These Highlights Do Not Include All The Information Needed To Use Ganciclovir For Injection safely And Effectively. See Full Prescribing Information For Ganciclovir For Injection.
87afd103-6e64-444f-971d-0e732e9ae995
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Hematologic Toxicity: Granulocytopenia, anemia, thrombocytopenia, and pancytopenia have been reported in patients treated with ganciclovir [see Warnings and Precautions (5.1) ]. Impairment of Fertility: Based on animal data and limited human data, ganciclovir may cause temporary or permanent inhibition of spermatogenesis in males and suppression of fertility in females [see Warnings and Precautions (5.3) ]. Fetal Toxicity: Based on animal data, ganciclovir has the potential to cause birth defects in humans [see Warnings and Precautions (5.4) ]. Mutagenesis and Carcinogenesis: Based on animal data, ganciclovir has the potential to cause cancers in humans [see Warnings and Precautions (5.5) ].
Indications and Usage
Ganciclovir is a deoxynucleoside analogue cytomegalovirus (CMV) DNA polymerase inhibitor indicated for the: treatment of CMV retinitis in immunocompromised adult patients, including patients with acquired immunodeficiency syndrome (AIDS). ( 1.1 ) prevention of CMV disease in adult transplant recipients at risk for CMV disease. ( 1.2 )
Dosage and Administration
Ganciclovir for injection is administered only intravenously. ( 2.1 ) Dosage in Adult Patients with Normal Renal Function Treatment of CMV retinitis ( 2.3 ) Induction: 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 14 to 21 days. Maintenance: 5 mg/kg (given intravenously at a constant rate over one hour) once daily for 7 days per week, or 6 mg/kg once daily for 5 days per week. Prevention of CMV disease in transplant recipients ( 2.4 ) Induction: 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 7 to 14 days. Maintenance: 5 mg/kg (given intravenously at a constant rate over one hour) once daily, 7 days per week, or 6 mg/kg once daily, 5 days per week until 100 to 120 days post-transplantation. Adults with renal impairment: Adjust dosage based on creatinine clearance. ( 2.5 ).
Warnings and Precautions
Renal Impairment: Increased serum creatinine levels have been observed with the use of ganciclovir, particularly in elderly patients and transplant recipients receiving concomitant nephrotoxic drugs. Monitor renal function during therapy with ganciclovir, particularly in elderly patients and in patients taking other nephrotoxic drugs, and reduce dosage in patients with renal impairment. ( 5.2 )
Contraindications
Ganciclovir for Injection, USP is contraindicated in patients who have experienced a clinically significant hypersensitivity reaction (e.g., anaphylaxis) to ganciclovir, valganciclovir, or any component of the formulation.
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: Hematologic Toxicity [see Warnings and Precautions (5.1) ] Renal Impairment [see Warnings and Precautions (5.2) ] Impairment of Fertility [see Warnings and Precautions (5.3) ] Fetal Toxicity [see Warnings and Precautions (5.4) ] Mutagenesis and Carcinogenesis [see Warnings and Precautions (5.5) ]
Drug Interactions
Drug-drug interaction studies were conducted in patients with normal renal function. Patients with impaired renal function may have increased concentrations of ganciclovir and the coadministered drug following concomitant administration of ganciclovir and drugs excreted by the same pathway as ganciclovir. Therefore, these patients should be closely monitored for toxicity of ganciclovir and the coadministered drug. Established and other potentially significant drug interactions conducted with ganciclovir are listed in Table 6 [see Clinical Pharmacology (12.3) ]. Table 6. Established and Other Potentially Significant Drug Interactions with Ganciclovir Name of the Concomitant Drug Change in the Concentration of Ganciclovir or Concomitant Drug Clinical Comment Imipenem-cilastatin Unknown Coadministration with imipenem-cilastatin is not recommended because generalized seizures have been reported in patients who received ganciclovir and imipenem-cilastatin. Cyclosporine or amphotericin B Unknown Monitor renal function when ganciclovir is coadministered with cyclosporine or amphotericin B because of potential increase in serum creatinine [see Warnings and Precautions (5.2) ] . Mycophenolate mofetil (MMF) ↔ Ganciclovir (in patients with normal renal function) ↔ MMF (in patients with normal renal function) Based on increased risk, patients should be monitored for hematological and renal toxicity. Other drugs associated with myelosuppresion or nephrotoxicity (e.g., dapsone, doxorubicin, flucytosine, hydroxyurea, pentamidine, tacrolimus, trimethoprim/ sulfamethoxazole, vinblastine, vincristine and zidovudine) Unknown Because of potential for higher toxicity, coadministration with ganciclovir should be considered only if the potential benefits are judged to outweigh the risks. Didanosine ↔ Ganciclovir ↑ Didanosine Patients should be closely monitored for didanosine toxicity (e.g., pancreatitis). Probenecid ↑ Ganciclovir Ganciclovir dose may need to be reduced. Monitor for evidence of ganciclovir toxicity.
Medication Information
Warnings and Precautions
Renal Impairment: Increased serum creatinine levels have been observed with the use of ganciclovir, particularly in elderly patients and transplant recipients receiving concomitant nephrotoxic drugs. Monitor renal function during therapy with ganciclovir, particularly in elderly patients and in patients taking other nephrotoxic drugs, and reduce dosage in patients with renal impairment. ( 5.2 )
Indications and Usage
Ganciclovir is a deoxynucleoside analogue cytomegalovirus (CMV) DNA polymerase inhibitor indicated for the: treatment of CMV retinitis in immunocompromised adult patients, including patients with acquired immunodeficiency syndrome (AIDS). ( 1.1 ) prevention of CMV disease in adult transplant recipients at risk for CMV disease. ( 1.2 )
Dosage and Administration
Ganciclovir for injection is administered only intravenously. ( 2.1 ) Dosage in Adult Patients with Normal Renal Function Treatment of CMV retinitis ( 2.3 ) Induction: 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 14 to 21 days. Maintenance: 5 mg/kg (given intravenously at a constant rate over one hour) once daily for 7 days per week, or 6 mg/kg once daily for 5 days per week. Prevention of CMV disease in transplant recipients ( 2.4 ) Induction: 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 7 to 14 days. Maintenance: 5 mg/kg (given intravenously at a constant rate over one hour) once daily, 7 days per week, or 6 mg/kg once daily, 5 days per week until 100 to 120 days post-transplantation. Adults with renal impairment: Adjust dosage based on creatinine clearance. ( 2.5 ).
Contraindications
Ganciclovir for Injection, USP is contraindicated in patients who have experienced a clinically significant hypersensitivity reaction (e.g., anaphylaxis) to ganciclovir, valganciclovir, or any component of the formulation.
