These Highlights Do Not Include All The Information Needed To Use Rivastigmine Tartrate Capsules Usp Safely And Effectively. See Full Prescribing Information For Rivastigmine Tartrate Capsules Usp.
83bd2ff6-1d8b-4cac-a37b-fd01dc3f34a1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Rivastigmine tartrate is an acetylcholinesterase inhibitor indicated for treatment of: Mild to moderate dementia of the Alzheimer's type ( 1.1 ) Mild to moderate dementia associated with Parkinson's disease ( 1.2 )
Indications and Usage
Rivastigmine tartrate is an acetylcholinesterase inhibitor indicated for treatment of: Mild to moderate dementia of the Alzheimer's type ( 1.1 ) Mild to moderate dementia associated with Parkinson's disease ( 1.2 )
Dosage and Administration
Alzheimer's Disease: Initiate treatment with 1.5 mg twice a day After a minimum of 2 weeks, if tolerated, increase dose to 3 mg twice a day and further to 4.5 mg twice a day and 6 mg twice a day if tolerated with a minimum of 2 weeks at each dose ( 2.1 ) Parkinson's Disease Dementia: Initiate treatment with 1.5 mg twice a day After a minimum of 4 weeks, if tolerated, increase dose to 3 mg twice a day and further to 4.5 mg twice a day and 6 mg twice a day if tolerated with a minimum of 4 weeks at each dose ( 2.2 ) Rivastigmine tartrate capsules should be taken with meals in divided doses in the morning and evening. ( 2.1 , 2.2 ) Rivastigmine tartrate oral solution and rivastigmine tartrate capsules may be interchanged at equal doses. ( 2.5 )
Warnings and Precautions
Gastrointestinal adverse reactions may include significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss, and may necessitate treatment interruption. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes ( 5.1 ) Discontinue rivastigmine in case of disseminated allergic dermatitis, which may occur after oral or transdermal administration. ( 4 , 5.2 ) In patients with suspected allergic contact dermatitis after transdermal rivastigmine use, switch to oral rivastigmine only after negative allergy testing
Contraindications
Rivastigmine tartrate capsules are contraindicated in patients with: known hypersensitivity to rivastigmine, other carbamate derivatives or other components of the formulation [see Description (11) ] . a previous history of application site reaction with rivastigmine transdermal patch suggestive of allergic contact dermatitis, in the absence of negative allergy testing [see Warnings and Precautions (5.2) ] . Isolated cases of generalized skin reactions have been described in postmarketing experience [see Adverse Reactions (6.2) ] .
Adverse Reactions
Rivastigmine tartrate can cause gastrointestinal adverse reactions, including significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose-related [see Adverse Reactions (6.1) ]. For this reason, patients should always be started at a dose of 1.5 mg twice a day and titrated to their maintenance dose. If treatment is interrupted for longer than 3 days, treatment should be reinitiated with the lowest daily dose [ see Dosage and Administration (2.1) ] to reduce the possibility of severe vomiting and its potentially serious sequelae (e.g., there has been one postmarketing report of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment with a 4.5 mg dose after 8 weeks of treatment interruption). Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than 3 days because of intolerance, the next dose should not be administered without contacting the physician regarding proper retitration.
Drug Interactions
Concomitant use with metoclopramide, beta-blockers, or cholinomimetic and anticholinergic drugs is not recommended ( 7.1 , 7.2 , 7.3 )
Medication Information
Warnings and Precautions
Gastrointestinal adverse reactions may include significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss, and may necessitate treatment interruption. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes ( 5.1 ) Discontinue rivastigmine in case of disseminated allergic dermatitis, which may occur after oral or transdermal administration. ( 4 , 5.2 ) In patients with suspected allergic contact dermatitis after transdermal rivastigmine use, switch to oral rivastigmine only after negative allergy testing
Indications and Usage
Rivastigmine tartrate is an acetylcholinesterase inhibitor indicated for treatment of: Mild to moderate dementia of the Alzheimer's type ( 1.1 ) Mild to moderate dementia associated with Parkinson's disease ( 1.2 )
Dosage and Administration
Alzheimer's Disease: Initiate treatment with 1.5 mg twice a day After a minimum of 2 weeks, if tolerated, increase dose to 3 mg twice a day and further to 4.5 mg twice a day and 6 mg twice a day if tolerated with a minimum of 2 weeks at each dose ( 2.1 ) Parkinson's Disease Dementia: Initiate treatment with 1.5 mg twice a day After a minimum of 4 weeks, if tolerated, increase dose to 3 mg twice a day and further to 4.5 mg twice a day and 6 mg twice a day if tolerated with a minimum of 4 weeks at each dose ( 2.2 ) Rivastigmine tartrate capsules should be taken with meals in divided doses in the morning and evening. ( 2.1 , 2.2 ) Rivastigmine tartrate oral solution and rivastigmine tartrate capsules may be interchanged at equal doses. ( 2.5 )
Contraindications
Rivastigmine tartrate capsules are contraindicated in patients with: known hypersensitivity to rivastigmine, other carbamate derivatives or other components of the formulation [see Description (11) ] . a previous history of application site reaction with rivastigmine transdermal patch suggestive of allergic contact dermatitis, in the absence of negative allergy testing [see Warnings and Precautions (5.2) ] . Isolated cases of generalized skin reactions have been described in postmarketing experience [see Adverse Reactions (6.2) ] .
