These Highlights Do Not Include All The Information Needed To Use Ibrance Safely And Effectively. See Full Prescribing Information For Ibrance.
86718885-da44-4d3d-b3ca-06cbcedcffd8
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indications and Usage ( 1 ) 4/2025 Dosage and Administration ( 2.1 , 2.2 ) 4/2025
Indications and Usage
IBRANCE is indicated for the treatment of adult patients with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer in combination with: • an aromatase inhibitor as initial endocrine-based therapy; or • fulvestrant in patients with disease progression following endocrine therapy. IBRANCE is indicated in combination with inavolisib and fulvestrant for the treatment of adult patients with endocrine-resistant, PIK3CA -mutated, HR-positive, HER2-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant endocrine therapy.
Dosage and Administration
Warnings and Precautions
• Neutropenia: Monitor complete blood count prior to start of IBRANCE therapy and at the beginning of each cycle, as well as on Day 15 of the first 2 cycles, and as clinically indicated. ( 2.2 , 5.1 ) • Interstitial Lung Disease (ILD)/Pneumonitis: Severe and fatal cases of ILD/pneumonitis have been reported. Monitor for pulmonary symptoms of ILD/pneumonitis. Interrupt IBRANCE immediately in patients with suspected ILD/pneumonitis. Permanently discontinue IBRANCE if severe ILD/pneumonitis occurs. ( 5.2 ) • Embryo-Fetal Toxicity: IBRANCE can cause fetal harm. Advise patients of potential risk to a fetus and to use effective contraception. ( 5.3 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Neutropenia [see Warnings and Precautions (5.1) ] • ILD/Pneumonitis [see Warnings and Precautions (5.2) ]
Drug Interactions
Palbociclib is primarily metabolized by CYP3A and sulfotransferase (SULT) enzyme SULT2A1. In vivo, palbociclib is a time-dependent inhibitor of CYP3A.
Storage and Handling
IBRANCE is supplied in the following strengths and package configurations: IBRANCE Capsules Package Configuration Capsule Strength (mg) NDC Capsule Description Bottles of 21 capsules 125 NDC 0069-0189-21 opaque, hard gelatin capsules, size 0, with caramel cap and body, printed with white ink "Pfizer" on the cap, "PBC 125" on the body Bottles of 21 capsules 100 NDC 0069-0188-21 opaque, hard gelatin capsules, size 1, with caramel cap and light orange body, printed with white ink "Pfizer" on the cap, "PBC 100" on the body Bottles of 21 capsules 75 NDC 0069-0187-21 opaque, hard gelatin capsules, size 2, with light orange cap and body, printed with white ink "Pfizer" on the cap, "PBC 75" on the body
How Supplied
IBRANCE is supplied in the following strengths and package configurations: IBRANCE Capsules Package Configuration Capsule Strength (mg) NDC Capsule Description Bottles of 21 capsules 125 NDC 0069-0189-21 opaque, hard gelatin capsules, size 0, with caramel cap and body, printed with white ink "Pfizer" on the cap, "PBC 125" on the body Bottles of 21 capsules 100 NDC 0069-0188-21 opaque, hard gelatin capsules, size 1, with caramel cap and light orange body, printed with white ink "Pfizer" on the cap, "PBC 100" on the body Bottles of 21 capsules 75 NDC 0069-0187-21 opaque, hard gelatin capsules, size 2, with light orange cap and body, printed with white ink "Pfizer" on the cap, "PBC 75" on the body
Medication Information
Warnings and Precautions
• Neutropenia: Monitor complete blood count prior to start of IBRANCE therapy and at the beginning of each cycle, as well as on Day 15 of the first 2 cycles, and as clinically indicated. ( 2.2 , 5.1 ) • Interstitial Lung Disease (ILD)/Pneumonitis: Severe and fatal cases of ILD/pneumonitis have been reported. Monitor for pulmonary symptoms of ILD/pneumonitis. Interrupt IBRANCE immediately in patients with suspected ILD/pneumonitis. Permanently discontinue IBRANCE if severe ILD/pneumonitis occurs. ( 5.2 ) • Embryo-Fetal Toxicity: IBRANCE can cause fetal harm. Advise patients of potential risk to a fetus and to use effective contraception. ( 5.3 , 8.1 , 8.3 )
Indications and Usage
IBRANCE is indicated for the treatment of adult patients with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer in combination with: • an aromatase inhibitor as initial endocrine-based therapy; or • fulvestrant in patients with disease progression following endocrine therapy. IBRANCE is indicated in combination with inavolisib and fulvestrant for the treatment of adult patients with endocrine-resistant, PIK3CA -mutated, HR-positive, HER2-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant endocrine therapy.
Dosage and Administration
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: • Neutropenia [see Warnings and Precautions (5.1) ] • ILD/Pneumonitis [see Warnings and Precautions (5.2) ]
Drug Interactions
Palbociclib is primarily metabolized by CYP3A and sulfotransferase (SULT) enzyme SULT2A1. In vivo, palbociclib is a time-dependent inhibitor of CYP3A.
