These Highlights Do Not Include All The Information Needed To Use Nexletol®
88d06d89-a3da-40b4-b273-8f4f7d56c4c9
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 11/2025
Indications and Usage
NEXLETOL is indicated: to reduce the risk of major adverse cardiovascular events (cardiovascular death, myocardial infarction, stroke, or coronary revascularization) in adults at increased risk for these events who are unable to take recommended statin therapy (including those not taking a statin). as an adjunct to diet and exercise, in combination with other low-density lipoprotein cholesterol (LDL-C) lowering therapies, or alone when concomitant LDL-C lowering therapy is not possible, to reduce LDL-C in adults with hypercholesterolemia, including heterozygous familial hypercholesterolemia (HeFH).
Dosage and Administration
Administer 180 mg orally once daily with or without food. ( 2.1 )
Warnings and Precautions
Hyperuricemia: Elevations in serum uric acid have occurred. Assess uric acid levels periodically as clinically indicated. Monitor for signs and symptoms of hyperuricemia, and initiate treatment with urate-lowering drugs as appropriate. ( 5.1 ) Tendon Rupture: Tendon rupture has occurred. Discontinue NEXLETOL at the first sign of tendon rupture. Avoid NEXLETOL in patients who have a history of tendon disorders or tendon rupture. ( 5.2 )
Contraindications
NEXLETOL is contraindicated in patients with a prior serious hypersensitivity reaction to bempedoic acid or any of the excipients in NEXLETOL. Serious hypersensitivity reactions, such as angioedema, have occurred [see Adverse Reactions (6.2) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hyperuricemia [see Warnings and Precautions (5.1) ] Tendon Rupture [see Warnings and Precautions (5.2) ]
Drug Interactions
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with NEXLETOL and instructions for preventing or managing them. Table 3. Clinically Important Drug Interactions with NEXLETOL Simvastatin Clinical Impact: Concomitant use of NEXLETOL with simvastatin causes an increase in simvastatin concentration and may increase the risk of simvastatin-related myopathy [see Clinical Pharmacology (12.3) ] . Intervention : Avoid concomitant use of NEXLETOL with simvastatin greater than 20 mg. Pravastatin Clinical Impact: Concomitant use of NEXLETOL with pravastatin causes an increase in pravastatin concentration and may increase the risk of pravastatin-related myopathy [see Clinical Pharmacology (12.3) ] . Intervention: Avoid concomitant use of NEXLETOL with pravastatin greater than 40 mg. Fibrates Clinical Impact: Concomitant administration of fibrates with NEXLETOL resulted in increased triglycerides and decreased high-density lipoprotein cholesterol (HDL-C) in some patients in clinical studies and post-marketing reports. Reversibility of both increased triglycerides and decreased HDL-C levels was observed when either NEXLETOL or fibrate therapy was discontinued. Intervention: Monitor triglycerides and HDL-C four weeks after initial concomitant use of NEXLETOL and a fibrate and periodically thereafter. If increased triglycerides or decreased HDL-C levels are detected, discontinue NEXLETOL or fibrate therapy based on clinical judgment. Monitor triglycerides and HDL-C levels until levels return to baseline.
Medication Information
Warnings and Precautions
Hyperuricemia: Elevations in serum uric acid have occurred. Assess uric acid levels periodically as clinically indicated. Monitor for signs and symptoms of hyperuricemia, and initiate treatment with urate-lowering drugs as appropriate. ( 5.1 ) Tendon Rupture: Tendon rupture has occurred. Discontinue NEXLETOL at the first sign of tendon rupture. Avoid NEXLETOL in patients who have a history of tendon disorders or tendon rupture. ( 5.2 )
Indications and Usage
NEXLETOL is indicated: to reduce the risk of major adverse cardiovascular events (cardiovascular death, myocardial infarction, stroke, or coronary revascularization) in adults at increased risk for these events who are unable to take recommended statin therapy (including those not taking a statin). as an adjunct to diet and exercise, in combination with other low-density lipoprotein cholesterol (LDL-C) lowering therapies, or alone when concomitant LDL-C lowering therapy is not possible, to reduce LDL-C in adults with hypercholesterolemia, including heterozygous familial hypercholesterolemia (HeFH).
Dosage and Administration
Administer 180 mg orally once daily with or without food. ( 2.1 )
Contraindications
NEXLETOL is contraindicated in patients with a prior serious hypersensitivity reaction to bempedoic acid or any of the excipients in NEXLETOL. Serious hypersensitivity reactions, such as angioedema, have occurred [see Adverse Reactions (6.2) ].
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hyperuricemia [see Warnings and Precautions (5.1) ] Tendon Rupture [see Warnings and Precautions (5.2) ]
Drug Interactions
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with NEXLETOL and instructions for preventing or managing them. Table 3. Clinically Important Drug Interactions with NEXLETOL Simvastatin Clinical Impact: Concomitant use of NEXLETOL with simvastatin causes an increase in simvastatin concentration and may increase the risk of simvastatin-related myopathy [see Clinical Pharmacology (12.3) ] . Intervention : Avoid concomitant use of NEXLETOL with simvastatin greater than 20 mg. Pravastatin Clinical Impact: Concomitant use of NEXLETOL with pravastatin causes an increase in pravastatin concentration and may increase the risk of pravastatin-related myopathy [see Clinical Pharmacology (12.3) ] . Intervention: Avoid concomitant use of NEXLETOL with pravastatin greater than 40 mg. Fibrates Clinical Impact: Concomitant administration of fibrates with NEXLETOL resulted in increased triglycerides and decreased high-density lipoprotein cholesterol (HDL-C) in some patients in clinical studies and post-marketing reports. Reversibility of both increased triglycerides and decreased HDL-C levels was observed when either NEXLETOL or fibrate therapy was discontinued. Intervention: Monitor triglycerides and HDL-C four weeks after initial concomitant use of NEXLETOL and a fibrate and periodically thereafter. If increased triglycerides or decreased HDL-C levels are detected, discontinue NEXLETOL or fibrate therapy based on clinical judgment. Monitor triglycerides and HDL-C levels until levels return to baseline.
