These Highlights Do Not Include All The Information Needed To Use Imaavy Safely And Effectively. See Full Prescribing Information For Imaavy.
8886274c-f2b2-48af-85c1-2f90bfe304b8
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
IMAAVY is indicated for the treatment of generalized myasthenia gravis (gMG) in adult and pediatric patients 12 years of age and older who are anti-acetylcholine receptor (AChR) or anti-muscle-specific tyrosine kinase (MuSK) antibody positive.
Indications and Usage
IMAAVY is indicated for the treatment of generalized myasthenia gravis (gMG) in adult and pediatric patients 12 years of age and older who are anti-acetylcholine receptor (AChR) or anti-muscle-specific tyrosine kinase (MuSK) antibody positive.
Dosage and Administration
See Full Prescribing Information for instructions on dosage, preparation, and administration. ( 2.1 , 2.2 , 2.3 ) Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of IMAAVY. ( 2.1 ) Administer via intravenous infusion only. ( 2.2 ) The recommended initial dosage is 30 mg/kg once via intravenous infusion over at least 30 minutes. Two weeks after the initial dosage, administer a maintenance dosage of 15 mg/kg via intravenous infusion over at least 15 minutes, and continue every two weeks thereafter. ( 2.2 ) Must be diluted with 0.9% sodium chloride injection prior to administration. ( 2.3 ) Administer as an intravenous infusion via a 0.2 micron in-line or add-on filter. ( 2.3 )
Warnings and Precautions
Infections: Delay administration of IMAAVY to patients with an active infection. Monitor for signs and symptoms of infection in patients treated with IMAAVY. If serious infection occurs, administer appropriate treatment and consider withholding IMAAVY until the infection has resolved. ( 5.1 ) Hypersensitivity Reactions: Angioedema, anaphylaxis, rash, urticaria, and eczema have occurred in patients treated with IMAAVY. If a hypersensitivity reaction occurs, discontinue the infusion and institute appropriate therapy. ( 5.2 ) Infusion-Related Reactions: If a severe infusion-related reaction occurs, discontinue the infusion and initiate appropriate therapy; consider the risks and benefits of readministering. If a mild to moderate infusion-related reaction occurs, may rechallenge with close clinical observation, slower infusion rates, and pre-medication. ( 5.3 )
Contraindications
IMAAVY is contraindicated in patients with a history of serious hypersensitivity reaction to nipocalimab or any of the excipients in IMAAVY. Reactions have included anaphylaxis and angioedema [see Warnings and Precautions (5.2) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling Infections [see Warnings and Precautions (5.1) ] Hypersensitivity Reactions [see Warnings and Precautions (5.2) ] Infusion-related Reactions [see Warnings and Precautions (5.3) ]
Drug Interactions
Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing IMAAVY and using alternative therapies. ( 7 )
Medication Information
Warnings and Precautions
Infections: Delay administration of IMAAVY to patients with an active infection. Monitor for signs and symptoms of infection in patients treated with IMAAVY. If serious infection occurs, administer appropriate treatment and consider withholding IMAAVY until the infection has resolved. ( 5.1 ) Hypersensitivity Reactions: Angioedema, anaphylaxis, rash, urticaria, and eczema have occurred in patients treated with IMAAVY. If a hypersensitivity reaction occurs, discontinue the infusion and institute appropriate therapy. ( 5.2 ) Infusion-Related Reactions: If a severe infusion-related reaction occurs, discontinue the infusion and initiate appropriate therapy; consider the risks and benefits of readministering. If a mild to moderate infusion-related reaction occurs, may rechallenge with close clinical observation, slower infusion rates, and pre-medication. ( 5.3 )
Indications and Usage
IMAAVY is indicated for the treatment of generalized myasthenia gravis (gMG) in adult and pediatric patients 12 years of age and older who are anti-acetylcholine receptor (AChR) or anti-muscle-specific tyrosine kinase (MuSK) antibody positive.
Dosage and Administration
See Full Prescribing Information for instructions on dosage, preparation, and administration. ( 2.1 , 2.2 , 2.3 ) Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of IMAAVY. ( 2.1 ) Administer via intravenous infusion only. ( 2.2 ) The recommended initial dosage is 30 mg/kg once via intravenous infusion over at least 30 minutes. Two weeks after the initial dosage, administer a maintenance dosage of 15 mg/kg via intravenous infusion over at least 15 minutes, and continue every two weeks thereafter. ( 2.2 ) Must be diluted with 0.9% sodium chloride injection prior to administration. ( 2.3 ) Administer as an intravenous infusion via a 0.2 micron in-line or add-on filter. ( 2.3 )
Contraindications
IMAAVY is contraindicated in patients with a history of serious hypersensitivity reaction to nipocalimab or any of the excipients in IMAAVY. Reactions have included anaphylaxis and angioedema [see Warnings and Precautions (5.2) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling Infections [see Warnings and Precautions (5.1) ] Hypersensitivity Reactions [see Warnings and Precautions (5.2) ] Infusion-related Reactions [see Warnings and Precautions (5.3) ]
Drug Interactions
Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing IMAAVY and using alternative therapies. ( 7 )
Description
IMAAVY is indicated for the treatment of generalized myasthenia gravis (gMG) in adult and pediatric patients 12 years of age and older who are anti-acetylcholine receptor (AChR) or anti-muscle-specific tyrosine kinase (MuSK) antibody positive.