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: Hematologic Toxicity [see Warnings and Precautions (5.1) ] Renal Impairment [see Warnings and Precautions (5.2) ] Impairment of Fertility [see Warnings and Precautions (5.3) ] Fetal Toxicity [see Warnings and Precautions (5.4) ] Mutagenesis and Carcinogenesis [see Warnings and Precautions (5.5) ]
Drug Interactions
Drug-drug interaction studies were conducted in patients with normal renal function. Patients with impaired renal function may have increased concentrations of ganciclovir and the coadministered drug following concomitant administration of ganciclovir and drugs excreted by the same pathway as ganciclovir. Therefore, these patients should be closely monitored for toxicity of ganciclovir and the coadministered drug. Established and other potentially significant drug interactions conducted with ganciclovir are listed in Table 6 [see Clinical Pharmacology (12.3) ]. Table 6. Established and Other Potentially Significant Drug Interactions with Ganciclovir Name of the Concomitant Drug Change in the Concentration of Ganciclovir or Concomitant Drug Clinical Comment Imipenem-cilastatin Unknown Coadministration with imipenem-cilastatin is not recommended because generalized seizures have been reported in patients who received ganciclovir and imipenem-cilastatin. Cyclosporine or amphotericin B Unknown Monitor renal function when ganciclovir is coadministered with cyclosporine or amphotericin B because of potential increase in serum creatinine [see Warnings and Precautions (5.2) ] . Mycophenolate mofetil (MMF) ↔ Ganciclovir (in patients with normal renal function) ↔ MMF (in patients with normal renal function) Based on increased risk, patients should be monitored for hematological and renal toxicity. Other drugs associated with myelosuppresion or nephrotoxicity (e.g., dapsone, doxorubicin, flucytosine, hydroxyurea, pentamidine, tacrolimus, trimethoprim/ sulfamethoxazole, vinblastine, vincristine and zidovudine) Unknown Because of potential for higher toxicity, coadministration with ganciclovir should be considered only if the potential benefits are judged to outweigh the risks. Didanosine ↔ Ganciclovir ↑ Didanosine Patients should be closely monitored for didanosine toxicity (e.g., pancreatitis). Probenecid ↑ Ganciclovir Ganciclovir dose may need to be reduced. Monitor for evidence of ganciclovir toxicity.
Description
Hematologic Toxicity: Granulocytopenia, anemia, thrombocytopenia, and pancytopenia have been reported in patients treated with ganciclovir [see Warnings and Precautions (5.1) ]. Impairment of Fertility: Based on animal data and limited human data, ganciclovir may cause temporary or permanent inhibition of spermatogenesis in males and suppression of fertility in females [see Warnings and Precautions (5.3) ]. Fetal Toxicity: Based on animal data, ganciclovir has the potential to cause birth defects in humans [see Warnings and Precautions (5.4) ]. Mutagenesis and Carcinogenesis: Based on animal data, ganciclovir has the potential to cause cancers in humans [see Warnings and Precautions (5.5) ].
Section 42229-5
Induction Dosage: The recommended initial dosage of ganciclovir for patients with normal renal function is 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 14 to 21 days.
Section 43679-0
Mechanism of Action
Ganciclovir is a synthetic analogue of 2'-deoxyguanosine, which inhibits replication of human CMV in cell culture and in vivo. In CMV-infected cells, ganciclovir is initially phosphorylated to ganciclovir monophosphate by the viral protein kinase, pUL97. Further phosphorylation occurs by cellular kinases to produce ganciclovir triphosphate, which is then slowly metabolized intracellularly. As the phosphorylation is largely dependent on the viral kinase, phosphorylation of ganciclovir occurs preferentially in virus-infected cells. The virustatic activity of ganciclovir is due to inhibition of the viral DNA polymerase, pUL54, by ganciclovir triphosphate.
Section 44425-7
Storage
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Protect from moisture.
Store reconstituted solution in the vial at 25°C (77°F) for no longer than 12 hours. Do not refrigerate or freeze.
Store diluted infusion solution under refrigeration at 2° to 8°C (36° to 46°F) for no longer than 24 hours. Do not freeze.
10 Overdosage
Reports of adverse reactions after overdoses with ganciclovir, some with fatal outcomes, have been received from clinical trials and during postmarketing experience. One or more of the following adverse reactions has been reported with overdoses:
Hematological toxicity: myelosuppression including pancytopenia, leukopenia, neutropenia, granulocytopenia, thrombocytopenia, bone marrow failure
Hepatotoxicity: hepatitis, liver function disorder
Renal toxicity: worsening of hematuria in a patient with preexisting renal impairment, acute kidney injury, elevated creatinine
Gastrointestinal toxicity: abdominal pain, diarrhea, vomiting
Neurotoxicity: seizure
Since ganciclovir is dialyzable, dialysis may be useful in reducing serum concentrations in patients who have received an overdose of ganciclovir [see Clinical Pharmacology (12.3)]. Adequate hydration should be maintained. The use of hematopoietic growth factors should be considered in patients with cytopenias [see Warnings and Precautions (5.1)].
15 References
1. "OSHA Hazardous Drugs." OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html .
11 Description
Ganciclovir for Injection contains ganciclovir, in the form of the sodium salt for intravenous injection. Ganciclovir is a synthetic guanine derivative active against cytomegalovirus (CMV).
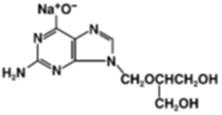
Chemically, ganciclovir is 9-[[2-hydroxy-1-(hydroxymethyl)-ethoxy]methyl]guanine and ganciclovir sodium is 9-[[2-hydroxy-1-(hydroxymethyl)-ethoxy]methyl]guanine, monosodium salt. The chemical structures of ganciclovir sodium and ganciclovir are:
| ganciclovir sodium C9H12N5NaO4, M.W.=277.22 |
ganciclovir C9H13N5O4 M.W.=255.23 |
Ganciclovir is a white to off-white crystalline powder. Ganciclovir is a polar hydrophilic compound with a solubility of 2.6 mg/mL in water at 25°C and an n-octanol/water partition coefficient of 0.022. The pKas for ganciclovir are 2.2 and 9.4.
Ganciclovir for Injection USP, formulated as monosodium salt, using sodium hydroxide as a salt forming agent, is a sterile white to off-white lyophilized powder. The lyophilized powder has an aqueous solubility of greater than 50 mg/mL at 25°C. At physiological pH, ganciclovir sodium exists as the un-ionized form with a solubility of approximately 6 mg/mL at 37°C.
Each vial contains ganciclovir sodium equivalent to 500 mg ganciclovir.
Inactive ingredients may include hydrochloric acid (QS) and sodium hydroxide (QS) added to adjust the pH.
All doses in this package insert are specified in terms of ganciclovir.
8.4 Pediatric Use
Safety and efficacy of ganciclovir have not been established in pediatric patients.
A total of 120 pediatric patients with serious CMV infections participated in clinical trials. Granulocytopenia and thrombocytopenia were the most common adverse reactions. The pharmacokinetic characteristics of ganciclovir after administration of ganciclovir for injection were studied in 27 neonates (aged 2 to 49 days) and ten pediatric patients, aged 9 months to 12 years. In neonates, the pharmacokinetic parameters after ganciclovir intravenous doses of 4 mg/kg (n = 14) and 6 mg/kg (n = 13) were Cmax 5.5 ± 1.6 and 7.0 ± 1.6 mcg/mL, systemic clearance 3.14 ± 1.75 and 3.56 ± 1.27 mL/min/kg, and t1/2 of 2.4 hours (harmonic mean) for both doses, respectively.
In pediatric patients 9 months to 12 years of age, the pharmacokinetic characteristics of ganciclovir were the same after single and multiple (every 12 hours) intravenous doses (5 mg/kg). The steady-state volume of distribution was 0.64 ± 0.22 L/kg, Cmax was 7.9 ± 3.9 mcg/mL, systemic clearance was 4.7 ± 2.2 mL/min/kg, and t1/2 was 2.4 ± 0.7 hours.
Although the pharmacokinetics of ganciclovir in pediatric patients were similar to those observed in adults, the safety and efficacy of ganciclovir at these exposures in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ganciclovir did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. Ganciclovir is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because renal clearance decreases with age, ganciclovir should be administered to elderly patients with special consideration of their renal status. Renal function should be monitored and dosage adjustments should be made accordingly [see Dosage and Administration (2.5), Warnings and Precautions (5.2), Use in Specific Populations (8.6)].