Adverse Reactions
Rivastigmine tartrate can cause gastrointestinal adverse reactions, including significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose-related [see Adverse Reactions (6.1) ]. For this reason, patients should always be started at a dose of 1.5 mg twice a day and titrated to their maintenance dose. If treatment is interrupted for longer than 3 days, treatment should be reinitiated with the lowest daily dose [ see Dosage and Administration (2.1) ] to reduce the possibility of severe vomiting and its potentially serious sequelae (e.g., there has been one postmarketing report of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment with a 4.5 mg dose after 8 weeks of treatment interruption). Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than 3 days because of intolerance, the next dose should not be administered without contacting the physician regarding proper retitration.
Drug Interactions
Concomitant use with metoclopramide, beta-blockers, or cholinomimetic and anticholinergic drugs is not recommended ( 7.1 , 7.2 , 7.3 )
Description
Rivastigmine tartrate is an acetylcholinesterase inhibitor indicated for treatment of: Mild to moderate dementia of the Alzheimer's type ( 1.1 ) Mild to moderate dementia associated with Parkinson's disease ( 1.2 )
Section 42229-5
Initial Dose
Initiate treatment with the 1.5 mg twice a day with rivastigmine tartrate capsules.
Section 44425-7
Store at 25°C (77°F); excursions permitted to 15°C-30°C (59°F-86°F) [see USP Controlled RoomTemperature]. Store in a tight container.
10 Overdosage
Because strategies for the management of overdose are continually evolving, it is advisable to contact a Poison Control Center to determine the latest recommendations for the management of an overdose of any drug.
As rivastigmine has a short plasma half-life of about 1 hour and a moderate duration of acetylcholinesterase inhibition of 8 to 10 hours, it is recommended that in cases of asymptomatic overdoses, no further dose of rivastigmine tartrate should be administered for the next 24 hours.
As in any case of overdose, general supportive measures should be utilized.
Overdosage with cholinesterase inhibitors can result in cholinergic crisis characterized by severe nausea, vomiting, salivation, sweating, bradycardia, hypotension, respiratory depression, collapse and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Atypical responses in blood pressure and heart rate have been reported with other drugs that increase cholinergic activity when coadministered with quaternary anticholinergics such as glycopyrrolate. Additional symptoms associated with rivastigmine overdose are diarrhea, abdominal pain, dizziness, tremor, headache, somnolence, confusional state, hyperhidrosis, hypertension, hallucinations and malaise. Due to the short half-life of rivastigmine, dialysis (hemodialysis, peritoneal dialysis, or hemofiltration) would not be clinically indicated in the event of an overdose.
In overdoses accompanied by severe nausea and vomiting, the use of antiemetics should be considered. A fatal outcome has been rarely reported with rivastigmine.
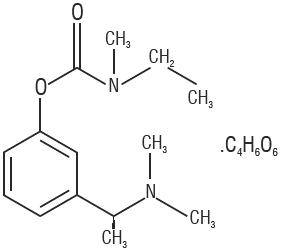
11 Description
Rivastigmine tartrate is a reversible cholinesterase inhibitor and is known chemically as (S)-N-Ethyl-N-methyl-3-[1-(dimethylamino)ethyl]-phenyl carbamate hydrogen-(2R,3R)-tartrate. Rivastigmine tartrate is commonly referred to in the pharmacological literature as SDZ ENA 713 or ENA 713. It has an empirical formula of C14H22N2O2 ∙ C4H6O6 (hydrogen tartrate salt-hta salt) and a molecular weight of 400.43 (hta salt). Rivastigmine tartrate is a white to off-white, fine crystalline powder that is very soluble in water, soluble in ethanol and acetonitrile, slightly soluble in n-octanol and very slightly soluble in ethyl acetate.
