Cinchona Officinalis 30c
852d03b8-4af0-6c68-e053-2991aa0aee56
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
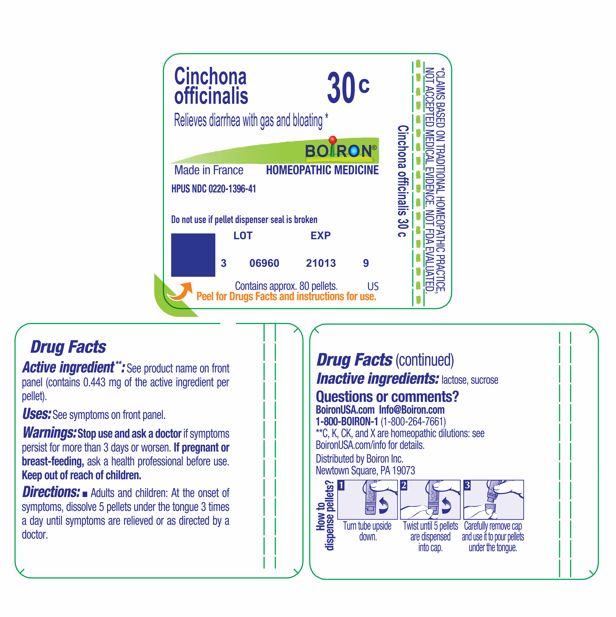
Active ingredient** (in each pellet) Cinchona officinalis 30C HPUS (0.44 mg) The letters "HPUS" indicate that the component in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
Medication Information
Indications and Usage
temporarily relieves symptoms of diarrheas such as:
- bloating
- gas
- fatigue
- physical weakness
Dosage and Administration
- Drink plenty or clear fluids to help prevent dehydration cause by diarrhea.
- Adults and children: At the onset of symptoms, dissolve 5 pellets under the tongue 3 times a day until symptoms are relieved or as directed by a doctor.
Description
Active ingredient** (in each pellet) Cinchona officinalis 30C HPUS (0.44 mg) The letters "HPUS" indicate that the component in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
Section 42229-5
Do not use if pellet dispenser seal is broken.
- do not use if glued carton end flaps are open or if pellet dispenser seal is broken
- contains 0.25 g of sugar per dose
- retain carton for full drug facts
DIARRHEA RELIEF & RECOVERY*
Bloating & Gas Fatigue Physical Weakness*
1 TUBE
3 TUBES
APPROX. 80 PELLETS EACH
TOTAL 240 PELLETS
No Known Drug Interactions
How to dispense pellets? Turn tube upside down. Twist until 5 pellets are dispensed. Remove cap and pour pellets under the tongue.
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
*C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details.
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if
- symptoms get worse
- diarrhea lasts for more than 2 days.
Section 50569-3
Ask a doctor before use if you have
- fever
- mucus in stool.
Section 50570-1
Do not use if you have
- bloody or black stool.
Section 51727-6
lactose, sucrose
Section 51945-4
Section 53413-1
BoironUSA.com [email protected]
1-800-BOIRON-1 (1-800-264-7661)
Distributed by Boiron, Inc. Newtown Square, PA 19073
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purpose*
Cinchona officinalis 30C HPUS (0.44 mg) .... Relieves diarrhea with gas and bloating
Section 55106-9
Active ingredient** (in each pellet)
Cinchona officinalis 30C HPUS (0.44 mg)
The letters "HPUS" indicate that the component in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
Structured Label Content
Indications and Usage (34067-9)
temporarily relieves symptoms of diarrheas such as:
- bloating
- gas
- fatigue
- physical weakness
Dosage and Administration (34068-7)
- Drink plenty or clear fluids to help prevent dehydration cause by diarrhea.
- Adults and children: At the onset of symptoms, dissolve 5 pellets under the tongue 3 times a day until symptoms are relieved or as directed by a doctor.
Section 42229-5 (42229-5)
Do not use if pellet dispenser seal is broken.
- do not use if glued carton end flaps are open or if pellet dispenser seal is broken
- contains 0.25 g of sugar per dose
- retain carton for full drug facts
DIARRHEA RELIEF & RECOVERY*
Bloating & Gas Fatigue Physical Weakness*
1 TUBE
3 TUBES
APPROX. 80 PELLETS EACH
TOTAL 240 PELLETS
No Known Drug Interactions
How to dispense pellets? Turn tube upside down. Twist until 5 pellets are dispensed. Remove cap and pour pellets under the tongue.
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
*C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details.
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- symptoms get worse
- diarrhea lasts for more than 2 days.
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- fever
- mucus in stool.
Section 50570-1 (50570-1)
Do not use if you have
- bloody or black stool.
Section 51727-6 (51727-6)
lactose, sucrose
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
BoironUSA.com [email protected]
1-800-BOIRON-1 (1-800-264-7661)
Distributed by Boiron, Inc. Newtown Square, PA 19073
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purpose*
Cinchona officinalis 30C HPUS (0.44 mg) .... Relieves diarrhea with gas and bloating
Section 55106-9 (55106-9)
Active ingredient** (in each pellet)
Cinchona officinalis 30C HPUS (0.44 mg)
The letters "HPUS" indicate that the component in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:12.239760 · Updated: 2026-03-14T23:17:37.584721