5.4 Fetal Toxicity
Ganciclovir may cause fetal toxicity when administered to pregnant women based on findings in animal studies. Systemic exposure of ganciclovir in animals at approximately 2 times the RHD caused fetal growth retardation, embryolethality, teratogenicity, and/or maternal toxicity. Teratogenic changes in animals included cleft palate, anophthalmia/microphthalmia, aplastic organs (kidney and pancreas), hydrocephaly and brachygnathia. Women of childbearing potential should be advised to use effective contraception during treatment and for at least 30 days following treatment with ganciclovir. Similarly, men should be advised to practice barrier contraception during and for at least 90 days following treatment with ganciclovir [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
4 Contraindications
Ganciclovir for Injection, USP is contraindicated in patients who have experienced a clinically significant hypersensitivity reaction (e.g., anaphylaxis) to ganciclovir, valganciclovir, or any component of the formulation.
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Hematologic Toxicity [see Warnings and Precautions (5.1)]
- Renal Impairment [see Warnings and Precautions (5.2)]
- Impairment of Fertility [see Warnings and Precautions (5.3)]
- Fetal Toxicity [see Warnings and Precautions (5.4)]
- Mutagenesis and Carcinogenesis [see Warnings and Precautions (5.5)]
7 Drug Interactions
Drug-drug interaction studies were conducted in patients with normal renal function. Patients with impaired renal function may have increased concentrations of ganciclovir and the coadministered drug following concomitant administration of ganciclovir and drugs excreted by the same pathway as ganciclovir. Therefore, these patients should be closely monitored for toxicity of ganciclovir and the coadministered drug.
Established and other potentially significant drug interactions conducted with ganciclovir are listed in Table 6 [see Clinical Pharmacology (12.3)].
| Name of the Concomitant Drug | Change in the Concentration of Ganciclovir or Concomitant Drug | Clinical Comment |
| Imipenem-cilastatin | Unknown | Coadministration with imipenem-cilastatin is not recommended because generalized seizures have been reported in patients who received ganciclovir and imipenem-cilastatin. |
| Cyclosporine or amphotericin B | Unknown | Monitor renal function when ganciclovir is coadministered with cyclosporine or amphotericin B because of potential increase in serum creatinine [see Warnings and Precautions (5.2)]. |
| Mycophenolate mofetil (MMF) |
↔ Ganciclovir (in patients with normal renal function) ↔ MMF (in patients with normal renal function) |
Based on increased risk, patients should be monitored for hematological and renal toxicity. |
| Other drugs associated with myelosuppresion or nephrotoxicity (e.g., dapsone, doxorubicin, flucytosine, hydroxyurea, pentamidine, tacrolimus, trimethoprim/ sulfamethoxazole, vinblastine, vincristine and zidovudine) | Unknown | Because of potential for higher toxicity, coadministration with ganciclovir should be considered only if the potential benefits are judged to outweigh the risks. |
| Didanosine |
↔ Ganciclovir ↑ Didanosine |
Patients should be closely monitored for didanosine toxicity (e.g., pancreatitis). |
| Probenecid | ↑ Ganciclovir | Ganciclovir dose may need to be reduced. Monitor for evidence of ganciclovir toxicity. |
5.2 Renal Impairment
Ganciclovir should be used with caution in patients with impaired renal function because the half-life and plasma/serum concentrations of ganciclovir will be increased due to reduced renal clearance. If renal function is impaired, dosage adjustments are recommended [see Dosage and Administration (2.5), Use in Specific Populations (8.5, 8.6)].
Increased serum creatinine levels have been reported in elderly patients and in transplant recipients receiving concomitant nephrotoxic medications (i.e., cyclosporine and amphotericin B). Monitoring renal function during therapy with ganciclovir is essential, especially for elderly patients and those patients receiving concomitant agents that may cause nephrotoxicity [see Dosage and Administration (2.5), Drug Interactions (7), Use in Specific Populations (8.5)].
8.6 Renal Impairment
Dose reduction is recommended when administering ganciclovir to patients with renal impairment [see Dosage and Administration (2.5), Warnings and Precautions (5.2)].
8.7 Hepatic Impairment
The safety and efficacy of ganciclovir have not been studied in patients with hepatic impairment.
1 Indications and Usage
Ganciclovir is a deoxynucleoside analogue cytomegalovirus (CMV) DNA polymerase inhibitor indicated for the:
12.1 Mechanism of Action
Ganciclovir is an antiviral drug with activity against CMV [see Microbiology (12.4)].
5.1 Hematologic Toxicity
Granulocytopenia (neutropenia), anemia, thrombocytopenia and pancytopenia, have been observed in patients treated with ganciclovir. The frequency and severity of these events vary widely in different patient populations [see Adverse Reactions (6.1)]. Ganciclovir is not recommended if the absolute neutrophil count is less than 500 cells/µL, hemoglobin is less than 8 g/dL, or the platelet count is less than 25,000 cells/µL. Ganciclovir should also be used with caution in patients with preexisting cytopenias and in patients receiving myelosuppressive drugs or irradiation. Granulocytopenia (neutropenia) usually occurs during the first or second week of treatment but may occur at any time during treatment. Cell counts usually begin to recover within 3 to 7 days after discontinuing drug. Colony-stimulating factors have been shown to increase neutrophil and white blood cell counts in patients receiving ganciclovir solution for treatment of CMV retinitis.
Due to the frequency of neutropenia, anemia and thrombocytopenia in patients receiving ganciclovir [see Adverse Reactions (6.1)], complete blood counts with differential and platelet counts should be performed frequently in all patients, especially in patients with renal impairment and in patients in whom ganciclovir or other nucleoside analogues have previously resulted in leukopenia, or in whom neutrophil counts are less than 1000 cells/µL at the beginning of treatment [see Dosage and Administration (2.2)].
2.7 Handling and Disposal
Caution should be exercised in the handling and preparation of solutions of ganciclovir. Solutions of ganciclovir are alkaline (pH 11). Avoid direct contact of the skin or mucous membranes with ganciclovir solution. If such contact occurs, wash thoroughly with soap and water; rinse eyes thoroughly with plain water. Wearing disposable gloves is recommended.
Because ganciclovir shares some of the properties of antitumor agents (i.e., carcinogenicity and mutagenicity), consideration should be given to handling and disposal according to guidelines issued for antineoplastic drugs1 [see How Supplied/Storage and Handling (16)].
5 Warnings and Precautions
- Renal Impairment: Increased serum creatinine levels have been observed with the use of ganciclovir, particularly in elderly patients and transplant recipients receiving concomitant nephrotoxic drugs. Monitor renal function during therapy with ganciclovir, particularly in elderly patients and in patients taking other nephrotoxic drugs, and reduce dosage in patients with renal impairment. (5.2)
2 Dosage and Administration
- Ganciclovir for injection is administered only intravenously. (2.1)
| Dosage in Adult Patients with Normal Renal Function | |
|---|---|
| Treatment of CMV retinitis (2.3) | Induction: 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 14 to 21 days. Maintenance: 5 mg/kg (given intravenously at a constant rate over one hour) once daily for 7 days per week, or 6 mg/kg once daily for 5 days per week. |
| Prevention of CMV disease in transplant recipients (2.4) | Induction: 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 7 to 14 days. Maintenance: 5 mg/kg (given intravenously at a constant rate over one hour) once daily, 7 days per week, or 6 mg/kg once daily, 5 days per week until 100 to 120 days post-transplantation. |
Adults with renal impairment: Adjust dosage based on creatinine clearance. (2.5).