The distribution coefficient at 37°C in n-octanol/phosphate buffer solution pH 7 is 3.0.
Rivastigmine tartrate capsules, USP contain rivastigmine tartrate, equivalent to 1.5 mg, 3 mg, 4.5 mg, and 6 mg of rivastigmine base for oral administration. Inactive ingredients are hydroxypropyl methylcellulose, magnesium stearate, microcrystalline cellulose, and colloidal silicon dioxide. Each hard-gelatin capsule contains gelatin, titanium dioxide and red and/or yellow iron oxides.
7.3 Beta Blockers
Additive bradycardic effects resulting in syncope may occur when rivastigmine tartrate is used concomitantly with beta-blockers, especially cardioselective beta-blockers (including atenolol). Concomitant use of rivastigmine tartrate with beta-blockers is not recommended.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. The use of rivastigmine tartrate in pediatric patients (below 18 years of age) is not recommended.
8.5 Geriatric Use
Of the total number of patients in clinical studies of rivastigmine tartrate, 86 % were 65 years and older while 46 % were 75 years and older. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
7.1 Metoclopramide
Due to the risk of additive extrapyramidal adverse reactions, the concomitant use of metoclopramide and rivastigmine tartrate is not recommended.
4 Contraindications
Rivastigmine tartrate capsules are contraindicated in patients with:
- known hypersensitivity to rivastigmine, other carbamate derivatives or other components of the formulation [see Description (11)].
- a previous history of application site reaction with rivastigmine transdermal patch suggestive of allergic contact dermatitis, in the absence of negative allergy testing [see Warnings and Precautions (5.2)].
Isolated cases of generalized skin reactions have been described in postmarketing experience [see Adverse Reactions (6.2)].
6 Adverse Reactions
The following adverse reactions are described below and elsewhere in the labeling:
- Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.1)].
- Allergic Dermatitis [see Warnings and Precautions (5.2)].
- Other Adverse Reactions from Increased Cholinergic Activity [see Warnings and Precautions (5.3)].
7 Drug Interactions
8.3 Nursing Mothers
Rivastigmine and its metabolites are excreted in rat milk following oral administration of rivastigmine; levels of rivastigmine plus metabolites in rat milk are approximately 2 times that in maternal plasma. It is not known whether rivastigmine is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from rivastigmine tartrate, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.6 Renal Impairment
Patients with moderate to severe renal impairment may be able to only tolerate lower doses [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
After a 6 mg dose of rivastigmine, anticholinesterase activity is present in cerebrospinal fluid (CSF) for about 10 hours, with a maximum inhibition of about 60% 5 hours after dosing.
In vitro and in vivo studies demonstrate that the inhibition of cholinesterase by rivastigmine is not affected by the concomitant administration of memantine, an N-methyl-D-aspartate receptor antagonist.
12.3 Pharmacokinetics
Rivastigmine shows linear pharmacokinetics up to 3 mg twice a day but is nonlinear at higher doses. Doubling the dose from 3 mg to 6 mg twice a day results in a 3-fold increase in AUC. The elimination half-life is about 1.5 hours, with most elimination as metabolites via the urine.
8.7 Hepatic Impairment
Patients with mild or moderate hepatic impairment may be able to only tolerate lower doses [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)]. No data are available on the use of rivastigmine tartrate in patients with severe hepatic impairment.
1 Indications and Usage
1.1 Alzheimer's Disease
Rivastigmine tartrate capsules, USP are indicated for the treatment of mild to moderate dementia of the Alzheimer's type.
5.2 Allergic Dermatitis
There have been isolated postmarketing reports of patients experiencing disseminated allergic dermatitis when administered rivastigmine irrespective of the route of administration (oral or transdermal). Treatment should be discontinued if disseminated allergic dermatitis occurs [see Contraindications (4)]. Patients and caregivers should be instructed accordingly [see Patient Counseling Information (17)].
In patients who develop application site reactions suggestive of allergic contact dermatitis to rivastigmine tartrate patch and who still require rivastigmine, treatment should be switched to oral rivastigmine only after negative allergy testing and under close medical supervision. It is possible that some patients sensitized to rivastigmine by exposure to rivastigmine patch may not be able to take rivastigmine in any form.
