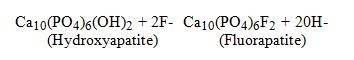Multi-vitamin Drops With Fluoride 025 Mg
880df628-5bba-4dc4-b557-1fcc822adba8
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Multi-Vitamin Drops with Fluoride 0 .25 mg Supplemental Facts Percentage of U.S. Recommended Daily Allowance Each 1.0 mL s upp li e s : Children 6 mos to 4 Years old Vitamin A (as Vitamin A Palmitate) 1500 IU 60% Vitamin C (as Ascorbic Acid) 35 mg 88% Vitamin D (as Cholecalciferol) 400 IU 100% Vitamin E (as D-Alpha-Tocopheryl Acid Succinate) 5 IU 50% Vitamin B1 (as Thiamine HCl) 0.5 mg 71% Vitamin B2 (as Riboflavin Phosphate Sodium) 0.6 mg 75% Vitamin B12 (as Cyanocobalamin) 2 mcg 67% Niacin (as Niacinamide) 8 mg 89% Vitamin B6 (as Pyridoxine HCl) 0.4 mg 57% Fluoride (as Sodium Fluoride) 0.25 mg *Daily Value not established. See INDICATIONS AND USAGE section for use by children 6 months to 6 years of age. This product does not contain the essential vitamin folic acid. Active ingredient for caries p r o phy l ax i s : Fluoride as sodium fluoride. This product does not contain Folic Acid. Other ingredients: Cherry Flavor, Citric Acid Anhydrous, Glycerin, Orange Flavor, Potassium Sorbate, Propylene Glycol, Purified Water, Sodium Benzoate, Sorbitol Solution, Sucralose.
Indications and Usage
Supplementation of the diet with vitamins A, C and D. Multi-Vitamin Drops with Fluoride 0.25 mg also provides fluoride for caries prophylaxis. The American Academy of Pediatrics recommends that children up to age 16, in areas where drinking water contains less than optimal levels of fluoride, receive daily fluoride supplementation. The American Academy of Pediatrics recommend that infants and young children 6 months to 3 years of age, in areas where the drinking water contains less than 0.3 ppm of fluoride, and children 3-6 years of age, in areas where the drinking water contains 0.3 through 0.6 ppm of fluoride, receive 0.25 mg of supplemental fluoride daily which is provided in a dose of 1 mL of Multi-Vitamin Drops with Fluoride 0.25 mg (See Dosage and Administration ). Multi-Vitamin Drops with Fluoride 0.25 mg supply significant amounts of vitamins A, C and D to supplement the diet, and to help assure that nutritional deficiencies of these vitamins will not develop. Thus, in a single easy-to-use preparation, children obtain essential vitamins and fluoride.
Dosage and Administration
See following chart. May be dropped directly into the mouth with dropper; or mixed with cereal, fruit juice or other food. Fluoride Ion Level in Drinking Water (ppm) * A g e <0.3 ppm 0.3 - 0.6 ppm >0.6 ppm Birth - 6 months None None None 6 mos - 3 years 0.25 mg (1 mL) / day † None None 3 - 6 years 0.50 mg (2 mL) / day 0.25 mg (1 mL) / day None * 1.0 ppm = 1 mg/liter † 2.2 mg sodium fluoride contains 1 mg fluoride ion.
Adverse Reactions
Allergic rash and other idiosyncrasies have been rarely reported.
How Supplied
Multi-Vitamin and Fluoride 0.25 mg drops is available in 50 mL bottles with accompanying calibrated dropper.
Medication Information
Indications and Usage
Supplementation of the diet with vitamins A, C and D.
Multi-Vitamin Drops with Fluoride 0.25 mg also provides fluoride for caries prophylaxis.
The American Academy of Pediatrics recommends that children up to age 16, in areas where drinking water contains less than optimal levels of fluoride, receive daily fluoride supplementation.
The American Academy of Pediatrics recommend that infants and young children 6 months to 3 years of age, in areas where the drinking water contains less than 0.3 ppm of fluoride, and children 3-6 years of age, in areas where the drinking water contains 0.3 through 0.6 ppm of fluoride, receive 0.25 mg of supplemental fluoride daily which is provided in a dose of 1 mL of Multi-Vitamin Drops with Fluoride 0.25 mg (See Dosage and Administration ).
