tablet orally, once daily or
- 400 mg film-coated tablet orally, twice daily (2.2).
- Treatment-experienced patients:
- 400 mg film-coated tablet orally, twice daily (2.2).
- During coadministration with rifampin in adults, 800 mg (2 × 400 mg) twice daily (2.2).
Pediatrics
- If weighing at least 40 kg, and either
- treatment-naïve patients or
- patients who are virologically suppressed on an initial regimen of ISENTRESS 400 mg twice daily:
- 1200 mg (2 × 600 mg) film-coated tablet orally, once daily or
- 400 mg film-coated tablet orally, twice daily or
- 300 mg (3 × 100 mg) chewable tablets, twice daily (2.3).
- If weighing at least 25 kg: One 400 mg film-coated tablet orally, twice daily. If unable to swallow a tablet, consider the chewable tablet, as specified in Table 2 (2.3).
- If weighing at least 3 kg to less than 25 kg: Weight-based dosing using the chewable tablet or oral suspension, as specified in Table 4 (2.3).
- For neonates (birth to 4 weeks [28 days] of age): Weight-based dosing of the oral suspension as specified in Table 5 (2.3).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Film-coated Tablets
- 400 mg pink, oval-shaped, film-coated tablets with "227" on one side (ISENTRESS).
- 600 mg yellow, oval-shaped, film-coated tablets with corporate logo and "242" on one side and plain on the other side (ISENTRESS HD).
- Chewable Tablets
- 100 mg pale orange, oval-shaped, orange-banana flavored, chewable tablets scored on both sides and imprinted on one face with the corporate logo and "477" on opposite sides of the score.
- 25 mg pale yellow, round, orange-banana flavored, chewable tablets with the corporate logo on one side and "473" on the other side.
- For Oral Suspension
- 100 mg white to off-white, banana flavored, granular powder that may contain yellow or beige to tan particles in a child resistant single-use foil packet.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ISENTRESS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: thrombocytopenia
Gastrointestinal Disorders: diarrhea
Hepatobiliary Disorders: hepatic failure (with and without associated hypersensitivity) in patients with underlying liver disease and/or concomitant medications
Musculoskeletal and Connective Tissue Disorders: rhabdomyolysis
Nervous System Disorders: cerebellar ataxia
Psychiatric Disorders: anxiety, paranoia
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Women infected with HIV should be instructed not to breastfeed due to the potential for HIV transmission (8.2).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
2.1 General Dosing Recommendations
- Because the formulations have different pharmacokinetic profiles, do not substitute ISENTRESS chewable tablets or ISENTRESS for oral suspension for the ISENTRESS 400 mg film-coated tablet or the ISENTRESS HD 600 mg film-coated tablet. See specific dosing guidance for chewable tablets and the formulation for oral suspension.
- Because the extent to which ISENTRESS may be dialyzable is unknown, dosing before a dialysis session should be avoided [see Clinical Pharmacology (12.3)].
- ISENTRESS film-coated tablets must be swallowed whole.
- ISENTRESS chewable tablets may be chewed or swallowed whole. Maximum daily dose is 300 mg taken by mouth twice daily.
- For children who have difficulty chewing the 25 mg chewable tablet, the tablet may be crushed.
- Preparation of the crushed 25 mg chewable tablet:
- Place the tablet(s) in a small, clean cup. For each tablet, add a teaspoonful (~5 mL) of liquid (for example, water, juice, or breast milk).
- Within 2 minutes, the tablet(s) will absorb the liquid and fall apart.
- Using a spoon, crush any remaining pieces of the tablet(s). Immediately administer the entire dose orally.
- If any portion of the dose is left in the cup, add another teaspoonful (~5 mL) of liquid, swirl and administer immediately.
- Preparation of the crushed 25 mg chewable tablet:
- ISENTRESS for oral suspension:
- See Instructions for Use for details on preparation and administration of ISENTRESS for oral suspension.
- Using the provided mixing cup, combine 10 mL of water and the entire contents of one packet of ISENTRESS for oral suspension and mix. Each single-use packet for oral suspension contains 100 mg of raltegravir which is suspended in 10 mL of water giving a final concentration of 10 mg per mL. Maximum daily dose is 100 mg taken by mouth twice daily.
- Gently swirl the mixing cup for 45 seconds in a circular motion to mix the powder into a uniform suspension. Do not shake.
- Once mixed, measure the prescribed dose volume of suspension with a syringe and administer the dose orally. The dose should be administered orally within 30 minutes of mixing.
- Discard any remaining suspension into the trash.