12.1 Mechanism of Action
Although the precise mechanism of action of rivastigmine is unknown, it is thought to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by cholinesterase. Therefore, the effect of rivastigmine may lessen as the disease process advances and fewer cholinergic neurons remain functionally intact. There is no evidence that rivastigmine alters the course of the underlying dementing process.
5 Warnings and Precautions
- Gastrointestinal adverse reactions may include significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss, and may necessitate treatment interruption. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes (5.1)
- Discontinue rivastigmine in case of disseminated allergic dermatitis, which may occur after oral or transdermal administration. (4, 5.2) In patients with suspected allergic contact dermatitis after transdermal rivastigmine use, switch to oral rivastigmine only after negative allergy testing
2 Dosage and Administration
Alzheimer's Disease:
- Initiate treatment with 1.5 mg twice a day
- After a minimum of 2 weeks, if tolerated, increase dose to 3 mg twice a day and further to 4.5 mg twice a day and 6 mg twice a day if tolerated with a minimum of 2 weeks at each dose (2.1)
Parkinson's Disease Dementia:
- Initiate treatment with 1.5 mg twice a day
- After a minimum of 4 weeks, if tolerated, increase dose to 3 mg twice a day and further to 4.5 mg twice a day and 6 mg twice a day if tolerated with a minimum of 4 weeks at each dose (2.2)
Rivastigmine tartrate capsules should be taken with meals in divided doses in the morning and evening. (2.1, 2.2) Rivastigmine tartrate oral solution and rivastigmine tartrate capsules may be interchanged at equal doses. (2.5)
8.8 Low Or High Body Weight
Because rivastigmine blood levels vary with weight, careful titration and monitoring should be performed in patients with low or high body weights [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths
- Capsules: 1.5 mg, 3 mg, 4.5 mg, or 6 mg (3.1)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of rivastigmine tartrate capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: Tachycardia
Hepatobiliary Disorders: Abnormal liver function tests, hepatitis.
Nervous System Disorders: seizure.
Psychiatric Disorders: Aggression, nightmares.
Skin and Subcutaneous Tissue Disorders: Allergic dermatitis, application site hypersensitivity (patch), blister, disseminated allergic dermatitis, Stevens-Johnson syndrome, urticaria.
2.3 Interruption of Treatment
If adverse effects (e.g., nausea, vomiting, abdominal pain, loss of appetite) cause intolerance during treatment, the patient should be instructed to discontinue treatment for several doses and then restart at the same or next lower dose level.
If dosing is interrupted for 3 days or fewer, restart treatment with the same or lower dose of rivastigmine tartrate capsules. If dosing is interrupted for more than 3 days, treatment should be restarted with 1.5 mg twice a day and titrated as described above [see Warnings and Precautions (5.1) ].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Rivastigmine tartrate capsules have been administered to over 5,297 individuals during clinical trials worldwide. Of these, 4,326 patients have been treated for at least 3 months, 3,407 patients have been treated for at least 6 months, 2,150 patients have been treated for 1 year, 1,250 patients have been treated for 2 years, and 168 patients have been treated for over 3 years. With regard to exposure to the highest dose, 2,809 patients were exposed to doses of 10 mg to 12 mg, 2,615 patients treated for 3 months, 2,328 patients treated for 6 months, 1,378 patients treated for 1 year, 917 patients treated for 2 years, and 129 patients treated for over 3 years.
1.2 Parkinson's Disease Dementia
Rivastigmine tartrate capsules, USP are indicated for the treatment of mild to moderate dementia associated with Parkinson's disease.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Instructions for Use).
2.1 Dosing in Alzheimer's Disease
Rivastigmine tartrate capsules should be taken with meals in divided doses in the morning and evening.
The recommended dosage of rivastigmine tartrate capsules in Alzheimer's disease is 6 mg to 12 mg per day, administered twice a day (daily doses of 3 mg to 6 mg twice a day). There is evidence from the clinical trials that doses at the higher end of this range may be more beneficial.
3.1 Rivastigmine Tartrate Capsules
Capsules, containing rivastigmine tartrate equivalent to 1.5 mg, 3 mg, 4.5 mg, or 6 mg of rivastigmine base, are available as follows:
-
1.5 mg capsule – white to off white powder filled in size "2" hard gelatin capsules with yellow opaque color cap and yellow opaque color body imprinted "C 91".
-
3 mg capsule –white to off white powder filled in size "2" hard gelatin capsules with orange opaque color cap and orange opaque color body imprinted "C 92".
