822d25c0-71b3-421d-b702-4cded0c4ca32
34390-5
HUMAN OTC DRUG LABEL
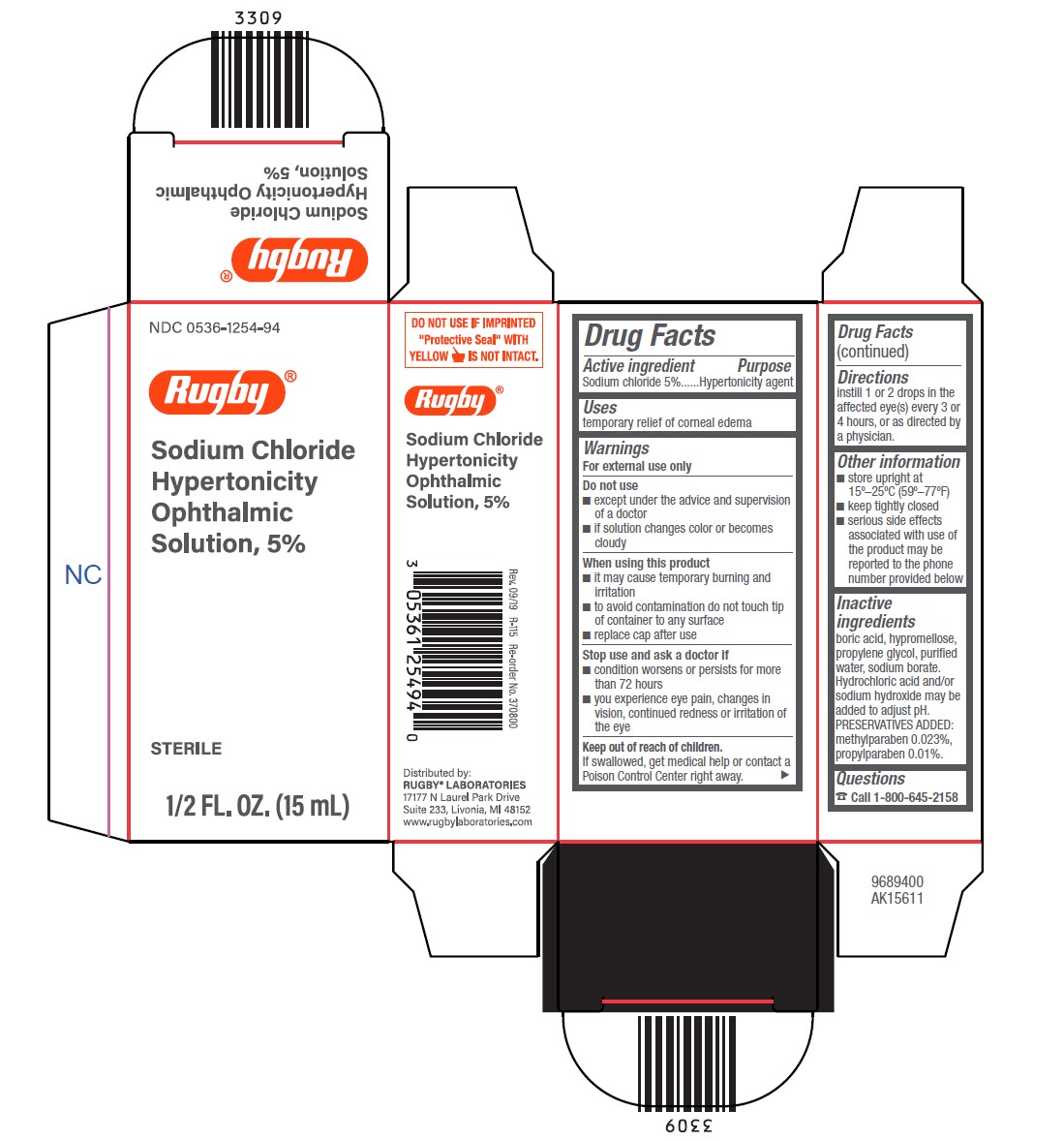
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Sodium chloride 5%
Purpose
Hypertonicity agent
Medication Information
Purpose
Hypertonicity agent
Description
Sodium chloride 5%
Uses
temporary relief of corneal edema
Warnings
Do not use
- except under the advice and supervision of a doctor
- if solution changes color or becomes cloudy
When using this product
- it may cause temporary burning and irritation
- to avoid contamination do not touch tip of container to any surface
- replace cap after use
Stop use and ask a doctor if
- condition worsens or persists for more than 72 hours
- you experience eye pain, changes in vision, continued redness or irritation of the eye
Directions
- instill 1 or 2 drops in the affected eye(s) every 3 or 4 hours, or as directed by a physician.
Questions?
[phone icon] Call 1-800-645-2158
Other Information
- store upright at 15° - 25°C (59° - 77°F)
- keep tightly closed
- serious side effects associated with use of the product may be reported to the phone number provided below
Active Ingredients
Sodium chloride 5%
Inactive Ingredients
boric acid, hypromellose, propylene glycol, purified water, sodium borate. Hydrochloric acid and/or sodium hydroxide may be added to adjust pH. PRESERVATIVES ADDED: methylparaben 0.023%, propylparaben 0.01%.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel – Carton
NDC 0536-1254-94
Rugby ®
Sodium Chloride
Hypertonicity
Ophthalmic
Solution, 5%
STERILE
1/2 FL. OZ. (15 mL)
Structured Label Content
Uses
temporary relief of corneal edema
Purpose
Hypertonicity agent
Warnings
Do not use
- except under the advice and supervision of a doctor
- if solution changes color or becomes cloudy
When using this product
- it may cause temporary burning and irritation
- to avoid contamination do not touch tip of container to any surface
- replace cap after use
Stop use and ask a doctor if
- condition worsens or persists for more than 72 hours
- you experience eye pain, changes in vision, continued redness or irritation of the eye
Directions
- instill 1 or 2 drops in the affected eye(s) every 3 or 4 hours, or as directed by a physician.
Questions?
[phone icon] Call 1-800-645-2158
Other Information (Other information)
- store upright at 15° - 25°C (59° - 77°F)
- keep tightly closed
- serious side effects associated with use of the product may be reported to the phone number provided below
Active Ingredients (Active ingredients)
Sodium chloride 5%
Inactive Ingredients (Inactive ingredients)
boric acid, hypromellose, propylene glycol, purified water, sodium borate. Hydrochloric acid and/or sodium hydroxide may be added to adjust pH. PRESERVATIVES ADDED: methylparaben 0.023%, propylparaben 0.01%.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Package/label Principal Display Panel – Carton (Package/Label Principal Display Panel – carton)
NDC 0536-1254-94
Rugby ®
Sodium Chloride
Hypertonicity
Ophthalmic
Solution, 5%
STERILE
1/2 FL. OZ. (15 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:36.585238 · Updated: 2026-03-14T23:03:47.044278