Description
Indications and Usage ( 1 ) 11/2025
Section 42229-5
Other Adverse Reactions
Section 42230-3
| PATIENT INFORMATION NEXLETOL® (NEX-le-tol) (bempedoic acid) tablets, for oral use |
|||||
|---|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: 01/2026 | ||||
|
What is NEXLETOL?
NEXLETOL is a prescription medicine used:
|
|||||
| Do not take NEXLETOL if you are allergic to bempedoic acid or any of the ingredients in NEXLETOL. See the end of this leaflet for a complete list of ingredients in NEXLETOL. Stop taking NEXLETOL and call your healthcare provider or go to the nearest hospital emergency room right away if you have any signs or symptoms of an allergic reaction including: | |||||
|
|
||||
Before you start taking NEXLETOL, tell your healthcare provider about all your medical conditions, including if you:
Especially tell your healthcare provider if you take or plan to take: |
|||||
|
|||||
| Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. | |||||
How should I take NEXLETOL?
|
|||||
|
What are possible side effects of NEXLETOL? NEXLETOL may cause serious side effects, including:
|
|||||
|
|
||||
|
|||||
|
|
||||
|
|||||
| The most common side effects of NEXLETOL in people with primary hypercholesterolemia include: | |||||
|
|
||||
| The most common side effects of NEXLETOL in people with heart problems include: | |||||
|
|
||||
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of NEXLETOL. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||||
How should I store NEXLETOL?
|
|||||
|
General information about the safe and effective use of NEXLETOL.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use NEXLETOL for a condition for which it was not prescribed. Do not give NEXLETOL to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about NEXLETOL that is written for healthcare professionals. |
|||||
What are the ingredients in NEXLETOL?
|
|||||
| Manufactured for: Esperion Therapeutics, Inc. 3891 Ranchero Drive, Suite 150 Ann Arbor, MI 48108 © 2026 Esperion Therapeutics, Inc. |
Section 43683-2
| Indications and Usage (1) | 11/2025 |
Section 44425-7
Storage and Handling
Store at 68°F to 77°F (20°C to 25°C); excursions permitted to 59°F to 86°F (15°C to 30°C) [see USP Controlled Room Temperature]. Store and dispense in the original package.
10 Overdosage
There is no clinical experience with NEXLETOL overdose. In the event of an overdosage, consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
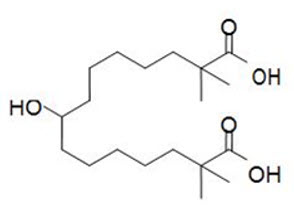
11 Description
NEXLETOL tablets, for oral use, contain bempedoic acid, an adenosine triphosphate-citrate lyase (ACL) inhibitor. The chemical name for bempedoic acid is 8-hydroxy-2,2,14,14-tetramethyl-pentadecanedioic acid. The molecular formula is C19H36O5, and the molecular weight is 344.5 grams per mole. Bempedoic acid is a white to off-white crystalline powder that is highly soluble in ethanol, isopropanol and pH 8 phosphate buffer, and insoluble in water and aqueous solutions below pH 5.
Structural formula:
Each film-coated tablet of NEXLETOL contains 180 mg of bempedoic acid and the following inactive ingredients: colloidal silicon dioxide, hydroxyl propyl cellulose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The film coating comprises of partially hydrolyzed polyvinyl alcohol, polyethylene glycol, talc, and titanium dioxide.
5.1 Hyperuricemia
NEXLETOL inhibits renal tubular OAT2 and may increase blood uric acid levels [see Clinical Pharmacology (12.3)]. In the primary hypercholesterolemia trials [see Clinical Studies (14.2)], 26% of NEXLETOL-treated patients with normal baseline uric acid values (versus 9.5% placebo) experienced hyperuricemia one or more times, and 3.5% of patients experienced clinically significant hyperuricemia reported as an adverse reaction (versus 1.1% placebo). Increases in uric acid levels usually occurred within the first 4 weeks of treatment initiation, persisted throughout treatment, and returned to baseline following discontinuation of treatment. After 12 weeks of treatment, the mean placebo-adjusted increase in uric acid compared to baseline was 0.8 mg/dL for patients treated with NEXLETOL. In the cardiovascular outcomes trial [see Clinical Studies (14.1)], 16.4% of NEXLETOL-treated patients experienced clinically significant hyperuricemia reported as an adverse reaction (versus 8.2% placebo).
Elevated blood uric acid may lead to the development of gout. In the primary hypercholesterolemia trials, gout was reported in 1.5% of patients treated with NEXLETOL and 0.4% of patients treated with placebo. In the cardiovascular outcomes trial, gout was reported in 3.2% of patients treated with NEXLETOL and 2.2% treated with placebo.
Advise patients to contact their healthcare provider if symptoms of hyperuricemia occur. Assess serum uric acid when clinically indicated. Monitor patients for signs and symptoms of hyperuricemia, and initiate treatment with urate-lowering drugs as appropriate.
8.4 Pediatric Use
The safety and effectiveness of NEXLETOL have not been established in pediatric patients.
8.5 Geriatric Use
Of the 3,009 adult patients in the primary hypercholesterolemia trials of NEXLETOL, 1,753 (58%) were 65 years of age and older, while 478 (16%) were 75 years of age and older.
Of the 13,970 adult patients in the cardiovascular outcomes trial [see Clinical Studies (14.1)], 8,204 (59%) were 65 years of age and older, while 2,107 (15%) were 75 years of age and older.