-
4.5 mg capsule – white to off white powder filled in size "2" hard gelatin capsules with red opaque color cap and red opaque color body imprinted "C 93".
-
6 mg capsule –white to off white powder filled in size "2" hard gelatin capsules with red opaque color cap and orange opaque color body imprinted "C 94".
5.1 Gastrointestinal Adverse Reactions
Rivastigmine tartrate can cause gastrointestinal adverse reactions, including significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose-related [see Adverse Reactions (6.1)]. For this reason, patients should always be started at a dose of 1.5 mg twice a day and titrated to their maintenance dose.
If treatment is interrupted for longer than 3 days, treatment should be reinitiated with the lowest daily dose [see Dosage and Administration (2.1) ] to reduce the possibility of severe vomiting and its potentially serious sequelae (e.g., there has been one postmarketing report of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment with a 4.5 mg dose after 8 weeks of treatment interruption).
Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than 3 days because of intolerance, the next dose should not be administered without contacting the physician regarding proper retitration.
2.5 Important Administration Instructions
Rivastigmine tartrate oral solution and rivastigmine tartrate capsules may be interchanged at equal doses.
2.2 Dosing in Parkinson's Disease Dementia
Rivastigmine tartrate capsules should be taken with meals in divided doses in the morning and evening.
The dosage of rivastigmine tartrate capsules shown to be effective in the single controlled clinical trial conducted in dementia associated with Parkinson's disease is 3 mg to 12 mg per day, administered twice a day (daily doses of 1.5 mg to 6 mg twice a day).
7.2 Cholinomimetic and Anticholinergic Drugs
Rivastigmine tartrate may increase the cholinergic effects of other cholinomimetic medications and may also interfere with the activity of anticholinergic medications (e.g., oxybutynin, tolterodine). Concomitant use of rivastigmine tartrate with medications having these pharmacologic effects is not recommended unless deemed clinically necessary [see Warnings and Precautions (5.3)].
5.4 Impairment in Driving Or Use of Machinery
Dementia may cause gradual impairment of driving performance or compromise the ability to use machinery. The administration of rivastigmine may also result in adverse reactions that are detrimental to these functions. During treatment with the rivastigmine tartrate capsules, routinely evaluate the patient's ability to continue driving or operating machinery.
Principal Display Panel 3 Mg Capsule Bottle Label
Rx only
NDC 75834-134-60
Rivastigmine Tartrate
Capsules USP
3 mg*
60 CAPSULES
NIVAGEN
PHARMACEUTICALS
Principal Display Panel 6 Mg Capsule Bottle Label
Rx only
NDC 75834-136-60
Rivastigmine Tartrate
Capsules USP
6 mg*
60 CAPSULES
NIVAGEN
PHARMACEUTICALS
Principal Display Panel 1.5 Mg Capsule Bottle Label
Rx only
NDC 75834-133-60
Rivastigmine Tartrate
Capsules USP
1.5 mg*
60 CAPSULES
NIVAGEN
PHARMACEUTICALS
Principal Display Panel 4.5 Mg Capsule Bottle Label
Rx only
NDC 75834-135-60
Rivastigmine Tartrate
Capsules USP
4.5 mg*
60 CAPSULES
NIVAGEN
PHARMACEUTICALS
Structured Label Content
Section 42229-5 (42229-5)
Initial Dose
Initiate treatment with the 1.5 mg twice a day with rivastigmine tartrate capsules.
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted to 15°C-30°C (59°F-86°F) [see USP Controlled RoomTemperature]. Store in a tight container.
10 Overdosage (10 OVERDOSAGE)
Because strategies for the management of overdose are continually evolving, it is advisable to contact a Poison Control Center to determine the latest recommendations for the management of an overdose of any drug.
As rivastigmine has a short plasma half-life of about 1 hour and a moderate duration of acetylcholinesterase inhibition of 8 to 10 hours, it is recommended that in cases of asymptomatic overdoses, no further dose of rivastigmine tartrate should be administered for the next 24 hours.
As in any case of overdose, general supportive measures should be utilized.
Overdosage with cholinesterase inhibitors can result in cholinergic crisis characterized by severe nausea, vomiting, salivation, sweating, bradycardia, hypotension, respiratory depression, collapse and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Atypical responses in blood pressure and heart rate have been reported with other drugs that increase cholinergic activity when coadministered with quaternary anticholinergics such as glycopyrrolate. Additional symptoms associated with rivastigmine overdose are diarrhea, abdominal pain, dizziness, tremor, headache, somnolence, confusional state, hyperhidrosis, hypertension, hallucinations and malaise. Due to the short half-life of rivastigmine, dialysis (hemodialysis, peritoneal dialysis, or hemofiltration) would not be clinically indicated in the event of an overdose.