Storage and Handling
IBRANCE is supplied in the following strengths and package configurations: IBRANCE Capsules Package Configuration Capsule Strength (mg) NDC Capsule Description Bottles of 21 capsules 125 NDC 0069-0189-21 opaque, hard gelatin capsules, size 0, with caramel cap and body, printed with white ink "Pfizer" on the cap, "PBC 125" on the body Bottles of 21 capsules 100 NDC 0069-0188-21 opaque, hard gelatin capsules, size 1, with caramel cap and light orange body, printed with white ink "Pfizer" on the cap, "PBC 100" on the body Bottles of 21 capsules 75 NDC 0069-0187-21 opaque, hard gelatin capsules, size 2, with light orange cap and body, printed with white ink "Pfizer" on the cap, "PBC 75" on the body
How Supplied
IBRANCE is supplied in the following strengths and package configurations: IBRANCE Capsules Package Configuration Capsule Strength (mg) NDC Capsule Description Bottles of 21 capsules 125 NDC 0069-0189-21 opaque, hard gelatin capsules, size 0, with caramel cap and body, printed with white ink "Pfizer" on the cap, "PBC 125" on the body Bottles of 21 capsules 100 NDC 0069-0188-21 opaque, hard gelatin capsules, size 1, with caramel cap and light orange body, printed with white ink "Pfizer" on the cap, "PBC 100" on the body Bottles of 21 capsules 75 NDC 0069-0187-21 opaque, hard gelatin capsules, size 2, with light orange cap and body, printed with white ink "Pfizer" on the cap, "PBC 75" on the body
Description
Indications and Usage ( 1 ) 4/2025 Dosage and Administration ( 2.1 , 2.2 ) 4/2025
Section 42229-5
Dose Modifications for Use With Strong CYP3A Inhibitors
Avoid concomitant use of strong CYP3A inhibitors and consider an alternative concomitant medication with no or minimal CYP3A inhibition. If patients must be coadministered a strong CYP3A inhibitor, reduce the IBRANCE dose to 75 mg once daily. If the strong inhibitor is discontinued, increase the IBRANCE dose (after 3 to 5 half-lives of the inhibitor) to the dose used prior to the initiation of the strong CYP3A inhibitor [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. Revised September 2025 | ||
|
PATIENT INFORMATION
|
||
|
What is the most important information I should know about IBRANCE? IBRANCE may cause serious side effects, including: Low white blood cell counts (neutropenia). Low white blood cell counts are very common when taking IBRANCE and may cause serious infections that can lead to death. Your healthcare provider should check your white blood cell counts before and during treatment. If you develop low white blood cell counts during treatment with IBRANCE, your healthcare provider may stop your treatment, decrease your dose, or may tell you to wait to begin your treatment cycle. Tell your healthcare provider right away if you have signs and symptoms of low white blood cell counts or infections such as fever and chills. Lung problems (pneumonitis). IBRANCE may cause severe or life-threatening inflammation of the lungs during treatment that can lead to death. Tell your healthcare provider right away if you have any new or worsening symptoms, including:
Your healthcare provider may interrupt or stop treatment with IBRANCE completely if your symptoms are severe. See "What are the possible side effects of IBRANCE?" for more information about side effects. |
||
|
What is IBRANCE? IBRANCE is a prescription medicine used: In adults to treat hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer that has spread to other parts of the body (metastatic) in combination with:
In adults with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative and with an abnormal phosphatidylinositol-3-kinase catalytic subunit alpha (PIK3CA) gene breast cancer that has spread to nearby tissue or lymph nodes (locally advanced), or to other parts of the body (metastatic) in combination with:
It is not known if IBRANCE is safe and effective in children. |
||
|
What should I tell my healthcare provider before taking IBRANCE? Before taking IBRANCE, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. IBRANCE and other medicines may affect each other causing side effects. Know the medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get a new medicine. |
||
|
How should I take IBRANCE?
|
||
|
What are the possible side effects of IBRANCE? IBRANCE may cause serious side effects. See "What is the most important information I should know about IBRANCE?" The most common side effects of IBRANCE when used with either letrozole or fulvestrant include:
|
||
|
|
|
|
|
|
|
The most common side effects of IBRANCE when used in combination with inavolisib plus fulvestrant include:
IBRANCE may cause fertility problems in males. This may affect your ability to father a child. Talk to your healthcare provider about family planning options before starting IBRANCE if this is a concern for you. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of IBRANCE. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
How should I store IBRANCE?
Keep IBRANCE and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of IBRANCE Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use IBRANCE for a condition for which it was not prescribed. Do not give IBRANCE to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for more information about IBRANCE that is written for health professionals. |
||
|
What are the ingredients in IBRANCE? Active ingredient: palbociclib Inactive ingredients: microcrystalline cellulose, lactose monohydrate, sodium starch glycolate, colloidal silicon dioxide, magnesium stearate, and hard gelatin capsule shells. The light orange, light orange/caramel and caramel opaque capsule shells contain: gelatin, red iron oxide, yellow iron oxide, and titanium dioxide. The printing ink contains: shellac, titanium dioxide, ammonium hydroxide, propylene glycol, and simethicone.
For more information, go to www.pfizer.com or call 1-800-438-1985. |
Section 44425-7
Store at 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 °F to 86 °F) [see USP Controlled Room Temperature].
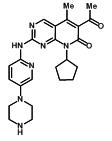
11 Description
IBRANCE capsules for oral administration contain 125 mg, 100 mg, or 75 mg of palbociclib, a kinase inhibitor. The molecular formula for palbociclib is C24H29N7O2. The molecular weight is 447.54 daltons. The chemical name is 6-acetyl-8-cyclopentyl-5-methyl-2-{[5-(piperazin-1-yl)pyridin-2-yl]amino}pyrido[2,3-d]pyrimidin-7(8H)-one, and its structural formula is:
Palbociclib is a yellow to orange powder with pKa of 7.4 (the secondary piperazine nitrogen) and 3.9 (the pyridine nitrogen). At or below pH 4, palbociclib behaves as a high-solubility compound. Above pH 4, the solubility of the drug substance reduces significantly.
Inactive ingredients: Microcrystalline cellulose, lactose monohydrate, sodium starch glycolate, colloidal silicon dioxide, magnesium stearate, and hard gelatin capsule shells. The light orange, light orange/caramel, and caramel opaque capsule shells contain gelatin, red iron oxide, yellow iron oxide, and titanium dioxide; the printing ink contains shellac, titanium dioxide, ammonium hydroxide, propylene glycol, and simethicone.
5.1 Neutropenia
Neutropenia was the most frequently reported adverse reaction in PALOMA-2 with an incidence of 80% and PALOMA-3 with an incidence of 83%. A Grade ≥3 decrease in neutrophil counts was reported in 66% of patients receiving IBRANCE plus letrozole in PALOMA-2 and 66% of patients receiving IBRANCE plus fulvestrant in PALOMA-3. In PALOMA-2 and PALOMA-3, the median time to first episode of any grade neutropenia was 15 days and the median duration of Grade ≥3 neutropenia was 7 days [see Adverse Reactions (6.1)].
Monitor complete blood counts prior to starting IBRANCE therapy and at the beginning of each cycle, as well as on Day 15 of the first 2 cycles, and as clinically indicated. Dose interruption, dose reduction, or delay in starting treatment cycles is recommended for patients who develop Grade 3 or 4 neutropenia [see Dosage and Administration (2.2)].
Febrile neutropenia has been reported in 1.8% of patients exposed to IBRANCE across PALOMA-2 and PALOMA-3. One death due to neutropenic sepsis was observed in PALOMA-3. Physicians should inform patients to promptly report any episodes of fever [see Patient Counseling Information (17)].
8.4 Pediatric Use
The safety and effectiveness of IBRANCE in pediatric patients have not been established.
The safety and effectiveness of IBRANCE were assessed but not established in three trials: one open-label trial [A5481092, (NCT03709680)] that included 98 pediatric patients 2 to <17 years of age who received IBRANCE in combination with chemotherapy for recurrent or refractory solid tumors and two open-label trials that included 42 pediatric patients 4 to <17 years of age who received IBRANCE as a single agent for recurrent or refractory solid tumors [APEC1621I, (NCT03526250)] or primary central nervous system (CNS) tumors [PBTC-042, (NCT02255461)].
No new safety signals were observed in these trials. Palbociclib exposures in pediatric patients who received IBRANCE as a single agent or in combination were within range of those observed in adults given a similar dose based on body surface area.