Section 42229-5
Preparation
Prepare the solution for infusion using aseptic technique as follows:
- Calculate the dosage (mg), total drug volume (mL) of IMAAVY solution required, and the number of IMAAVY vials needed, based on the patient's current weight [see Dosage and Administration (2.2)] . Each single-dose vial of IMAAVY is at a concentration of 185 mg/mL.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Check that the solution in each vial is colorless to slightly brownish, clear to slightly opalescent, and free of visible particles. Do not use if visible particles are present or if the solution is discolored (other than colorless to slightly brownish).
- Gently withdraw the calculated volume of IMAAVY from the vial(s). Discard any unused portion of the vials.
- Dilute total volume withdrawn of IMAAVY by adding to an infusion container containing 0.9% sodium chloride injection to a final volume of:
- 250 mL for patients who weigh 40 kg or more, or
- 100 mL for patients who weigh less than 40 kg.
- Gently invert the infusion container at least 10 times to mix the solution. Do not shake.
- Verify that a uniform solution has been achieved by visual inspection. Do not use if particulate matter or discoloration is present.
Section 42230-3
| PATIENT INFORMATION
IMAAVY™ [im-AH-vee] (nipocalimab-aahu) injection, for intravenous use |
||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Issued: 4/2025 | |
|
What is IMAAVY?
IMAAVY is a prescription medicine used to treat adults and children 12 years of age and older with a disease called generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) or anti-muscle-specific tyrosine kinase (MuSK) antibody positive. It is not known if IMAAVY is safe and effective in children under 12 years of age. |
||
Do not receive IMAAVY if you:
|
||
Before receiving IMAAVY, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How will I receive IMAAVY?
|
||
What should I avoid while receiving IMAAVY?
|
||
|
What are the possible side effects of IMAAVY?
IMAAVY may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
|
||
|
|
|
The most common side effects in people with gMG treated with IMAAVY include:
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
General information about the safe and effective use of IMAAVY.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about IMAAVY that is written for health professionals. |
||
|
What are the ingredients of IMAAVY?
Active ingredient : nipocalimab-aahu Inactive ingredients: arginine hydrochloride, histidine, L-histidine monohydrochloride monohydrate, methionine, polysorbate 80, sucrose, and water for injection. |
||
| Manufactured by: Janssen Biotech, Inc., Horsham, PA 19044, USA
U.S. License Number 1864 For patent information: www.janssenpatents.com © 2025 Janssen Pharmaceutical Companies For more information, call 1-800-526-7736 or go to www.IMAAVY.com |
Section 44425-7
Storage and Handling
11 Description
Nipocalimab-aahu, a neonatal Fc receptor blocker, is a recombinant human immunoglobulin G1 lambda (IgG1λ) monoclonal antibody, expressed in a genetically engineered Chinese hamster ovary cell line. Nipocalimab-aahu has an aglycosylated Fc region, therefore it lacks effector functions. Nipocalimab-aahu has an approximate molecular weight of 142 kilodaltons (kDa).
IMAAVY™ (nipocalimab-aahu) injection is a sterile, preservative-free, colorless to slightly brownish, clear to slightly opalescent solution, supplied in a single-dose vial for intravenous infusion after dilution.
Each single-dose vial contains either 300 mg/1.62 mL or 1,200 mg/6.5 mL of nipocalimab-aahu at a concentration of 185 mg/mL. In addition, each mL of solution contains arginine hydrochloride (25.35 mg), histidine (0.77 mg), L-histidine monohydrochloride monohydrate (1.07 mg), methionine (1.0 mg), polysorbate 80 (0.60 mg), sucrose (64.3 mg), and water for injection, USP, at a pH of 6.0.
5.1 Infections
IMAAVY may increase the risk of infection [see Adverse Reactions (6.1)] . In Study 1 [see Clinical Studies (14)] , 42 (43%) out of 98 patients treated with IMAAVY reported 71 events of infection. Across Study 1 (double blind period) and its extension study (open label-period), out of 186 patients treated with IMAAVY, 132 (71%) patients reported 360 events of infection. Serious infections were reported in 7% of patients treated with IMAAVY. Delay IMAAVY administration in patients with an active infection until the infection is resolved. During treatment with IMAAVY, monitor for clinical signs and symptoms of infection. If serious infection occurs, administer appropriate treatment and consider withholding IMAAVY until the infection has resolved.
8.4 Pediatric Use
The safety and effectiveness of IMAAVY for the treatment of gMG have been established in pediatric patients 12 years of age and older. Use of IMAAVY in pediatric patients for this indication is supported by evidence from an adequate and well-controlled trial in adults with additional pharmacokinetic and safety data in pediatric patients who are 12 years of age and older [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)] .
Safety and effectiveness of IMAAVY for the treatment of gMG in pediatric patients below the age of 12 years have not been established.
8.5 Geriatric Use
Clinical studies of IMAAVY did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger adult patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of nipocalimab-aahu or of other nipocalimab products.
In clinical trials, antibodies to nipocalimab-aahu were detected in 49/102 (48%) adult and pediatric patients 12 years of age and older during 24-week treatment period.