No overall differences in safety or effectiveness of NEXLETOL have been observed between patients 65 years of age and older and younger adult patients.
5.2 Tendon Rupture
NEXLETOL is associated with an increased risk of tendon rupture or injury. In the primary hypercholesterolemia trials [see Clinical Studies (14.2)], tendon rupture occurred in 0.5% of patients treated with NEXLETOL versus 0% of placebo-treated patients and involved the rotator cuff (the shoulder), biceps tendon, or Achilles tendon. Tendon rupture occurred within weeks to months of starting NEXLETOL. In the cardiovascular outcomes trial [see Clinical Studies (14.1)], tendon rupture events occurred in 1.2% of NEXLETOL-treated patients versus 0.9% of placebo-treated patients. Tendon rupture may occur more frequently in patients over 60 years of age, in those taking corticosteroid or fluoroquinolone drugs, in patients with renal failure, and in patients with previous tendon disorders.
Discontinue NEXLETOL immediately if the patient experiences rupture of a tendon. Consider discontinuing NEXLETOL if the patient experiences joint pain, swelling, or inflammation. Advise patients to rest at the first sign of tendinitis or tendon rupture and to contact their healthcare provider if tendinitis or tendon rupture symptoms occur. Consider alternative therapy in patients with a history of tendon disorders or tendon rupture.
4 Contraindications
NEXLETOL is contraindicated in patients with a prior serious hypersensitivity reaction to bempedoic acid or any of the excipients in NEXLETOL. Serious hypersensitivity reactions, such as angioedema, have occurred [see Adverse Reactions (6.2)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hyperuricemia [see Warnings and Precautions (5.1)]
- Tendon Rupture [see Warnings and Precautions (5.2)]
7 Drug Interactions
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with NEXLETOL and instructions for preventing or managing them.
| Simvastatin | |
| Clinical Impact: | Concomitant use of NEXLETOL with simvastatin causes an increase in simvastatin concentration and may increase the risk of simvastatin-related myopathy [see Clinical Pharmacology (12.3)]. |
| Intervention: | Avoid concomitant use of NEXLETOL with simvastatin greater than 20 mg. |
| Pravastatin | |
| Clinical Impact: | Concomitant use of NEXLETOL with pravastatin causes an increase in pravastatin concentration and may increase the risk of pravastatin-related myopathy [see Clinical Pharmacology (12.3)]. |
| Intervention: | Avoid concomitant use of NEXLETOL with pravastatin greater than 40 mg. |
| Fibrates | |
| Clinical Impact: | Concomitant administration of fibrates with NEXLETOL resulted in increased triglycerides and decreased high-density lipoprotein cholesterol (HDL-C) in some patients in clinical studies and post-marketing reports. Reversibility of both increased triglycerides and decreased HDL-C levels was observed when either NEXLETOL or fibrate therapy was discontinued. |
| Intervention: | Monitor triglycerides and HDL-C four weeks after initial concomitant use of NEXLETOL and a fibrate and periodically thereafter. If increased triglycerides or decreased HDL-C levels are detected, discontinue NEXLETOL or fibrate therapy based on clinical judgment. Monitor triglycerides and HDL-C levels until levels return to baseline. |
12.2 Pharmacodynamics
Administration of bempedoic acid alone, or in combination with other lipid modifying agents, decreases LDL-C, non-high density lipoprotein cholesterol (non-HDL-C), apolipoprotein B (apo B), and total cholesterol (TC) in patients with hypercholesterolemia.
12.3 Pharmacokinetics
Bempedoic acid pharmacokinetic parameters are presented as the mean [± standard deviation (SD)] unless otherwise specified. The steady-state maximum plasma concentration (Cmax) and area under the curve (AUC) following multiple-dose administration of bempedoic acid at 180 mg/day were 20.6 ± 6.1 µg/mL and 289.0 ± 96.4 µg∙h/mL, respectively. Bempedoic acid steady-state pharmacokinetics were generally linear over a range of > 60 mg to 220 mg (approximately 33% to 122% of the recommended dosage of 180 mg daily). There were no time-dependent changes in bempedoic acid pharmacokinetics following repeat administration at the recommended dosage, and bempedoic acid steady-state was achieved after 7 days. The mean accumulation ratio was approximately 2.3-fold.
The steady-state Cmax and AUC of the active metabolite (ESP15228) of bempedoic acid were 2.8 ± 0.9 µg/mL and 51.2 ± 17.2 µg∙h/mL, respectively. ESP15228 likely made a minor contribution to the overall clinical activity of bempedoic acid based on systemic exposure, relative potency, and pharmacokinetic properties.
2.1 Recommended Dosage
The recommended dosage of NEXLETOL is 180 mg administered orally once daily. NEXLETOL can be taken with or without food.
After initiation of NEXLETOL, analyze lipid levels within 8 to 12 weeks.
8.7 Hepatic Impairment
No dosage adjustment is necessary in patients with mild or moderate hepatic impairment (Child-Pugh A or B) [see Clinical Pharmacology (12.3)]. Patients with severe hepatic impairment (Child-Pugh C) have not been studied.
1 Indications and Usage
NEXLETOL is indicated:
- to reduce the risk of major adverse cardiovascular events (cardiovascular death, myocardial infarction, stroke, or coronary revascularization) in adults at increased risk for these events who are unable to take recommended statin therapy (including those not taking a statin).
- as an adjunct to diet and exercise, in combination with other low-density lipoprotein cholesterol (LDL-C) lowering therapies, or alone when concomitant LDL-C lowering therapy is not possible, to reduce LDL-C in adults with hypercholesterolemia, including heterozygous familial hypercholesterolemia (HeFH).
