These Highlights Do Not Include All The Information Needed To Use Cerdelga®
819f828a-b888-4e46-83fc-94d774a28a83
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
CERDELGA is indicated for the long-term treatment of adult patients with Gaucher disease type 1 (GD1) who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) as detected by an FDA-cleared test [see Dosage and Administration (2.1) ] .
Indications and Usage
CERDELGA is indicated for the long-term treatment of adult patients with Gaucher disease type 1 (GD1) who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) as detected by an FDA-cleared test [see Dosage and Administration (2.1) ] .
Dosage and Administration
Patient Selection ( 2.1 ) : Select patients using an FDA-cleared test for determining CYP2D6 genotype. Recommended Dosage Based on CYP2D6 Metabolizer Status ( 2.2 ) : EMs and IMs: 84 mg orally twice daily. PMs: 84 mg orally once daily. See the Full Prescribing Information for dosing recommendations in patients receiving CYP2D6 and/or CYP3A inhibitors, or with renal or hepatic impairment. ( 2.2 , 4 , 7.1 , 8.6 , 8.7 ) Administration ( 2.4 ) : Swallow capsules whole, do not crush, dissolve or open capsules. ( 2.4 ) Avoid eating grapefruit or drinking grapefruit juice. ( 2.4 )
Warnings and Precautions
ECG Changes and Potential for Cardiac Arrhythmias : Not recommended in patients with pre-existing cardiac disease, long QT syndrome, and concomitant use of Class IA and Class III antiarrhythmics. ( 5.1 )
Contraindications
CERDELGA is contraindicated in the following patients based on CYP2D6 metabolizer status due to the risk of cardiac arrhythmias from prolongation of the PR, QTc, and/or QRS cardiac intervals.
Adverse Reactions
Most common adverse reactions (≥10%) are: fatigue, headache, nausea, diarrhea, back pain, pain in extremities, and upper abdominal pain. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Genzyme Corporation at 1-800-745-4447 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
See Full Prescribing Information for a list of clinically significant drug interactions. ( 7.1 , 7.2 )
Storage and Handling
CERDELGA is supplied as 84 mg eliglustat in a capsule with a pearl blue-green opaque cap and pearl white opaque body imprinted with "GZ02" in black. CERDELGA 84 mg capsules are supplied as: NDC 58468-0220-1 – Carton containing 4 packs of capsules (56 capsules total). Each pack is composed of 1 blister card of 14 capsules and a cardboard wallet. NDC 58468-0220-2 – Carton containing 1 pack of capsules (14 capsules total). Each pack is comprised of 1 blister card of 14 capsules and a cardboard wallet.
How Supplied
CERDELGA is supplied as 84 mg eliglustat in a capsule with a pearl blue-green opaque cap and pearl white opaque body imprinted with "GZ02" in black. CERDELGA 84 mg capsules are supplied as: NDC 58468-0220-1 – Carton containing 4 packs of capsules (56 capsules total). Each pack is composed of 1 blister card of 14 capsules and a cardboard wallet. NDC 58468-0220-2 – Carton containing 1 pack of capsules (14 capsules total). Each pack is comprised of 1 blister card of 14 capsules and a cardboard wallet.
Medication Information
Warnings and Precautions
ECG Changes and Potential for Cardiac Arrhythmias : Not recommended in patients with pre-existing cardiac disease, long QT syndrome, and concomitant use of Class IA and Class III antiarrhythmics. ( 5.1 )
Indications and Usage
CERDELGA is indicated for the long-term treatment of adult patients with Gaucher disease type 1 (GD1) who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) as detected by an FDA-cleared test [see Dosage and Administration (2.1) ] .
Dosage and Administration
Patient Selection ( 2.1 ) : Select patients using an FDA-cleared test for determining CYP2D6 genotype. Recommended Dosage Based on CYP2D6 Metabolizer Status ( 2.2 ) : EMs and IMs: 84 mg orally twice daily. PMs: 84 mg orally once daily. See the Full Prescribing Information for dosing recommendations in patients receiving CYP2D6 and/or CYP3A inhibitors, or with renal or hepatic impairment. ( 2.2 , 4 , 7.1 , 8.6 , 8.7 ) Administration ( 2.4 ) : Swallow capsules whole, do not crush, dissolve or open capsules. ( 2.4 ) Avoid eating grapefruit or drinking grapefruit juice. ( 2.4 )
Contraindications
CERDELGA is contraindicated in the following patients based on CYP2D6 metabolizer status due to the risk of cardiac arrhythmias from prolongation of the PR, QTc, and/or QRS cardiac intervals.
Adverse Reactions
Most common adverse reactions (≥10%) are: fatigue, headache, nausea, diarrhea, back pain, pain in extremities, and upper abdominal pain. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Genzyme Corporation at 1-800-745-4447 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
See Full Prescribing Information for a list of clinically significant drug interactions. ( 7.1 , 7.2 )
Storage and Handling
CERDELGA is supplied as 84 mg eliglustat in a capsule with a pearl blue-green opaque cap and pearl white opaque body imprinted with "GZ02" in black. CERDELGA 84 mg capsules are supplied as: NDC 58468-0220-1 – Carton containing 4 packs of capsules (56 capsules total). Each pack is composed of 1 blister card of 14 capsules and a cardboard wallet. NDC 58468-0220-2 – Carton containing 1 pack of capsules (14 capsules total). Each pack is comprised of 1 blister card of 14 capsules and a cardboard wallet.