5.3 Impairment of Fertility
Based on animal data and limited human data, ganciclovir at the recommended human dose (RHD) may cause temporary or permanent inhibition of spermatogenesis in males, and may cause suppression of fertility in females. Advise patients that fertility may be impaired with the use of ganciclovir [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
3 Dosage Forms and Strengths
For injection: Single dose vial containing 500 mg of ganciclovir as a sterile lyophilized white to off-white powder for reconstitution with 10 mL of preservative-free Sterile Water for Injection for intravenous use [see Dosage and Administration (2.6)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ganciclovir for injection or ganciclovir capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic disorders: hemolytic anemia, agranulocytosis, granulocytopenia
Cardiac disorders: cardiac arrest, conduction disorder, torsade de pointes, ventricular tachycardia
Congenital, familial and genetic disorders: congenital anomaly
Endocrine disorders: inappropriate antidiuretic hormone secretion
Eye disorders: cataracts, dry eyes
Gastrointestinal disorders: intestinal ulcer
Hepatobiliary disorders: cholelithiasis, cholestasis, hepatic failure, hepatitis
Immune system disorders: anaphylactic reaction, allergic reaction, vasculitis
Investigations: blood triglycerides increased
Metabolism and nutrition disorders: acidosis, hypercalcemia, hyponatremia
Musculoskeletal and connective tissue disorders: arthritis, rhabdomyolysis
Nervous system disorders: dysesthesia, dysphasia, extrapyramidal disorder, facial paralysis, amnesia, anosmia, myelopathy, cerebrovascular accident, third cranial nerve paralysis, aphasia, encephalopathy, intracranial hypertension
Psychiatric disorders: irritability, hallucinations
Renal and urinary disorders: renal tubular disorder, hemolytic uremic syndrome
Reproductive system and breast disorders: infertility, testicular hypotrophy
Respiratory, thoracic and mediastinal disorders: bronchospasm, pulmonary fibrosis
Skin and subcutaneous tissues disorders: exfoliative dermatitis, Stevens-Johnson syndrome
Vascular disorders: peripheral ischemia
8 Use in Specific Populations
- Lactation: Breastfeeding is not recommended with use of ganciclovir. (8.2)
1.1 Treatment of Cmv Retinitis
Ganciclovir is indicated for the treatment of cytomegalovirus (CMV) retinitis in immunocompromised adult patients, including patients with acquired immunodeficiency syndrome (AIDS) [see Clinical Studies (14.1)].
14.1 Treatment of Cmv Retinitis
In a retrospective, non-randomized, single-center analysis of 41 patients with AIDS and CMV retinitis diagnosed by ophthalmologic examination between August 1983 and April 1988, treatment with ganciclovir for injection solution resulted in a delay in mean (median) time to first retinitis progression compared to untreated controls [105 (71) days from diagnosis vs. 35 (29) days from diagnosis]. Patients in this series received induction treatment of ganciclovir for injection 5 mg/kg twice daily for 14 to 21 days followed by maintenance treatment with either 5 mg/kg once daily, 7 days per week or 6 mg/kg once daily, 5 days per week.
In a controlled, randomized study conducted between February 1989 and December 1990, immediate treatment with ganciclovir was compared to delayed treatment in 42 patients with AIDS and peripheral CMV retinitis; 35 of 42 patients (13 in the immediate-treatment group and 22 in the delayed-treatment group) were included in the analysis of time to retinitis progression. Based on masked assessment of fundus photographs, the mean [95% CI] and median [95% CI] times to progression of retinitis were 66 days [39, 94] and 50 days [40, 84], respectively, in the immediate-treatment group compared to 19 days [11, 27] and 13.5 days [8, 18], respectively, in the delayed-treatment group.
Data from trials ICM 1653, ICM 1774, and AVI 034, which were performed comparing ganciclovir for injection to oral ganciclovir for treatment of CMV retinitis in patients with AIDS, are shown in Table 12 and Figures 1, 2, and 3, and are discussed below.
|
Demographics |
ICM 1653 (n = 121) |
ICM 1774 (n = 225) |
AVI 034 (n = 159) |
|
|
Median age (years) Range |
38 24 to 62 |
37 22 to 56 |
39 23 to 62 |
|
|
Sex |
Males |
116 (96%) |
222 (99%) |
148 (93%) |
|
Females |
5 (4%) |
3 (1%) |
10 (6%) |
|
|
Ethnicity |
Asian |
3 (3%) |
5 (2%) |
7 (4%) |
|
Black |
11 (9%) |
9 (4%) |
3 (2%) |
|
|
Caucasian |
98 (81%) |
186 (83%) |
140 (88%) |
|
|
Other |
9 (7%) |
25 (11%) |
8 (5%) |
|
|
Median CD4 Count Range |
9.5 0 to 141 |
7.0 0 to 80 |
10.0 0 to 320 |
|
|
Mean (SD) Observation Time (days) |
107.9 (43.0) |
97.6 (42.5) |
80.9 (47.0) |
5.5 Mutagenesis and Carcinogenesis
Animal data indicate that ganciclovir is mutagenic and carcinogenic. Ganciclovir should therefore be considered a potential carcinogen in humans [see Dosage and Administration (2.7), Nonclinical Toxicology (13.1)].
2.2 Testing Before and During Treatment
- Females of reproductive potential should undergo pregnancy testing before initiation of treatment with ganciclovir [see Warnings and Precautions (5.4), Use in Specific Populations (8.1, 8.3)].
- Complete blood counts with differential and platelet counts should be performed frequently, especially in patients in whom ganciclovir or other nucleoside analogues have previously resulted in cytopenias, or in whom absolute neutrophil counts are less than 1000 cells/μL at the beginning of treatment [see Warnings and Precautions (5.1)].
- All patients should be monitored for renal function before and during treatment with ganciclovir and dose should be adjusted as needed [see Dosage and Administration (2.5), Warnings and Precautions (5.2)].
- Patients with CMV retinitis should have frequent ophthalmological examinations during treatment with ganciclovir solution to monitor disease status and for other retinal abnormalities [see Adverse Reactions (6.1)].
Principal Display Panel 500 Mg Carton
2.6 Preparation of Ganciclovir for Injection
Ganciclovir for injection must be reconstituted and diluted under the supervision of a healthcare provider and administered as intravenous infusion. Each 10 mL clear glass vial contains ganciclovir sodium equivalent to 500 mg of ganciclovir. Wearing disposable gloves is recommended during reconstitution and when wiping the outer surface of the vial and the table after reconstitution. The contents of the vial should be prepared for administration in the following manner:
- Reconstitution Instructions:
a) Reconstitute lyophilized ganciclovir for injection by injecting 10 mL of Sterile Water for Injection, USP, into the vial. Do not use bacteriostatic water for injection containing parabens. It is incompatible with ganciclovir for injection and may cause precipitation.
b) Gently swirl the vial in order to ensure complete wetting of the product. Continue swirling until a clear reconstituted solution is obtained.
c) Visually inspect the reconstituted solution for particulate matter and discoloration prior to proceeding with infusion. Discard the vial if particulate matter or discoloration is observed.
d) Reconstituted solution in the vial is stable at room temperature (25°C) for 12 hours. Do not refrigerate or freeze. - Infusion Instructions:
a) Based on patient weight, the appropriate volume of the reconstituted solution (ganciclovir concentration 50 mg/mL) should be removed from the vial and added to an acceptable infusion fluid (typically 100 mL) for delivery over the course of one hour. Infusion concentrations greater than 10 mg/mL are not recommended. The following infusion fluids have been determined to be chemically and physically compatible with ganciclovir for injection solution: 0.9% Sodium Chloride, 5% Dextrose, Ringer's Injection and Lactated Ringer's Injection, USP.
b) Ganciclovir for injection, when reconstituted with Sterile Water for Injection (non-bacteriostatic) and further diluted with 0.9% sodium chloride injection or other acceptable infusion fluid as specified above, should be used within 24 hours of dilution to reduce the risk of bacterial contamination. The diluted infusion solution should be refrigerated (2°C to 8°C). Do not freeze.