12.1 Mechanism of Action
Bempedoic acid is an adenosine triphosphate-citrate lyase (ACL) inhibitor that lowers LDL-C by inhibition of cholesterol synthesis in the liver. ACL is an enzyme upstream of 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase in the cholesterol biosynthesis pathway. Bempedoic acid and its active metabolite, ESP15228, require coenzyme A (CoA) activation by very long-chain acyl-CoA synthetase 1 (ACSVL1) to ETC-1002-CoA and ESP15228-CoA, respectively. ACSVL1 is expressed primarily in the liver. Inhibition of ACL by ETC-1002-CoA results in decreased cholesterol synthesis in the liver and lowers LDL-C in blood via upregulation of low-density lipoprotein receptors.
5 Warnings and Precautions
Hyperuricemia: Elevations in serum uric acid have occurred. Assess uric acid levels periodically as clinically indicated. Monitor for signs and symptoms of hyperuricemia, and initiate treatment with urate-lowering drugs as appropriate. (5.1)
Tendon Rupture: Tendon rupture has occurred. Discontinue NEXLETOL at the first sign of tendon rupture. Avoid NEXLETOL in patients who have a history of tendon disorders or tendon rupture. (5.2)
2 Dosage and Administration
Administer 180 mg orally once daily with or without food. (2.1)
3 Dosage Forms and Strengths
NEXLETOL is available as:
- Tablets: 180 mg, white to off-white, oval shaped, debossed with "180" on one side and "ESP" on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of NEXLETOL. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Hypersensitivity reactions including: angioedema, wheezing, rash, and urticaria.
8 Use in Specific Populations
- Pregnancy: Based on mechanism of action, may cause fetal harm. (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data in Table 1 reflect exposure to NEXLETOL in two placebo-controlled primary hypercholesterolemia trials that included 2,009 patients treated with NEXLETOL for 52 weeks (median treatment duration of 52 weeks) [see Clinical Studies (14.2)]. The mean age for NEXLETOL-treated patients was 65 years, 29% were female, 95% were White, 3% were Black or African American, 1% were Asian, and 1% were other races; 3% identified as Hispanic or Latino ethnicity. All patients received NEXLETOL 180 mg orally once daily plus maximally tolerated statin therapy alone or in combination with other lipid-lowering therapies. At baseline, 97% of patients had CVD and about 4% had a diagnosis of HeFH. Patients on simvastatin 40 mg/day or higher were excluded from the trials.
In the primary hypercholesterolemia trials, adverse reactions led to discontinuation of treatment in 11% of NEXLETOL-treated patients and 8% of placebo-treated patients. The most common reasons for NEXLETOL treatment discontinuation were muscle spasms (0.5% versus 0.3% placebo), diarrhea (0.4% versus 0.1% placebo), and pain in extremity (0.3% versus 0.0% placebo). Adverse reactions reported in at least 2% of NEXLETOL-treated patients and more frequently than in placebo-treated patients are shown in Table 1.
| Adverse Reaction | Placebo Background therapy included statin and ± other lipid-lowering therapies
(N = 999) % |
NEXLETOL
(N = 2,009) % |
|---|---|---|
| Upper respiratory tract infection | 4.0 | 4.5 |
| Muscle spasms | 2.3 | 3.6 |
| Hyperuricemia Grouped term that includes other related terms
|
1.1 | 3.5 |
| Back pain | 2.2 | 3.3 |
| Abdominal pain or discomfort | 2.2 | 3.1 |
| Bronchitis | 2.5 | 3.0 |
| Pain in extremity | 1.7 | 3.0 |
| Anemia | 1.9 | 2.8 |
| Elevated liver enzymes | 0.8 | 2.1 |
In the cardiovascular outcomes trial, in which 7,001 patients were exposed to NEXLETOL and 6,964 patients were exposed to placebo for a median of 3.1 years [see Clinical Studies, (14.1)], adverse reactions led to discontinuation of treatment in 11% of NEXLETOL-treated patients and 10% of placebo-treated patients. Adverse reactions reported in at least 2% of NEXLETOL-treated patients and 0.5% greater than placebo are shown in Table 2.
| Adverse Reaction | Placebo (N=6,964) % |
NEXLETOL (N=7,001) % |
|---|---|---|
| Hyperuricemia Grouped term that includes other related terms
|
8 | 16 |
| Renal impairment Renal impairment includes laboratory related terms including glomerular filtration rate decreased, blood creatinine increased and hematuria
|
9 | 11 |
| Anemia | 4 | 5 |
| Elevated liver enzymes | 3 | 4 |
| Muscle spasms | 3 | 4 |
| Gout | 2 | 3 |
| Cholelithiasis | 1 | 2 |
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Patient Information).
14.2 Primary Hypercholesterolemia Trials in Adults
The efficacy of NEXLETOL as an adjunct to diet and statin therapy, to reduce elevated LDL-C in adults with primary hypercholesterolemia (including HeFH) was investigated in two multi-center, randomized, double-blind, placebo-controlled trials that enrolled 3,009 adult patients with HeFH or established CVD who were on maximally tolerated statin therapy (Trials 2 and 3). Demographics and baseline disease characteristics were balanced between the treatment arms in these trials. In both trials, the maximum LDL-C lowering effects occurred at Week 4. These results were consistent across all subgroups studied in any of the trials, including age, sex, race, ethnicity, region, history of diabetes, baseline LDL-C, body mass index (BMI), HeFH status, and background therapies.