Out of the 49 patients who were positive for antibodies to nipocalimab-aahu, 19 (38.8%) patients had neutralizing antibodies to nipocalimab-aahu. There was no identified clinically relevant effect of antibodies, including neutralizing antibodies, to nipocalimab-aahu on the pharmacokinetics, pharmacodynamics, safety, or effectiveness of IMAAVY.
14 Clinical Studies
The efficacy of IMAAVY for the treatment of gMG in adults who are anti-AChR or anti-MuSK antibody positive was established in a 24-week, multicenter, randomized, double-blind, placebo-controlled study (Study 1; NCT04951622). Patients were treated with IMAAVY with the recommended dosage regimen [see Dosage and Administration (2.1)] .
Study 1 enrolled patients with gMG who met the following criteria:
- Myasthenia Gravis Foundation of America (MGFA) Clinical Classification Class II to IV
- Myasthenia Gravis-Activities of Daily Living (MG-ADL) total score of at least 6
- On stable dose of standard of care MG therapy prior to baseline that included acetylcholinesterase (AChE) inhibitors, steroids or non-steroidal immunosuppressive therapies (NSISTs), either in combination or alone.
In Study 1, a total of 196 patients were randomized 1:1 to receive IMAAVY (n=98) or placebo (n=98). Baseline characteristics were similar between treatment groups. For the primary efficacy analysis population (n=153), patients had a median age of 52 years at screening (range 20 to 81 years) and a median time since diagnosis of 6 years. Sixty percent of patients were female; 63% were White; 32% were Asian; 1% were Black or African-American; and <1% were American Indian or Alaskan Native. At baseline, median MG-ADL total score was 9, and median Quantitative Myasthenia Gravis (QMG) total score was 15. Eighty-eight percent (n=134) of patients were positive for AChR antibodies and 10% (n=16) were positive for MuSK antibodies.
At baseline, in each group, 85% of patients received AChE inhibitors, 66% of patients received steroids, and 54% of patients received NSISTs at stable doses.
The efficacy of IMAAVY was measured using the MG-ADL scale, which assesses the impact of gMG on daily functions of 8 signs and symptoms that are typically affected in gMG. Each item is assessed on a 4-point scale, where a score of 0 represents normal function and a score of 3 represents loss of ability to perform that function. A total score ranges from 0 to 24, with the higher scores indicating more impairment.
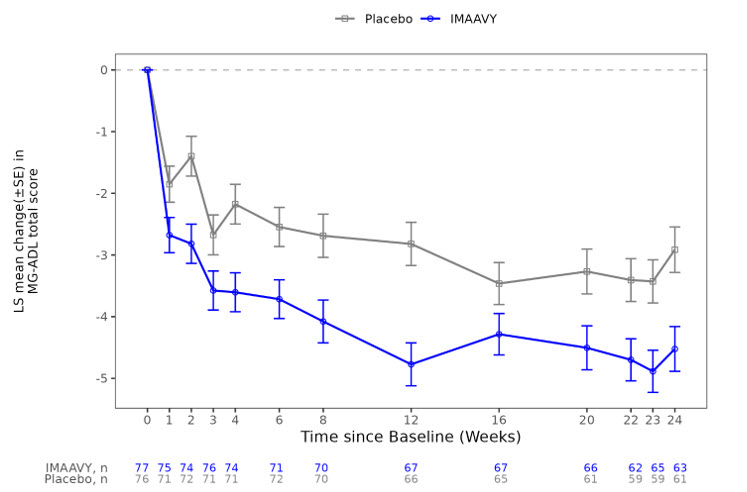
The primary efficacy endpoint was the comparison of the mean change from baseline to Weeks 22, 23, and 24 between treatment groups in the MG-ADL total score. A statistically significant difference favoring IMAAVY was observed in MG-ADL total score change from baseline (p=0.002; see Table 2 and Figure 1).
The efficacy of IMAAVY was also measured using the QMG total score, which is a 13-item categorial grading system that assesses muscle weakness. Each item is assessed on a 4 -point scale, where a score of 0 represents no weakness, and a score of 3 represents severe weakness. A total possible score ranges from 0 to 39, where higher scores indicate more severe impairment.
The secondary endpoint was the comparison of the mean change from baseline to Weeks 22 and 24 between treatment groups in the QMG total score. A statistically significant difference favoring IMAAVY was observed in the QMG total score change from baseline (p<0.001; see Table 2).
The results are presented shown in Table 2.
| Efficacy Endpoints | IMAAVY
N = 77 LS Mean (SE) |
Placebo
N = 76 LS Mean (SE) |
IMAAVY Change Relative to Placebo
LS Mean Difference (95% CI) |
p-value |
|---|---|---|---|---|
| Key: CI=confidence interval; MG-ADL = Myasthenia Gravis – Activities of Daily Living; QMG = Quantitative Myasthenia Gravis; LS mean = Least squares mean; SE = standard error | ||||
| Primary Endpoint | ||||
| MG-ADL Total Score
Mean change from baseline over weeks 22, 23, and 24
|
-4.7 (0.33) | -3.3 (0.34) | -1.5 (-2.4, -0.5) | 0.002 |
| Secondary Endpoint | ||||
| QMG
Total Score Mean change from baseline over weeks 22 and 24
|
-4.9 (0.5) | -2.1 (0.5) | -2.8 (-4.2, -1.4) | <0.001 |
Figure 1 shows the mean change from baseline to Week 24 in MG-ADL total score in Study 1, and Figure 2 shows the mean change from baseline to Week 24 in QMG total score in Study 1.