6.1 Clinical Trial Experience in Adult Patients
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect rates observed in practice. The most common adverse reactions and laboratory abnormalities reported in at least 20% of patients were pyrexia, diarrhea, leukopenia, nausea, anemia, asthenia, headache, cough, decreased appetite, dyspnea, abdominal pain, sepsis, hyperhidrosis, and blood creatinine increased.
Selected adverse reactions that occurred during clinical trials of ganciclovir for injection are summarized below, according to the participating study patient population.
2.1 Important Dosing and Administration Information
- To avoid phlebitis/pain at the infusion site, ganciclovir for injection must only be administered by intravenous infusion over one hour, preferably via plastic cannula, into a vein with adequate blood flow to permit rapid dilution and distribution.
- Do not administer ganciclovir for injection by rapid or bolus intravenous injection which may increase toxicity as a result of excessive plasma levels.
- The recommended dosage and infusion rate for ganciclovir should not be exceeded.
- Do not administer the reconstituted ganciclovir for injection solution intramuscularly or subcutaneously because it may result in severe tissue irritation due to high pH [see Description (11)].
- Administration of ganciclovir should be accompanied by adequate hydration.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
1.2 Prevention of Cmv Disease in Transplant Recipients
Ganciclovir is indicated for the prevention of CMV disease in adult transplant recipients at risk for CMV disease [see Clinical Studies (14.2)].
14.2 Prevention of Cmv Disease in Transplant Recipients
Ganciclovir was evaluated in three randomized, controlled trials of prevention of CMV disease in organ transplant recipients.
2.5 Recommended Dosage in Adult Patients With Renal Impairment
For patients with impairment of renal function, refer to Table 1 for recommended doses of ganciclovir for induction and maintenance dosage for treatment of CMV retinitis and prevention of CMV disease in transplant recipients. Carefully monitor serum creatinine or creatinine clearance before and during treatment to allow for dosage adjustments in patients with impaired renal function.
| Creatinine Clearance Creatinine clearance can be related to serum creatinine by the formulas given below. (mL/min) |
Ganciclovir Induction Dose (mg/kg) |
Dosing Interval (hours) for Induction | Ganciclovir Maintenance Dose (mg/kg) | Dosing Interval (hours) for Maintenance |
| Greater than or equal to 70 | 5 | 12 | 5 | 24 |
| 50 to 69 | 2.5 | 12 | 2.5 | 24 |
| 25 to 49 | 2.5 | 24 | 1.25 | 24 |
| 10 to 24 | 1.25 | 24 | 0.625 | 24 |
| Less than 10 | 1.25 | 3 times per week, following hemodialysis | 0.625 | 3 times per week, following hemodialysis |
| Creatinine clearance for males = | (140 - age [yrs]) (body wt [kg]) |
| (72) (serum creatinine [mg/dL]) |
Creatinine clearance for females = 0.85 × male value
Warning: Hematologic Toxicity, Impairment of Fertility, Fetal Toxicity, Mutagenesis and Carcinogenesis
- Hematologic Toxicity: Granulocytopenia, anemia, thrombocytopenia, and pancytopenia have been reported in patients treated with ganciclovir [see Warnings and Precautions (5.1)].
- Impairment of Fertility: Based on animal data and limited human data, ganciclovir may cause temporary or permanent inhibition of spermatogenesis in males and suppression of fertility in females [see Warnings and Precautions (5.3)].
- Fetal Toxicity: Based on animal data, ganciclovir has the potential to cause birth defects in humans [see Warnings and Precautions (5.4)].
- Mutagenesis and Carcinogenesis: Based on animal data, ganciclovir has the potential to cause cancers in humans [see Warnings and Precautions (5.5)].
Structured Label Content
Section 42229-5 (42229-5)
Induction Dosage: The recommended initial dosage of ganciclovir for patients with normal renal function is 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 14 to 21 days.
Section 43679-0 (43679-0)
Mechanism of Action
Ganciclovir is a synthetic analogue of 2'-deoxyguanosine, which inhibits replication of human CMV in cell culture and in vivo. In CMV-infected cells, ganciclovir is initially phosphorylated to ganciclovir monophosphate by the viral protein kinase, pUL97. Further phosphorylation occurs by cellular kinases to produce ganciclovir triphosphate, which is then slowly metabolized intracellularly. As the phosphorylation is largely dependent on the viral kinase, phosphorylation of ganciclovir occurs preferentially in virus-infected cells. The virustatic activity of ganciclovir is due to inhibition of the viral DNA polymerase, pUL54, by ganciclovir triphosphate.
Section 44425-7 (44425-7)
Storage
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Protect from moisture.
Store reconstituted solution in the vial at 25°C (77°F) for no longer than 12 hours. Do not refrigerate or freeze.
Store diluted infusion solution under refrigeration at 2° to 8°C (36° to 46°F) for no longer than 24 hours. Do not freeze.
10 Overdosage (10 OVERDOSAGE)
Reports of adverse reactions after overdoses with ganciclovir, some with fatal outcomes, have been received from clinical trials and during postmarketing experience. One or more of the following adverse reactions has been reported with overdoses:
Hematological toxicity: myelosuppression including pancytopenia, leukopenia, neutropenia, granulocytopenia, thrombocytopenia, bone marrow failure
Hepatotoxicity: hepatitis, liver function disorder
Renal toxicity: worsening of hematuria in a patient with preexisting renal impairment, acute kidney injury, elevated creatinine
Gastrointestinal toxicity: abdominal pain, diarrhea, vomiting
Neurotoxicity: seizure
Since ganciclovir is dialyzable, dialysis may be useful in reducing serum concentrations in patients who have received an overdose of ganciclovir [see Clinical Pharmacology (12.3)]. Adequate hydration should be maintained. The use of hematopoietic growth factors should be considered in patients with cytopenias [see Warnings and Precautions (5.1)].
15 References (15 REFERENCES)
1. "OSHA Hazardous Drugs." OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html .
11 Description (11 DESCRIPTION)
Ganciclovir for Injection contains ganciclovir, in the form of the sodium salt for intravenous injection. Ganciclovir is a synthetic guanine derivative active against cytomegalovirus (CMV).
Chemically, ganciclovir is 9-[[2-hydroxy-1-(hydroxymethyl)-ethoxy]methyl]guanine and ganciclovir sodium is 9-[[2-hydroxy-1-(hydroxymethyl)-ethoxy]methyl]guanine, monosodium salt. The chemical structures of ganciclovir sodium and ganciclovir are:
| ganciclovir sodium C9H12N5NaO4, M.W.=277.22 |
ganciclovir C9H13N5O4 M.W.=255.23 |
Ganciclovir is a white to off-white crystalline powder. Ganciclovir is a polar hydrophilic compound with a solubility of 2.6 mg/mL in water at 25°C and an n-octanol/water partition coefficient of 0.022. The pKas for ganciclovir are 2.2 and 9.4.