Principal Display Panel 180 Mg Tablet Bottle Label
NDC 72426-118-03
Rx only
NEXLETOL®
(bempedoic acid) tablets
Contains
30 Tablets
180 mg
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Bempedoic acid was negative for mutagenicity in an in vitro Ames assay and negative for clastogenicity in the vitro human lymphocyte chromosome aberration assay. Bempedoic acid was negative in both in vivo mouse micronucleus and in vivo rat bone marrow micronucleus/liver comet assay. In a 2-year rat carcinogenicity study, Wistar rats were given oral doses of bempedoic acid at 3, 10 and 30 mg/kg/day. An increased incidence of liver hepatocellular adenomas and hepatocellular adenomas combined with carcinomas, thyroid gland follicular cell adenoma and follicular cell adenomas combined with carcinomas, and pancreatic islet cell adenomas combined with carcinomas were observed in male rats at the dose of 30 mg/kg/day (exposure equivalent to the maximum recommended human dose (MRHD), based on AUC). In a 2-year mice carcinogenicity study, CD-1 mice were given oral doses of bempedoic acid at 25, 75 and 150 mg/kg/day. Bempedoic acid-related increases in the incidence of liver hepatocellular adenomas, hepatocellular carcinomas and hepatocellular adenomas combined with carcinomas in male mice were observed at 75 and 150 mg/kg/day (exposures equivalent to the MRHD). Observations of liver and thyroid tumors are consistent with PPAR alpha agonism in rodents. The human relevance of pancreatic islet cell tumor findings is unknown.
In fertility and early embryofetal development study in rats, bempedoic acid was given orally to male and female rats at 10, 30 and 60 mg/kg/day. Males were dosed for 28 days prior to mating and females were dosed 14 days prior to mating through gestation day 7. No adverse effects on fertility were observed in females in the absence of maternal toxicity. No effects were observed on male fertility outcomes, but decreases in sperm counts were observed at 60 mg/kg/day (9 times the MRHD).
14.1 Cardiovascular Outcomes Trial in Adults With Cvd Or At High Risk for Cvd
Trial 1 (NCT02993406) was a randomized, double-blind, placebo-controlled, event-driven trial in 13,970 adult patients with established CVD (70%) or at high risk for a CVD event but without CVD (30%) who were not receiving recommended statin dosages. Patients with established CVD had documented history of coronary artery disease, symptomatic peripheral arterial disease, and/or cerebrovascular atherosclerotic disease. Patients without established CVD were considered at high risk for CVD based on meeting at least one of the following criteria:
-
(1)Diabetes mellitus (type 1 or type 2) in females over 65 years of age or males over 60 years of age;
-
(2)A Reynolds Risk score > 30% or a SCORE Risk score > 7.5% over 10 years. Reynolds risk score and SCORE risk score evaluate a 10-year risk of having a cardiovascular (CV) event. The Reynolds risk score is based on the following risk factors: sex, age, smoking status, systolic blood pressure, total cholesterol, HDL cholesterol, high sensitivity C-reactive protein (hsCRP), and familial history of CVD events. LDL-C is an additional risk factor considered in SCORE risk score; or
-
(3)A coronary artery calcium score >400 Agatston units at any time in the past.
Patients were randomized 1:1 to receive either oral NEXLETOL 180 mg per day (n = 6,992) or placebo (n = 6,978), alone or as an add on to other background lipid-lowering therapies. Background therapy could include less than low-intensity statin dosages. Overall, 95.3% of adult patients were followed until the end of the trial or death. The median follow-up duration was 3.4 years.
Structured Label Content
Section 42229-5 (42229-5)
Other Adverse Reactions
Section 42230-3 (42230-3)
| PATIENT INFORMATION NEXLETOL® (NEX-le-tol) (bempedoic acid) tablets, for oral use |
|||||
|---|---|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Revised: 01/2026 | ||||
|
What is NEXLETOL?
NEXLETOL is a prescription medicine used:
|
|||||
| Do not take NEXLETOL if you are allergic to bempedoic acid or any of the ingredients in NEXLETOL. See the end of this leaflet for a complete list of ingredients in NEXLETOL. Stop taking NEXLETOL and call your healthcare provider or go to the nearest hospital emergency room right away if you have any signs or symptoms of an allergic reaction including: | |||||
|
|
||||
Before you start taking NEXLETOL, tell your healthcare provider about all your medical conditions, including if you:
Especially tell your healthcare provider if you take or plan to take: |
|||||
|
|||||
| Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. | |||||
How should I take NEXLETOL?
|
|||||
|
What are possible side effects of NEXLETOL? NEXLETOL may cause serious side effects, including:
|
|||||
|
|
||||
|
|||||
|
|
||||
|
|||||
| The most common side effects of NEXLETOL in people with primary hypercholesterolemia include: | |||||
|
|
||||
| The most common side effects of NEXLETOL in people with heart problems include: | |||||
|
|
||||
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of NEXLETOL. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||||
How should I store NEXLETOL?
|
|||||
|
General information about the safe and effective use of NEXLETOL.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use NEXLETOL for a condition for which it was not prescribed. Do not give NEXLETOL to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about NEXLETOL that is written for healthcare professionals. |
|||||
What are the ingredients in NEXLETOL?
|
|||||
| Manufactured for: Esperion Therapeutics, Inc. 3891 Ranchero Drive, Suite 150 Ann Arbor, MI 48108 © 2026 Esperion Therapeutics, Inc. |
Section 43683-2 (43683-2)
| Indications and Usage (1) | 11/2025 |
Section 44425-7 (44425-7)
Storage and Handling
Store at 68°F to 77°F (20°C to 25°C); excursions permitted to 59°F to 86°F (15°C to 30°C) [see USP Controlled Room Temperature]. Store and dispense in the original package.
10 Overdosage (10 OVERDOSAGE)
There is no clinical experience with NEXLETOL overdose. In the event of an overdosage, consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
11 Description (11 DESCRIPTION)
NEXLETOL tablets, for oral use, contain bempedoic acid, an adenosine triphosphate-citrate lyase (ACL) inhibitor. The chemical name for bempedoic acid is 8-hydroxy-2,2,14,14-tetramethyl-pentadecanedioic acid. The molecular formula is C19H36O5, and the molecular weight is 344.5 grams per mole. Bempedoic acid is a white to off-white crystalline powder that is highly soluble in ethanol, isopropanol and pH 8 phosphate buffer, and insoluble in water and aqueous solutions below pH 5.