Multi-Vitamin Drops with Fluoride 0.25 mg supply significant amounts of vitamins A, C and D to supplement the diet, and to help assure that nutritional deficiencies of these vitamins will not develop.
Thus, in a single easy-to-use preparation, children obtain essential vitamins and fluoride.
Dosage and Administration
See following chart. May be dropped directly into the mouth with dropper; or mixed with cereal, fruit juice or other food. Fluoride Ion Level in Drinking Water (ppm) * A g e <0.3 ppm 0.3 - 0.6 ppm >0.6 ppm Birth - 6 months None None None 6 mos - 3 years 0.25 mg (1 mL) / day † None None 3 - 6 years 0.50 mg (2 mL) / day 0.25 mg (1 mL) / day None * 1.0 ppm = 1 mg/liter † 2.2 mg sodium fluoride contains 1 mg fluoride ion.
Adverse Reactions
Allergic rash and other idiosyncrasies have been rarely reported.
How Supplied
Multi-Vitamin and Fluoride 0.25 mg drops is available in 50 mL bottles with accompanying calibrated dropper.
Description
Multi-Vitamin Drops with Fluoride 0 .25 mg Supplemental Facts Percentage of U.S. Recommended Daily Allowance Each 1.0 mL s upp li e s : Children 6 mos to 4 Years old Vitamin A (as Vitamin A Palmitate) 1500 IU 60% Vitamin C (as Ascorbic Acid) 35 mg 88% Vitamin D (as Cholecalciferol) 400 IU 100% Vitamin E (as D-Alpha-Tocopheryl Acid Succinate) 5 IU 50% Vitamin B1 (as Thiamine HCl) 0.5 mg 71% Vitamin B2 (as Riboflavin Phosphate Sodium) 0.6 mg 75% Vitamin B12 (as Cyanocobalamin) 2 mcg 67% Niacin (as Niacinamide) 8 mg 89% Vitamin B6 (as Pyridoxine HCl) 0.4 mg 57% Fluoride (as Sodium Fluoride) 0.25 mg *Daily Value not established. See INDICATIONS AND USAGE section for use by children 6 months to 6 years of age. This product does not contain the essential vitamin folic acid. Active ingredient for caries p r o phy l ax i s : Fluoride as sodium fluoride. This product does not contain Folic Acid. Other ingredients: Cherry Flavor, Citric Acid Anhydrous, Glycerin, Orange Flavor, Potassium Sorbate, Propylene Glycol, Purified Water, Sodium Benzoate, Sorbitol Solution, Sucralose.
Section 42229-5
Multi-Vitamin Drops with Fluoride 0 .25 mg
Supplemental Facts
| Percentage of U.S. Recommended Daily Allowance | ||
| Each 1.0 mL s upp li e s : | Children 6 mos to 4 Years old | |
| Vitamin A (as Vitamin A Palmitate) | 1500 IU | 60% |
| Vitamin C (as Ascorbic Acid) | 35 mg | 88% |
| Vitamin D (as Cholecalciferol) | 400 IU | 100% |
| Vitamin E (as D-Alpha-Tocopheryl
Acid Succinate) |
5 IU | 50% |
| Vitamin B1 (as Thiamine HCl) | 0.5 mg | 71% |
| Vitamin B2 (as Riboflavin Phosphate Sodium)
|
0.6 mg | 75% |
| Vitamin B12 (as Cyanocobalamin) | 2 mcg | 67% |
| Niacin (as Niacinamide) | 8 mg | 89% |
| Vitamin B6 (as Pyridoxine HCl) | 0.4 mg | 57% |
| Fluoride (as Sodium Fluoride) | 0.25 mg |
*Daily Value not established.
See INDICATIONS AND USAGE section for use by children 6 months to 6 years of age.
This product does not contain the essential vitamin folic acid.
Active ingredient for caries p r o phy l ax i s : Fluoride as sodium fluoride. This product does not contain Folic Acid.
Other ingredients: Cherry Flavor, Citric Acid Anhydrous, Glycerin, Orange Flavor, Potassium Sorbate, Propylene Glycol, Purified Water, Sodium Benzoate, Sorbitol Solution, Sucralose.
Warnings
As in the case of all medications, keep out of reach of children.