Ganciclovir for Injection USP, formulated as monosodium salt, using sodium hydroxide as a salt forming agent, is a sterile white to off-white lyophilized powder. The lyophilized powder has an aqueous solubility of greater than 50 mg/mL at 25°C. At physiological pH, ganciclovir sodium exists as the un-ionized form with a solubility of approximately 6 mg/mL at 37°C.
Each vial contains ganciclovir sodium equivalent to 500 mg ganciclovir.
Inactive ingredients may include hydrochloric acid (QS) and sodium hydroxide (QS) added to adjust the pH.
All doses in this package insert are specified in terms of ganciclovir.
8.4 Pediatric Use
Safety and efficacy of ganciclovir have not been established in pediatric patients.
A total of 120 pediatric patients with serious CMV infections participated in clinical trials. Granulocytopenia and thrombocytopenia were the most common adverse reactions. The pharmacokinetic characteristics of ganciclovir after administration of ganciclovir for injection were studied in 27 neonates (aged 2 to 49 days) and ten pediatric patients, aged 9 months to 12 years. In neonates, the pharmacokinetic parameters after ganciclovir intravenous doses of 4 mg/kg (n = 14) and 6 mg/kg (n = 13) were Cmax 5.5 ± 1.6 and 7.0 ± 1.6 mcg/mL, systemic clearance 3.14 ± 1.75 and 3.56 ± 1.27 mL/min/kg, and t1/2 of 2.4 hours (harmonic mean) for both doses, respectively.
In pediatric patients 9 months to 12 years of age, the pharmacokinetic characteristics of ganciclovir were the same after single and multiple (every 12 hours) intravenous doses (5 mg/kg). The steady-state volume of distribution was 0.64 ± 0.22 L/kg, Cmax was 7.9 ± 3.9 mcg/mL, systemic clearance was 4.7 ± 2.2 mL/min/kg, and t1/2 was 2.4 ± 0.7 hours.
Although the pharmacokinetics of ganciclovir in pediatric patients were similar to those observed in adults, the safety and efficacy of ganciclovir at these exposures in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ganciclovir did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. Ganciclovir is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because renal clearance decreases with age, ganciclovir should be administered to elderly patients with special consideration of their renal status. Renal function should be monitored and dosage adjustments should be made accordingly [see Dosage and Administration (2.5), Warnings and Precautions (5.2), Use in Specific Populations (8.6)].
5.4 Fetal Toxicity
Ganciclovir may cause fetal toxicity when administered to pregnant women based on findings in animal studies. Systemic exposure of ganciclovir in animals at approximately 2 times the RHD caused fetal growth retardation, embryolethality, teratogenicity, and/or maternal toxicity. Teratogenic changes in animals included cleft palate, anophthalmia/microphthalmia, aplastic organs (kidney and pancreas), hydrocephaly and brachygnathia. Women of childbearing potential should be advised to use effective contraception during treatment and for at least 30 days following treatment with ganciclovir. Similarly, men should be advised to practice barrier contraception during and for at least 90 days following treatment with ganciclovir [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
4 Contraindications (4 CONTRAINDICATIONS)
Ganciclovir for Injection, USP is contraindicated in patients who have experienced a clinically significant hypersensitivity reaction (e.g., anaphylaxis) to ganciclovir, valganciclovir, or any component of the formulation.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Hematologic Toxicity [see Warnings and Precautions (5.1)]
- Renal Impairment [see Warnings and Precautions (5.2)]
- Impairment of Fertility [see Warnings and Precautions (5.3)]
- Fetal Toxicity [see Warnings and Precautions (5.4)]
- Mutagenesis and Carcinogenesis [see Warnings and Precautions (5.5)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Drug-drug interaction studies were conducted in patients with normal renal function. Patients with impaired renal function may have increased concentrations of ganciclovir and the coadministered drug following concomitant administration of ganciclovir and drugs excreted by the same pathway as ganciclovir. Therefore, these patients should be closely monitored for toxicity of ganciclovir and the coadministered drug.
Established and other potentially significant drug interactions conducted with ganciclovir are listed in Table 6 [see Clinical Pharmacology (12.3)].
| Name of the Concomitant Drug | Change in the Concentration of Ganciclovir or Concomitant Drug | Clinical Comment |
| Imipenem-cilastatin | Unknown | Coadministration with imipenem-cilastatin is not recommended because generalized seizures have been reported in patients who received ganciclovir and imipenem-cilastatin. |
| Cyclosporine or amphotericin B | Unknown | Monitor renal function when ganciclovir is coadministered with cyclosporine or amphotericin B because of potential increase in serum creatinine [see Warnings and Precautions (5.2)]. |
| Mycophenolate mofetil (MMF) |
↔ Ganciclovir (in patients with normal renal function) ↔ MMF (in patients with normal renal function) |
Based on increased risk, patients should be monitored for hematological and renal toxicity. |
| Other drugs associated with myelosuppresion or nephrotoxicity (e.g., dapsone, doxorubicin, flucytosine, hydroxyurea, pentamidine, tacrolimus, trimethoprim/ sulfamethoxazole, vinblastine, vincristine and zidovudine) | Unknown | Because of potential for higher toxicity, coadministration with ganciclovir should be considered only if the potential benefits are judged to outweigh the risks. |
| Didanosine |
↔ Ganciclovir ↑ Didanosine |
Patients should be closely monitored for didanosine toxicity (e.g., pancreatitis). |
| Probenecid | ↑ Ganciclovir | Ganciclovir dose may need to be reduced. Monitor for evidence of ganciclovir toxicity. |
5.2 Renal Impairment
Ganciclovir should be used with caution in patients with impaired renal function because the half-life and plasma/serum concentrations of ganciclovir will be increased due to reduced renal clearance. If renal function is impaired, dosage adjustments are recommended [see Dosage and Administration (2.5), Use in Specific Populations (8.5, 8.6)].
Increased serum creatinine levels have been reported in elderly patients and in transplant recipients receiving concomitant nephrotoxic medications (i.e., cyclosporine and amphotericin B). Monitoring renal function during therapy with ganciclovir is essential, especially for elderly patients and those patients receiving concomitant agents that may cause nephrotoxicity [see Dosage and Administration (2.5), Drug Interactions (7), Use in Specific Populations (8.5)].
8.6 Renal Impairment
Dose reduction is recommended when administering ganciclovir to patients with renal impairment [see Dosage and Administration (2.5), Warnings and Precautions (5.2)].
8.7 Hepatic Impairment
The safety and efficacy of ganciclovir have not been studied in patients with hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Ganciclovir is a deoxynucleoside analogue cytomegalovirus (CMV) DNA polymerase inhibitor indicated for the:
12.1 Mechanism of Action
Ganciclovir is an antiviral drug with activity against CMV [see Microbiology (12.4)].
5.1 Hematologic Toxicity
Granulocytopenia (neutropenia), anemia, thrombocytopenia and pancytopenia, have been observed in patients treated with ganciclovir. The frequency and severity of these events vary widely in different patient populations [see Adverse Reactions (6.1)]. Ganciclovir is not recommended if the absolute neutrophil count is less than 500 cells/µL, hemoglobin is less than 8 g/dL, or the platelet count is less than 25,000 cells/µL. Ganciclovir should also be used with caution in patients with preexisting cytopenias and in patients receiving myelosuppressive drugs or irradiation. Granulocytopenia (neutropenia) usually occurs during the first or second week of treatment but may occur at any time during treatment. Cell counts usually begin to recover within 3 to 7 days after discontinuing drug. Colony-stimulating factors have been shown to increase neutrophil and white blood cell counts in patients receiving ganciclovir solution for treatment of CMV retinitis.