5.2 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including ISENTRESS. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jiroveci pneumonia, tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of treatment.
14.1 Description of Clinical Studies
The evidence of durable efficacy of ISENTRESS 400 mg twice daily is based on the analyses of 240-week data from a randomized, double-blind, active-controlled trial, STARTMRK evaluating ISENTRESS 400 mg twice daily in antiretroviral treatment-naïve HIV-1 infected adult subjects, the analysis of 96-week data from a randomized, double-blind, active-control trial, ONCEMRK evaluating ISENTRESS HD 1200 mg (2 × 600 mg) once daily in treatment-naïve adult subjects, and 96-week data from 2 randomized, double-blind, placebo-controlled studies, BENCHMRK 1 and BENCHMRK 2, evaluating ISENTRESS 400 mg twice daily in antiretroviral treatment-experienced HIV-1 infected adult subjects. See Table 18.
| Trial | Study Type | Population | Study Arms (N) | Dose/Formulation | Timepoint |
|---|---|---|---|---|---|
| STARTMRK | Randomized, double-blind, active-controlled | Treatment-Naïve Adults | ISENTRESS 400 mg Twice Daily (281) Efavirenz 600 mg At Bedtime (282) Both in combination with emtricitabine (+) tenofovir disoproxil fumarate |
400 mg film-coated tablet | Week 240 |
| ONCEMRK | Randomized, double-blind, active-controlled | Treatment-Naïve Adults | ISENTRESS HD 1200 mg Once Daily (531) | 600 mg film-coated tablet | Week 96 |
| ISENTRESS 400 mg Twice Daily (266) Both in combination with emtricitabine (+) tenofovir disoproxil fumarate |
400 mg film-coated tablet | ||||
| BENCHMRK 1 | Randomized, double-blind, placebo-controlled | Treatment-Experienced Adults | ISENTRESS 400 mg Twice Daily (232) Placebo (118) Both in combination with optimized background therapy |
400 mg film-coated tablet | Week 240 (Week 156 on double-blind plus Week 84 on open-label) |
| BENCHMRK 2 | Randomized, double-blind, placebo-controlled | Treatment-Experienced Adults | ISENTRESS 400 mg Twice Daily (230) Placebo (119) Both in combination with optimized background therapy |
400 mg film-coated tablet | Week 240 (Week 156 on double-blind plus Week 84 on open-label) |
| IMPAACT P1066 | Open-label, non-comparative | Pediatric Patients – 4 weeks to 18 years of age (Treatment-Experienced or Failed Prior PMTCT) |
ISENTRESS 400 mg tablet Twice Daily – 12 to 18 years or 6 to <12 years and ≥25 kg (87) ISENTRESS chewable tablet- Weight-Based Dose to Approximate 6 mg/kg Twice Daily – 2 to <12 years (39) ISENTRESS for oral suspension- Weight-Based Dose to Approximate 6 mg/kg Twice Daily – 4 weeks to <2 years (26) In combination with optimized background therapy |
400 mg film-coated tablet 25 mg and 100 mg chewable tablet 100 mg sachet for oral suspension |
Week 240 |
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ISENTRESS tablets 400 mg are pink, oval-shaped, film-coated tablets with "227" on one side. They are supplied as follows:
-
NDC 0006-0227-61 unit-of-use bottles of 60.
ISENTRESS HD tablets 600 mg are yellow, oval-shaped, film-coated tablets with corporate logo and "242" on one side and plain on the other side. They are supplied as follows:
-
NDC 0006-3080-01 unit-of-use bottles of 60.
ISENTRESS tablets 100 mg are pale orange, oval-shaped, orange-banana flavored, chewable tablets scored on both sides and imprinted on one face with the corporate logo and "477" on opposite sides of the score. They are supplied as follows:
-
NDC 0006-0477-61 unit-of-use bottles of 60.
ISENTRESS tablets 25 mg are pale yellow, round, orange-banana flavored, chewable tablets with the corporate logo on one side and "473" on the other side. They are supplied as follows:
-
NDC 0006-0473-61 unit-of-use bottles of 60.
ISENTRESS for oral suspension 100 mg is a white to off-white granular powder that may contain yellow or beige to tan particles, in child resistant single-use foil packets, packaged as
A kit with two 1 mL syringes, two 3 mL syringes, two 10 mL syringes and two mixing cups. It is supplied as follows:
-
NDC 0006-3603-61 unit of use carton with 60 packets.
-
NDC 0006-3603-01 individual packet.