In overdoses accompanied by severe nausea and vomiting, the use of antiemetics should be considered. A fatal outcome has been rarely reported with rivastigmine.
11 Description (11 DESCRIPTION)
Rivastigmine tartrate is a reversible cholinesterase inhibitor and is known chemically as (S)-N-Ethyl-N-methyl-3-[1-(dimethylamino)ethyl]-phenyl carbamate hydrogen-(2R,3R)-tartrate. Rivastigmine tartrate is commonly referred to in the pharmacological literature as SDZ ENA 713 or ENA 713. It has an empirical formula of C14H22N2O2 ∙ C4H6O6 (hydrogen tartrate salt-hta salt) and a molecular weight of 400.43 (hta salt). Rivastigmine tartrate is a white to off-white, fine crystalline powder that is very soluble in water, soluble in ethanol and acetonitrile, slightly soluble in n-octanol and very slightly soluble in ethyl acetate.
The distribution coefficient at 37°C in n-octanol/phosphate buffer solution pH 7 is 3.0.
Rivastigmine tartrate capsules, USP contain rivastigmine tartrate, equivalent to 1.5 mg, 3 mg, 4.5 mg, and 6 mg of rivastigmine base for oral administration. Inactive ingredients are hydroxypropyl methylcellulose, magnesium stearate, microcrystalline cellulose, and colloidal silicon dioxide. Each hard-gelatin capsule contains gelatin, titanium dioxide and red and/or yellow iron oxides.
7.3 Beta Blockers (7.3 Beta-blockers)
Additive bradycardic effects resulting in syncope may occur when rivastigmine tartrate is used concomitantly with beta-blockers, especially cardioselective beta-blockers (including atenolol). Concomitant use of rivastigmine tartrate with beta-blockers is not recommended.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established. The use of rivastigmine tartrate in pediatric patients (below 18 years of age) is not recommended.
8.5 Geriatric Use
Of the total number of patients in clinical studies of rivastigmine tartrate, 86 % were 65 years and older while 46 % were 75 years and older. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
7.1 Metoclopramide
Due to the risk of additive extrapyramidal adverse reactions, the concomitant use of metoclopramide and rivastigmine tartrate is not recommended.
4 Contraindications (4 CONTRAINDICATIONS)
Rivastigmine tartrate capsules are contraindicated in patients with:
- known hypersensitivity to rivastigmine, other carbamate derivatives or other components of the formulation [see Description (11)].
- a previous history of application site reaction with rivastigmine transdermal patch suggestive of allergic contact dermatitis, in the absence of negative allergy testing [see Warnings and Precautions (5.2)].
Isolated cases of generalized skin reactions have been described in postmarketing experience [see Adverse Reactions (6.2)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described below and elsewhere in the labeling:
- Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.1)].
- Allergic Dermatitis [see Warnings and Precautions (5.2)].
- Other Adverse Reactions from Increased Cholinergic Activity [see Warnings and Precautions (5.3)].
7 Drug Interactions (7 DRUG INTERACTIONS)
8.3 Nursing Mothers
Rivastigmine and its metabolites are excreted in rat milk following oral administration of rivastigmine; levels of rivastigmine plus metabolites in rat milk are approximately 2 times that in maternal plasma. It is not known whether rivastigmine is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from rivastigmine tartrate, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.6 Renal Impairment
Patients with moderate to severe renal impairment may be able to only tolerate lower doses [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
After a 6 mg dose of rivastigmine, anticholinesterase activity is present in cerebrospinal fluid (CSF) for about 10 hours, with a maximum inhibition of about 60% 5 hours after dosing.
In vitro and in vivo studies demonstrate that the inhibition of cholinesterase by rivastigmine is not affected by the concomitant administration of memantine, an N-methyl-D-aspartate receptor antagonist.
12.3 Pharmacokinetics
Rivastigmine shows linear pharmacokinetics up to 3 mg twice a day but is nonlinear at higher doses. Doubling the dose from 3 mg to 6 mg twice a day results in a 3-fold increase in AUC. The elimination half-life is about 1.5 hours, with most elimination as metabolites via the urine.