Figure 1: Least Squares Mean Change from Baseline in MG-ADL Total Score Over 24 Weeks in Study 1
LS = least squares, SE = standard error, MG-ADL = Myasthenia Gravis Activities of Daily Living
Figure 2: Least Squares Mean Change from Baseline in QMG Total Score Over 24 Weeks in Study 1
LS = least squares, SE = standard error, QMG = Quantitative Myasthenia Gravis.
4 Contraindications
IMAAVY is contraindicated in patients with a history of serious hypersensitivity reaction to nipocalimab or any of the excipients in IMAAVY. Reactions have included anaphylaxis and angioedema [see Warnings and Precautions (5.2)] .
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling
- Infections [see Warnings and Precautions (5.1)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
- Infusion-related Reactions [see Warnings and Precautions (5.3)]
7 Drug Interactions
Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing IMAAVY and using alternative therapies. ( 7)
12.2 Pharmacodynamics
In Study 1 [see Clinical Studies (14)], the pharmacological effect of nipocalimab-aahu was assessed by measuring the decrease in serum IgG levels and anti-AChR and anti-MuSK autoantibody levels. In patients positive for AChR and MuSK autoantibodies who were treated with IMAAVY, there was a reduction in AChR and MuSK autoantibodies relative to baseline. Decreases in total IgG levels followed a similar pattern. A similar reduction in AChR autoantibodies was observed in adolescent patients with gMG compared to adults.
12.3 Pharmacokinetics
Nipocalimab exhibits nonlinear pharmacokinetics. Following a single intravenous infusion of IMAAVY at doses ranging from 0.3 to 60 mg/kg (4 times the recommended maintenance dosage) in healthy participants, C max of nipocalimab-aahu increased in a dose-proportional manner while AUC increased in a greater than dose-proportional manner.
2.2 Recommended Dosage
Dilute IMAAVY prior to administration. Administer via intravenous infusion only [see Dosage and Administration (2.3)] .
The recommended initial dosage of IMAAVY is 30 mg/kg administered once via intravenous infusion over at least 30 minutes. Two weeks after the initial dosage administer a maintenance dosage of 15 mg/kg via intravenous infusion over at least 15 minutes. Continue the maintenance dosage every two weeks thereafter.
If a scheduled infusion appointment is missed, the maintenance dosage of IMAAVY should be administered as soon as possible. Resume dosing every two weeks thereafter.
1 Indications and Usage
IMAAVY is indicated for the treatment of generalized myasthenia gravis (gMG) in adult and pediatric patients 12 years of age and older who are anti-acetylcholine receptor (AChR) or anti-muscle-specific tyrosine kinase (MuSK) antibody positive.
12.1 Mechanism of Action
Nipocalimab-aahu is a human IgG1 monoclonal antibody that binds to neonatal Fc receptor (FcRn), resulting in the reduction of circulating IgG levels.
5 Warnings and Precautions
- Infections: Delay administration of IMAAVY to patients with an active infection. Monitor for signs and symptoms of infection in patients treated with IMAAVY. If serious infection occurs, administer appropriate treatment and consider withholding IMAAVY until the infection has resolved. ( 5.1)
- Hypersensitivity Reactions: Angioedema, anaphylaxis, rash, urticaria, and eczema have occurred in patients treated with IMAAVY. If a hypersensitivity reaction occurs, discontinue the infusion and institute appropriate therapy. ( 5.2)
- Infusion-Related Reactions: If a severe infusion-related reaction occurs, discontinue the infusion and initiate appropriate therapy; consider the risks and benefits of readministering. If a mild to moderate infusion-related reaction occurs, may rechallenge with close clinical observation, slower infusion rates, and pre-medication. ( 5.3)
2 Dosage and Administration
- See Full Prescribing Information for instructions on dosage, preparation, and administration. ( 2.1, 2.2, 2.3)
- Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of IMAAVY. ( 2.1)
- Administer via intravenous infusion only. ( 2.2)
- The recommended initial dosage is 30 mg/kg once via intravenous infusion over at least 30 minutes. Two weeks after the initial dosage, administer a maintenance dosage of 15 mg/kg via intravenous infusion over at least 15 minutes, and continue every two weeks thereafter. ( 2.2)
- Must be diluted with 0.9% sodium chloride injection prior to administration. ( 2.3)
- Administer as an intravenous infusion via a 0.2 micron in-line or add-on filter. ( 2.3)
2.1 Recommended Vaccination
Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of IMAAVY. Because IMAAVY causes transient reduction in IgG levels, vaccination with live vaccines is not recommended during treatment with IMAAVY [see Warnings and Precautions (5.1)].
3 Dosage Forms and Strengths
Injection: colorless to slightly brownish, clear to slightly opalescent solution available as:
- 300 mg/1.62 mL (185 mg/mL) in a single-dose vial
- 1,200 mg/6.5 mL (185 mg/mL) in a single-dose vial
5.2 Hypersensitivity Reactions
In clinical trials, hypersensitivity reactions, including angioedema, anaphylaxis, rash, urticaria, and eczema were observed in patients treated with IMAAVY. In Study 1, hypersensitivity reactions were mild or moderate, occurred within one hour to 2 weeks of administration [see Adverse Reactions (6.1)] . One patient experienced a hypersensitivity reaction (urticaria) that led to treatment discontinuation.