14.3 Treatment Experienced Adult Subjects (14.3 Treatment-Experienced Adult Subjects)
BENCHMRK 1 and BENCHMRK 2 are Phase 3 studies to evaluate the safety and antiretroviral activity of ISENTRESS 400 mg twice daily in combination with an optimized background therapy (OBT), versus OBT alone, in HIV-1-infected subjects, 16 years or older, with documented resistance to at least 1 drug in each of 3 classes (NNRTIs, NRTIs, PIs) of antiretroviral therapies. Randomization was stratified by degree of resistance to PI (1PI vs. >1PI) and the use of enfuvirtide in the OBT. Prior to randomization, OBT was selected by the investigator based on genotypic/phenotypic resistance testing and prior ART history.
Table 20 shows the demographic characteristics of subjects in the group receiving ISENTRESS 400 mg twice daily and subjects in the placebo group.
| Randomized Studies BENCHMRK 1 and BENCHMRK 2 |
ISENTRESS 400 mg Twice Daily + OBT (N = 462) |
Placebo + OBT (N = 237) |
|---|---|---|
| Gender | ||
| Male | 88% | 89% |
| Female | 12% | 11% |
| Race | ||
| White | 65% | 73% |
| Black | 14% | 11% |
| Asian | 3% | 3% |
| Hispanic | 11% | 8% |
| Others | 6% | 5% |
| Age (years) | ||
| Median (min, max) | 45 (16 to 74) | 45 (17 to 70) |
| CD4+ Cell Count | ||
| Median (min, max), cells/mm3 | 119 (1 to 792) | 123 (0 to 759) |
| ≤50 cells/mm3 | 32% | 33% |
| >50 and ≤200 cells/mm3 | 37% | 36% |
| Plasma HIV-1 RNA | ||
| Median (min, max), log10 copies/mL | 4.8 (2 to 6) | 4.7 (2 to 6) |
| >100,000 copies/mL | 36% | 33% |
| History of AIDS | ||
| Yes | 92% | 91% |
| Prior Use of ART, Median (1st Quartile, 3rd Quartile) | ||
| Years of ART Use | 10 (7 to 12) | 10 (8 to 12) |
| Number of ART | 12 (9 to 15) | 12 (9 to 14) |
|
Hepatitis Co-infection Hepatitis B virus surface antigen positive or hepatitis C virus antibody positive.
|
||
| No Hepatitis B or C virus | 83% | 84% |
| Hepatitis B virus only | 8% | 3% |
| Hepatitis C virus only | 8% | 12% |
| Co-infection of Hepatitis B and C virus | 1% | 1% |
| Stratum | ||
| Enfuvirtide in OBT | 38% | 38% |
| Resistant to ≥2 PI | 97% | 95% |
Table 21 compares the characteristics of optimized background therapy at baseline in the group receiving ISENTRESS 400 mg twice daily and subjects in the control group.
| Randomized Studies BENCHMRK 1 and BENCHMRK 2 |
ISENTRESS 400 mg Twice Daily + OBT (N = 462) |
Placebo + OBT (N = 237) |
|---|---|---|
| Number of ARTs in OBT | ||
| Median (min, max) | 4 (1 to 7) | 4 (2 to 7) |
|
Number of Active PI in OBT by Phenotypic Resistance Test Darunavir use in OBT in darunavir-naïve subjects was counted as one active PI.
|
||
| 0 | 36% | 41% |
| 1 or more | 60% | 58% |
|
Phenotypic Sensitivity Score (PSS) The Phenotypic Sensitivity Score (PSS) and the Genotypic Sensitivity Score (GSS) were defined as the total oral ARTs in OBT to which a subject's viral isolate showed phenotypic sensitivity and genotypic sensitivity, respectively, based upon phenotypic and genotypic resistance tests. Enfuvirtide use in OBT in enfuvirtide-naïve subjects was counted as one active drug in OBT in the GSS and PSS. Similarly, darunavir use in OBT in darunavir-naïve subjects was counted as one active drug in OBT.
|
||
| 0 | 15% | 18% |
| 1 | 31% | 30% |
| 2 | 31% | 28% |
| 3 or more | 18% | 20% |
| Genotypic Sensitivity Score (GSS) | ||
| 0 | 25% | 27% |
| 1 | 38% | 40% |
| 2 | 24% | 21% |
| 3 or more | 11% | 10% |
Week 96 outcomes for the 699 subjects randomized and treated with the recommended dose of ISENTRESS 400 mg twice daily or placebo in the pooled BENCHMRK 1 and 2 studies are shown in Table 22.