How Supplied
CERDELGA is supplied as 84 mg eliglustat in a capsule with a pearl blue-green opaque cap and pearl white opaque body imprinted with "GZ02" in black. CERDELGA 84 mg capsules are supplied as: NDC 58468-0220-1 – Carton containing 4 packs of capsules (56 capsules total). Each pack is composed of 1 blister card of 14 capsules and a cardboard wallet. NDC 58468-0220-2 – Carton containing 1 pack of capsules (14 capsules total). Each pack is comprised of 1 blister card of 14 capsules and a cardboard wallet.
Description
CERDELGA is indicated for the long-term treatment of adult patients with Gaucher disease type 1 (GD1) who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) as detected by an FDA-cleared test [see Dosage and Administration (2.1) ] .
Section 42229-5
Limitations of Use:
- Patients who are CYP2D6 ultra-rapid metabolizers (URMs) may not achieve adequate concentrations of CERDELGA to achieve a therapeutic effect [see Clinical Studies (14)].
- A specific dosage cannot be recommended for those patients whose CYP2D6 genotype cannot be determined (indeterminate metabolizers) [see Clinical Studies (14)].
Section 42231-1
| MEDICATION GUIDE CERDELGA® (sir-DEL-guh) (eliglustat) capsules |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Issued: January 2024 |
|
What is the most important information I should know about CERDELGA?
CERDELGA can affect the way other medicines work and other medicines can affect how CERDELGA works. Using CERDELGA with other medicines or herbal supplements may cause an increased risk of side effects. Especially tell your doctor if you take:
|
|
|
What is CERDELGA?
CERDELGA is a prescription medicine used for the long-term treatment of Gaucher disease type 1 (GD1) in adults. CERDELGA is not used in certain people with Gaucher disease type 1. Your doctor will perform a test to make sure that CERDELGA is right for you. It is not known if CERDELGA is safe and effective in children. |
|
|
What should I tell my doctor before taking CERDELGA? Before taking CERDELGA, tell your doctor about all of your medical conditions, including if you:
|
|
How should I take CERDELGA?
|
|
|
What should I avoid while taking CERDELGA?
Avoid eating or drinking grapefruit products while taking CERDELGA. Grapefruit products can increase the amount of CERDELGA in your body. |
|
|
What are the possible side effects of CERDELGA?
See "What is the most important information I should know about CERDELGA?"
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of CERDELGA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store CERDELGA?
|
|
|
General information about the safe and effective use of CERDELGA.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CERDELGA for a condition for which it was not prescribed. Do not give CERDELGA to other people, even if they have the same symptoms you have. It may harm them. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about CERDELGA that is written for health professionals. For more information, go to www.cerdelga.com or call 1-800-745-4447. |
|
|
What are the ingredients in CERDELGA? Active ingredient: eliglustat Inactive ingredients: microcrystalline cellulose, lactose monohydrate, hypromellose, glyceryl behenate, gelatin, candurin silver fine, yellow iron oxide, and FD&C blue 2. Genzyme Corporation, 450 Water Street, Cambridge, MA 02141 A SANOFI COMPANY CERDELGA is a registered trademark of Genzyme Corporation. ©2024 Genzyme Corporation. All rights reserved. |
Section 44425-7
Store at 68°F–77°F (20°C–25°C) with excursions permitted between 59°F and 86°F (15°C to 30°C) [see USP Controlled Room Temperature].
10 Overdosage
The highest eliglustat plasma concentration experienced to date occurred in a single-dose, dose escalation study in healthy subjects, in a subject taking a dose equivalent to approximately 21 times the recommended dose for GD1 patients. At the time of the highest plasma concentration (59-fold higher than normal therapeutic conditions), the subject experienced dizziness marked by disequilibrium, hypotension, bradycardia, nausea, and vomiting.
In the event of acute overdose, the patient should be carefully observed and given symptomatic and supportive treatment.
Hemodialysis is unlikely to be beneficial given that eliglustat has a large volume of distribution [see Clinical Pharmacology (12.3)].
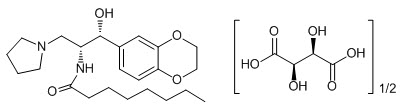
11 Description
CERDELGA (eliglustat) capsules contain eliglustat tartrate, which is a small molecule inhibitor of glucosylceramide synthase that resembles the ceramide substrate for the enzyme, with the chemical name N-((1R,2R)-1-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-1-hydroxy-3-(pyrrolidin-1-yl)propan-2-yl)octanamide (2R,3R)-2,3-dihydroxysuccinate. Its molecular weight is 479.59, and the empirical formula is C23H36N2O4+½(C4H6O6) with the following chemical structure:
Each capsule of CERDELGA for oral use contains 84 mg of eliglustat (equivalent to 100 mg of eliglustat tartrate). The inactive ingredients are candurin silver fine, FD&C blue 2, gelatin, glyceryl behenate, hypromellose, lactose monohydrate, microcrystalline cellulose, and yellow iron oxide.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of CERDELGA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Clinical experience has not identified differences in responses between the elderly and younger patients.