Structural formula:
Each film-coated tablet of NEXLETOL contains 180 mg of bempedoic acid and the following inactive ingredients: colloidal silicon dioxide, hydroxyl propyl cellulose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate. The film coating comprises of partially hydrolyzed polyvinyl alcohol, polyethylene glycol, talc, and titanium dioxide.
5.1 Hyperuricemia
NEXLETOL inhibits renal tubular OAT2 and may increase blood uric acid levels [see Clinical Pharmacology (12.3)]. In the primary hypercholesterolemia trials [see Clinical Studies (14.2)], 26% of NEXLETOL-treated patients with normal baseline uric acid values (versus 9.5% placebo) experienced hyperuricemia one or more times, and 3.5% of patients experienced clinically significant hyperuricemia reported as an adverse reaction (versus 1.1% placebo). Increases in uric acid levels usually occurred within the first 4 weeks of treatment initiation, persisted throughout treatment, and returned to baseline following discontinuation of treatment. After 12 weeks of treatment, the mean placebo-adjusted increase in uric acid compared to baseline was 0.8 mg/dL for patients treated with NEXLETOL. In the cardiovascular outcomes trial [see Clinical Studies (14.1)], 16.4% of NEXLETOL-treated patients experienced clinically significant hyperuricemia reported as an adverse reaction (versus 8.2% placebo).
Elevated blood uric acid may lead to the development of gout. In the primary hypercholesterolemia trials, gout was reported in 1.5% of patients treated with NEXLETOL and 0.4% of patients treated with placebo. In the cardiovascular outcomes trial, gout was reported in 3.2% of patients treated with NEXLETOL and 2.2% treated with placebo.
Advise patients to contact their healthcare provider if symptoms of hyperuricemia occur. Assess serum uric acid when clinically indicated. Monitor patients for signs and symptoms of hyperuricemia, and initiate treatment with urate-lowering drugs as appropriate.
8.4 Pediatric Use
The safety and effectiveness of NEXLETOL have not been established in pediatric patients.
8.5 Geriatric Use
Of the 3,009 adult patients in the primary hypercholesterolemia trials of NEXLETOL, 1,753 (58%) were 65 years of age and older, while 478 (16%) were 75 years of age and older.
Of the 13,970 adult patients in the cardiovascular outcomes trial [see Clinical Studies (14.1)], 8,204 (59%) were 65 years of age and older, while 2,107 (15%) were 75 years of age and older.
No overall differences in safety or effectiveness of NEXLETOL have been observed between patients 65 years of age and older and younger adult patients.
5.2 Tendon Rupture
NEXLETOL is associated with an increased risk of tendon rupture or injury. In the primary hypercholesterolemia trials [see Clinical Studies (14.2)], tendon rupture occurred in 0.5% of patients treated with NEXLETOL versus 0% of placebo-treated patients and involved the rotator cuff (the shoulder), biceps tendon, or Achilles tendon. Tendon rupture occurred within weeks to months of starting NEXLETOL. In the cardiovascular outcomes trial [see Clinical Studies (14.1)], tendon rupture events occurred in 1.2% of NEXLETOL-treated patients versus 0.9% of placebo-treated patients. Tendon rupture may occur more frequently in patients over 60 years of age, in those taking corticosteroid or fluoroquinolone drugs, in patients with renal failure, and in patients with previous tendon disorders.
Discontinue NEXLETOL immediately if the patient experiences rupture of a tendon. Consider discontinuing NEXLETOL if the patient experiences joint pain, swelling, or inflammation. Advise patients to rest at the first sign of tendinitis or tendon rupture and to contact their healthcare provider if tendinitis or tendon rupture symptoms occur. Consider alternative therapy in patients with a history of tendon disorders or tendon rupture.
4 Contraindications (4 CONTRAINDICATIONS)
NEXLETOL is contraindicated in patients with a prior serious hypersensitivity reaction to bempedoic acid or any of the excipients in NEXLETOL. Serious hypersensitivity reactions, such as angioedema, have occurred [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hyperuricemia [see Warnings and Precautions (5.1)]
- Tendon Rupture [see Warnings and Precautions (5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with NEXLETOL and instructions for preventing or managing them.
| Simvastatin | |
| Clinical Impact: | Concomitant use of NEXLETOL with simvastatin causes an increase in simvastatin concentration and may increase the risk of simvastatin-related myopathy [see Clinical Pharmacology (12.3)]. |
| Intervention: | Avoid concomitant use of NEXLETOL with simvastatin greater than 20 mg. |
| Pravastatin | |
| Clinical Impact: | Concomitant use of NEXLETOL with pravastatin causes an increase in pravastatin concentration and may increase the risk of pravastatin-related myopathy [see Clinical Pharmacology (12.3)]. |
| Intervention: | Avoid concomitant use of NEXLETOL with pravastatin greater than 40 mg. |
| Fibrates | |
| Clinical Impact: | Concomitant administration of fibrates with NEXLETOL resulted in increased triglycerides and decreased high-density lipoprotein cholesterol (HDL-C) in some patients in clinical studies and post-marketing reports. Reversibility of both increased triglycerides and decreased HDL-C levels was observed when either NEXLETOL or fibrate therapy was discontinued. |
| Intervention: | Monitor triglycerides and HDL-C four weeks after initial concomitant use of NEXLETOL and a fibrate and periodically thereafter. If increased triglycerides or decreased HDL-C levels are detected, discontinue NEXLETOL or fibrate therapy based on clinical judgment. Monitor triglycerides and HDL-C levels until levels return to baseline. |
12.2 Pharmacodynamics
Administration of bempedoic acid alone, or in combination with other lipid modifying agents, decreases LDL-C, non-high density lipoprotein cholesterol (non-HDL-C), apolipoprotein B (apo B), and total cholesterol (TC) in patients with hypercholesterolemia.