Due to the frequency of neutropenia, anemia and thrombocytopenia in patients receiving ganciclovir [see Adverse Reactions (6.1)], complete blood counts with differential and platelet counts should be performed frequently in all patients, especially in patients with renal impairment and in patients in whom ganciclovir or other nucleoside analogues have previously resulted in leukopenia, or in whom neutrophil counts are less than 1000 cells/µL at the beginning of treatment [see Dosage and Administration (2.2)].
2.7 Handling and Disposal
Caution should be exercised in the handling and preparation of solutions of ganciclovir. Solutions of ganciclovir are alkaline (pH 11). Avoid direct contact of the skin or mucous membranes with ganciclovir solution. If such contact occurs, wash thoroughly with soap and water; rinse eyes thoroughly with plain water. Wearing disposable gloves is recommended.
Because ganciclovir shares some of the properties of antitumor agents (i.e., carcinogenicity and mutagenicity), consideration should be given to handling and disposal according to guidelines issued for antineoplastic drugs1 [see How Supplied/Storage and Handling (16)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Renal Impairment: Increased serum creatinine levels have been observed with the use of ganciclovir, particularly in elderly patients and transplant recipients receiving concomitant nephrotoxic drugs. Monitor renal function during therapy with ganciclovir, particularly in elderly patients and in patients taking other nephrotoxic drugs, and reduce dosage in patients with renal impairment. (5.2)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Ganciclovir for injection is administered only intravenously. (2.1)
| Dosage in Adult Patients with Normal Renal Function | |
|---|---|
| Treatment of CMV retinitis (2.3) | Induction: 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 14 to 21 days. Maintenance: 5 mg/kg (given intravenously at a constant rate over one hour) once daily for 7 days per week, or 6 mg/kg once daily for 5 days per week. |
| Prevention of CMV disease in transplant recipients (2.4) | Induction: 5 mg/kg (given intravenously at a constant rate over one hour) every 12 hours for 7 to 14 days. Maintenance: 5 mg/kg (given intravenously at a constant rate over one hour) once daily, 7 days per week, or 6 mg/kg once daily, 5 days per week until 100 to 120 days post-transplantation. |
Adults with renal impairment: Adjust dosage based on creatinine clearance. (2.5).
5.3 Impairment of Fertility
Based on animal data and limited human data, ganciclovir at the recommended human dose (RHD) may cause temporary or permanent inhibition of spermatogenesis in males, and may cause suppression of fertility in females. Advise patients that fertility may be impaired with the use of ganciclovir [see Use in Specific Populations (8.1, 8.3), Nonclinical Toxicology (13.1)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: Single dose vial containing 500 mg of ganciclovir as a sterile lyophilized white to off-white powder for reconstitution with 10 mL of preservative-free Sterile Water for Injection for intravenous use [see Dosage and Administration (2.6)].
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ganciclovir for injection or ganciclovir capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic disorders: hemolytic anemia, agranulocytosis, granulocytopenia
Cardiac disorders: cardiac arrest, conduction disorder, torsade de pointes, ventricular tachycardia
Congenital, familial and genetic disorders: congenital anomaly
Endocrine disorders: inappropriate antidiuretic hormone secretion
Eye disorders: cataracts, dry eyes
Gastrointestinal disorders: intestinal ulcer
Hepatobiliary disorders: cholelithiasis, cholestasis, hepatic failure, hepatitis
Immune system disorders: anaphylactic reaction, allergic reaction, vasculitis
Investigations: blood triglycerides increased
Metabolism and nutrition disorders: acidosis, hypercalcemia, hyponatremia
Musculoskeletal and connective tissue disorders: arthritis, rhabdomyolysis
Nervous system disorders: dysesthesia, dysphasia, extrapyramidal disorder, facial paralysis, amnesia, anosmia, myelopathy, cerebrovascular accident, third cranial nerve paralysis, aphasia, encephalopathy, intracranial hypertension
Psychiatric disorders: irritability, hallucinations
Renal and urinary disorders: renal tubular disorder, hemolytic uremic syndrome
Reproductive system and breast disorders: infertility, testicular hypotrophy
Respiratory, thoracic and mediastinal disorders: bronchospasm, pulmonary fibrosis
Skin and subcutaneous tissues disorders: exfoliative dermatitis, Stevens-Johnson syndrome
Vascular disorders: peripheral ischemia
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Breastfeeding is not recommended with use of ganciclovir. (8.2)
1.1 Treatment of Cmv Retinitis (1.1 Treatment of CMV Retinitis)
Ganciclovir is indicated for the treatment of cytomegalovirus (CMV) retinitis in immunocompromised adult patients, including patients with acquired immunodeficiency syndrome (AIDS) [see Clinical Studies (14.1)].
14.1 Treatment of Cmv Retinitis (14.1 Treatment of CMV Retinitis)
In a retrospective, non-randomized, single-center analysis of 41 patients with AIDS and CMV retinitis diagnosed by ophthalmologic examination between August 1983 and April 1988, treatment with ganciclovir for injection solution resulted in a delay in mean (median) time to first retinitis progression compared to untreated controls [105 (71) days from diagnosis vs. 35 (29) days from diagnosis]. Patients in this series received induction treatment of ganciclovir for injection 5 mg/kg twice daily for 14 to 21 days followed by maintenance treatment with either 5 mg/kg once daily, 7 days per week or 6 mg/kg once daily, 5 days per week.
In a controlled, randomized study conducted between February 1989 and December 1990, immediate treatment with ganciclovir was compared to delayed treatment in 42 patients with AIDS and peripheral CMV retinitis; 35 of 42 patients (13 in the immediate-treatment group and 22 in the delayed-treatment group) were included in the analysis of time to retinitis progression. Based on masked assessment of fundus photographs, the mean [95% CI] and median [95% CI] times to progression of retinitis were 66 days [39, 94] and 50 days [40, 84], respectively, in the immediate-treatment group compared to 19 days [11, 27] and 13.5 days [8, 18], respectively, in the delayed-treatment group.
Data from trials ICM 1653, ICM 1774, and AVI 034, which were performed comparing ganciclovir for injection to oral ganciclovir for treatment of CMV retinitis in patients with AIDS, are shown in Table 12 and Figures 1, 2, and 3, and are discussed below.
|
Demographics |
ICM 1653 (n = 121) |
ICM 1774 (n = 225) |
AVI 034 (n = 159) |
|
|
Median age (years) Range |
38 24 to 62 |
37 22 to 56 |
39 23 to 62 |
|
|
Sex |
Males |
116 (96%) |
222 (99%) |
148 (93%) |
|
Females |
5 (4%) |
3 (1%) |
10 (6%) |
|
|
Ethnicity |
Asian |
3 (3%) |
5 (2%) |
7 (4%) |
|
Black |
11 (9%) |
9 (4%) |
3 (2%) |
|
|
Caucasian |
98 (81%) |
186 (83%) |
140 (88%) |
|
|
Other |
9 (7%) |
25 (11%) |
8 (5%) |
|
|
Median CD4 Count Range |
9.5 0 to 141 |
7.0 0 to 80 |
10.0 0 to 320 |
|
|
Mean (SD) Observation Time (days) |
107.9 (43.0) |
97.6 (42.5) |
80.9 (47.0) |
5.5 Mutagenesis and Carcinogenesis
Animal data indicate that ganciclovir is mutagenic and carcinogenic. Ganciclovir should therefore be considered a potential carcinogen in humans [see Dosage and Administration (2.7), Nonclinical Toxicology (13.1)].