4 Contraindications
CERDELGA is contraindicated in the following patients based on CYP2D6 metabolizer status due to the risk of cardiac arrhythmias from prolongation of the PR, QTc, and/or QRS cardiac intervals.
6 Adverse Reactions
Most common adverse reactions (≥10%) are: fatigue, headache, nausea, diarrhea, back pain, pain in extremities, and upper abdominal pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Genzyme Corporation at 1-800-745-4447 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
8.6 Renal Impairment
Use CERDELGA in patients with renal impairment based on the patient's CYP2D6 metabolizer status [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Effects on spleen and liver volume, hemoglobin, and platelets increased with increasing steady-state average trough concentrations of eliglustat ranging up to 14 ng/mL in treatment naive patients in Trial 1. In patients previously treated with enzyme-replacement therapy in Trial 2 [see Clinical Studies (14.2)], no clinically relevant exposure-response relationship was observed.
2.1 Patient Selection
Select patients with Gaucher disease type 1 based on their CYP2D6 metabolizer status. It is recommended patient genotypes be established using an FDA-cleared test for determining CYP2D6 genotype [see Indications and Usage (1)].
8.7 Hepatic Impairment
Use CERDELGA in patients with hepatic impairment based on CYP2D6 metabolizer status and concomitant use of CYP2D6 or CYP3A inhibitors [see Clinical Pharmacology (12.3)].
1 Indications and Usage
CERDELGA is indicated for the long-term treatment of adult patients with Gaucher disease type 1 (GD1) who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) as detected by an FDA-cleared test [see Dosage and Administration (2.1)].
12.1 Mechanism of Action
Gaucher disease is caused by a deficiency of the lysosomal enzyme acid β-glucosidase. Acid β-glucosidase catalyzes the conversion of the sphingolipid glucocerebroside into glucose and ceramide. The enzymatic deficiency causes an accumulation of glucosylceramide (GL-1) primarily in the lysosomal compartment of macrophages, giving rise to foam cells or "Gaucher cells."
The clinical features of this lysosomal storage disorder (LSD) are reflective of the accumulation of Gaucher cells in the reticuloendothelial system (liver, spleen, bone marrow, and other organs). The accumulation of Gaucher cells in the liver, spleen, and bone marrow leads to organomegaly and skeletal disease. Presence of Gaucher cells in the bone marrow and spleen leads to clinically significant anemia and thrombocytopenia.
CERDELGA is a specific inhibitor of glucosylceramide synthase (IC50=10 ng/mL) and acts as a substrate reduction therapy for GD1 by reducing the production of GL-1. By reducing GL-1 production, CERDELGA alleviates the accumulation of GL-1 in the target organs.
5 Warnings and Precautions
ECG Changes and Potential for Cardiac Arrhythmias: Not recommended in patients with pre-existing cardiac disease, long QT syndrome, and concomitant use of Class IA and Class III antiarrhythmics. (5.1)
2 Dosage and Administration
Patient Selection ( 2.1 ):
- Select patients using an FDA-cleared test for determining CYP2D6 genotype.
Recommended Dosage Based on CYP2D6 Metabolizer Status ( 2.2 ):
- EMs and IMs: 84 mg orally twice daily.
- PMs: 84 mg orally once daily.
- See the Full Prescribing Information for dosing recommendations in patients receiving CYP2D6 and/or CYP3A inhibitors, or with renal or hepatic impairment. (2.2, 4, 7.1, 8.6, 8.7)
Administration ( 2.4 ):
2.2 Recommended Adult Dosage
The recommended dosage of CERDELGA in adults is based on the patient's CYP2D6 metabolizer status.
| CYP2D6 Metabolizer Status | CERDELGA Dosage |
|---|---|
| EMs | 84 mg twice daily |
| IMs | |
| PMs | 84 mg once daily |
3 Dosage Forms and Strengths
Capsules: 84 mg of eliglustat is in a capsule with a pearl blue-green opaque cap and pearl white opaque body imprinted with "GZ02" in black.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions to CERDELGA (occurring in ≥10% of the 126 GD1 patients treated with CERDELGA across Trials 1 and 2) were fatigue, headache, nausea, diarrhea, back pain, pain in extremities, and upper abdominal pain.