12.3 Pharmacokinetics
Bempedoic acid pharmacokinetic parameters are presented as the mean [± standard deviation (SD)] unless otherwise specified. The steady-state maximum plasma concentration (Cmax) and area under the curve (AUC) following multiple-dose administration of bempedoic acid at 180 mg/day were 20.6 ± 6.1 µg/mL and 289.0 ± 96.4 µg∙h/mL, respectively. Bempedoic acid steady-state pharmacokinetics were generally linear over a range of > 60 mg to 220 mg (approximately 33% to 122% of the recommended dosage of 180 mg daily). There were no time-dependent changes in bempedoic acid pharmacokinetics following repeat administration at the recommended dosage, and bempedoic acid steady-state was achieved after 7 days. The mean accumulation ratio was approximately 2.3-fold.
The steady-state Cmax and AUC of the active metabolite (ESP15228) of bempedoic acid were 2.8 ± 0.9 µg/mL and 51.2 ± 17.2 µg∙h/mL, respectively. ESP15228 likely made a minor contribution to the overall clinical activity of bempedoic acid based on systemic exposure, relative potency, and pharmacokinetic properties.
2.1 Recommended Dosage
The recommended dosage of NEXLETOL is 180 mg administered orally once daily. NEXLETOL can be taken with or without food.
After initiation of NEXLETOL, analyze lipid levels within 8 to 12 weeks.
8.7 Hepatic Impairment
No dosage adjustment is necessary in patients with mild or moderate hepatic impairment (Child-Pugh A or B) [see Clinical Pharmacology (12.3)]. Patients with severe hepatic impairment (Child-Pugh C) have not been studied.
1 Indications and Usage (1 INDICATIONS AND USAGE)
NEXLETOL is indicated:
- to reduce the risk of major adverse cardiovascular events (cardiovascular death, myocardial infarction, stroke, or coronary revascularization) in adults at increased risk for these events who are unable to take recommended statin therapy (including those not taking a statin).
- as an adjunct to diet and exercise, in combination with other low-density lipoprotein cholesterol (LDL-C) lowering therapies, or alone when concomitant LDL-C lowering therapy is not possible, to reduce LDL-C in adults with hypercholesterolemia, including heterozygous familial hypercholesterolemia (HeFH).
12.1 Mechanism of Action
Bempedoic acid is an adenosine triphosphate-citrate lyase (ACL) inhibitor that lowers LDL-C by inhibition of cholesterol synthesis in the liver. ACL is an enzyme upstream of 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase in the cholesterol biosynthesis pathway. Bempedoic acid and its active metabolite, ESP15228, require coenzyme A (CoA) activation by very long-chain acyl-CoA synthetase 1 (ACSVL1) to ETC-1002-CoA and ESP15228-CoA, respectively. ACSVL1 is expressed primarily in the liver. Inhibition of ACL by ETC-1002-CoA results in decreased cholesterol synthesis in the liver and lowers LDL-C in blood via upregulation of low-density lipoprotein receptors.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Hyperuricemia: Elevations in serum uric acid have occurred. Assess uric acid levels periodically as clinically indicated. Monitor for signs and symptoms of hyperuricemia, and initiate treatment with urate-lowering drugs as appropriate. (5.1)
Tendon Rupture: Tendon rupture has occurred. Discontinue NEXLETOL at the first sign of tendon rupture. Avoid NEXLETOL in patients who have a history of tendon disorders or tendon rupture. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Administer 180 mg orally once daily with or without food. (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
NEXLETOL is available as:
- Tablets: 180 mg, white to off-white, oval shaped, debossed with "180" on one side and "ESP" on the other side.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of NEXLETOL. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune System Disorders: Hypersensitivity reactions including: angioedema, wheezing, rash, and urticaria.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Based on mechanism of action, may cause fetal harm. (8.1)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data in Table 1 reflect exposure to NEXLETOL in two placebo-controlled primary hypercholesterolemia trials that included 2,009 patients treated with NEXLETOL for 52 weeks (median treatment duration of 52 weeks) [see Clinical Studies (14.2)]. The mean age for NEXLETOL-treated patients was 65 years, 29% were female, 95% were White, 3% were Black or African American, 1% were Asian, and 1% were other races; 3% identified as Hispanic or Latino ethnicity. All patients received NEXLETOL 180 mg orally once daily plus maximally tolerated statin therapy alone or in combination with other lipid-lowering therapies. At baseline, 97% of patients had CVD and about 4% had a diagnosis of HeFH. Patients on simvastatin 40 mg/day or higher were excluded from the trials.
In the primary hypercholesterolemia trials, adverse reactions led to discontinuation of treatment in 11% of NEXLETOL-treated patients and 8% of placebo-treated patients. The most common reasons for NEXLETOL treatment discontinuation were muscle spasms (0.5% versus 0.3% placebo), diarrhea (0.4% versus 0.1% placebo), and pain in extremity (0.3% versus 0.0% placebo). Adverse reactions reported in at least 2% of NEXLETOL-treated patients and more frequently than in placebo-treated patients are shown in Table 1.
