meclizine hydrochloride tablets, usp
87210edf-0bf9-2577-e053-2991aa0aaceb
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Meclizine hydrochloride tablets are indicated for the treatment of vertigo associated with diseases affecting the vestibular system.
Dosage and Administration
The recommended dose is 25 to 100 mg daily administered orally, in divided dosage, depending upon clinical response.
Contraindications
Meclizine hydrochloride tablets are contraindicated in individuals who have shown a previous hypersensitivity to it.
Adverse Reactions
Anaphylactoid reaction, drowsiness, dry mouth, headache, fatigue, vomiting and, on rare occasions, blurred vision have been reported. To report SUSPECTED ADVERSE REACTIONS, contact Jubilant Cadista Pharmaceuticals Inc. at 1-800-313-4623 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
25 mg (Yellow, oval shaped tablets, debossed with “TL 121” with score on one side and plain on the other side.) NDC 60760-087-90 BOTTLES OF 90 Store at 20 to 25°C (68 to 77°F) [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container (USP). Keep this and all medication out of the reach of children. Manufactured By: Jubilant Cadista Pharmaceuticals Inc. Salisbury, MD 21801, USA Revised 01/2019
Description
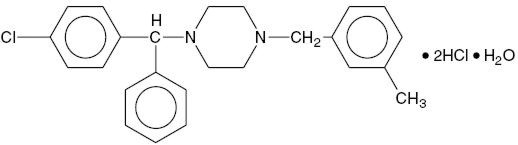
Meclizine HCl, USP an oral antiemetic, is a white or slightly yellowish, crystalline powder. It has the following structural formula: Chemically, Meclizine HCl is 1-( p -chloro-α-phenylbenzyl)-4-( m -methylbenzyl) piperazine dihydrochloride monohydrate. Meclizine hydrochloride tablets, USP are available in two different strengths: 12.5 mg and 25 mg. In addition each tablet contains the following inactive ingredients: Colloidal Silicon Dioxide, Croscarmellose Sodium, Lactose Monohydrate, Magnesium Stearate, Microcrystalline Cellulose. Also, Meclizine hydrochloride tablets USP, 12.5 mg contains FD&C Blue #1 Aluminum Lake (11-13%) and Meclizine hydrochloride tablets USP, 25 mg contains D&C Yellow #10 Aluminum Lake (15-20%). Each meclizine HCl 12.5 mg tablet contains 12.5 mg of meclizine dihydrochloride equivalent to 10.53 mg of meclizine free base. Each meclizine HCl 25 mg tablet contains 25 mg of meclizine dihydrochloride equivalent to 21.07 mg of meclizine free base.
Medication Information
Warnings
Since drowsiness may, on occasion, occur with use of this drug, patients should be warned of this possibility and cautioned against driving a car or operating dangerous machinery.
Patients should avoid alcoholic beverages while taking this drug.
Due to its potential anticholinergic action, this drug should be used with caution in patients with asthma, glaucoma, or enlargement of the prostate gland.
Indications and Usage
Meclizine hydrochloride tablets are indicated for the treatment of vertigo associated with diseases affecting the vestibular system.
Dosage and Administration
The recommended dose is 25 to 100 mg daily administered orally, in divided dosage, depending upon clinical response.
Contraindications
Meclizine hydrochloride tablets are contraindicated in individuals who have shown a previous hypersensitivity to it.
Adverse Reactions
Anaphylactoid reaction, drowsiness, dry mouth, headache, fatigue, vomiting and, on rare occasions, blurred vision have been reported.
To report SUSPECTED ADVERSE REACTIONS, contact Jubilant Cadista Pharmaceuticals Inc. at 1-800-313-4623 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied
25 mg (Yellow, oval shaped tablets, debossed with “TL 121” with score on one side and plain on the other side.)
NDC
60760-087-90 BOTTLES OF 90
Store at 20 to 25°C (68 to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container (USP).
Keep this and all medication out of the reach of children.
Manufactured By:
Jubilant Cadista Pharmaceuticals Inc.
Salisbury, MD 21801, USA
Revised 01/2019
Description
Meclizine HCl, USP an oral antiemetic, is a white or slightly yellowish, crystalline powder. It has the following structural formula:
Chemically, Meclizine HCl is 1-( p-chloro-α-phenylbenzyl)-4-( m-methylbenzyl) piperazine dihydrochloride monohydrate.
Meclizine hydrochloride tablets, USP are available in two different strengths: 12.5 mg and 25 mg. In addition each tablet contains the following inactive ingredients: Colloidal Silicon Dioxide, Croscarmellose Sodium, Lactose Monohydrate, Magnesium Stearate, Microcrystalline Cellulose. Also, Meclizine hydrochloride tablets USP, 12.5 mg contains FD&C Blue #1 Aluminum Lake (11-13%) and Meclizine hydrochloride tablets USP, 25 mg contains D&C Yellow #10 Aluminum Lake (15-20%).