Juvenile Animal Toxicity Data
Altered glucose metabolism (glycosuria, hyperglycemia, decreased insulin) associated with changes in the pancreas (islet cell vacuolation), eye (cataracts, lens degeneration), kidney (tubule vacuolation, chronic progressive nephropathy) and adipose tissue (atrophy) were identified in a 27 week repeat-dose toxicology study in rats that were immature at the beginning of the studies and were most prevalent in males at oral palbociclib doses ≥30 mg/kg/day (approximately 11 times the adult human exposure [AUC] at the recommended dose). Some of these findings (glycosuria/hyperglycemia, pancreatic islet cell vacuolation, and kidney tubule vacuolation) were present with lower incidence and severity in a 15 week repeat-dose toxicology study in immature rats. Altered glucose metabolism or associated changes in the pancreas, eye, kidney and adipose tissue were not identified in a 27-week repeat-dose toxicology study in rats that were mature at the beginning of the study and in dogs in repeat-dose toxicology studies up to 39 weeks duration.
Toxicities in teeth independent of altered glucose metabolism were observed in rats. Administration of 100 mg/kg palbociclib for 27 weeks (approximately 15 times the adult human exposure [AUC] at the recommended dose) resulted in abnormalities in growing incisor teeth (discolored, ameloblast degeneration/necrosis, mononuclear cell infiltrate). Other toxicities of potential concern to pediatric patients have not been evaluated in juvenile animals.
8.5 Geriatric Use
Of 444 patients who received IBRANCE in PALOMA-2, 181 patients (41%) were ≥65 years of age and 48 patients (11%) were ≥75 years of age. Of 347 patients who received IBRANCE in PALOMA-3, 86 patients (25%) were ≥65 years of age and 27 patients (8%) were ≥75 years of age. No overall differences in safety or effectiveness of IBRANCE were observed between these patients and younger patients.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Neutropenia [see Warnings and Precautions (5.1)]
-
•ILD/Pneumonitis [see Warnings and Precautions (5.2)]
7 Drug Interactions
Palbociclib is primarily metabolized by CYP3A and sulfotransferase (SULT) enzyme SULT2A1. In vivo, palbociclib is a time-dependent inhibitor of CYP3A.
8.7 Renal Impairment
No dose adjustment is required in patients with mild, moderate, or severe renal impairment (CrCl >15 mL/min). Based on a pharmacokinetic trial in subjects with varying degrees of renal function, the total palbociclib exposure (AUCINF) increased by 39%, 42%, and 31% with mild (60 mL/min ≤ CrCl <90 mL/min), moderate (30 mL/min ≤ CrCl <60 mL/min), and severe (CrCl <30 mL/min) renal impairment, respectively, relative to subjects with normal renal function. Peak palbociclib exposure (Cmax) increased by 17%, 12%, and 15% for mild, moderate, and severe renal impairment, respectively, relative to subjects with normal renal function. The pharmacokinetics of palbociclib have not been studied in patients requiring hemodialysis [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics (PK) of palbociclib were characterized in patients with solid tumors including advanced breast cancer and in healthy subjects.
2.2 Dose Modification
The recommended dose modifications for adverse reactions are listed in Tables 1, 2, and 3.
| Dose Level | Dose |
|---|---|
|
Recommended starting dose |
125 mg/day |
|
First dose reduction |
100 mg/day |
|
Second dose reduction |
75 mg/day If further dose reduction below 75 mg/day is required, discontinue.
|
| Grading according to CTCAE 4.0. CTCAE=Common Terminology Criteria for Adverse Events; LLN=lower limit of normal. |
|
|
Monitor complete blood counts prior to the start of IBRANCE therapy and at the beginning of each cycle, as well as on Day 15 of the first 2 cycles, and as clinically indicated. |
|
|
CTCAE Grade |
Dose Modifications |
|
Grade 1 or 2 |
No dose adjustment is required. |
|
Grade 3 |
Day 1 of cycle: |
|
Grade 3 neutropenia Absolute neutrophil count (ANC): Grade 1: ANC < LLN - 1500/mm3; Grade 2: ANC 1000 - <1500/mm3; Grade 3: ANC 500 - <1000/mm3; Grade 4: ANC <500/mm3. with fever ≥38.5 ºC and/or infection
|
At any time: |
|
Grade 4 |
At any time: |
| CTCAE Grade | Dose Modifications |
|---|---|
| Grading according to CTCAE 4.0. CTCAE=Common Terminology Criteria for Adverse Events. |
|
|
Grade 1 or 2 |
No dose adjustment is required. |
|
Grade ≥3 non-hematologic toxicity (if persisting despite optimal medical treatment) |
Withhold until symptoms resolve to:
Resume at the next lower dose. |
Permanently discontinue IBRANCE in patients with severe interstitial lung disease (ILD)/pneumonitis.
Refer to the Full Prescribing Information for coadministered endocrine therapy and/or inavolisib dose adjustment guidelines in the event of toxicity and other relevant safety information or contraindications.
8.6 Hepatic Impairment
No dose adjustment is required in patients with mild or moderate hepatic impairment (Child-Pugh classes A and B). For patients with severe hepatic impairment (Child-Pugh class C), the recommended dose of IBRANCE is 75 mg once daily for 21 consecutive days followed by 7 days off treatment to comprise a complete cycle of 28 days [see Dosage and Administration (2.2)]. Based on a pharmacokinetic trial in subjects with varying degrees of hepatic function, the palbociclib unbound exposure (unbound AUCINF) decreased by 17% in subjects with mild hepatic impairment (Child-Pugh class A), and increased by 34% and 77% in subjects with moderate (Child-Pugh class B) and severe (Child-Pugh class C) hepatic impairment, respectively, relative to subjects with normal hepatic function. Peak palbociclib unbound exposure (unbound Cmax) increased by 7%, 38% and 72% for mild, moderate and severe hepatic impairment, respectively, relative to subjects with normal hepatic function [see Clinical Pharmacology (12.3)].
Review the Full Prescribing Information for the aromatase inhibitor or fulvestrant for dose modifications related to hepatic impairment.
1 Indications and Usage
IBRANCE is indicated for the treatment of adult patients with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer in combination with:
-
•an aromatase inhibitor as initial endocrine-based therapy; or
-
•fulvestrant in patients with disease progression following endocrine therapy.
IBRANCE is indicated in combination with inavolisib and fulvestrant for the treatment of adult patients with endocrine-resistant, PIK3CA-mutated, HR-positive, HER2-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant endocrine therapy.