References
- Brudevoid F, McCann HG: Fluoride and caries control - Mechanism of action, in Nizel AE (ed):
The
Science
of
Nutrition
and
its
Application
in
Clinical
Dentistry.
Philadelphia, WB Saunders Co, 1966, pp 331-347.
- American Academy of Pediatrics Committee on Nutrition: Fluoride supplementation,
Pediatrics 1986; 77:758.
- American Dental Association Council on Dental Therapeutics:
Accepted
Dental
Therapeutics, ed 38, Chicago, 1979, p321.
- Hennon DK, Stookey GK, Muhler JC: The clinical anticariogenic effectiveness of supplementary fluoride-vitamin preparations - Results at the end of three years.
J
Dent
Children
1966; 33 January: 3-12.
- Hennon DK, Stookey GK, Muhler JC: The clinical anticariogenic effectiveness of supplementary fluoride-vitamin preparations - Results at the end of four years.
J
Dent
Children
1967; 34 November; 439- 443.
- Hennon DK, Stookey GK, Muhler JC: The clinical anticariogenic effectiveness of supplementary fluoride-vitamin preparations - Results at the end of five and a half years.
Phar
and
Ther
in
Dent
1970; 1:1.
- Hennon DK, Stookey GK, Beiswanger BB: Fluoride-vitamin supplements: Effects on dental caries and fluorosis when used in areas with suboptimum fluoride in the water supply. J Am Dent Assoc 1977; 95-965
Distributed by:
Method Pharmaceuticals, LLC
Fort Worth, TX 76118
877-250-3427
Rev. 01/17
Made in the USA
Precautions
The suggested dose should not be exceeded since dental fluorosis may result from continued ingestion of large amounts of fluoride.
When prescribing vitamin fluoride products:
- Determine the fluoride content of the drinking water.
- Make sure the child is not receiving significant amounts of fluoride from other medications and swallowed toothpaste.
- Periodically check to make sure that the child does not develop significant dental fluorosis.
Multi-Vitamin Drops with Fluoride 0.25 mg should be dispensed in the original plastic container, since contact with glass leads to instability and precipitation. (The amount of sodium fluoride in the 50 mL size is well below the maximum to be dispensed at one time according to recommendations of the American Dental Association.)
Important Considerations When Using Dosage Schedule:
- If fluoride level is unknown, drinking water should be tested for fluoride content before supplements are prescribed. For testing of fluoride content, contact the local or state health department.
- All sources of fluoride should be evaluated with a thorough fluoride history. Patient exposure to multiple water sources can make proper prescribing complex.
- Ingestion of higher than recommended levels of fluoride by children has been associated with an increase in mild dental fluorosis in developing, unerupted teeth.
Fluoride supplements require long-term compliance on a daily basis.
Recommended Storage
Store at controlled room temperature 15°-25°C (between 59°F and 77°F). Excursions Permitted. After opening store away from direct light. Close tightly after each use. Occasional deepening of color has no significant effect on vitamin potency.
REFRIGERATION IS NOT REQUIRED.
SHAKE WELL.
Clinical Pharmacology
It is well established that fluoridation of the water supply (1 ppm fluoride) during the period of tooth development leads to a significant decrease in the incidence of dental caries.
Hydroxyapatite is the principal crystal for all calcified tissue in the human body. The fluoride ion reacts with hydroxyapatite in the tooth as it is formed to produce the more caries-resistant crystal, fluorapatite.
The reaction may be expressed by the equation:
Three stages of fluoride deposition in tooth enamel can be distinguished:
- Small amounts (reflecting the low levels of fluoride in tissue fluids) are incorporated into the enamel crystals while they are being formed.
- After enamel has been laid down, fluoride deposition continues in the surface enamel. Diffusion of fluoride from the surface inward is apparently restricted.
- After eruption, the surface enamel acquires fluoride from water, food, supplementary fluoride and smaller amounts from saliva.