2.2 Testing Before and During Treatment
- Females of reproductive potential should undergo pregnancy testing before initiation of treatment with ganciclovir [see Warnings and Precautions (5.4), Use in Specific Populations (8.1, 8.3)].
- Complete blood counts with differential and platelet counts should be performed frequently, especially in patients in whom ganciclovir or other nucleoside analogues have previously resulted in cytopenias, or in whom absolute neutrophil counts are less than 1000 cells/μL at the beginning of treatment [see Warnings and Precautions (5.1)].
- All patients should be monitored for renal function before and during treatment with ganciclovir and dose should be adjusted as needed [see Dosage and Administration (2.5), Warnings and Precautions (5.2)].
- Patients with CMV retinitis should have frequent ophthalmological examinations during treatment with ganciclovir solution to monitor disease status and for other retinal abnormalities [see Adverse Reactions (6.1)].
Principal Display Panel 500 Mg Carton (PRINCIPAL DISPLAY PANEL - 500 mg Carton)
2.6 Preparation of Ganciclovir for Injection
Ganciclovir for injection must be reconstituted and diluted under the supervision of a healthcare provider and administered as intravenous infusion. Each 10 mL clear glass vial contains ganciclovir sodium equivalent to 500 mg of ganciclovir. Wearing disposable gloves is recommended during reconstitution and when wiping the outer surface of the vial and the table after reconstitution. The contents of the vial should be prepared for administration in the following manner:
- Reconstitution Instructions:
a) Reconstitute lyophilized ganciclovir for injection by injecting 10 mL of Sterile Water for Injection, USP, into the vial. Do not use bacteriostatic water for injection containing parabens. It is incompatible with ganciclovir for injection and may cause precipitation.
b) Gently swirl the vial in order to ensure complete wetting of the product. Continue swirling until a clear reconstituted solution is obtained.
c) Visually inspect the reconstituted solution for particulate matter and discoloration prior to proceeding with infusion. Discard the vial if particulate matter or discoloration is observed.
d) Reconstituted solution in the vial is stable at room temperature (25°C) for 12 hours. Do not refrigerate or freeze. - Infusion Instructions:
a) Based on patient weight, the appropriate volume of the reconstituted solution (ganciclovir concentration 50 mg/mL) should be removed from the vial and added to an acceptable infusion fluid (typically 100 mL) for delivery over the course of one hour. Infusion concentrations greater than 10 mg/mL are not recommended. The following infusion fluids have been determined to be chemically and physically compatible with ganciclovir for injection solution: 0.9% Sodium Chloride, 5% Dextrose, Ringer's Injection and Lactated Ringer's Injection, USP.
b) Ganciclovir for injection, when reconstituted with Sterile Water for Injection (non-bacteriostatic) and further diluted with 0.9% sodium chloride injection or other acceptable infusion fluid as specified above, should be used within 24 hours of dilution to reduce the risk of bacterial contamination. The diluted infusion solution should be refrigerated (2°C to 8°C). Do not freeze.
6.1 Clinical Trial Experience in Adult Patients
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect rates observed in practice. The most common adverse reactions and laboratory abnormalities reported in at least 20% of patients were pyrexia, diarrhea, leukopenia, nausea, anemia, asthenia, headache, cough, decreased appetite, dyspnea, abdominal pain, sepsis, hyperhidrosis, and blood creatinine increased.
Selected adverse reactions that occurred during clinical trials of ganciclovir for injection are summarized below, according to the participating study patient population.
2.1 Important Dosing and Administration Information
- To avoid phlebitis/pain at the infusion site, ganciclovir for injection must only be administered by intravenous infusion over one hour, preferably via plastic cannula, into a vein with adequate blood flow to permit rapid dilution and distribution.
- Do not administer ganciclovir for injection by rapid or bolus intravenous injection which may increase toxicity as a result of excessive plasma levels.
- The recommended dosage and infusion rate for ganciclovir should not be exceeded.
- Do not administer the reconstituted ganciclovir for injection solution intramuscularly or subcutaneously because it may result in severe tissue irritation due to high pH [see Description (11)].
- Administration of ganciclovir should be accompanied by adequate hydration.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
1.2 Prevention of Cmv Disease in Transplant Recipients (1.2 Prevention of CMV Disease in Transplant Recipients)
Ganciclovir is indicated for the prevention of CMV disease in adult transplant recipients at risk for CMV disease [see Clinical Studies (14.2)].
14.2 Prevention of Cmv Disease in Transplant Recipients (14.2 Prevention of CMV Disease in Transplant Recipients)
Ganciclovir was evaluated in three randomized, controlled trials of prevention of CMV disease in organ transplant recipients.
2.5 Recommended Dosage in Adult Patients With Renal Impairment (2.5 Recommended Dosage in Adult Patients with Renal Impairment)
For patients with impairment of renal function, refer to Table 1 for recommended doses of ganciclovir for induction and maintenance dosage for treatment of CMV retinitis and prevention of CMV disease in transplant recipients. Carefully monitor serum creatinine or creatinine clearance before and during treatment to allow for dosage adjustments in patients with impaired renal function.
| Creatinine Clearance Creatinine clearance can be related to serum creatinine by the formulas given below. (mL/min) |
Ganciclovir Induction Dose (mg/kg) |
Dosing Interval (hours) for Induction | Ganciclovir Maintenance Dose (mg/kg) | Dosing Interval (hours) for Maintenance |
| Greater than or equal to 70 | 5 | 12 | 5 | 24 |
| 50 to 69 | 2.5 | 12 | 2.5 | 24 |
| 25 to 49 | 2.5 | 24 | 1.25 | 24 |
| 10 to 24 | 1.25 | 24 | 0.625 | 24 |
| Less than 10 | 1.25 | 3 times per week, following hemodialysis | 0.625 | 3 times per week, following hemodialysis |
| Creatinine clearance for males = | (140 - age [yrs]) (body wt [kg]) |
| (72) (serum creatinine [mg/dL]) |
Creatinine clearance for females = 0.85 × male value
Warning: Hematologic Toxicity, Impairment of Fertility, Fetal Toxicity, Mutagenesis and Carcinogenesis (WARNING: HEMATOLOGIC TOXICITY, IMPAIRMENT OF FERTILITY, FETAL TOXICITY, MUTAGENESIS AND CARCINOGENESIS)
- Hematologic Toxicity: Granulocytopenia, anemia, thrombocytopenia, and pancytopenia have been reported in patients treated with ganciclovir [see Warnings and Precautions (5.1)].
- Impairment of Fertility: Based on animal data and limited human data, ganciclovir may cause temporary or permanent inhibition of spermatogenesis in males and suppression of fertility in females [see Warnings and Precautions (5.3)].
- Fetal Toxicity: Based on animal data, ganciclovir has the potential to cause birth defects in humans [see Warnings and Precautions (5.4)].
- Mutagenesis and Carcinogenesis: Based on animal data, ganciclovir has the potential to cause cancers in humans [see Warnings and Precautions (5.5)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:44.117220 · Updated: 2026-03-14T22:17:14.177741