12.1 Mechanism of Action
Palbociclib is an inhibitor of cyclin-dependent kinases (CDK) 4 and 6. Cyclin D1 and CDK4/6 are downstream of signaling pathways which lead to cellular proliferation. In vitro, palbociclib reduced cellular proliferation of estrogen receptor (ER)-positive breast cancer cell lines by blocking progression of the cell from G1 into S phase of the cell cycle. Treatment of breast cancer cell lines with the combination of palbociclib and antiestrogens leads to decreased retinoblastoma (Rb) protein phosphorylation resulting in reduced E2F expression and signaling, and increased growth arrest compared to treatment with each drug alone. In vitro treatment of ER-positive breast cancer cell lines with the combination of palbociclib and antiestrogens led to increased cell senescence compared to each drug alone, which was sustained for up to 6 days following palbociclib removal and was greater if antiestrogen treatment was continued. In vivo studies using a patient-derived ER-positive breast cancer xenograft model demonstrated that the combination of palbociclib and letrozole increased the inhibition of Rb phosphorylation, downstream signaling, and tumor growth compared to each drug alone.
Human bone marrow mononuclear cells treated with palbociclib in the presence or absence of an anti-estrogen in vitro did not become senescent and resumed proliferation following palbociclib withdrawal.
5.3 Embryo Fetal Toxicity
Based on findings from animal studies and its mechanism of action, IBRANCE can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of palbociclib to pregnant rats and rabbits during organogenesis resulted in embryo-fetal toxicity at maternal exposures that were ≥4 times the human clinical exposure based on area under the curve (AUC). Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with IBRANCE and for at least 3 weeks after the last dose [see Use in Specific Populations (8.1 and 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions
-
•Neutropenia: Monitor complete blood count prior to start of IBRANCE therapy and at the beginning of each cycle, as well as on Day 15 of the first 2 cycles, and as clinically indicated. (2.2, 5.1)
-
•Interstitial Lung Disease (ILD)/Pneumonitis: Severe and fatal cases of ILD/pneumonitis have been reported. Monitor for pulmonary symptoms of ILD/pneumonitis. Interrupt IBRANCE immediately in patients with suspected ILD/pneumonitis. Permanently discontinue IBRANCE if severe ILD/pneumonitis occurs. (5.2)
-
•Embryo-Fetal Toxicity: IBRANCE can cause fetal harm. Advise patients of potential risk to a fetus and to use effective contraception. (5.3, 8.1, 8.3)
2 Dosage and Administration
3 Dosage Forms and Strengths
125 mg capsules: opaque, hard gelatin capsules, size 0, with caramel cap and body, printed with white ink "Pfizer" on the cap, "PBC 125" on the body.
100 mg capsules: opaque, hard gelatin capsules, size 1, with caramel cap and light orange body, printed with white ink "Pfizer" on the cap, "PBC 100" on the body.
75 mg capsules: opaque, hard gelatin capsules, size 2, with light orange cap and body, printed with white ink "Pfizer" on the cap, "PBC 75" on the body.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of IBRANCE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Respiratory Disorders: Interstitial lung disease (ILD)/non-infectious pneumonitis
Skin and Subcutaneous Tissue Disorders: Palmar-plantar erythrodysesthesia syndrome (PPES)
8 Use in Specific Populations
-
•Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.1 Recommended Dose and Schedule
The recommended dose of IBRANCE is a 125 mg capsule taken orally once daily for 21 consecutive days followed by 7 days off treatment to comprise a complete cycle of 28 days. IBRANCE capsule should be taken with food [see Clinical Pharmacology (12.3)].
Administer the recommended dose of an aromatase inhibitor when given with IBRANCE. Please refer to the Full Prescribing Information for the aromatase inhibitor being used.
When given with IBRANCE, the recommended dose of fulvestrant is 500 mg administered on Days 1, 15, 29, and once monthly thereafter. Please refer to the Full Prescribing Information of fulvestrant.
Refer to the Full Prescribing Information for inavolisib and fulvestrant for dosing information.
Advise patients to take their dose of IBRANCE at approximately the same time each day.
If the patient vomits or misses a dose, an additional dose should not be taken. The next prescribed dose should be taken at the usual time. IBRANCE capsules should be swallowed whole (do not chew, crush, or open them prior to swallowing). Capsules should not be ingested if they are broken, cracked, or otherwise not intact.
Pre/perimenopausal women treated with the combination IBRANCE plus an aromatase inhibitor or fulvestrant or IBRANCE plus inavolisib and fulvestrant therapy should also be treated with luteinizing hormone-releasing hormone (LHRH) agonists according to current clinical practice standards.
For men treated with combination IBRANCE plus aromatase inhibitor or IBRANCE plus inavolisib and fulvestrant therapy, consider treatment with an LHRH agonist according to current clinical practice standards.
16 How Supplied/storage and Handling
IBRANCE is supplied in the following strengths and package configurations:
| IBRANCE Capsules | |||
|---|---|---|---|
| Package Configuration | Capsule Strength (mg) | NDC | Capsule Description |
|
Bottles of 21 capsules |
125 |
NDC 0069-0189-21 |
opaque, hard gelatin capsules, size 0, with caramel cap and body, printed with white ink "Pfizer" on the cap, "PBC 125" on the body |
|
Bottles of 21 capsules |
100 |
NDC 0069-0188-21 |
opaque, hard gelatin capsules, size 1, with caramel cap and light orange body, printed with white ink "Pfizer" on the cap, "PBC 100" on the body |
|
Bottles of 21 capsules |
75 |
NDC 0069-0187-21 |
opaque, hard gelatin capsules, size 2, with light orange cap and body, printed with white ink "Pfizer" on the cap, "PBC 75" on the body |
5.2 Interstitial Lung Disease (ild)/pneumonitis
Severe, life-threatening, or fatal interstitial lung disease (ILD) and/or pneumonitis can occur in patients treated with cyclin-dependent kinase 4/6 (CDK4/6) inhibitors, including IBRANCE when taken in combination with endocrine therapy.
Across clinical trials (PALOMA-1, PALOMA-2, PALOMA-3), 1% of IBRANCE-treated patients had ILD/pneumonitis of any grade, 0.1% had Grade 3 or 4 and no fatal cases were reported. Additional cases of ILD/pneumonitis have been observed in the postmarketing setting, with fatalities reported [see Adverse Reactions (6.2)].
Monitor patients for pulmonary symptoms indicative of ILD/pneumonitis (e.g. hypoxia, cough, dyspnea). In patients who have new or worsening respiratory symptoms and are suspected to have developed pneumonitis, interrupt IBRANCE immediately and evaluate the patient. Permanently discontinue IBRANCE in patients with severe ILD or pneumonitis [see Dosage and Administration (2.2)].