| Adverse Reaction | Placebo Background therapy included statin and ± other lipid-lowering therapies
(N = 999) % |
NEXLETOL
(N = 2,009) % |
|---|---|---|
| Upper respiratory tract infection | 4.0 | 4.5 |
| Muscle spasms | 2.3 | 3.6 |
| Hyperuricemia Grouped term that includes other related terms
|
1.1 | 3.5 |
| Back pain | 2.2 | 3.3 |
| Abdominal pain or discomfort | 2.2 | 3.1 |
| Bronchitis | 2.5 | 3.0 |
| Pain in extremity | 1.7 | 3.0 |
| Anemia | 1.9 | 2.8 |
| Elevated liver enzymes | 0.8 | 2.1 |
In the cardiovascular outcomes trial, in which 7,001 patients were exposed to NEXLETOL and 6,964 patients were exposed to placebo for a median of 3.1 years [see Clinical Studies, (14.1)], adverse reactions led to discontinuation of treatment in 11% of NEXLETOL-treated patients and 10% of placebo-treated patients. Adverse reactions reported in at least 2% of NEXLETOL-treated patients and 0.5% greater than placebo are shown in Table 2.
| Adverse Reaction | Placebo (N=6,964) % |
NEXLETOL (N=7,001) % |
|---|---|---|
| Hyperuricemia Grouped term that includes other related terms
|
8 | 16 |
| Renal impairment Renal impairment includes laboratory related terms including glomerular filtration rate decreased, blood creatinine increased and hematuria
|
9 | 11 |
| Anemia | 4 | 5 |
| Elevated liver enzymes | 3 | 4 |
| Muscle spasms | 3 | 4 |
| Gout | 2 | 3 |
| Cholelithiasis | 1 | 2 |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Patient Information).
14.2 Primary Hypercholesterolemia Trials in Adults
The efficacy of NEXLETOL as an adjunct to diet and statin therapy, to reduce elevated LDL-C in adults with primary hypercholesterolemia (including HeFH) was investigated in two multi-center, randomized, double-blind, placebo-controlled trials that enrolled 3,009 adult patients with HeFH or established CVD who were on maximally tolerated statin therapy (Trials 2 and 3). Demographics and baseline disease characteristics were balanced between the treatment arms in these trials. In both trials, the maximum LDL-C lowering effects occurred at Week 4. These results were consistent across all subgroups studied in any of the trials, including age, sex, race, ethnicity, region, history of diabetes, baseline LDL-C, body mass index (BMI), HeFH status, and background therapies.
Principal Display Panel 180 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 180 mg Tablet Bottle Label)
NDC 72426-118-03
Rx only
NEXLETOL®
(bempedoic acid) tablets
Contains
30 Tablets
180 mg
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Bempedoic acid was negative for mutagenicity in an in vitro Ames assay and negative for clastogenicity in the vitro human lymphocyte chromosome aberration assay. Bempedoic acid was negative in both in vivo mouse micronucleus and in vivo rat bone marrow micronucleus/liver comet assay. In a 2-year rat carcinogenicity study, Wistar rats were given oral doses of bempedoic acid at 3, 10 and 30 mg/kg/day. An increased incidence of liver hepatocellular adenomas and hepatocellular adenomas combined with carcinomas, thyroid gland follicular cell adenoma and follicular cell adenomas combined with carcinomas, and pancreatic islet cell adenomas combined with carcinomas were observed in male rats at the dose of 30 mg/kg/day (exposure equivalent to the maximum recommended human dose (MRHD), based on AUC). In a 2-year mice carcinogenicity study, CD-1 mice were given oral doses of bempedoic acid at 25, 75 and 150 mg/kg/day. Bempedoic acid-related increases in the incidence of liver hepatocellular adenomas, hepatocellular carcinomas and hepatocellular adenomas combined with carcinomas in male mice were observed at 75 and 150 mg/kg/day (exposures equivalent to the MRHD). Observations of liver and thyroid tumors are consistent with PPAR alpha agonism in rodents. The human relevance of pancreatic islet cell tumor findings is unknown.
In fertility and early embryofetal development study in rats, bempedoic acid was given orally to male and female rats at 10, 30 and 60 mg/kg/day. Males were dosed for 28 days prior to mating and females were dosed 14 days prior to mating through gestation day 7. No adverse effects on fertility were observed in females in the absence of maternal toxicity. No effects were observed on male fertility outcomes, but decreases in sperm counts were observed at 60 mg/kg/day (9 times the MRHD).
14.1 Cardiovascular Outcomes Trial in Adults With Cvd Or At High Risk for Cvd (14.1 Cardiovascular Outcomes Trial in Adults With CVD or at High Risk for CVD)
Trial 1 (NCT02993406) was a randomized, double-blind, placebo-controlled, event-driven trial in 13,970 adult patients with established CVD (70%) or at high risk for a CVD event but without CVD (30%) who were not receiving recommended statin dosages. Patients with established CVD had documented history of coronary artery disease, symptomatic peripheral arterial disease, and/or cerebrovascular atherosclerotic disease. Patients without established CVD were considered at high risk for CVD based on meeting at least one of the following criteria:
-
(1)Diabetes mellitus (type 1 or type 2) in females over 65 years of age or males over 60 years of age;
-
(2)A Reynolds Risk score > 30% or a SCORE Risk score > 7.5% over 10 years. Reynolds risk score and SCORE risk score evaluate a 10-year risk of having a cardiovascular (CV) event. The Reynolds risk score is based on the following risk factors: sex, age, smoking status, systolic blood pressure, total cholesterol, HDL cholesterol, high sensitivity C-reactive protein (hsCRP), and familial history of CVD events. LDL-C is an additional risk factor considered in SCORE risk score; or
-
(3)A coronary artery calcium score >400 Agatston units at any time in the past.
Patients were randomized 1:1 to receive either oral NEXLETOL 180 mg per day (n = 6,992) or placebo (n = 6,978), alone or as an add on to other background lipid-lowering therapies. Background therapy could include less than low-intensity statin dosages. Overall, 95.3% of adult patients were followed until the end of the trial or death. The median follow-up duration was 3.4 years.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:26.037968 · Updated: 2026-03-14T22:43:43.392452