8.7 Hepatic Impairment
Patients with mild or moderate hepatic impairment may be able to only tolerate lower doses [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)]. No data are available on the use of rivastigmine tartrate in patients with severe hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
1.1 Alzheimer's Disease
Rivastigmine tartrate capsules, USP are indicated for the treatment of mild to moderate dementia of the Alzheimer's type.
5.2 Allergic Dermatitis
There have been isolated postmarketing reports of patients experiencing disseminated allergic dermatitis when administered rivastigmine irrespective of the route of administration (oral or transdermal). Treatment should be discontinued if disseminated allergic dermatitis occurs [see Contraindications (4)]. Patients and caregivers should be instructed accordingly [see Patient Counseling Information (17)].
In patients who develop application site reactions suggestive of allergic contact dermatitis to rivastigmine tartrate patch and who still require rivastigmine, treatment should be switched to oral rivastigmine only after negative allergy testing and under close medical supervision. It is possible that some patients sensitized to rivastigmine by exposure to rivastigmine patch may not be able to take rivastigmine in any form.
12.1 Mechanism of Action
Although the precise mechanism of action of rivastigmine is unknown, it is thought to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by cholinesterase. Therefore, the effect of rivastigmine may lessen as the disease process advances and fewer cholinergic neurons remain functionally intact. There is no evidence that rivastigmine alters the course of the underlying dementing process.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Gastrointestinal adverse reactions may include significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss, and may necessitate treatment interruption. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes (5.1)
- Discontinue rivastigmine in case of disseminated allergic dermatitis, which may occur after oral or transdermal administration. (4, 5.2) In patients with suspected allergic contact dermatitis after transdermal rivastigmine use, switch to oral rivastigmine only after negative allergy testing
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Alzheimer's Disease:
- Initiate treatment with 1.5 mg twice a day
- After a minimum of 2 weeks, if tolerated, increase dose to 3 mg twice a day and further to 4.5 mg twice a day and 6 mg twice a day if tolerated with a minimum of 2 weeks at each dose (2.1)
Parkinson's Disease Dementia:
- Initiate treatment with 1.5 mg twice a day
- After a minimum of 4 weeks, if tolerated, increase dose to 3 mg twice a day and further to 4.5 mg twice a day and 6 mg twice a day if tolerated with a minimum of 4 weeks at each dose (2.2)
Rivastigmine tartrate capsules should be taken with meals in divided doses in the morning and evening. (2.1, 2.2) Rivastigmine tartrate oral solution and rivastigmine tartrate capsules may be interchanged at equal doses. (2.5)
8.8 Low Or High Body Weight (8.8 Low or High Body Weight)
Because rivastigmine blood levels vary with weight, careful titration and monitoring should be performed in patients with low or high body weights [see Dosage and Administration (2.3) and Clinical Pharmacology (12.3)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Capsules: 1.5 mg, 3 mg, 4.5 mg, or 6 mg (3.1)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of rivastigmine tartrate capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiac Disorders: Tachycardia
Hepatobiliary Disorders: Abnormal liver function tests, hepatitis.
Nervous System Disorders: seizure.
Psychiatric Disorders: Aggression, nightmares.
Skin and Subcutaneous Tissue Disorders: Allergic dermatitis, application site hypersensitivity (patch), blister, disseminated allergic dermatitis, Stevens-Johnson syndrome, urticaria.
2.3 Interruption of Treatment
If adverse effects (e.g., nausea, vomiting, abdominal pain, loss of appetite) cause intolerance during treatment, the patient should be instructed to discontinue treatment for several doses and then restart at the same or next lower dose level.
If dosing is interrupted for 3 days or fewer, restart treatment with the same or lower dose of rivastigmine tartrate capsules. If dosing is interrupted for more than 3 days, treatment should be restarted with 1.5 mg twice a day and titrated as described above [see Warnings and Precautions (5.1) ].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Rivastigmine tartrate capsules have been administered to over 5,297 individuals during clinical trials worldwide. Of these, 4,326 patients have been treated for at least 3 months, 3,407 patients have been treated for at least 6 months, 2,150 patients have been treated for 1 year, 1,250 patients have been treated for 2 years, and 168 patients have been treated for over 3 years. With regard to exposure to the highest dose, 2,809 patients were exposed to doses of 10 mg to 12 mg, 2,615 patients treated for 3 months, 2,328 patients treated for 6 months, 1,378 patients treated for 1 year, 917 patients treated for 2 years, and 129 patients treated for over 3 years.
1.2 Parkinson's Disease Dementia
Rivastigmine tartrate capsules, USP are indicated for the treatment of mild to moderate dementia associated with Parkinson's disease.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Instructions for Use).