Principal Display Panel 75 Mg Capsule Bottle Label
PROFESSIONAL SAMPLE - NOT FOR SALE
NDC 63539-187-11
Pfizer
Ibrance
®
(palbociclib)
capsules
75 mg
21 Capsules
Rx only
Principal Display Panel 100 Mg Capsule Bottle Label
PROFESSIONAL SAMPLE - NOT FOR SALE
NDC 63539-188-11
Pfizer
Ibrance
®
(palbociclib)
capsules
100 mg
21 Capsules
Rx only
Principal Display Panel 125 Mg Capsule Bottle Label
PROFESSIONAL SAMPLE - NOT FOR SALE
NDC 63539-189-11
Pfizer
Ibrance™
(palbociclib)
capsules
125 mg
For Oncology Use Only
21 Capsules
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Palbociclib was assessed for carcinogenicity in a 6-month transgenic mouse study and in a 2-year rat study. Oral administration of palbociclib for 2 years resulted in an increased incidence of microglial cell tumors in the central nervous system of male rats at a dose of 30 mg/kg/day (approximately 8 times the human clinical exposure based on AUC). There were no neoplastic findings in female rats at doses up to 200 mg/kg/day (approximately 5 times the human clinical exposure based on AUC). Oral administration of palbociclib to male and female rasH2 transgenic mice for 6 months did not result in increased incidence of neoplasms at doses up to 60 mg/kg/day.
Palbociclib was aneugenic in Chinese Hamster Ovary cells in vitro and in the bone marrow of male rats at doses ≥100 mg/kg/day for 3 weeks. Palbociclib was not mutagenic in an in vitro bacterial reverse mutation (Ames) assay and was not clastogenic in the in vitro human lymphocyte chromosome aberration assay.
In a fertility study in female rats, palbociclib did not affect mating or fertility at any dose up to 300 mg/kg/day (approximately 4 times human clinical exposure based on AUC) and no adverse effects were observed in the female reproductive tissues in repeat-dose toxicity studies up to 300 mg/kg/day in the rat and 3 mg/kg/day in the dog (approximately 6 times and similar to human exposure [AUC], at the recommended dose, respectively).
The adverse effects of palbociclib on male reproductive function and fertility were observed in the repeat-dose toxicology studies in rats and dogs and a male fertility study in rats. In repeat-dose toxicology studies, palbociclib-related findings in the testis, epididymis, prostate, and seminal vesicle at ≥30 mg/kg/day in rats and ≥0.2 mg/kg/day in dogs included decreased organ weight, atrophy or degeneration, hypospermia, intratubular cellular debris, and decreased secretion. Partial reversibility of male reproductive organ effects was observed in the rat and dog following a 4- and 12-week non-dosing period, respectively. These doses in rats and dogs resulted in approximately ≥10 and 0.1 times, respectively, the exposure [AUC] in humans at the recommended dose. In the fertility and early embryonic development study in male rats, palbociclib caused no effects on mating but resulted in a slight decrease in fertility in association with lower sperm motility and density at 100 mg/kg/day with projected exposure levels [AUC] of 20 times the exposure in humans at the recommended dose.
7.3 Drugs That May Have Their Plasma Concentrations Altered By Palbociclib
Coadministration of midazolam with multiple doses of IBRANCE increased the midazolam plasma exposure by 61%, in healthy subjects, compared to administration of midazolam alone. The dose of the sensitive CYP3A substrate with a narrow therapeutic index (e.g., alfentanil, cyclosporine, dihydroergotamine, ergotamine, everolimus, fentanyl, pimozide, quinidine, sirolimus, and tacrolimus) may need to be reduced, as IBRANCE may increase its exposure [see Clinical Pharmacology (12.3)].
Structured Label Content
Section 42229-5 (42229-5)
Dose Modifications for Use With Strong CYP3A Inhibitors
Avoid concomitant use of strong CYP3A inhibitors and consider an alternative concomitant medication with no or minimal CYP3A inhibition. If patients must be coadministered a strong CYP3A inhibitor, reduce the IBRANCE dose to 75 mg once daily. If the strong inhibitor is discontinued, increase the IBRANCE dose (after 3 to 5 half-lives of the inhibitor) to the dose used prior to the initiation of the strong CYP3A inhibitor [see Drug Interactions (7.1) and Clinical Pharmacology (12.3)].
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. Revised September 2025 | ||
|
PATIENT INFORMATION
|
||
|
What is the most important information I should know about IBRANCE? IBRANCE may cause serious side effects, including: Low white blood cell counts (neutropenia). Low white blood cell counts are very common when taking IBRANCE and may cause serious infections that can lead to death. Your healthcare provider should check your white blood cell counts before and during treatment. If you develop low white blood cell counts during treatment with IBRANCE, your healthcare provider may stop your treatment, decrease your dose, or may tell you to wait to begin your treatment cycle. Tell your healthcare provider right away if you have signs and symptoms of low white blood cell counts or infections such as fever and chills. Lung problems (pneumonitis). IBRANCE may cause severe or life-threatening inflammation of the lungs during treatment that can lead to death. Tell your healthcare provider right away if you have any new or worsening symptoms, including:
Your healthcare provider may interrupt or stop treatment with IBRANCE completely if your symptoms are severe. See "What are the possible side effects of IBRANCE?" for more information about side effects. |
||
|
What is IBRANCE? IBRANCE is a prescription medicine used: In adults to treat hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative breast cancer that has spread to other parts of the body (metastatic) in combination with:
In adults with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative and with an abnormal phosphatidylinositol-3-kinase catalytic subunit alpha (PIK3CA) gene breast cancer that has spread to nearby tissue or lymph nodes (locally advanced), or to other parts of the body (metastatic) in combination with:
It is not known if IBRANCE is safe and effective in children. |
||
|
What should I tell my healthcare provider before taking IBRANCE? Before taking IBRANCE, tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. IBRANCE and other medicines may affect each other causing side effects. Know the medicines you take. Keep a list of them to show your healthcare provider or pharmacist when you get a new medicine. |
||
|
How should I take IBRANCE?
|
||
|
What are the possible side effects of IBRANCE? IBRANCE may cause serious side effects. See "What is the most important information I should know about IBRANCE?" The most common side effects of IBRANCE when used with either letrozole or fulvestrant include:
|
||
|
|
|
|
|
|
|
The most common side effects of IBRANCE when used in combination with inavolisib plus fulvestrant include:
IBRANCE may cause fertility problems in males. This may affect your ability to father a child. Talk to your healthcare provider about family planning options before starting IBRANCE if this is a concern for you. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all of the possible side effects of IBRANCE. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
|
How should I store IBRANCE?
Keep IBRANCE and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of IBRANCE Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use IBRANCE for a condition for which it was not prescribed. Do not give IBRANCE to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for more information about IBRANCE that is written for health professionals. |
||
|
What are the ingredients in IBRANCE? Active ingredient: palbociclib Inactive ingredients: microcrystalline cellulose, lactose monohydrate, sodium starch glycolate, colloidal silicon dioxide, magnesium stearate, and hard gelatin capsule shells. The light orange, light orange/caramel and caramel opaque capsule shells contain: gelatin, red iron oxide, yellow iron oxide, and titanium dioxide. The printing ink contains: shellac, titanium dioxide, ammonium hydroxide, propylene glycol, and simethicone.