Management of hypersensitivity reactions depends on the type and severity of the reaction. Monitor the patient during treatment with IMAAVY and for 30 minutes after the administration is complete for clinical signs and symptoms of hypersensitivity reactions [see Dosage and Administration (2.3)] . If a hypersensitivity reaction occurs during administration, discontinue IMAAVY infusion and institute appropriate supportive measures if needed. IMAAVY is contraindicated in patients with a history of serious hypersensitivity to nipocalimab or any of the excipients of IMAAVY [see Contraindications (4)] .
5.3 Infusion Related Reactions
In clinical trials, infusion-related reactions, including headache, influenza-like illness, rash, nausea, fatigue, dizziness, chills, and erythema were observed in patients treated with IMAAVY. In Study 1, infusion-related reactions were mild to moderate in severity and occurred within one hour to 2 days of administration [see Adverse Reactions (6.1)] .
Monitor patients during treatment with IMAAVY and for 30 minutes after each infusion [see Dosage and Administration (2.3)] . If a severe infusion-related reaction occurs, discontinue IMAAVY infusion and initiate appropriate therapy. Consider the risks and benefits of readministering IMAAVY following a severe infusion-related reaction. If a mild to moderate infusion related reaction occurs, patients may be rechallenged with close clinical observation, slower infusion rates, and pre-medication.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Patient Information).
7.1 Effect of Imaavy On Other Drugs
Concomitant use of IMAAVY with medications that bind to the human neonatal Fc receptor (FcRn) (e.g., immunoglobulin products, monoclonal antibodies, or antibody derivates containing the human Fc domain of the IgG subclass) may lower systemic exposures and reduce effectiveness of such medications. Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing IMAAVY, and using alternative therapies [see Clinical Pharmacology (12.3)] .
2.3 Preparation and Administration Instructions
Prior to administration, dilute IMAAVY single-dose vials with only 0.9% sodium chloride injection using the instructions below. For patients who weigh 40 kg or more, the total volume to be administered is 250 mL; for patients who are 12 years or older and weigh less than 40 kg, the total volume to be administered is 100 mL (see Preparation) .
Principal Display Panel 300 Mg / 1.62 Ml Vial Box
NDC 57894-800-01
imaavy™
(nipocalimab-aahu)
Injection
300 mg / 1.62 mL
(185 mg / mL)
For Intravenous Infusion
After Dilution
Rx only
One single-dose vial.
Discard unused portion.
Johnson
& Johnson
Principal Display Panel 1,200 Mg / 6.5 Ml Vial Box
NDC 57894-801-01
imaavy™
(nipocalimab-aahu)
Injection
1,200 mg / 6.5 mL
(185 mg / mL)
For Intravenous Infusion
After Dilution
Rx only
One single-dose vial.
Discard unused portion.
Johnson
& Johnson
Structured Label Content
Section 42229-5 (42229-5)
Preparation
Prepare the solution for infusion using aseptic technique as follows:
- Calculate the dosage (mg), total drug volume (mL) of IMAAVY solution required, and the number of IMAAVY vials needed, based on the patient's current weight [see Dosage and Administration (2.2)] . Each single-dose vial of IMAAVY is at a concentration of 185 mg/mL.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Check that the solution in each vial is colorless to slightly brownish, clear to slightly opalescent, and free of visible particles. Do not use if visible particles are present or if the solution is discolored (other than colorless to slightly brownish).
- Gently withdraw the calculated volume of IMAAVY from the vial(s). Discard any unused portion of the vials.
- Dilute total volume withdrawn of IMAAVY by adding to an infusion container containing 0.9% sodium chloride injection to a final volume of:
- 250 mL for patients who weigh 40 kg or more, or
- 100 mL for patients who weigh less than 40 kg.
- Gently invert the infusion container at least 10 times to mix the solution. Do not shake.
- Verify that a uniform solution has been achieved by visual inspection. Do not use if particulate matter or discoloration is present.
Section 42230-3 (42230-3)
| PATIENT INFORMATION
IMAAVY™ [im-AH-vee] (nipocalimab-aahu) injection, for intravenous use |
||
|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration | Issued: 4/2025 | |
|
What is IMAAVY?
IMAAVY is a prescription medicine used to treat adults and children 12 years of age and older with a disease called generalized myasthenia gravis (gMG) who are anti-acetylcholine receptor (AChR) or anti-muscle-specific tyrosine kinase (MuSK) antibody positive. It is not known if IMAAVY is safe and effective in children under 12 years of age. |
||
Do not receive IMAAVY if you:
|
||
Before receiving IMAAVY, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How will I receive IMAAVY?
|
||
What should I avoid while receiving IMAAVY?
|
||
|
What are the possible side effects of IMAAVY?
IMAAVY may cause serious side effects, including:
|
||
|
|
|
|
||
|
|
|
|
||
|
|
|
The most common side effects in people with gMG treated with IMAAVY include:
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
General information about the safe and effective use of IMAAVY.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. You can ask your pharmacist or healthcare provider for information about IMAAVY that is written for health professionals. |
||
|
What are the ingredients of IMAAVY?