| ISENTRESS 400 mg Twice Daily + OBT (N = 462) |
Placebo + OBT (N = 237) |
|
|---|---|---|
| Subjects with HIV-1 RNA less than 50 copies/mL | 55% | 27% |
|
Virologic Failure Includes subjects who switched to open-label raltegravir after Week 16 due to the protocol-defined virologic failure, subjects who discontinued prior to Week 96 for lack of efficacy, subjects changed OBT due to lack of efficacy prior to Week 96, or subjects who were ≥50 copies in the 96 week window.
|
35% | 66% |
| No virologic data at Week 96 Window | ||
| Reasons | ||
| Discontinued study due to AE or death Includes subjects who discontinued due to AE or death at any time point from day 1 through the Week 96 window if this resulted in no virologic data on treatment during the Week 96 window.
|
3% | 3% |
| Discontinued study for other reasons Other includes: withdrew consent, loss to follow-up, moved etc., if the viral load at the time of discontinuation was <50 copies/mL.
|
4% | 4% |
| Missing data during window but on study | 4% | <1% |
The mean changes in CD4 count from baseline were 118 cells/mm3 in the group receiving ISENTRESS 400 mg twice daily and 47 cells/mm3 for the control group.
Treatment-emergent CDC Category C events occurred in 4% of the group receiving ISENTRESS 400 mg twice daily and 5% of the control group.
Virologic responses at Week 96 by baseline genotypic and phenotypic sensitivity score are shown in Table 23.
| Percent with HIV-1 RNA <50 copies/mL At Week 96 | ||||
|---|---|---|---|---|
| n | ISENTRESS 400 mg Twice Daily + OBT (N = 462) |
n | Placebo + OBT (N = 237) |
|
|
Phenotypic Sensitivity Score (PSS) The Phenotypic Sensitivity Score (PSS) and the Genotypic Sensitivity Score (GSS) were defined as the total oral ARTs in OBT to which a subject's viral isolate showed phenotypic sensitivity and genotypic sensitivity, respectively, based upon phenotypic and genotypic resistance tests. Enfuvirtide use in OBT in enfuvirtide-naïve subjects was counted as one active drug in OBT in the GSS and PSS. Similarly, darunavir use in OBT in darunavir-naïve subjects was counted as one active drug in OBT.
|
||||
| 0 | 67 | 43 | 43 | 5 |
| 1 | 144 | 58 | 71 | 23 |
| 2 | 142 | 61 | 66 | 32 |
| 3 or more | 85 | 48 | 48 | 42 |
| Genotypic Sensitivity Score (GSS) | ||||
| 0 | 116 | 39 | 65 | 5 |
| 1 | 177 | 62 | 95 | 26 |
| 2 | 111 | 61 | 49 | 53 |
| 3 or more | 51 | 49 | 23 | 35 |
8.7 Use in Patients With Renal Impairment (8.7 Use in Patients with Renal Impairment)
No dosage adjustment of ISENTRESS or ISENTRESS HD is necessary in patients with any degree of renal impairment [see Clinical Pharmacology (12.3)]. The extent to which ISENTRESS may be dialyzable is unknown; therefore, dosing before a dialysis session should be avoided.
8.6 Use in Patients With Hepatic Impairment (8.6 Use in Patients with Hepatic Impairment)
No dosage adjustment of ISENTRESS is necessary for patients with mild to moderate (Child-Pugh A and B) hepatic impairment. No hepatic impairment study has been conducted with ISENTRESS HD and therefore administration in subjects with hepatic impairment is not recommended. The effect of severe hepatic impairment on the pharmacokinetics of raltegravir has not been studied [see Clinical Pharmacology (12.3)].
Principal Display Panel 600 Mg Bottle Label (PRINCIPAL DISPLAY PANEL - 600 mg Bottle Label)
NDC 0006-3080-01
Isentress
®
HD
(raltegravir) tablets
600 mg
RECOMMENDED DOSAGE: Two tablets once daily.
See Package Insert.
Each tablet contains 651.6 mg raltegravir
potassium, equivalent to
600 mg raltegravir.
Rx only
60 Tablets
5.1 Severe Skin and Hypersensitivity Reactions
Severe, potentially life-threatening, and fatal skin reactions have been reported. These include cases of Stevens-Johnson syndrome and toxic epidermal necrolysis. Hypersensitivity reactions have also been reported and were characterized by rash, constitutional findings, and sometimes, organ dysfunction, including hepatic failure. Discontinue ISENTRESS or ISENTRESS HD and other suspect agents immediately if signs or symptoms of severe skin reactions or hypersensitivity reactions develop (including, but not limited to, severe rash or rash accompanied by fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, facial edema, hepatitis, eosinophilia, angioedema). Clinical status including liver aminotransferases should be monitored and appropriate therapy initiated. Delay in stopping ISENTRESS or ISENTRESS HD treatment or other suspect agents after the onset of severe rash may result in a life-threatening reaction.