For more information, go to www.pfizer.com or call 1-800-438-1985. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store at 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 °F to 86 °F) [see USP Controlled Room Temperature].
11 Description (11 DESCRIPTION)
IBRANCE capsules for oral administration contain 125 mg, 100 mg, or 75 mg of palbociclib, a kinase inhibitor. The molecular formula for palbociclib is C24H29N7O2. The molecular weight is 447.54 daltons. The chemical name is 6-acetyl-8-cyclopentyl-5-methyl-2-{[5-(piperazin-1-yl)pyridin-2-yl]amino}pyrido[2,3-d]pyrimidin-7(8H)-one, and its structural formula is:
Palbociclib is a yellow to orange powder with pKa of 7.4 (the secondary piperazine nitrogen) and 3.9 (the pyridine nitrogen). At or below pH 4, palbociclib behaves as a high-solubility compound. Above pH 4, the solubility of the drug substance reduces significantly.
Inactive ingredients: Microcrystalline cellulose, lactose monohydrate, sodium starch glycolate, colloidal silicon dioxide, magnesium stearate, and hard gelatin capsule shells. The light orange, light orange/caramel, and caramel opaque capsule shells contain gelatin, red iron oxide, yellow iron oxide, and titanium dioxide; the printing ink contains shellac, titanium dioxide, ammonium hydroxide, propylene glycol, and simethicone.
5.1 Neutropenia
Neutropenia was the most frequently reported adverse reaction in PALOMA-2 with an incidence of 80% and PALOMA-3 with an incidence of 83%. A Grade ≥3 decrease in neutrophil counts was reported in 66% of patients receiving IBRANCE plus letrozole in PALOMA-2 and 66% of patients receiving IBRANCE plus fulvestrant in PALOMA-3. In PALOMA-2 and PALOMA-3, the median time to first episode of any grade neutropenia was 15 days and the median duration of Grade ≥3 neutropenia was 7 days [see Adverse Reactions (6.1)].
Monitor complete blood counts prior to starting IBRANCE therapy and at the beginning of each cycle, as well as on Day 15 of the first 2 cycles, and as clinically indicated. Dose interruption, dose reduction, or delay in starting treatment cycles is recommended for patients who develop Grade 3 or 4 neutropenia [see Dosage and Administration (2.2)].
Febrile neutropenia has been reported in 1.8% of patients exposed to IBRANCE across PALOMA-2 and PALOMA-3. One death due to neutropenic sepsis was observed in PALOMA-3. Physicians should inform patients to promptly report any episodes of fever [see Patient Counseling Information (17)].
8.4 Pediatric Use
The safety and effectiveness of IBRANCE in pediatric patients have not been established.
The safety and effectiveness of IBRANCE were assessed but not established in three trials: one open-label trial [A5481092, (NCT03709680)] that included 98 pediatric patients 2 to <17 years of age who received IBRANCE in combination with chemotherapy for recurrent or refractory solid tumors and two open-label trials that included 42 pediatric patients 4 to <17 years of age who received IBRANCE as a single agent for recurrent or refractory solid tumors [APEC1621I, (NCT03526250)] or primary central nervous system (CNS) tumors [PBTC-042, (NCT02255461)].
No new safety signals were observed in these trials. Palbociclib exposures in pediatric patients who received IBRANCE as a single agent or in combination were within range of those observed in adults given a similar dose based on body surface area.
Juvenile Animal Toxicity Data
Altered glucose metabolism (glycosuria, hyperglycemia, decreased insulin) associated with changes in the pancreas (islet cell vacuolation), eye (cataracts, lens degeneration), kidney (tubule vacuolation, chronic progressive nephropathy) and adipose tissue (atrophy) were identified in a 27 week repeat-dose toxicology study in rats that were immature at the beginning of the studies and were most prevalent in males at oral palbociclib doses ≥30 mg/kg/day (approximately 11 times the adult human exposure [AUC] at the recommended dose). Some of these findings (glycosuria/hyperglycemia, pancreatic islet cell vacuolation, and kidney tubule vacuolation) were present with lower incidence and severity in a 15 week repeat-dose toxicology study in immature rats. Altered glucose metabolism or associated changes in the pancreas, eye, kidney and adipose tissue were not identified in a 27-week repeat-dose toxicology study in rats that were mature at the beginning of the study and in dogs in repeat-dose toxicology studies up to 39 weeks duration.
Toxicities in teeth independent of altered glucose metabolism were observed in rats. Administration of 100 mg/kg palbociclib for 27 weeks (approximately 15 times the adult human exposure [AUC] at the recommended dose) resulted in abnormalities in growing incisor teeth (discolored, ameloblast degeneration/necrosis, mononuclear cell infiltrate). Other toxicities of potential concern to pediatric patients have not been evaluated in juvenile animals.
8.5 Geriatric Use
Of 444 patients who received IBRANCE in PALOMA-2, 181 patients (41%) were ≥65 years of age and 48 patients (11%) were ≥75 years of age. Of 347 patients who received IBRANCE in PALOMA-3, 86 patients (25%) were ≥65 years of age and 27 patients (8%) were ≥75 years of age. No overall differences in safety or effectiveness of IBRANCE were observed between these patients and younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
-
•Neutropenia [see Warnings and Precautions (5.1)]
-
•ILD/Pneumonitis [see Warnings and Precautions (5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Palbociclib is primarily metabolized by CYP3A and sulfotransferase (SULT) enzyme SULT2A1. In vivo, palbociclib is a time-dependent inhibitor of CYP3A.
8.7 Renal Impairment
No dose adjustment is required in patients with mild, moderate, or severe renal impairment (CrCl >15 mL/min). Based on a pharmacokinetic trial in subjects with varying degrees of renal function, the total palbociclib exposure (AUCINF) increased by 39%, 42%, and 31% with mild (60 mL/min ≤ CrCl <90 mL/min), moderate (30 mL/min ≤ CrCl <60 mL/min), and severe (CrCl <30 mL/min) renal impairment, respectively, relative to subjects with normal renal function. Peak palbociclib exposure (Cmax) increased by 17%, 12%, and 15% for mild, moderate, and severe renal impairment, respectively, relative to subjects with normal renal function. The pharmacokinetics of palbociclib have not been studied in patients requiring hemodialysis [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics (PK) of palbociclib were characterized in patients with solid tumors including advanced breast cancer and in healthy subjects.
