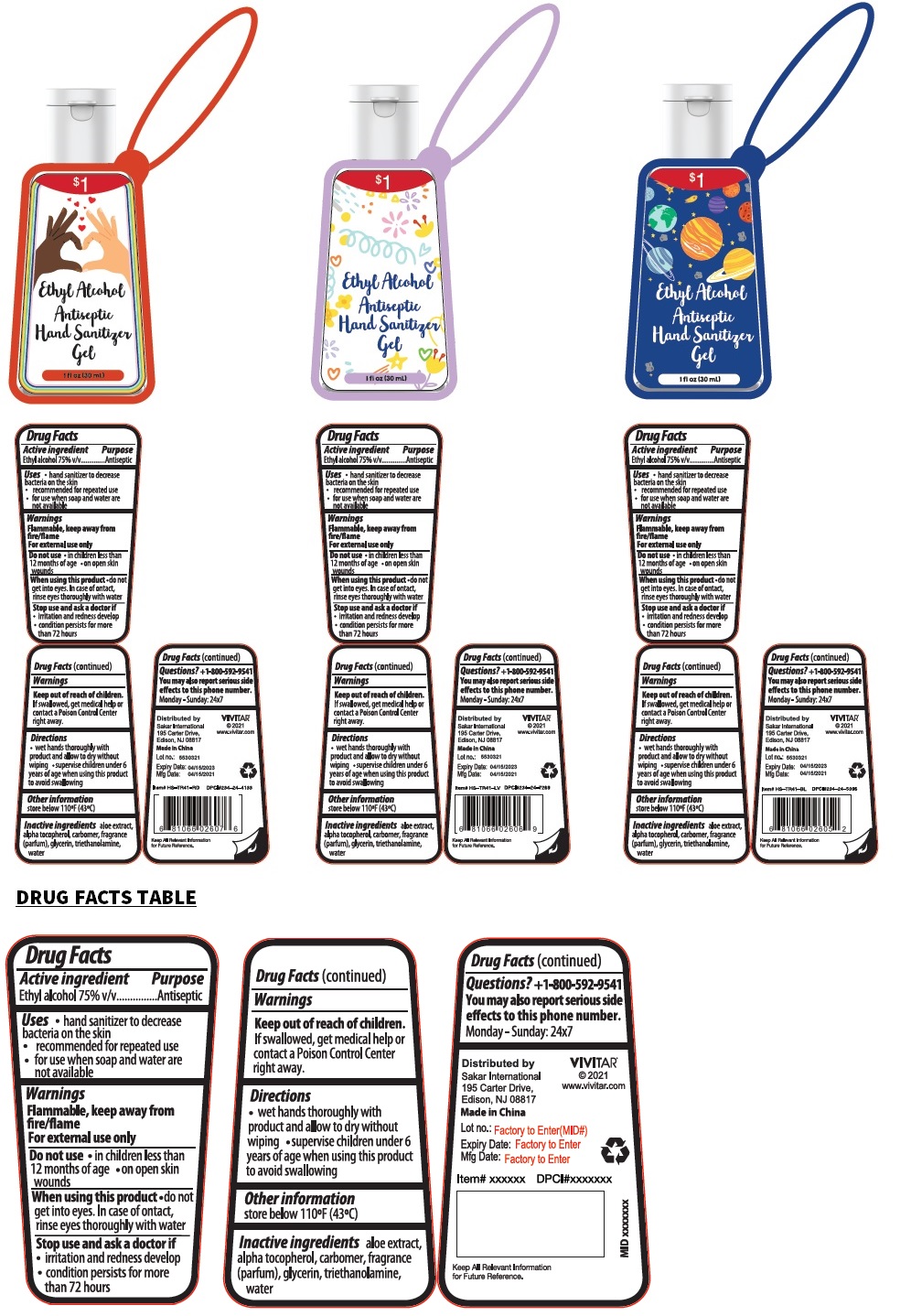
Ethyl Alcohol antiseptic hand Sanitizer gel
82c1d871-61b0-4ab9-b67b-8db506ef7271
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl alcohol 75% v/v
Purpose
Antiseptic
Medication Information
Purpose
Antiseptic
Description
Ethyl alcohol 75% v/v
Uses
• hand sanitizer to decrease bacteria on the skin
• recommended for repeated use
• for use when soap and water are not available
Section 42229-5
Distributed by
Sakar International
195 Carter Drive,
Edison, NJ 08817
www.vivitar.com
Made in China
VIVITAR®
© 2021
Keep All Relevant Information for Future Reference.
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
Flammable, keep away from fire/flame
For external use only
Do not use
• in children less than 12 months of age • on open skin wounds
When using this product • do not get into eyes. In case of contact, rinse eyes thoroughly with water
Stop use and ask a doctor if
• irritation and redness develop
• condition persists for more than 72 hours
Packaging
Directions
• wet hands thoroughly with product and allow to dry without wiping • supervise children under 6 years of age when using this product to avoid swallowing
Drug Facts
Questions?
You may also report serious side effects to this phone number.
We are available 24x7
Active Ingredient
Ethyl alcohol 75% v/v
Other Information
store below 110ºF (43ºC)
Inactive Ingredients
aloe extract, alpha tocopherol, carbomer, fragrance (parfum), glycerin, triethanolamine, water
Structured Label Content
Uses
• hand sanitizer to decrease bacteria on the skin
• recommended for repeated use
• for use when soap and water are not available
Section 42229-5 (42229-5)
Distributed by
Sakar International
195 Carter Drive,
Edison, NJ 08817
www.vivitar.com
Made in China
VIVITAR®
© 2021
Keep All Relevant Information for Future Reference.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Antiseptic
Warnings
Flammable, keep away from fire/flame
For external use only
Do not use
• in children less than 12 months of age • on open skin wounds
When using this product • do not get into eyes. In case of contact, rinse eyes thoroughly with water
Stop use and ask a doctor if
• irritation and redness develop
• condition persists for more than 72 hours
Packaging
Directions
• wet hands thoroughly with product and allow to dry without wiping • supervise children under 6 years of age when using this product to avoid swallowing
Drug Facts
Questions?
You may also report serious side effects to this phone number.
We are available 24x7
Active Ingredient (Active ingredient)
Ethyl alcohol 75% v/v
Other Information (Other information )
store below 110ºF (43ºC)
Inactive Ingredients (Inactive ingredients)
aloe extract, alpha tocopherol, carbomer, fragrance (parfum), glycerin, triethanolamine, water
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:59:32.253492 · Updated: 2026-03-14T22:59:32.588894