Giltuss Multi-symptom Cold & Flu
8518fd91-981e-de78-e053-2a91aa0afcb2
34390-5
HUMAN OTC DRUG LABEL
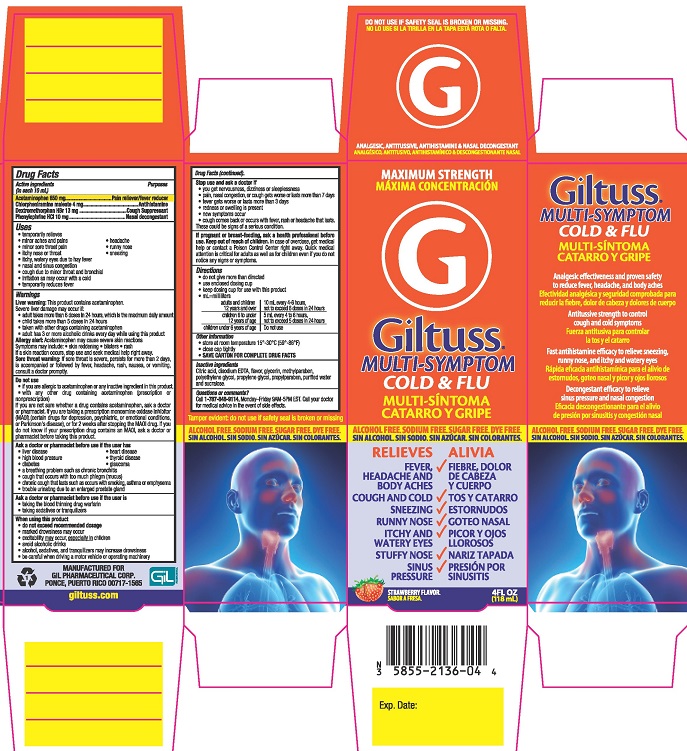
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Medication Information
Warnings and Precautions
Warnings
Liver warning:This product contains acetaminophen. Severe liver damage may occur if:
- adult takes more than 6 doses in 24 hours, which is the maximum daily amount
- child takes more than 5 doses in 24 hours
- taken with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product
Allergy alert:Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
- hives
- facial swelling
- asthma (wheezing)
- shock
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning:If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Indications and Usage
Uses
- temporarily relieves:
- minor aches and pains
- headache
- minor sore throat pain
- runny nose
- itchy nose or throat
- sneezing
- itchy watery eyes due to hay fever
- nasal and sinus congestion
- cough due to minor throat and bronchial irritation as may occur with a cold
- temporarily reduces fever
Dosage and Administration
Directions
- do not give more than directed
- use enclosed dosing cup
- keep dosing cup for use with this product
- mL = milliliter
| adults and children 12 years and over | 10 mL every 4-6 hours, not to exceed 6 doses in 2 hours |
| children 6 to under 12 years of age | 5 mL every 4-6 hours, not to exceed 5 doses in 24 hours |
| children under 6 years of age | Do not use |
Description
Drug Facts
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Section 50566-9
Stop use and ask a doctor if
- you get nervousness, dizzyness or sleeplessness
- pain, nasal congestion, or cough gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
- cough comes back or occurs with fever, rash or headache that lasts.
These could be signs of a serious condition.
Section 50567-7
When using this product
- do not exceed recommended dosage
- marked drowsiness may occur
- excitability may occur, especially in children
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Section 50568-5
Ask a doctor or pharmacist before use if the user is
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers
Section 50569-3
Ask a doctor or pharmacist before use if the user has
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- glaucoma
- a breathing problem such as chronic bronchitis
- cough that occurs with too much phlegm (mucus)
- chronic cough that lasts such as occurs with smoking, asthma, or emphysema
- trouble urinating due to enlargement of the prostate gland
Section 50570-1
Do not use
- if you are allergic to acetaminophen or any inactive ingredient in this product
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Section 51727-6
Inactive ingredients citric acid, disodium EDTA, flavor, glycerin, methylparaben, polyethylene glycol, propylene glycol, propylparaben, purified water and sucralose
Section 51945-4
MAXIMUM STRENGTH
Giltuss® MULTI-SYMPTOM
COLD & FLU
ANALGESIC, ANTITUSSIVE, ANTIHISTAMINE & NASAL DECONGESTANT
ALCOHOL FREE. SODIUM FREE. SUGAR FREE. DYE FREE
RELIEVES
- FEVER, HEADACHE AND BODY ACHES
- COUGH AND COLD
- SNEEZING
- RUNNY NOSE
- ITCHY AND WATERY EYES
- STUFFY NOSE
- SINUS PRESSURE
STRAWBERRY FLAVOR
4FL OZ (118 mL)
Section 53413-1
Questions or comments?
