fexofenadine hydrochloride and pseudoephedrine hydrochloride extended-release tablets, 60 mg/120 mg
87ab4578-84de-475d-a4b0-eec969f17253
34390-5
HUMAN OTC DRUG LABEL
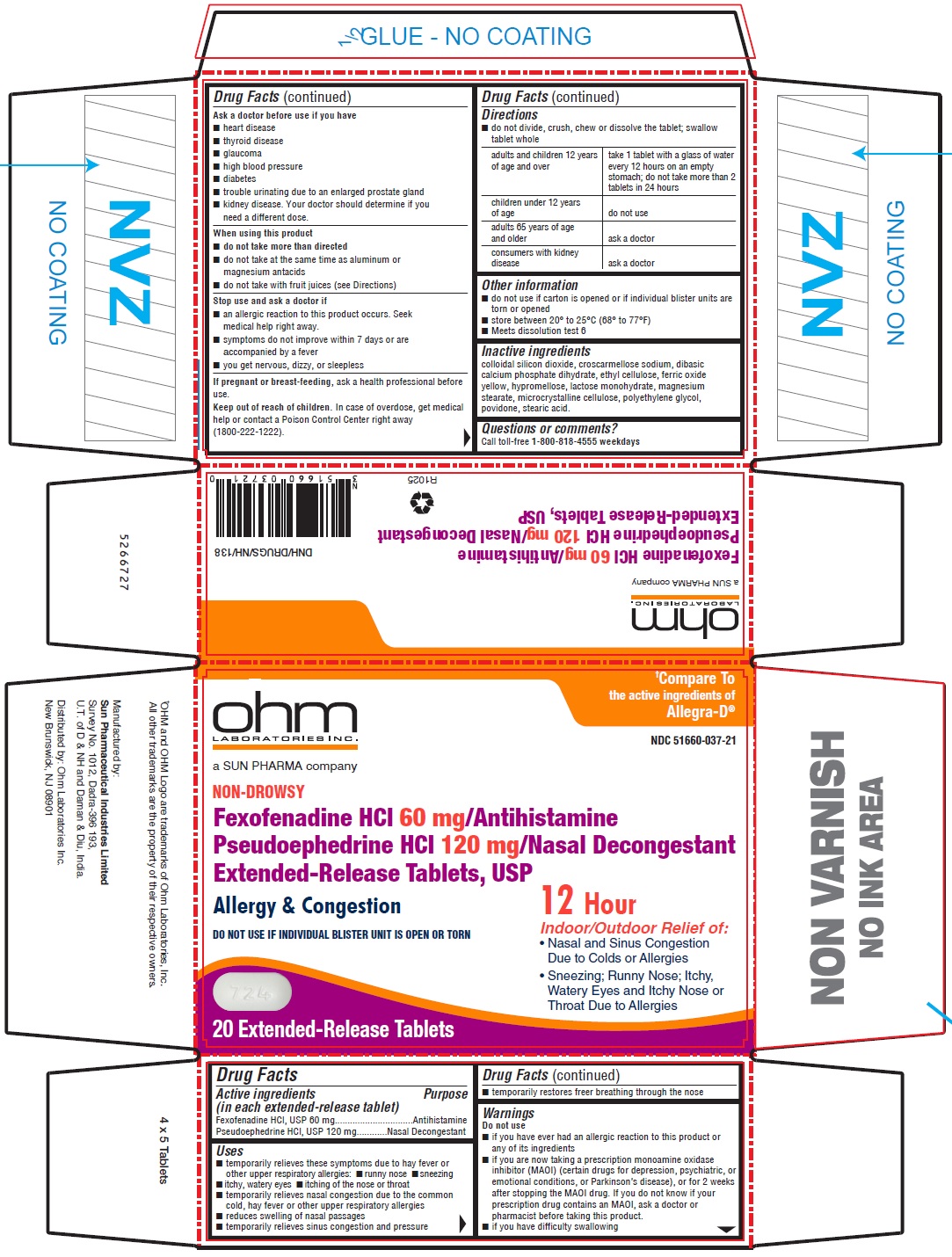
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- reduces swelling of nasal passages
- temporarily relieves sinus congestion and pressure
- temporarily restores freer breathing through the nose
Directions
- do not divide, crush, chew or dissolve the tablet; swallow tablet whole
| adults and children 12 years of age and over | take 1 tablet with a glass of water every 12 hours on an empty stomach; do not take more than 2 tablets in 24 hours |
| children under 12 years of age | do not use |
| adults 65 years of age and older | ask a doctor |
| consumers with kidney disease | ask a doctor |
Other Information
- do not use if carton is opened or if individual blister units are torn or opened
- store between 20° to 25°C (68° to 77°F)
- Meets dissolution test 6
Inactive Ingredients
colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate dihydrate, ethyl cellulose, ferric oxide yellow, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, stearic acid.