2.2 Dose Modification
The recommended dose modifications for adverse reactions are listed in Tables 1, 2, and 3.
| Dose Level | Dose |
|---|---|
|
Recommended starting dose |
125 mg/day |
|
First dose reduction |
100 mg/day |
|
Second dose reduction |
75 mg/day If further dose reduction below 75 mg/day is required, discontinue.
|
| Grading according to CTCAE 4.0. CTCAE=Common Terminology Criteria for Adverse Events; LLN=lower limit of normal. |
|
|
Monitor complete blood counts prior to the start of IBRANCE therapy and at the beginning of each cycle, as well as on Day 15 of the first 2 cycles, and as clinically indicated. |
|
|
CTCAE Grade |
Dose Modifications |
|
Grade 1 or 2 |
No dose adjustment is required. |
|
Grade 3 |
Day 1 of cycle: |
|
Grade 3 neutropenia Absolute neutrophil count (ANC): Grade 1: ANC < LLN - 1500/mm3; Grade 2: ANC 1000 - <1500/mm3; Grade 3: ANC 500 - <1000/mm3; Grade 4: ANC <500/mm3. with fever ≥38.5 ºC and/or infection
|
At any time: |
|
Grade 4 |
At any time: |
| CTCAE Grade | Dose Modifications |
|---|---|
| Grading according to CTCAE 4.0. CTCAE=Common Terminology Criteria for Adverse Events. |
|
|
Grade 1 or 2 |
No dose adjustment is required. |
|
Grade ≥3 non-hematologic toxicity (if persisting despite optimal medical treatment) |
Withhold until symptoms resolve to:
Resume at the next lower dose. |
Permanently discontinue IBRANCE in patients with severe interstitial lung disease (ILD)/pneumonitis.
Refer to the Full Prescribing Information for coadministered endocrine therapy and/or inavolisib dose adjustment guidelines in the event of toxicity and other relevant safety information or contraindications.
8.6 Hepatic Impairment
No dose adjustment is required in patients with mild or moderate hepatic impairment (Child-Pugh classes A and B). For patients with severe hepatic impairment (Child-Pugh class C), the recommended dose of IBRANCE is 75 mg once daily for 21 consecutive days followed by 7 days off treatment to comprise a complete cycle of 28 days [see Dosage and Administration (2.2)]. Based on a pharmacokinetic trial in subjects with varying degrees of hepatic function, the palbociclib unbound exposure (unbound AUCINF) decreased by 17% in subjects with mild hepatic impairment (Child-Pugh class A), and increased by 34% and 77% in subjects with moderate (Child-Pugh class B) and severe (Child-Pugh class C) hepatic impairment, respectively, relative to subjects with normal hepatic function. Peak palbociclib unbound exposure (unbound Cmax) increased by 7%, 38% and 72% for mild, moderate and severe hepatic impairment, respectively, relative to subjects with normal hepatic function [see Clinical Pharmacology (12.3)].
Review the Full Prescribing Information for the aromatase inhibitor or fulvestrant for dose modifications related to hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
IBRANCE is indicated for the treatment of adult patients with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer in combination with:
-
•an aromatase inhibitor as initial endocrine-based therapy; or
-
•fulvestrant in patients with disease progression following endocrine therapy.
IBRANCE is indicated in combination with inavolisib and fulvestrant for the treatment of adult patients with endocrine-resistant, PIK3CA-mutated, HR-positive, HER2-negative, locally advanced or metastatic breast cancer, as detected by an FDA-approved test, following recurrence on or after completing adjuvant endocrine therapy.
12.1 Mechanism of Action
Palbociclib is an inhibitor of cyclin-dependent kinases (CDK) 4 and 6. Cyclin D1 and CDK4/6 are downstream of signaling pathways which lead to cellular proliferation. In vitro, palbociclib reduced cellular proliferation of estrogen receptor (ER)-positive breast cancer cell lines by blocking progression of the cell from G1 into S phase of the cell cycle. Treatment of breast cancer cell lines with the combination of palbociclib and antiestrogens leads to decreased retinoblastoma (Rb) protein phosphorylation resulting in reduced E2F expression and signaling, and increased growth arrest compared to treatment with each drug alone. In vitro treatment of ER-positive breast cancer cell lines with the combination of palbociclib and antiestrogens led to increased cell senescence compared to each drug alone, which was sustained for up to 6 days following palbociclib removal and was greater if antiestrogen treatment was continued. In vivo studies using a patient-derived ER-positive breast cancer xenograft model demonstrated that the combination of palbociclib and letrozole increased the inhibition of Rb phosphorylation, downstream signaling, and tumor growth compared to each drug alone.
Human bone marrow mononuclear cells treated with palbociclib in the presence or absence of an anti-estrogen in vitro did not become senescent and resumed proliferation following palbociclib withdrawal.
5.3 Embryo Fetal Toxicity (5.3 Embryo-Fetal Toxicity)
Based on findings from animal studies and its mechanism of action, IBRANCE can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, administration of palbociclib to pregnant rats and rabbits during organogenesis resulted in embryo-fetal toxicity at maternal exposures that were ≥4 times the human clinical exposure based on area under the curve (AUC). Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with IBRANCE and for at least 3 weeks after the last dose [see Use in Specific Populations (8.1 and 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Neutropenia: Monitor complete blood count prior to start of IBRANCE therapy and at the beginning of each cycle, as well as on Day 15 of the first 2 cycles, and as clinically indicated. (2.2, 5.1)
-
•Interstitial Lung Disease (ILD)/Pneumonitis: Severe and fatal cases of ILD/pneumonitis have been reported. Monitor for pulmonary symptoms of ILD/pneumonitis. Interrupt IBRANCE immediately in patients with suspected ILD/pneumonitis. Permanently discontinue IBRANCE if severe ILD/pneumonitis occurs. (5.2)
-
•Embryo-Fetal Toxicity: IBRANCE can cause fetal harm. Advise patients of potential risk to a fetus and to use effective contraception. (5.3, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
125 mg capsules: opaque, hard gelatin capsules, size 0, with caramel cap and body, printed with white ink "Pfizer" on the cap, "PBC 125" on the body.
100 mg capsules: opaque, hard gelatin capsules, size 1, with caramel cap and light orange body, printed with white ink "Pfizer" on the cap, "PBC 100" on the body.
75 mg capsules: opaque, hard gelatin capsules, size 2, with light orange cap and body, printed with white ink "Pfizer" on the cap, "PBC 75" on the body.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of IBRANCE. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Respiratory Disorders: Interstitial lung disease (ILD)/non-infectious pneumonitis
Skin and Subcutaneous Tissue Disorders: Palmar-plantar erythrodysesthesia syndrome (PPES)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
-
•Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.1 Recommended Dose and Schedule
The recommended dose of IBRANCE is a 125 mg capsule taken orally once daily for 21 consecutive days followed by 7 days off treatment to comprise a complete cycle of 28 days. IBRANCE capsule should be taken with food [see Clinical Pharmacology (12.3)].
Administer the recommended dose of an aromatase inhibitor when given with IBRANCE. Please refer to the Full Prescribing Information for the aromatase inhibitor being used.