Active ingredient : nipocalimab-aahu Inactive ingredients: arginine hydrochloride, histidine, L-histidine monohydrochloride monohydrate, methionine, polysorbate 80, sucrose, and water for injection. |
||
| Manufactured by: Janssen Biotech, Inc., Horsham, PA 19044, USA
U.S. License Number 1864 For patent information: www.janssenpatents.com © 2025 Janssen Pharmaceutical Companies For more information, call 1-800-526-7736 or go to www.IMAAVY.com |
Section 44425-7 (44425-7)
Storage and Handling
11 Description (11 DESCRIPTION)
Nipocalimab-aahu, a neonatal Fc receptor blocker, is a recombinant human immunoglobulin G1 lambda (IgG1λ) monoclonal antibody, expressed in a genetically engineered Chinese hamster ovary cell line. Nipocalimab-aahu has an aglycosylated Fc region, therefore it lacks effector functions. Nipocalimab-aahu has an approximate molecular weight of 142 kilodaltons (kDa).
IMAAVY™ (nipocalimab-aahu) injection is a sterile, preservative-free, colorless to slightly brownish, clear to slightly opalescent solution, supplied in a single-dose vial for intravenous infusion after dilution.
Each single-dose vial contains either 300 mg/1.62 mL or 1,200 mg/6.5 mL of nipocalimab-aahu at a concentration of 185 mg/mL. In addition, each mL of solution contains arginine hydrochloride (25.35 mg), histidine (0.77 mg), L-histidine monohydrochloride monohydrate (1.07 mg), methionine (1.0 mg), polysorbate 80 (0.60 mg), sucrose (64.3 mg), and water for injection, USP, at a pH of 6.0.
5.1 Infections
IMAAVY may increase the risk of infection [see Adverse Reactions (6.1)] . In Study 1 [see Clinical Studies (14)] , 42 (43%) out of 98 patients treated with IMAAVY reported 71 events of infection. Across Study 1 (double blind period) and its extension study (open label-period), out of 186 patients treated with IMAAVY, 132 (71%) patients reported 360 events of infection. Serious infections were reported in 7% of patients treated with IMAAVY. Delay IMAAVY administration in patients with an active infection until the infection is resolved. During treatment with IMAAVY, monitor for clinical signs and symptoms of infection. If serious infection occurs, administer appropriate treatment and consider withholding IMAAVY until the infection has resolved.
8.4 Pediatric Use
The safety and effectiveness of IMAAVY for the treatment of gMG have been established in pediatric patients 12 years of age and older. Use of IMAAVY in pediatric patients for this indication is supported by evidence from an adequate and well-controlled trial in adults with additional pharmacokinetic and safety data in pediatric patients who are 12 years of age and older [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14)] .
Safety and effectiveness of IMAAVY for the treatment of gMG in pediatric patients below the age of 12 years have not been established.
8.5 Geriatric Use
Clinical studies of IMAAVY did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger adult patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of nipocalimab-aahu or of other nipocalimab products.
In clinical trials, antibodies to nipocalimab-aahu were detected in 49/102 (48%) adult and pediatric patients 12 years of age and older during 24-week treatment period.
Out of the 49 patients who were positive for antibodies to nipocalimab-aahu, 19 (38.8%) patients had neutralizing antibodies to nipocalimab-aahu. There was no identified clinically relevant effect of antibodies, including neutralizing antibodies, to nipocalimab-aahu on the pharmacokinetics, pharmacodynamics, safety, or effectiveness of IMAAVY.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of IMAAVY for the treatment of gMG in adults who are anti-AChR or anti-MuSK antibody positive was established in a 24-week, multicenter, randomized, double-blind, placebo-controlled study (Study 1; NCT04951622). Patients were treated with IMAAVY with the recommended dosage regimen [see Dosage and Administration (2.1)] .
Study 1 enrolled patients with gMG who met the following criteria:
- Myasthenia Gravis Foundation of America (MGFA) Clinical Classification Class II to IV
- Myasthenia Gravis-Activities of Daily Living (MG-ADL) total score of at least 6
- On stable dose of standard of care MG therapy prior to baseline that included acetylcholinesterase (AChE) inhibitors, steroids or non-steroidal immunosuppressive therapies (NSISTs), either in combination or alone.
In Study 1, a total of 196 patients were randomized 1:1 to receive IMAAVY (n=98) or placebo (n=98). Baseline characteristics were similar between treatment groups. For the primary efficacy analysis population (n=153), patients had a median age of 52 years at screening (range 20 to 81 years) and a median time since diagnosis of 6 years. Sixty percent of patients were female; 63% were White; 32% were Asian; 1% were Black or African-American; and <1% were American Indian or Alaskan Native. At baseline, median MG-ADL total score was 9, and median Quantitative Myasthenia Gravis (QMG) total score was 15. Eighty-eight percent (n=134) of patients were positive for AChR antibodies and 10% (n=16) were positive for MuSK antibodies.
At baseline, in each group, 85% of patients received AChE inhibitors, 66% of patients received steroids, and 54% of patients received NSISTs at stable doses.
The efficacy of IMAAVY was measured using the MG-ADL scale, which assesses the impact of gMG on daily functions of 8 signs and symptoms that are typically affected in gMG. Each item is assessed on a 4-point scale, where a score of 0 represents normal function and a score of 3 represents loss of ability to perform that function. A total score ranges from 0 to 24, with the higher scores indicating more impairment.