Each meclizine HCl 12.5 mg tablet contains 12.5 mg of meclizine dihydrochloride equivalent to 10.53 mg of meclizine free base.
Each meclizine HCl 25 mg tablet contains 25 mg of meclizine dihydrochloride equivalent to 21.07 mg of meclizine free base.
Section 42229-5
Absorption
Meclizine is absorbed after oral administration with maximum plasma concentrations reaching at a median T
max value of 3 hours post-dose (range: 1.5 to 6 hours) for the tablet dosage form.
Section 43682-4
Pharmacokinetics
The available pharmacokinetic information for meclizine following oral administration has been summarized from published literature.
Section 51945-4
Clinical Pharmacology
Meclizine hydrochloride is an antihistamine that shows marked protective activity against nebulized histamine and lethal doses of intravenously injected histamine in guinea pigs. It has a marked effect in blocking the vasodepressor response to histamine, but only a slight blocking action against acetylcholine. Its activity is relatively weak in inhibiting the spasmogenic action of histamine on isolated guinea pig ileum.
Structured Label Content
Warnings (WARNINGS)
Since drowsiness may, on occasion, occur with use of this drug, patients should be warned of this possibility and cautioned against driving a car or operating dangerous machinery.
Patients should avoid alcoholic beverages while taking this drug.
Due to its potential anticholinergic action, this drug should be used with caution in patients with asthma, glaucoma, or enlargement of the prostate gland.
Indications and Usage (INDICATIONS AND USAGE)
Meclizine hydrochloride tablets are indicated for the treatment of vertigo associated with diseases affecting the vestibular system.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
The recommended dose is 25 to 100 mg daily administered orally, in divided dosage, depending upon clinical response.
Contraindications (CONTRAINDICATIONS)
Meclizine hydrochloride tablets are contraindicated in individuals who have shown a previous hypersensitivity to it.
Adverse Reactions (ADVERSE REACTIONS)
Anaphylactoid reaction, drowsiness, dry mouth, headache, fatigue, vomiting and, on rare occasions, blurred vision have been reported.
To report SUSPECTED ADVERSE REACTIONS, contact Jubilant Cadista Pharmaceuticals Inc. at 1-800-313-4623 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
How Supplied (HOW SUPPLIED)
25 mg (Yellow, oval shaped tablets, debossed with “TL 121” with score on one side and plain on the other side.)
NDC
60760-087-90 BOTTLES OF 90
Store at 20 to 25°C (68 to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container (USP).
Keep this and all medication out of the reach of children.
Manufactured By:
Jubilant Cadista Pharmaceuticals Inc.
Salisbury, MD 21801, USA
Revised 01/2019
Description (DESCRIPTION)
Meclizine HCl, USP an oral antiemetic, is a white or slightly yellowish, crystalline powder. It has the following structural formula:
Chemically, Meclizine HCl is 1-( p-chloro-α-phenylbenzyl)-4-( m-methylbenzyl) piperazine dihydrochloride monohydrate.
Meclizine hydrochloride tablets, USP are available in two different strengths: 12.5 mg and 25 mg. In addition each tablet contains the following inactive ingredients: Colloidal Silicon Dioxide, Croscarmellose Sodium, Lactose Monohydrate, Magnesium Stearate, Microcrystalline Cellulose. Also, Meclizine hydrochloride tablets USP, 12.5 mg contains FD&C Blue #1 Aluminum Lake (11-13%) and Meclizine hydrochloride tablets USP, 25 mg contains D&C Yellow #10 Aluminum Lake (15-20%).
Each meclizine HCl 12.5 mg tablet contains 12.5 mg of meclizine dihydrochloride equivalent to 10.53 mg of meclizine free base.
Each meclizine HCl 25 mg tablet contains 25 mg of meclizine dihydrochloride equivalent to 21.07 mg of meclizine free base.
Section 42229-5 (42229-5)
Absorption
Meclizine is absorbed after oral administration with maximum plasma concentrations reaching at a median T
max value of 3 hours post-dose (range: 1.5 to 6 hours) for the tablet dosage form.
Section 43682-4 (43682-4)
Pharmacokinetics
The available pharmacokinetic information for meclizine following oral administration has been summarized from published literature.
Section 51945-4 (51945-4)
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Meclizine hydrochloride is an antihistamine that shows marked protective activity against nebulized histamine and lethal doses of intravenously injected histamine in guinea pigs. It has a marked effect in blocking the vasodepressor response to histamine, but only a slight blocking action against acetylcholine. Its activity is relatively weak in inhibiting the spasmogenic action of histamine on isolated guinea pig ileum.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:31.938286 · Updated: 2026-03-14T21:50:37.524392