2.1 Dosing in Alzheimer's Disease
Rivastigmine tartrate capsules should be taken with meals in divided doses in the morning and evening.
The recommended dosage of rivastigmine tartrate capsules in Alzheimer's disease is 6 mg to 12 mg per day, administered twice a day (daily doses of 3 mg to 6 mg twice a day). There is evidence from the clinical trials that doses at the higher end of this range may be more beneficial.
3.1 Rivastigmine Tartrate Capsules (3.1 Rivastigmine tartrate capsules)
Capsules, containing rivastigmine tartrate equivalent to 1.5 mg, 3 mg, 4.5 mg, or 6 mg of rivastigmine base, are available as follows:
-
1.5 mg capsule – white to off white powder filled in size "2" hard gelatin capsules with yellow opaque color cap and yellow opaque color body imprinted "C 91".
-
3 mg capsule –white to off white powder filled in size "2" hard gelatin capsules with orange opaque color cap and orange opaque color body imprinted "C 92".
-
4.5 mg capsule – white to off white powder filled in size "2" hard gelatin capsules with red opaque color cap and red opaque color body imprinted "C 93".
-
6 mg capsule –white to off white powder filled in size "2" hard gelatin capsules with red opaque color cap and orange opaque color body imprinted "C 94".
5.1 Gastrointestinal Adverse Reactions
Rivastigmine tartrate can cause gastrointestinal adverse reactions, including significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose-related [see Adverse Reactions (6.1)]. For this reason, patients should always be started at a dose of 1.5 mg twice a day and titrated to their maintenance dose.
If treatment is interrupted for longer than 3 days, treatment should be reinitiated with the lowest daily dose [see Dosage and Administration (2.1) ] to reduce the possibility of severe vomiting and its potentially serious sequelae (e.g., there has been one postmarketing report of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment with a 4.5 mg dose after 8 weeks of treatment interruption).
Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than 3 days because of intolerance, the next dose should not be administered without contacting the physician regarding proper retitration.
2.5 Important Administration Instructions
Rivastigmine tartrate oral solution and rivastigmine tartrate capsules may be interchanged at equal doses.
2.2 Dosing in Parkinson's Disease Dementia
Rivastigmine tartrate capsules should be taken with meals in divided doses in the morning and evening.
The dosage of rivastigmine tartrate capsules shown to be effective in the single controlled clinical trial conducted in dementia associated with Parkinson's disease is 3 mg to 12 mg per day, administered twice a day (daily doses of 1.5 mg to 6 mg twice a day).
7.2 Cholinomimetic and Anticholinergic Drugs
Rivastigmine tartrate may increase the cholinergic effects of other cholinomimetic medications and may also interfere with the activity of anticholinergic medications (e.g., oxybutynin, tolterodine). Concomitant use of rivastigmine tartrate with medications having these pharmacologic effects is not recommended unless deemed clinically necessary [see Warnings and Precautions (5.3)].
5.4 Impairment in Driving Or Use of Machinery (5.4 Impairment in Driving or Use of Machinery)
Dementia may cause gradual impairment of driving performance or compromise the ability to use machinery. The administration of rivastigmine may also result in adverse reactions that are detrimental to these functions. During treatment with the rivastigmine tartrate capsules, routinely evaluate the patient's ability to continue driving or operating machinery.
Principal Display Panel 3 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 3 mg Capsule Bottle Label)
Rx only
NDC 75834-134-60
Rivastigmine Tartrate
Capsules USP
3 mg*
60 CAPSULES
NIVAGEN
PHARMACEUTICALS
Principal Display Panel 6 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 6 mg Capsule Bottle Label)
Rx only
NDC 75834-136-60
Rivastigmine Tartrate
Capsules USP
6 mg*
60 CAPSULES
NIVAGEN
PHARMACEUTICALS
Principal Display Panel 1.5 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 1.5 mg Capsule Bottle Label)
Rx only
NDC 75834-133-60
Rivastigmine Tartrate
Capsules USP
1.5 mg*
60 CAPSULES
NIVAGEN
PHARMACEUTICALS
Principal Display Panel 4.5 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 4.5 mg Capsule Bottle Label)
Rx only
NDC 75834-135-60
Rivastigmine Tartrate
Capsules USP
4.5 mg*
60 CAPSULES
NIVAGEN
PHARMACEUTICALS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:36:39.928702 · Updated: 2026-03-14T21:47:16.707891