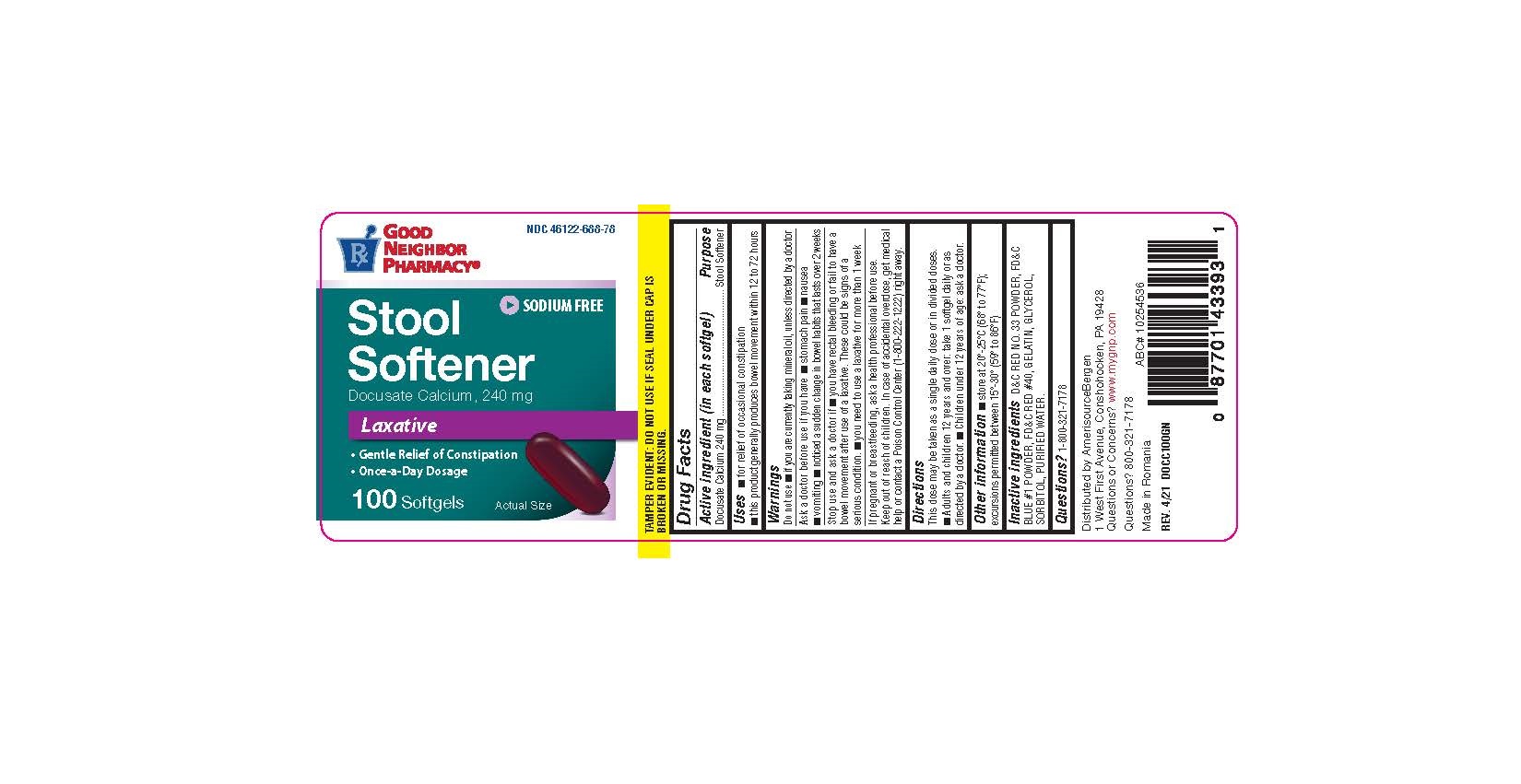
Docusate Calcium
871cdcd5-bf8e-4c70-8505-c81cf1c4fe33
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Docusate Calcium 240 mg
Purpose
Stool Softener
Medication Information
Purpose
Stool Softener
Description
Docusate Calcium 240 mg
Uses
- For the relief of occasional constipation.
- This product generally produces a bowel movement within 12 to 72 hours.
Directions
Adults and Children over 12 years of age
Take orally 1 softgel daily for 2 to 3 days or until bowl movements are normal, or as directed by a doctor.
Children under 12 years of age
Do not use this product for children under 12 years of age, unless directed by a doctor.
Active Ingredient
Docusate Calcium 240 mg
Other Information
- Store at room temperature between 15ºC to 25ºC (59ºF to 77ºF).
- Do not use if either seal is broken or missing.
Inactive Ingredients
FD&C Blue #1, FD&C Red #40, Gelatin, Glycerin, Sorbitol Special.
Warnings: Do Not Use
- If you are currently taking mineral oil, unless directed by a doctor.
- When abdominal pain, nausea or vomiting are present.
- For longer than one week unless directed by a doctor.
Questions Or Comments
Distributed by AmerisourceBergen
1300 Morris Drive, Chesterbrook, PA 19087
Questions or Concerns? www.mygnp.com
Questions? 800-321-7178
MADE IN ROMANIA
REV. 2/21 DOCC110GN
Ask A Doctor Before Use
■ stomach pain ■ nausea ■ vomiting ■ noticed a sudden change in bowel habits that lasts over 2 weeks
Stop Use and Ask A Doctor If
- You have rectal bleeding.
- You fail to have a bowel movement after use.
If Pregnant Or Breast Feeding
Ask a healthcare professional before use.
Keep Out of Reach of Children.
In case of accidental overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Package/label Principal Display Panel
Structured Label Content
Uses
- For the relief of occasional constipation.
- This product generally produces a bowel movement within 12 to 72 hours.
Purpose
Stool Softener
Directions
Adults and Children over 12 years of age
Take orally 1 softgel daily for 2 to 3 days or until bowl movements are normal, or as directed by a doctor.
Children under 12 years of age
Do not use this product for children under 12 years of age, unless directed by a doctor.
Active Ingredient (Active ingredient)
Docusate Calcium 240 mg
Other Information
- Store at room temperature between 15ºC to 25ºC (59ºF to 77ºF).
- Do not use if either seal is broken or missing.
Inactive Ingredients
FD&C Blue #1, FD&C Red #40, Gelatin, Glycerin, Sorbitol Special.
Warnings: Do Not Use (Warnings: Do not use)
- If you are currently taking mineral oil, unless directed by a doctor.
- When abdominal pain, nausea or vomiting are present.
- For longer than one week unless directed by a doctor.
Questions Or Comments (Questions or Comments)
Distributed by AmerisourceBergen
1300 Morris Drive, Chesterbrook, PA 19087
Questions or Concerns? www.mygnp.com
Questions? 800-321-7178
MADE IN ROMANIA
REV. 2/21 DOCC110GN
Ask A Doctor Before Use (Ask a Doctor Before Use)
■ stomach pain ■ nausea ■ vomiting ■ noticed a sudden change in bowel habits that lasts over 2 weeks
Stop Use and Ask A Doctor If (Stop Use and Ask a Doctor If)
- You have rectal bleeding.
- You fail to have a bowel movement after use.
If Pregnant Or Breast Feeding (If Pregnant or Breast Feeding)
Ask a healthcare professional before use.
Keep Out of Reach of Children.
In case of accidental overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Package/label Principal Display Panel (Package/Label Principal Display Panel)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:54.430627 · Updated: 2026-03-14T23:06:03.178139