The adverse reaction profile of CERDELGA is based on two controlled studies, Trials 1 and 2 [see Clinical Studies (14.1, 14.2)]. Table 3 presents the profile from the 9-month double-blind, randomized, placebo-controlled trial of 40 treatment-naive patients (Trial 1). Patients were between the ages of 16 and 63 on the date of the first dose of study drug, and included 20 males and 20 females.
| CERDELGA (N=20) |
Placebo (N=20) |
|
|---|---|---|
| Adverse Reaction | Patients n (%) |
Patients n (%) |
| Arthralgia | 9 (45) | 2 (10) |
| Headache | 8 (40) | 6 (30) |
| Migraine | 2 (10) | 0 (0) |
| Flatulence | 2 (10) | 1 (5) |
| Nausea | 2 (10) | 1 (5) |
| Oropharyngeal pain | 2 (10) | 1 (5) |
Table 4 presents the profile from the 12-month open-label, randomized, imiglucerase-controlled trial of 159 treated patients switching from enzyme replacement therapy (ERT) (Trial 2). Patients were between the ages of 18 and 69 on the date of the first dose of CERDELGA, and included 87 females and 72 males.
| CERDELGA (N=106) |
Imiglucerase (N=53) |
|
|---|---|---|
| Adverse Reaction | Patients n (%) |
Patients n (%) |
| Fatigue | 15 (14) | 1 (2) |
| Headache | 14 (13) | 1 (2) |
| Nausea | 13 (12) | 0 (0) |
| Diarrhea | 13 (12) | 2 (4) |
| Back pain | 13 (12) | 3 (6) |
| Pain in extremity | 12 (11) | 1 (2) |
| Upper abdominal pain | 11 (10) | 0 (0) |
| Dizziness | 9 (8) | 0 (0) |
| Asthenia | 9 (8) | 0 (0) |
| Cough | 7 (7) | 2 (4) |
| Dyspepsia | 7 (7) | 1 (2) |
| Gastroesophageal reflux disease | 7 (7) | 0 (0) |
| Constipation | 5 (5) | 0 (0) |
| Palpitations | 5 (5) | 0 (0) |
| Rash | 5 (5) | 0 (0) |
In a separate uncontrolled study, with up to 4 years of treatment in 26 naive GD1 patients, the types and incidences of adverse reactions were similar to Trials 1 and 2.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling
CERDELGA is supplied as 84 mg eliglustat in a capsule with a pearl blue-green opaque cap and pearl white opaque body imprinted with "GZ02" in black.
CERDELGA 84 mg capsules are supplied as:
NDC 58468-0220-1 – Carton containing 4 packs of capsules (56 capsules total). Each pack is composed of 1 blister card of 14 capsules and a cardboard wallet.
NDC 58468-0220-2 – Carton containing 1 pack of capsules (14 capsules total). Each pack is comprised of 1 blister card of 14 capsules and a cardboard wallet.
7.1 Effect of Other Drugs On Cerdelga
Coadministration of CERDELGA with:
- CYP2D6 or CYP3A inhibitors may increase eliglustat concentrations which may increase the risk of cardiac arrhythmias from prolongation of the PR, QTc, and/or QRS cardiac interval [see Warnings and Precautions (5.1), Clinical Pharmacology (12.3)].
- strong CYP3A inducers decrease eliglustat concentrations which may reduce CERDELGA efficacy [see Clinical Pharmacology (12.3)].
See Table 5 for prevention and management of interactions with drugs affecting CERDELGA. Use of CERDELGA is contraindicated, to be avoided, or may require dosage adjustment depending on the concomitant drug and CYP2D6 metabolizer status [see Dosage and Administration (2.2, 2.3), Contraindications (4), Drug Interactions (7.1)].
| Concomitant Drug(s) | CYP2D6 Metabolizer Status | |
|---|---|---|
| EMs | IMs | PMs |
| CYP2D6 Inhibitor | ||
| Strong | Reduce frequency of CERDELGA 84 mg to once daily | Continue CERDELGA 84 mg once daily No effect of CYP2D6 inhibitor due to little or no CYP2D6 activity in CYP2D6 PMs.
|
| Moderate | ||
| Weak | Continue CERDELGA 84 mg twice daily | |
| CYP3A Inhibitor | ||
| Strong | Reduce frequency of CERDELGA 84 mg to once daily | Contraindicated |
| Moderate | Avoid coadministration | |
| Weak | Continue CERDELGA 84 mg twice daily | Avoid coadministration |
| CYP2D6 Inhibitor Concomitantly with a strong CYP3A Inhibitor | ||
| Strong | Contraindicated | |
| Moderate | ||
| CYP2D6 Inhibitor Concomitantly with a moderate CYP3A Inhibitor | ||
| Strong | Contraindicated | Avoid coadministration |
| Moderate | ||
| CYP3A Inducer | ||
| Strong | Avoid coadministration |
7.2 Effect of Cerdelga On Other Drugs
See Table 6 for clinically relevant interactions affecting P-gp or CYP2D6 substrates when coadministered with CERDELGA.
| Substrates for P-gp or CYP2D6 | ||
|---|---|---|
| Clinical Impact | Coadministration of CERDELGA may increase concentrations of drugs that are substrates for P-gp or CYP2D6 [see Clinical Pharmacology (12.3)] and may increase the risk of toxicity of these drugs. | |
| Prevention or Management | Digoxin | Monitor serum digoxin concentrations before initiating CERDELGA. Reduce digoxin dose by 30% and continue monitoring. |
| Other P-gp substrates or CYP2D6 substrates | Monitor therapeutic drug concentrations, as indicated, or consider reducing the dosage of the concomitant drug and titrate to clinical effect. |
2.4 Important Administration Instructions
- Swallow capsules whole, preferably with water, and do not crush, dissolve, or open the capsules.