Principal Display Panel 25 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label)
NDC 0006-0473-61
Isentress®
(raltegravir) CHEWABLE Tablets
25 mg
For Pediatric Use
Phenylketonurics:
contains phenylalanine (a component
of aspartame) 0.05 mg per 25 mg chewable tablet.
Rx only
60 Tablets
Principal Display Panel 100 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label)
NDC 0006-0477-61
Isentress®
(raltegravir) CHEWABLE Tablets
100 mg
For Pediatric Use
Phenylketonurics:
contains phenylalanine (a component
of aspartame) 0.10 mg per 100 mg chewable tablet.
Rx only
60 Tablets
Principal Display Panel 400 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 400 mg Tablet Bottle Label)
NDC 0006-0227-61
Isentress®
(raltegravir) tablets
400 mg
Each tablet contains 434.4 mg raltegravir
potassium, equivalent to 400 mg raltegravir.
USUAL DOSAGE: See Package Insert.
Rx only
60 Tablets
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies of raltegravir in mice did not show any carcinogenic potential. At the highest dose levels, 400 mg/kg/day in females and 250 mg/kg/day in males, systemic exposure was 1.8-fold (females) or 1.2-fold (males) greater than the AUC (54 µM∙hr) at the 400-mg twice daily human dose. Treatment-related squamous cell carcinoma of nose/nasopharynx was observed in female rats dosed with 600 mg/kg/day raltegravir for 104 weeks. These tumors were possibly the result of local irritation and inflammation due to local deposition and/or aspiration of drug in the mucosa of the nose/nasopharynx during dosing. No tumors of the nose/nasopharynx were observed in rats dosed with 150 mg/kg/day (males) and 50 mg/kg/day (females) and the systemic exposure in rats was 1.7-fold (males) to 1.4-fold (females) greater than the AUC (54 µM∙hr) at the 400-mg twice daily human dose.
No evidence of mutagenicity or genotoxicity was observed in in vitro microbial mutagenesis (Ames) tests, in vitro alkaline elution assays for DNA breakage, and in vitro and in vivo chromosomal aberration studies.
No effect on fertility was seen in male and female rats at doses up to 600 mg/kg/day which resulted in a 3-fold exposure above the exposure at the recommended human dose.
7.1 Effect of Other Agents On the Pharmacokinetics of Raltegravir (7.1 Effect of Other Agents on the Pharmacokinetics of Raltegravir)
Raltegravir is not a substrate of cytochrome P450 (CYP) enzymes. Based on in vivo and in vitro studies, raltegravir is eliminated mainly by metabolism via a UGT1A1-mediated glucuronidation pathway. Coadministration of ISENTRESS with drugs that inhibit UGT1A1 may increase plasma levels of raltegravir and coadministration of ISENTRESS with drugs that induce UGT1A1, such as rifampin, may reduce plasma levels of raltegravir (see Table 11).
Selected drug interactions are presented in Table 11 [see Clinical Pharmacology (12.3)]. In some cases, recommendations differ for ISENTRESS versus ISENTRESS HD.
| Concomitant Drug Class: Drug Name |
Effect on Concentration of Raltegravir | Clinical Comment for ISENTRESS | Clinical Comment for ISENTRESS HD |
|---|---|---|---|
| Metal-Containing Antacids | |||
| Aluminum and/or magnesium-containing antacids | ↓ | Coadministration or staggered administration is not recommended. | |
| Calcium carbonate antacid | ↓ | No dose adjustment | Co-administration is not recommended |
| Other Agents | |||
| Rifampin | ↓ | The recommended dosage is 800 mg twice daily during coadministration with rifampin. There are no data to guide co-administration of ISENTRESS with rifampin in patients below 18 years of age [see Dosage and Administration (2.1)]. | Coadministration is not recommended. |
| Tipranavir/ritonavir | No dose adjustment | Coadministration is not recommended | |
| Etravirine | ↓ | No dose adjustment | Coadministration is not recommended. |
| Strong inducers of drug metabolizing enzymes not mentioned above e.g., Carbamazepine Phenobarbital Phenytoin |
↓↔ | The impact of other strong inducers of drug metabolizing enzymes on raltegravir is unknown. Coadministration is not recommended. |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:32.153263 · Updated: 2026-03-14T22:46:59.478961