Description
Drug Facts
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center right away (1800-222-1222).
Section 53414-9
If pregnant or breast-feeding,ask a health professional before use.
Section 55105-1
| Active ingredients (in each extended-release tablet) | Purpose |
|---|---|
| Fexofenadine HCl, USP 60 mg | Antihistamine |
| Pseudoephedrine HCl, USP 120 mg | Nasal Decongestant |
Do Not Use
- if you have ever had an allergic reaction to this product or any of its ingredients
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have difficulty swallowing
Questions Or Comments?
Call toll-free 1-800-818-4555 weekdays
When Using This Product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Stop Use and Ask A Doctor If
- an allergic reaction to this product occurs. Seek medical help right away.
- symptoms do not improve within 7 days or are accompanied by a fever
- you get nervous, dizzy, or sleepless
Ask A Doctor Before Use If You Have
- heart disease
- thyroid disease
- glaucoma
- high blood pressure
- diabetes
- trouble urinating due to an enlarged prostate gland
- kidney disease. Your doctor should determine if you need a different dose.
Principal Display Panel 20 Tablet Blister Pack Carton
†Compare To
the active ingredients of
Allegra-D
®
NDC 51660-037-21
ohm ®
NON-DROWSY
Original Prescription Strength
Fexofenadine HCl 60 mg/Antihistamine
Pseudoephedrine HCl 120 mg/Nasal Decongestant
Extended-Release Tablets, USP
Allergy & Congestion
Indoor and Outdoor Allergies
DO NOT USE IF INDIVIDUAL BLISTER UNIT IS OPEN OR TORN
12 Hour
Relief of:
- Nasal and Sinus Congestion
Due to Colds or Allergies - Sneezing; Runny Nose; Itchy,
Watery Eyes and Itchy Nose or
Throat Due to Allergies
20 Extended-Release Tablets
Structured Label Content
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
- temporarily relieves nasal congestion due to the common cold, hay fever or other upper respiratory allergies
- reduces swelling of nasal passages
- temporarily relieves sinus congestion and pressure
- temporarily restores freer breathing through the nose
Directions
- do not divide, crush, chew or dissolve the tablet; swallow tablet whole
| adults and children 12 years of age and over | take 1 tablet with a glass of water every 12 hours on an empty stomach; do not take more than 2 tablets in 24 hours |
| children under 12 years of age | do not use |
| adults 65 years of age and older | ask a doctor |
| consumers with kidney disease | ask a doctor |
Other Information (Other information)
- do not use if carton is opened or if individual blister units are torn or opened
- store between 20° to 25°C (68° to 77°F)
- Meets dissolution test 6
Inactive Ingredients (Inactive ingredients)
colloidal silicon dioxide, croscarmellose sodium, dibasic calcium phosphate dihydrate, ethyl cellulose, ferric oxide yellow, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, povidone, stearic acid.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center right away (1800-222-1222).
Section 53414-9 (53414-9)
If pregnant or breast-feeding,ask a health professional before use.
Section 55105-1 (55105-1)
| Active ingredients (in each extended-release tablet) | Purpose |
|---|---|
| Fexofenadine HCl, USP 60 mg | Antihistamine |
| Pseudoephedrine HCl, USP 120 mg | Nasal Decongestant |
Do Not Use (Do not use)
- if you have ever had an allergic reaction to this product or any of its ingredients
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- if you have difficulty swallowing
Questions Or Comments? (Questions or comments?)
Call toll-free 1-800-818-4555 weekdays
When Using This Product (When using this product)
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- an allergic reaction to this product occurs. Seek medical help right away.
- symptoms do not improve within 7 days or are accompanied by a fever
- you get nervous, dizzy, or sleepless
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- heart disease
- thyroid disease
- glaucoma
- high blood pressure
- diabetes
- trouble urinating due to an enlarged prostate gland
- kidney disease. Your doctor should determine if you need a different dose.
Principal Display Panel 20 Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 20 Tablet Blister Pack Carton)
†Compare To
the active ingredients of
Allegra-D
®
NDC 51660-037-21
ohm ®
NON-DROWSY
Original Prescription Strength
Fexofenadine HCl 60 mg/Antihistamine
Pseudoephedrine HCl 120 mg/Nasal Decongestant
Extended-Release Tablets, USP
Allergy & Congestion
Indoor and Outdoor Allergies
DO NOT USE IF INDIVIDUAL BLISTER UNIT IS OPEN OR TORN
12 Hour
Relief of:
- Nasal and Sinus Congestion
Due to Colds or Allergies - Sneezing; Runny Nose; Itchy,
Watery Eyes and Itchy Nose or
Throat Due to Allergies
20 Extended-Release Tablets
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:30.388200 · Updated: 2026-03-14T23:13:37.162375