- CERDELGA can be taken with or without food.
- Avoid the consumption of grapefruit or grapefruit juice (strong CYP3A inhibitors) with CERDELGA [see Drug Interactions (7.1)].
- If a dose of CERDELGA is missed, take the prescribed dose at the next scheduled time; do not double the next dose.
- For patients currently treated with imiglucerase, velaglucerase alfa, or taliglucerase alfa, CERDELGA may be administered 24 hours after the last dose of the previous enzyme replacement therapy (ERT).
Principal Display Panel 4 Blister Pack Carton
NDC 58468-0220-1
Cerdelga®
(eliglustat) capsules
84 mg*
*Each capsule contains 84 mg of eliglustat which is
equivalent to 100 mg of eliglustat tartrate
Dispense the enclosed Medication Guide to each patient
Four cartons of 14 capsules each
Rx Only
5.1 Ecg Changes and Potential for Cardiac Arrhythmias
CERDELGA is predicted to cause increases in ECG intervals (PR, QTc, and QRS) at substantially elevated eliglustat plasma concentrations and may increase the risk of cardiac arrhythmias.
- Use of CERDELGA is contraindicated, to be avoided, or requires dosage adjustment in patients taking CYP2D6 or CYP3A inhibitors, depending on CYP2D6 metabolizer status, type of inhibitor, or degree of hepatic impairment [see Dosage and Administration (2.3), Contraindications (4), Drug Interactions (7.1)].
Use of CERDELGA in patients with pre-existing cardiac conditions has not been studied during clinical trials. Avoid use of CERDELGA in patients with:
- pre-existing cardiac disease (congestive heart failure, recent acute myocardial infarction, bradycardia, heart block, ventricular arrhythmia)
- long QT syndrome
- in combination with Class IA (e.g., quinidine, procainamide) or Class III (e.g., amiodarone, sotalol) antiarrhythmic medications [see Clinical Pharmacology (12.2)]
14.1 Cerdelga in Treatment Naive Gd1 Patients – Trial 1
Trial 1 (NCT00891202) was a randomized, double-blind, placebo-controlled, multicenter clinical study evaluating the efficacy and safety of CERDELGA in 40 treatment- naive GD1 patients 16 years of age or older (median age 30.4 years) with pre-existing splenomegaly and hematological abnormalities. Patients were required to have received no treatment with substrate reduction therapy within 6 months or ERT within 9 months prior to randomization; all but 5 patients in the study had no prior therapy. Patients were stratified according to baseline spleen volume (≤20 or >20 multiples of normal [MN]) and randomized in a 1:1 ratio to receive CERDELGA or placebo for the duration of the 9-month blinded primary analysis period. The CERDELGA treatment group was comprised of IM (5%), EM (90%) and URM (5%) patients. Patients randomized to CERDELGA treatment received a starting dose of 42 mg twice daily, with a dose increase to 84 mg twice daily possible at Week 4 based on the plasma trough concentration at Week 2. The majority of patients (17 [85%]) received a dose escalation to 84 mg twice daily at Week 4, and 3 (15%) continued to receive 42 mg twice daily for the duration of the 9-month blinded primary analysis period.
The primary endpoint was the percentage change in spleen volume (in MN) from baseline to 9 months as compared to placebo. Secondary endpoints were absolute change in hemoglobin level, percentage change in liver volume (in MN), and percentage change in platelet count from baseline to 9 months compared to placebo.
At baseline, mean spleen volumes were 12.5 and 13.9 MN in the placebo and CERDELGA groups, respectively, and mean liver volumes were 1.4 MN for both groups. Mean hemoglobin levels were 12.8 and 12.1 g/dL, and platelet counts were 78.5 and 75.1 × 109/L, respectively.
During the 9-month primary analysis period, CERDELGA demonstrated statistically significant improvements in all primary and secondary endpoints compared to placebo, as shown in Table 10.
| Placebo (n=20) |
CERDELGA (n=20) | Difference (CERDELGA – Placebo) [95% CI] |
p value Estimates and p-value are based on ANCOVA model that includes treatment group, baseline spleen severity group (≤20 MN, >20 MN) and baseline parameter value.