Call 1-787-848-9114,Monday-Friday 9AM - 5PM EST. Call your doctor for medical advice in the event of side effects.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purposes
Pain reliever/fever reducer
Antihistamine
Cough Suppressant
Nasal decongestant
Section 55106-9
Active ingredients (in each 10 mL)
Acetaminophen 650 mg
Chlorpheniramine maleate 4 mg
Dextromethorphan hydrobromide 13 mg
Phenylephrine hydrochloride 10 mg
Section 60561-8
Other information
- store at room temperature 15° - 30°C (59° - 86°F)
- close cap tightly
- SAVE CARTON FOR COMPLETE DRUG FACTS
Structured Label Content
Indications and Usage (34067-9)
Uses
- temporarily relieves:
- minor aches and pains
- headache
- minor sore throat pain
- runny nose
- itchy nose or throat
- sneezing
- itchy watery eyes due to hay fever
- nasal and sinus congestion
- cough due to minor throat and bronchial irritation as may occur with a cold
- temporarily reduces fever
Dosage and Administration (34068-7)
Directions
- do not give more than directed
- use enclosed dosing cup
- keep dosing cup for use with this product
- mL = milliliter
| adults and children 12 years and over | 10 mL every 4-6 hours, not to exceed 6 doses in 2 hours |
| children 6 to under 12 years of age | 5 mL every 4-6 hours, not to exceed 5 doses in 24 hours |
| children under 6 years of age | Do not use |
Warnings and Precautions (34071-1)
Warnings
Liver warning:This product contains acetaminophen. Severe liver damage may occur if:
- adult takes more than 6 doses in 24 hours, which is the maximum daily amount
- child takes more than 5 doses in 24 hours
- taken with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product
Allergy alert:Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
- hives
- facial swelling
- asthma (wheezing)
- shock
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning:If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- you get nervousness, dizzyness or sleeplessness
- pain, nasal congestion, or cough gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
- cough comes back or occurs with fever, rash or headache that lasts.
These could be signs of a serious condition.
Section 50567-7 (50567-7)
When using this product
- do not exceed recommended dosage
- marked drowsiness may occur
- excitability may occur, especially in children
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if the user is
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers
Section 50569-3 (50569-3)
Ask a doctor or pharmacist before use if the user has
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- glaucoma
- a breathing problem such as chronic bronchitis
- cough that occurs with too much phlegm (mucus)
- chronic cough that lasts such as occurs with smoking, asthma, or emphysema
- trouble urinating due to enlargement of the prostate gland
Section 50570-1 (50570-1)
Do not use
- if you are allergic to acetaminophen or any inactive ingredient in this product
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Section 51727-6 (51727-6)
Inactive ingredients citric acid, disodium EDTA, flavor, glycerin, methylparaben, polyethylene glycol, propylene glycol, propylparaben, purified water and sucralose
Section 51945-4 (51945-4)
MAXIMUM STRENGTH
Giltuss® MULTI-SYMPTOM
COLD & FLU
ANALGESIC, ANTITUSSIVE, ANTIHISTAMINE & NASAL DECONGESTANT
ALCOHOL FREE. SODIUM FREE. SUGAR FREE. DYE FREE
RELIEVES
- FEVER, HEADACHE AND BODY ACHES
- COUGH AND COLD
- SNEEZING
- RUNNY NOSE
- ITCHY AND WATERY EYES
- STUFFY NOSE
- SINUS PRESSURE
STRAWBERRY FLAVOR
4FL OZ (118 mL)
Section 53413-1 (53413-1)
Questions or comments?
Call 1-787-848-9114,Monday-Friday 9AM - 5PM EST. Call your doctor for medical advice in the event of side effects.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purposes
Pain reliever/fever reducer
Antihistamine
Cough Suppressant
Nasal decongestant
Section 55106-9 (55106-9)
Active ingredients (in each 10 mL)
Acetaminophen 650 mg
Chlorpheniramine maleate 4 mg
Dextromethorphan hydrobromide 13 mg
Phenylephrine hydrochloride 10 mg
Section 60561-8 (60561-8)
Other information
- store at room temperature 15° - 30°C (59° - 86°F)
- close cap tightly
- SAVE CARTON FOR COMPLETE DRUG FACTS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:02.048142 · Updated: 2026-03-14T23:12:59.392851