The primary efficacy endpoint was the comparison of the mean change from baseline to Weeks 22, 23, and 24 between treatment groups in the MG-ADL total score. A statistically significant difference favoring IMAAVY was observed in MG-ADL total score change from baseline (p=0.002; see Table 2 and Figure 1).
The efficacy of IMAAVY was also measured using the QMG total score, which is a 13-item categorial grading system that assesses muscle weakness. Each item is assessed on a 4 -point scale, where a score of 0 represents no weakness, and a score of 3 represents severe weakness. A total possible score ranges from 0 to 39, where higher scores indicate more severe impairment.
The secondary endpoint was the comparison of the mean change from baseline to Weeks 22 and 24 between treatment groups in the QMG total score. A statistically significant difference favoring IMAAVY was observed in the QMG total score change from baseline (p<0.001; see Table 2).
The results are presented shown in Table 2.
| Efficacy Endpoints | IMAAVY
N = 77 LS Mean (SE) |
Placebo
N = 76 LS Mean (SE) |
IMAAVY Change Relative to Placebo
LS Mean Difference (95% CI) |
p-value |
|---|---|---|---|---|
| Key: CI=confidence interval; MG-ADL = Myasthenia Gravis – Activities of Daily Living; QMG = Quantitative Myasthenia Gravis; LS mean = Least squares mean; SE = standard error | ||||
| Primary Endpoint | ||||
| MG-ADL Total Score
Mean change from baseline over weeks 22, 23, and 24
|
-4.7 (0.33) | -3.3 (0.34) | -1.5 (-2.4, -0.5) | 0.002 |
| Secondary Endpoint | ||||
| QMG
Total Score Mean change from baseline over weeks 22 and 24
|
-4.9 (0.5) | -2.1 (0.5) | -2.8 (-4.2, -1.4) | <0.001 |
Figure 1 shows the mean change from baseline to Week 24 in MG-ADL total score in Study 1, and Figure 2 shows the mean change from baseline to Week 24 in QMG total score in Study 1.
Figure 1: Least Squares Mean Change from Baseline in MG-ADL Total Score Over 24 Weeks in Study 1
LS = least squares, SE = standard error, MG-ADL = Myasthenia Gravis Activities of Daily Living
Figure 2: Least Squares Mean Change from Baseline in QMG Total Score Over 24 Weeks in Study 1
LS = least squares, SE = standard error, QMG = Quantitative Myasthenia Gravis.
4 Contraindications (4 CONTRAINDICATIONS)
IMAAVY is contraindicated in patients with a history of serious hypersensitivity reaction to nipocalimab or any of the excipients in IMAAVY. Reactions have included anaphylaxis and angioedema [see Warnings and Precautions (5.2)] .
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling
- Infections [see Warnings and Precautions (5.1)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.2)]
- Infusion-related Reactions [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing IMAAVY and using alternative therapies. ( 7)
12.2 Pharmacodynamics
In Study 1 [see Clinical Studies (14)], the pharmacological effect of nipocalimab-aahu was assessed by measuring the decrease in serum IgG levels and anti-AChR and anti-MuSK autoantibody levels. In patients positive for AChR and MuSK autoantibodies who were treated with IMAAVY, there was a reduction in AChR and MuSK autoantibodies relative to baseline. Decreases in total IgG levels followed a similar pattern. A similar reduction in AChR autoantibodies was observed in adolescent patients with gMG compared to adults.
12.3 Pharmacokinetics
Nipocalimab exhibits nonlinear pharmacokinetics. Following a single intravenous infusion of IMAAVY at doses ranging from 0.3 to 60 mg/kg (4 times the recommended maintenance dosage) in healthy participants, C max of nipocalimab-aahu increased in a dose-proportional manner while AUC increased in a greater than dose-proportional manner.
2.2 Recommended Dosage
Dilute IMAAVY prior to administration. Administer via intravenous infusion only [see Dosage and Administration (2.3)] .
The recommended initial dosage of IMAAVY is 30 mg/kg administered once via intravenous infusion over at least 30 minutes. Two weeks after the initial dosage administer a maintenance dosage of 15 mg/kg via intravenous infusion over at least 15 minutes. Continue the maintenance dosage every two weeks thereafter.
If a scheduled infusion appointment is missed, the maintenance dosage of IMAAVY should be administered as soon as possible. Resume dosing every two weeks thereafter.
1 Indications and Usage (1 INDICATIONS AND USAGE)
IMAAVY is indicated for the treatment of generalized myasthenia gravis (gMG) in adult and pediatric patients 12 years of age and older who are anti-acetylcholine receptor (AChR) or anti-muscle-specific tyrosine kinase (MuSK) antibody positive.
