84a633dc-bd52-0571-e053-2a91aa0a6df8
84a633dc-bd52-0571-e053-2a91aa0a6df8
34390-5
HUMAN OTC DRUG LABEL
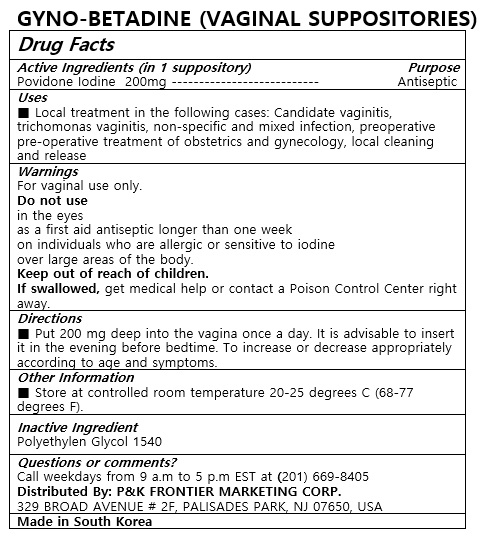
Drug Facts
Composition & Product
Identifiers & Packaging
Description
POVIDONE-IODINE
Medication Information
Warnings and Precautions
Do not use
in the eyes
as a first aid antiseptic longer than one week
on individuals who are allergic or sensitive to iodine
over large areas of the body
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Active Ingredient
POVIDONE-IODINE
Indications and Usage
Put 200 mg deep into the vagina once a day. It is advisable to insert it in the evening before bedtime. To increase or decrease appropriately according to age and symptoms.
Dosage and Administration
For vaginal use only
Description
POVIDONE-IODINE
Section 50565-1
Keep out of reach of children
Section 51727-6
POLYETHYLENE GLYCOL 1540
Section 51945-4
Section 55105-1
Local treatment in the following cases: Candidate vaginitis, trichomonas vaginitis, non-specific and mixed infection, preoperative pre-operative treatment of obstetrics and gynecology, local cleaning and release
Structured Label Content
Warnings and Precautions (34071-1)
Do not use
in the eyes
as a first aid antiseptic longer than one week
on individuals who are allergic or sensitive to iodine
over large areas of the body
Keep out of reach of children.
If swallowed, get medical help or contact a Poison Control Center right away.
Active Ingredient (55106-9)
POVIDONE-IODINE
Indications and Usage (34067-9)
Put 200 mg deep into the vagina once a day. It is advisable to insert it in the evening before bedtime. To increase or decrease appropriately according to age and symptoms.
Dosage and Administration (34068-7)
For vaginal use only
Section 50565-1 (50565-1)
Keep out of reach of children
Section 51727-6 (51727-6)
POLYETHYLENE GLYCOL 1540
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Local treatment in the following cases: Candidate vaginitis, trichomonas vaginitis, non-specific and mixed infection, preoperative pre-operative treatment of obstetrics and gynecology, local cleaning and release
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:47.034730 · Updated: 2026-03-14T22:55:27.684543