|
|
|---|---|---|---|---|
| MN = Multiples of Normal, CI = confidence interval, NA = Not applicable | ||||
| Percentage Change in Spleen Volume MN (%) | 2.3 | -27.8 | -30.0 [-36.8, -23.2] |
<0.0001 |
| Absolute Change in Spleen Volume (MN) | 0.3 | -3.7 | -4.1 [-5.3, -2.9] |
NA |
| Absolute Change in Hemoglobin Level (g/dL) | -0.5 | 0.7 | 1.2 [0.6, 1.9] |
0.0006 |
| Percentage Change in Liver Volume MN (%) | 1.4 | -5.2 | -6.6 [-11.4, -1.9] |
0.0072 |
| Absolute Change in Liver Volume (MN) | 0.0 | -0.1 | -0.1 [-0.2, 0.0] |
NA |
| Percentage Change in Platelet Count (%) | -9.1 | 32.0 | 41.1 [24.0, 58.2] |
<0.0001 |
| Absolute Change in Platelet Count (× 109/L) | -7.2 | 24.1 | 31.3 [18.8, 43.8] |
NA |
In the open-label extension phase of Trial 1 in naive GD1 patients, 38 of 40 patients who continued treatment with CERDELGA for 2 years demonstrated the following changes in clinical parameters from baseline to 2 years: mean (SD) percent change in spleen volume (MN) -51.1% (10.7); mean (SD) percent change in liver volume (MN) -16.1% (11.3); mean (SD) absolute change in hemoglobin level (g/dL) 1.3 (1.2), and mean (SD) percent change in platelet count (mm3) 65.3% (40.9).
In a separate uncontrolled study (NCT00358150) of treatment-naive GD1 patients, improvements in spleen and liver volume, hemoglobin level, and platelet count continued through the 4-year treatment period.
14.2 Patients Switching From Enzyme Replacement Therapy to Cerdelga – Trial 2
Trial 2 (NCT00943111) was a randomized, open-label, active-controlled, non- inferiority, multicenter clinical study evaluating the efficacy and safety of CERDELGA compared with imiglucerase in 159 treated GD1 patients (median age 37.4 years) previously treated with enzyme replacement therapy (≥3 years of enzyme replacement therapy, dosed at 30–130 U/kg/month in at least 6 of the prior 9 months) who met pre-specified therapeutic goals at baseline. Pre-specified baseline therapeutic goals included: no bone crisis and free of symptomatic bone disease within the last year; mean hemoglobin level of ≥11 g/dL in females and ≥12 g/dL in males; mean platelet count ≥100,000/mm3; spleen volume <10 times normal and liver volume <1.5 times normal.
Patients were randomized 2:1 to receive CERDELGA or imiglucerase for the duration of the 12-month primary analysis period. Seventy-five percent of patients randomized to CERDELGA were previously treated with imiglucerase; 21% with velaglucerase alfa and 4% were unreported. Patients randomized to CERDELGA treatment received a starting dose of 42 mg twice daily, with dose increases to 84 mg twice daily and 127 mg twice daily possible at Weeks 4 and 8 based on plasma trough concentrations of CERDELGA at Weeks 2 and 6, respectively. The percentage of patients receiving the 3 possible CERDELGA doses was: 42 mg twice daily (20%), 84 mg twice daily (32%) and 127 mg twice daily (48%). The CERDELGA treatment group was comprised of PM (4%), IM (10%), EM (80%) and URM (4%) patients.
At baseline, mean spleen volumes were 2.6 and 3.2 MN in the imiglucerase and CERDELGA groups, respectively, and liver volumes were 0.9 MN in both groups. Mean hemoglobin levels were 13.8 and 13.6 g/dL, and platelet counts were 192 and 207 × 109/L, respectively.
The primary composite endpoint required stability in all four component domains (hemoglobin level, platelet count, liver volume, and spleen volume) based on changes between baseline and 12 months. Stability was defined by the following pre-specified thresholds of change: hemoglobin level <1.5 g/dL decrease, platelet count <25% decrease, liver volume <20% increase and spleen volume <25% increase. The percentages of patients meeting the criteria for stability in the individual components of the composite endpoint were assessed as secondary efficacy endpoints.
CERDELGA met the criteria to be declared non-inferior to imiglucerase in maintaining patient stability. After 12 months of treatment, the percentage of patients meeting the primary composite endpoint was 84.8% for the CERDELGA group compared to 93.6% for the imiglucerase group. The lower bound of the 95% CI of the 8.8% difference, -17.6%, was within the pre-specified non-inferiority margin of -25%. At Month 12, the percentages of CERDELGA and imiglucerase patients respectively, who met stability criteria for the individual components of the composite endpoint were: hemoglobin level, 94.9% and 100%; platelet count, 92.9% and 100%; spleen volume, 95.8% and 100%; and liver volume, 96.0% and 93.6%. Of the patients who did not meet stability criteria for the individual components, 12 of 15 CERDELGA patients and 3 of 3 imiglucerase patients remained within therapeutic goals for GD1.