12.1 Mechanism of Action
Nipocalimab-aahu is a human IgG1 monoclonal antibody that binds to neonatal Fc receptor (FcRn), resulting in the reduction of circulating IgG levels.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Infections: Delay administration of IMAAVY to patients with an active infection. Monitor for signs and symptoms of infection in patients treated with IMAAVY. If serious infection occurs, administer appropriate treatment and consider withholding IMAAVY until the infection has resolved. ( 5.1)
- Hypersensitivity Reactions: Angioedema, anaphylaxis, rash, urticaria, and eczema have occurred in patients treated with IMAAVY. If a hypersensitivity reaction occurs, discontinue the infusion and institute appropriate therapy. ( 5.2)
- Infusion-Related Reactions: If a severe infusion-related reaction occurs, discontinue the infusion and initiate appropriate therapy; consider the risks and benefits of readministering. If a mild to moderate infusion-related reaction occurs, may rechallenge with close clinical observation, slower infusion rates, and pre-medication. ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- See Full Prescribing Information for instructions on dosage, preparation, and administration. ( 2.1, 2.2, 2.3)
- Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of IMAAVY. ( 2.1)
- Administer via intravenous infusion only. ( 2.2)
- The recommended initial dosage is 30 mg/kg once via intravenous infusion over at least 30 minutes. Two weeks after the initial dosage, administer a maintenance dosage of 15 mg/kg via intravenous infusion over at least 15 minutes, and continue every two weeks thereafter. ( 2.2)
- Must be diluted with 0.9% sodium chloride injection prior to administration. ( 2.3)
- Administer as an intravenous infusion via a 0.2 micron in-line or add-on filter. ( 2.3)
2.1 Recommended Vaccination
Evaluate the need to administer age-appropriate vaccines according to immunization guidelines before initiation of IMAAVY. Because IMAAVY causes transient reduction in IgG levels, vaccination with live vaccines is not recommended during treatment with IMAAVY [see Warnings and Precautions (5.1)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: colorless to slightly brownish, clear to slightly opalescent solution available as:
- 300 mg/1.62 mL (185 mg/mL) in a single-dose vial
- 1,200 mg/6.5 mL (185 mg/mL) in a single-dose vial
5.2 Hypersensitivity Reactions
In clinical trials, hypersensitivity reactions, including angioedema, anaphylaxis, rash, urticaria, and eczema were observed in patients treated with IMAAVY. In Study 1, hypersensitivity reactions were mild or moderate, occurred within one hour to 2 weeks of administration [see Adverse Reactions (6.1)] . One patient experienced a hypersensitivity reaction (urticaria) that led to treatment discontinuation.
Management of hypersensitivity reactions depends on the type and severity of the reaction. Monitor the patient during treatment with IMAAVY and for 30 minutes after the administration is complete for clinical signs and symptoms of hypersensitivity reactions [see Dosage and Administration (2.3)] . If a hypersensitivity reaction occurs during administration, discontinue IMAAVY infusion and institute appropriate supportive measures if needed. IMAAVY is contraindicated in patients with a history of serious hypersensitivity to nipocalimab or any of the excipients of IMAAVY [see Contraindications (4)] .
5.3 Infusion Related Reactions (5.3 Infusion-Related Reactions)
In clinical trials, infusion-related reactions, including headache, influenza-like illness, rash, nausea, fatigue, dizziness, chills, and erythema were observed in patients treated with IMAAVY. In Study 1, infusion-related reactions were mild to moderate in severity and occurred within one hour to 2 days of administration [see Adverse Reactions (6.1)] .
Monitor patients during treatment with IMAAVY and for 30 minutes after each infusion [see Dosage and Administration (2.3)] . If a severe infusion-related reaction occurs, discontinue IMAAVY infusion and initiate appropriate therapy. Consider the risks and benefits of readministering IMAAVY following a severe infusion-related reaction. If a mild to moderate infusion related reaction occurs, patients may be rechallenged with close clinical observation, slower infusion rates, and pre-medication.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient and/or caregiver to read the FDA-approved patient labeling (Patient Information).
7.1 Effect of Imaavy On Other Drugs (7.1 Effect of IMAAVY on Other Drugs)
Concomitant use of IMAAVY with medications that bind to the human neonatal Fc receptor (FcRn) (e.g., immunoglobulin products, monoclonal antibodies, or antibody derivates containing the human Fc domain of the IgG subclass) may lower systemic exposures and reduce effectiveness of such medications. Closely monitor for reduced effectiveness of medications that bind to the human neonatal Fc receptor. When concomitant long-term use of such medications is essential for patient care, consider discontinuing IMAAVY, and using alternative therapies [see Clinical Pharmacology (12.3)] .
2.3 Preparation and Administration Instructions
Prior to administration, dilute IMAAVY single-dose vials with only 0.9% sodium chloride injection using the instructions below. For patients who weigh 40 kg or more, the total volume to be administered is 250 mL; for patients who are 12 years or older and weigh less than 40 kg, the total volume to be administered is 100 mL (see Preparation) .
Principal Display Panel 300 Mg / 1.62 Ml Vial Box (PRINCIPAL DISPLAY PANEL - 300 mg / 1.62 mL Vial Box)
NDC 57894-800-01
imaavy™
(nipocalimab-aahu)
Injection
300 mg / 1.62 mL
(185 mg / mL)
For Intravenous Infusion
After Dilution
Rx only
One single-dose vial.
Discard unused portion.
Johnson
& Johnson
Principal Display Panel 1,200 Mg / 6.5 Ml Vial Box (PRINCIPAL DISPLAY PANEL - 1,200 mg / 6.5 mL Vial Box)
NDC 57894-801-01
imaavy™
(nipocalimab-aahu)
Injection
1,200 mg / 6.5 mL
(185 mg / mL)
For Intravenous Infusion
After Dilution
Rx only
One single-dose vial.
Discard unused portion.
Johnson
& Johnson
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:01.034431 · Updated: 2026-03-14T22:26:10.810615