When given with IBRANCE, the recommended dose of fulvestrant is 500 mg administered on Days 1, 15, 29, and once monthly thereafter. Please refer to the Full Prescribing Information of fulvestrant.
Refer to the Full Prescribing Information for inavolisib and fulvestrant for dosing information.
Advise patients to take their dose of IBRANCE at approximately the same time each day.
If the patient vomits or misses a dose, an additional dose should not be taken. The next prescribed dose should be taken at the usual time. IBRANCE capsules should be swallowed whole (do not chew, crush, or open them prior to swallowing). Capsules should not be ingested if they are broken, cracked, or otherwise not intact.
Pre/perimenopausal women treated with the combination IBRANCE plus an aromatase inhibitor or fulvestrant or IBRANCE plus inavolisib and fulvestrant therapy should also be treated with luteinizing hormone-releasing hormone (LHRH) agonists according to current clinical practice standards.
For men treated with combination IBRANCE plus aromatase inhibitor or IBRANCE plus inavolisib and fulvestrant therapy, consider treatment with an LHRH agonist according to current clinical practice standards.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
IBRANCE is supplied in the following strengths and package configurations:
| IBRANCE Capsules | |||
|---|---|---|---|
| Package Configuration | Capsule Strength (mg) | NDC | Capsule Description |
|
Bottles of 21 capsules |
125 |
NDC 0069-0189-21 |
opaque, hard gelatin capsules, size 0, with caramel cap and body, printed with white ink "Pfizer" on the cap, "PBC 125" on the body |
|
Bottles of 21 capsules |
100 |
NDC 0069-0188-21 |
opaque, hard gelatin capsules, size 1, with caramel cap and light orange body, printed with white ink "Pfizer" on the cap, "PBC 100" on the body |
|
Bottles of 21 capsules |
75 |
NDC 0069-0187-21 |
opaque, hard gelatin capsules, size 2, with light orange cap and body, printed with white ink "Pfizer" on the cap, "PBC 75" on the body |
5.2 Interstitial Lung Disease (ild)/pneumonitis (5.2 Interstitial Lung Disease (ILD)/Pneumonitis)
Severe, life-threatening, or fatal interstitial lung disease (ILD) and/or pneumonitis can occur in patients treated with cyclin-dependent kinase 4/6 (CDK4/6) inhibitors, including IBRANCE when taken in combination with endocrine therapy.
Across clinical trials (PALOMA-1, PALOMA-2, PALOMA-3), 1% of IBRANCE-treated patients had ILD/pneumonitis of any grade, 0.1% had Grade 3 or 4 and no fatal cases were reported. Additional cases of ILD/pneumonitis have been observed in the postmarketing setting, with fatalities reported [see Adverse Reactions (6.2)].
Monitor patients for pulmonary symptoms indicative of ILD/pneumonitis (e.g. hypoxia, cough, dyspnea). In patients who have new or worsening respiratory symptoms and are suspected to have developed pneumonitis, interrupt IBRANCE immediately and evaluate the patient. Permanently discontinue IBRANCE in patients with severe ILD or pneumonitis [see Dosage and Administration (2.2)].
Principal Display Panel 75 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 75 mg Capsule Bottle Label)
PROFESSIONAL SAMPLE - NOT FOR SALE
NDC 63539-187-11
Pfizer
Ibrance
®
(palbociclib)
capsules
75 mg
21 Capsules
Rx only
Principal Display Panel 100 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Capsule Bottle Label)
PROFESSIONAL SAMPLE - NOT FOR SALE
NDC 63539-188-11
Pfizer
Ibrance
®
(palbociclib)
capsules
100 mg
21 Capsules
Rx only
Principal Display Panel 125 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 125 mg Capsule Bottle Label)
PROFESSIONAL SAMPLE - NOT FOR SALE
NDC 63539-189-11
Pfizer
Ibrance™
(palbociclib)
capsules
125 mg
For Oncology Use Only
21 Capsules
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Palbociclib was assessed for carcinogenicity in a 6-month transgenic mouse study and in a 2-year rat study. Oral administration of palbociclib for 2 years resulted in an increased incidence of microglial cell tumors in the central nervous system of male rats at a dose of 30 mg/kg/day (approximately 8 times the human clinical exposure based on AUC). There were no neoplastic findings in female rats at doses up to 200 mg/kg/day (approximately 5 times the human clinical exposure based on AUC). Oral administration of palbociclib to male and female rasH2 transgenic mice for 6 months did not result in increased incidence of neoplasms at doses up to 60 mg/kg/day.
Palbociclib was aneugenic in Chinese Hamster Ovary cells in vitro and in the bone marrow of male rats at doses ≥100 mg/kg/day for 3 weeks. Palbociclib was not mutagenic in an in vitro bacterial reverse mutation (Ames) assay and was not clastogenic in the in vitro human lymphocyte chromosome aberration assay.
In a fertility study in female rats, palbociclib did not affect mating or fertility at any dose up to 300 mg/kg/day (approximately 4 times human clinical exposure based on AUC) and no adverse effects were observed in the female reproductive tissues in repeat-dose toxicity studies up to 300 mg/kg/day in the rat and 3 mg/kg/day in the dog (approximately 6 times and similar to human exposure [AUC], at the recommended dose, respectively).
The adverse effects of palbociclib on male reproductive function and fertility were observed in the repeat-dose toxicology studies in rats and dogs and a male fertility study in rats. In repeat-dose toxicology studies, palbociclib-related findings in the testis, epididymis, prostate, and seminal vesicle at ≥30 mg/kg/day in rats and ≥0.2 mg/kg/day in dogs included decreased organ weight, atrophy or degeneration, hypospermia, intratubular cellular debris, and decreased secretion. Partial reversibility of male reproductive organ effects was observed in the rat and dog following a 4- and 12-week non-dosing period, respectively. These doses in rats and dogs resulted in approximately ≥10 and 0.1 times, respectively, the exposure [AUC] in humans at the recommended dose. In the fertility and early embryonic development study in male rats, palbociclib caused no effects on mating but resulted in a slight decrease in fertility in association with lower sperm motility and density at 100 mg/kg/day with projected exposure levels [AUC] of 20 times the exposure in humans at the recommended dose.
7.3 Drugs That May Have Their Plasma Concentrations Altered By Palbociclib (7.3 Drugs That May Have Their Plasma Concentrations Altered by Palbociclib)
Coadministration of midazolam with multiple doses of IBRANCE increased the midazolam plasma exposure by 61%, in healthy subjects, compared to administration of midazolam alone. The dose of the sensitive CYP3A substrate with a narrow therapeutic index (e.g., alfentanil, cyclosporine, dihydroergotamine, ergotamine, everolimus, fentanyl, pimozide, quinidine, sirolimus, and tacrolimus) may need to be reduced, as IBRANCE may increase its exposure [see Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:11.840962 · Updated: 2026-03-14T22:36:18.567883