Mean changes from baseline in the hematological and visceral parameters through 12 months of treatment are shown in Table 11. There were no clinically meaningful differences between groups for any of the four parameters.
| Imiglucerase (N=47) Mean [95% CI] |
CERDELGA (N=99) Mean [95% CI] |
|
|---|---|---|
| MN = Multiples of Normal, CI = confidence interval | ||
| Percentage Change in Spleen Volume MN (%) Excludes patients with a total splenectomy.
|
-3.0 [-6.4, 0.4] |
-6.2 [-9.5, -2.8] |
| Absolute Change in Spleen Volume (MN) | -0.1 [-0.2, 0.0] |
-0.2 [-0.3, -0.1] |
| Absolute Change in Hemoglobin Level (g/dL) | 0.0 [-0.2, 0.2] |
-0.2 [-0.4, -0.1] |
| Percentage Change in Liver Volume MN (%) | 3.6 [0.6, 6.6] |
1.8 [-0.2, 3.7] |
| Absolute Change in Liver Volume (MN) | 0.0 [0.0, 0.1] |
0.0 [0.0, 0.0] |
| Percentage Change in Platelet Count (%) | 2.9 [-0.6, 6.4] |
3.8 [0.0, 7.6] |
| Absolute Change in Platelet Count (× 109/L) | 6.0 [-0.9, 13.0] |
9.5 [1.4, 17.6] |
| Patients Stable for 52 Weeks, n (%) (Composite Primary Endpoint) |
44 (93.6) | 84 (84.8) |
In the open-label extension phase of Trial 2, 141 of 146 patients (42 patients previously treated with enzyme treatment therapy and 99 who continued treatment with CERDELGA) were evaluated for stability, as defined in the initial 12 months of the trial, in clinical parameters (composite of spleen and liver volume, hemoglobin level, and platelet count). Stability was shown in 120/141 (85%) patients at one year and 111/129 (86%) patients at 2 years of CERDELGA exposure.
2.3 Dosage Adjustment in Ems and Ims With Or Without Hepatic Impairment and Concomitant Use of Cyp2d6 Or Cyp3a Inhibitors
Reduce dosage frequency of CERDELGA 84 mg to once daily in CYP2D6 EMs and IMs with or without hepatic impairment taking CYP2D6 or CYP3A inhibitors, as shown in Table 2 [see Warnings and Precautions (5.1), Drug Interactions (7.1), Use in Specific Populations (8.7)].
| CYP2D6 Metabolizer Status | Hepatic Impairment Status | Concomitant CYP Inhibitor |
|---|---|---|
| EMs | Without Hepatic Impairment |
|
| Mild (Child-Pugh Class A) Hepatic Impairment |
|
|
| IMs | Without Hepatic Impairment |
|
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use:
- Patients who are CYP2D6 ultra-rapid metabolizers (URMs) may not achieve adequate concentrations of CERDELGA to achieve a therapeutic effect [see Clinical Studies (14)].
- A specific dosage cannot be recommended for those patients whose CYP2D6 genotype cannot be determined (indeterminate metabolizers) [see Clinical Studies (14)].
Section 42231-1 (42231-1)
| MEDICATION GUIDE CERDELGA® (sir-DEL-guh) (eliglustat) capsules |
|
|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Issued: January 2024 |
|
What is the most important information I should know about CERDELGA?
CERDELGA can affect the way other medicines work and other medicines can affect how CERDELGA works. Using CERDELGA with other medicines or herbal supplements may cause an increased risk of side effects. Especially tell your doctor if you take:
|
|
|
What is CERDELGA?
CERDELGA is a prescription medicine used for the long-term treatment of Gaucher disease type 1 (GD1) in adults. CERDELGA is not used in certain people with Gaucher disease type 1. Your doctor will perform a test to make sure that CERDELGA is right for you. It is not known if CERDELGA is safe and effective in children. |
|
|
What should I tell my doctor before taking CERDELGA? Before taking CERDELGA, tell your doctor about all of your medical conditions, including if you:
|
|
How should I take CERDELGA?
|
|
|
What should I avoid while taking CERDELGA?
Avoid eating or drinking grapefruit products while taking CERDELGA. Grapefruit products can increase the amount of CERDELGA in your body. |
|
|
What are the possible side effects of CERDELGA?
See "What is the most important information I should know about CERDELGA?"
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of CERDELGA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|
How should I store CERDELGA?
|
|
|
General information about the safe and effective use of CERDELGA.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CERDELGA for a condition for which it was not prescribed. Do not give CERDELGA to other people, even if they have the same symptoms you have. It may harm them. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about CERDELGA that is written for health professionals. For more information, go to www.cerdelga.com or call 1-800-745-4447. |
|
|
What are the ingredients in CERDELGA? Active ingredient: eliglustat Inactive ingredients: microcrystalline cellulose, lactose monohydrate, hypromellose, glyceryl behenate, gelatin, candurin silver fine, yellow iron oxide, and FD&C blue 2. Genzyme Corporation, 450 Water Street, Cambridge, MA 02141 A SANOFI COMPANY CERDELGA is a registered trademark of Genzyme Corporation. ©2024 Genzyme Corporation. All rights reserved. |
Section 44425-7 (44425-7)
Store at 68°F–77°F (20°C–25°C) with excursions permitted between 59°F and 86°F (15°C to 30°C) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
The highest eliglustat plasma concentration experienced to date occurred in a single-dose, dose escalation study in healthy subjects, in a subject taking a dose equivalent to approximately 21 times the recommended dose for GD1 patients. At the time of the highest plasma concentration (59-fold higher than normal therapeutic conditions), the subject experienced dizziness marked by disequilibrium, hypotension, bradycardia, nausea, and vomiting.