Principal Display Panel
NDC 58657-325-50
Multi- Vitamin
Drops
With Fluoride
0.25 mg
1.69 FL. OZ. (50 mL)
Dosage and Administration:
See following chart. May be dropped directly into the mouth with dropper; or mixed with cereal, fruit juice or other food.
| Fluoride Ion Level in Drinking Water (ppm) * | |||
| A g e | <0.3 ppm | 0.3 - 0.6 ppm | >0.6 ppm |
| Birth - 6 months | None | None | None |
| 6 mos - 3 years | 0.25 mg (1 mL) / day † | None | None |
| 3 - 6 years | 0.50 mg (2 mL) / day | 0.25 mg (1 mL) / day | None |
* 1.0 ppm = 1 mg/liter
† 2.2 mg sodium fluoride contains 1 mg fluoride ion.
Structured Label Content
Section 42229-5 (42229-5)
Multi-Vitamin Drops with Fluoride 0 .25 mg
Supplemental Facts
| Percentage of U.S. Recommended Daily Allowance | ||
| Each 1.0 mL s upp li e s : | Children 6 mos to 4 Years old | |
| Vitamin A (as Vitamin A Palmitate) | 1500 IU | 60% |
| Vitamin C (as Ascorbic Acid) | 35 mg | 88% |
| Vitamin D (as Cholecalciferol) | 400 IU | 100% |
| Vitamin E (as D-Alpha-Tocopheryl
Acid Succinate) |
5 IU | 50% |
| Vitamin B1 (as Thiamine HCl) | 0.5 mg | 71% |
| Vitamin B2 (as Riboflavin Phosphate Sodium)
|
0.6 mg | 75% |
| Vitamin B12 (as Cyanocobalamin) | 2 mcg | 67% |
| Niacin (as Niacinamide) | 8 mg | 89% |
| Vitamin B6 (as Pyridoxine HCl) | 0.4 mg | 57% |
| Fluoride (as Sodium Fluoride) | 0.25 mg |
*Daily Value not established.
See INDICATIONS AND USAGE section for use by children 6 months to 6 years of age.
This product does not contain the essential vitamin folic acid.
Active ingredient for caries p r o phy l ax i s : Fluoride as sodium fluoride. This product does not contain Folic Acid.
Other ingredients: Cherry Flavor, Citric Acid Anhydrous, Glycerin, Orange Flavor, Potassium Sorbate, Propylene Glycol, Purified Water, Sodium Benzoate, Sorbitol Solution, Sucralose.
Warnings (WARNINGS)
As in the case of all medications, keep out of reach of children.
References (REFERENCES)
- Brudevoid F, McCann HG: Fluoride and caries control - Mechanism of action, in Nizel AE (ed):
The
Science
of
Nutrition
and
its
Application
in
Clinical
Dentistry.
Philadelphia, WB Saunders Co, 1966, pp 331-347.
- American Academy of Pediatrics Committee on Nutrition: Fluoride supplementation,
Pediatrics 1986; 77:758.
- American Dental Association Council on Dental Therapeutics:
Accepted
Dental
Therapeutics, ed 38, Chicago, 1979, p321.
- Hennon DK, Stookey GK, Muhler JC: The clinical anticariogenic effectiveness of supplementary fluoride-vitamin preparations - Results at the end of three years.
J
Dent
Children
1966; 33 January: 3-12.
- Hennon DK, Stookey GK, Muhler JC: The clinical anticariogenic effectiveness of supplementary fluoride-vitamin preparations - Results at the end of four years.
J
Dent
Children
1967; 34 November; 439- 443.
- Hennon DK, Stookey GK, Muhler JC: The clinical anticariogenic effectiveness of supplementary fluoride-vitamin preparations - Results at the end of five and a half years.
Phar
and
Ther
in
Dent
1970; 1:1.
- Hennon DK, Stookey GK, Beiswanger BB: Fluoride-vitamin supplements: Effects on dental caries and fluorosis when used in areas with suboptimum fluoride in the water supply. J Am Dent Assoc 1977; 95-965
Distributed by:
Method Pharmaceuticals, LLC
Fort Worth, TX 76118
877-250-3427
Rev. 01/17
Made in the USA
Precautions (PRECAUTIONS)
The suggested dose should not be exceeded since dental fluorosis may result from continued ingestion of large amounts of fluoride.
When prescribing vitamin fluoride products:
- Determine the fluoride content of the drinking water.
- Make sure the child is not receiving significant amounts of fluoride from other medications and swallowed toothpaste.
- Periodically check to make sure that the child does not develop significant dental fluorosis.
Multi-Vitamin Drops with Fluoride 0.25 mg should be dispensed in the original plastic container, since contact with glass leads to instability and precipitation. (The amount of sodium fluoride in the 50 mL size is well below the maximum to be dispensed at one time according to recommendations of the American Dental Association.)
Important Considerations When Using Dosage Schedule:
- If fluoride level is unknown, drinking water should be tested for fluoride content before supplements are prescribed. For testing of fluoride content, contact the local or state health department.
- All sources of fluoride should be evaluated with a thorough fluoride history. Patient exposure to multiple water sources can make proper prescribing complex.
- Ingestion of higher than recommended levels of fluoride by children has been associated with an increase in mild dental fluorosis in developing, unerupted teeth.
Fluoride supplements require long-term compliance on a daily basis.
How Supplied (HOW SUPPLIED)
Multi-Vitamin and Fluoride 0.25 mg drops is available in 50 mL bottles with accompanying calibrated dropper.
Adverse Reactions (ADVERSE REACTIONS)
Allergic rash and other idiosyncrasies have been rarely reported.
Recommended Storage (RECOMMENDED STORAGE)
Store at controlled room temperature 15°-25°C (between 59°F and 77°F). Excursions Permitted. After opening store away from direct light. Close tightly after each use. Occasional deepening of color has no significant effect on vitamin potency.
REFRIGERATION IS NOT REQUIRED.
SHAKE WELL.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
It is well established that fluoridation of the water supply (1 ppm fluoride) during the period of tooth development leads to a significant decrease in the incidence of dental caries.
Hydroxyapatite is the principal crystal for all calcified tissue in the human body. The fluoride ion reacts with hydroxyapatite in the tooth as it is formed to produce the more caries-resistant crystal, fluorapatite.
The reaction may be expressed by the equation:
Three stages of fluoride deposition in tooth enamel can be distinguished:
- Small amounts (reflecting the low levels of fluoride in tissue fluids) are incorporated into the enamel crystals while they are being formed.
- After enamel has been laid down, fluoride deposition continues in the surface enamel. Diffusion of fluoride from the surface inward is apparently restricted.
- After eruption, the surface enamel acquires fluoride from water, food, supplementary fluoride and smaller amounts from saliva.
Indications and Usage (INDICATIONS AND USAGE)
Supplementation of the diet with vitamins A, C and D.
Multi-Vitamin Drops with Fluoride 0.25 mg also provides fluoride for caries prophylaxis.
The American Academy of Pediatrics recommends that children up to age 16, in areas where drinking water contains less than optimal levels of fluoride, receive daily fluoride supplementation.
The American Academy of Pediatrics recommend that infants and young children 6 months to 3 years of age, in areas where the drinking water contains less than 0.3 ppm of fluoride, and children 3-6 years of age, in areas where the drinking water contains 0.3 through 0.6 ppm of fluoride, receive 0.25 mg of supplemental fluoride daily which is provided in a dose of 1 mL of Multi-Vitamin Drops with Fluoride 0.25 mg (See Dosage and Administration ).
Multi-Vitamin Drops with Fluoride 0.25 mg supply significant amounts of vitamins A, C and D to supplement the diet, and to help assure that nutritional deficiencies of these vitamins will not develop.
Thus, in a single easy-to-use preparation, children obtain essential vitamins and fluoride.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 58657-325-50
Multi- Vitamin
Drops
With Fluoride
0.25 mg
1.69 FL. OZ. (50 mL)
Dosage and Administration: (DOSAGE AND ADMINISTRATION:)
See following chart. May be dropped directly into the mouth with dropper; or mixed with cereal, fruit juice or other food.
| Fluoride Ion Level in Drinking Water (ppm) * | |||
| A g e | <0.3 ppm | 0.3 - 0.6 ppm | >0.6 ppm |
| Birth - 6 months | None | None | None |
| 6 mos - 3 years | 0.25 mg (1 mL) / day † | None | None |
| 3 - 6 years | 0.50 mg (2 mL) / day | 0.25 mg (1 mL) / day | None |
* 1.0 ppm = 1 mg/liter
† 2.2 mg sodium fluoride contains 1 mg fluoride ion.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:55.914839 · Updated: 2026-03-14T22:48:45.434128