In the event of acute overdose, the patient should be carefully observed and given symptomatic and supportive treatment.
Hemodialysis is unlikely to be beneficial given that eliglustat has a large volume of distribution [see Clinical Pharmacology (12.3)].
11 Description (11 DESCRIPTION)
CERDELGA (eliglustat) capsules contain eliglustat tartrate, which is a small molecule inhibitor of glucosylceramide synthase that resembles the ceramide substrate for the enzyme, with the chemical name N-((1R,2R)-1-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-1-hydroxy-3-(pyrrolidin-1-yl)propan-2-yl)octanamide (2R,3R)-2,3-dihydroxysuccinate. Its molecular weight is 479.59, and the empirical formula is C23H36N2O4+½(C4H6O6) with the following chemical structure:
Each capsule of CERDELGA for oral use contains 84 mg of eliglustat (equivalent to 100 mg of eliglustat tartrate). The inactive ingredients are candurin silver fine, FD&C blue 2, gelatin, glyceryl behenate, hypromellose, lactose monohydrate, microcrystalline cellulose, and yellow iron oxide.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of CERDELGA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Clinical experience has not identified differences in responses between the elderly and younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
CERDELGA is contraindicated in the following patients based on CYP2D6 metabolizer status due to the risk of cardiac arrhythmias from prolongation of the PR, QTc, and/or QRS cardiac intervals.
6 Adverse Reactions (6 ADVERSE REACTIONS)
Most common adverse reactions (≥10%) are: fatigue, headache, nausea, diarrhea, back pain, pain in extremities, and upper abdominal pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Genzyme Corporation at 1-800-745-4447 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
8.6 Renal Impairment
Use CERDELGA in patients with renal impairment based on the patient's CYP2D6 metabolizer status [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Effects on spleen and liver volume, hemoglobin, and platelets increased with increasing steady-state average trough concentrations of eliglustat ranging up to 14 ng/mL in treatment naive patients in Trial 1. In patients previously treated with enzyme-replacement therapy in Trial 2 [see Clinical Studies (14.2)], no clinically relevant exposure-response relationship was observed.
2.1 Patient Selection
Select patients with Gaucher disease type 1 based on their CYP2D6 metabolizer status. It is recommended patient genotypes be established using an FDA-cleared test for determining CYP2D6 genotype [see Indications and Usage (1)].
8.7 Hepatic Impairment
Use CERDELGA in patients with hepatic impairment based on CYP2D6 metabolizer status and concomitant use of CYP2D6 or CYP3A inhibitors [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
CERDELGA is indicated for the long-term treatment of adult patients with Gaucher disease type 1 (GD1) who are CYP2D6 extensive metabolizers (EMs), intermediate metabolizers (IMs), or poor metabolizers (PMs) as detected by an FDA-cleared test [see Dosage and Administration (2.1)].
12.1 Mechanism of Action
Gaucher disease is caused by a deficiency of the lysosomal enzyme acid β-glucosidase. Acid β-glucosidase catalyzes the conversion of the sphingolipid glucocerebroside into glucose and ceramide. The enzymatic deficiency causes an accumulation of glucosylceramide (GL-1) primarily in the lysosomal compartment of macrophages, giving rise to foam cells or "Gaucher cells."
The clinical features of this lysosomal storage disorder (LSD) are reflective of the accumulation of Gaucher cells in the reticuloendothelial system (liver, spleen, bone marrow, and other organs). The accumulation of Gaucher cells in the liver, spleen, and bone marrow leads to organomegaly and skeletal disease. Presence of Gaucher cells in the bone marrow and spleen leads to clinically significant anemia and thrombocytopenia.
CERDELGA is a specific inhibitor of glucosylceramide synthase (IC50=10 ng/mL) and acts as a substrate reduction therapy for GD1 by reducing the production of GL-1. By reducing GL-1 production, CERDELGA alleviates the accumulation of GL-1 in the target organs.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
ECG Changes and Potential for Cardiac Arrhythmias: Not recommended in patients with pre-existing cardiac disease, long QT syndrome, and concomitant use of Class IA and Class III antiarrhythmics. (5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Patient Selection ( 2.1 ):
- Select patients using an FDA-cleared test for determining CYP2D6 genotype.
Recommended Dosage Based on CYP2D6 Metabolizer Status ( 2.2 ):
- EMs and IMs: 84 mg orally twice daily.
- PMs: 84 mg orally once daily.
- See the Full Prescribing Information for dosing recommendations in patients receiving CYP2D6 and/or CYP3A inhibitors, or with renal or hepatic impairment. (2.2, 4, 7.1, 8.6, 8.7)
Administration ( 2.4 ):
2.2 Recommended Adult Dosage
The recommended dosage of CERDELGA in adults is based on the patient's CYP2D6 metabolizer status.
| CYP2D6 Metabolizer Status | CERDELGA Dosage |
|---|---|
| EMs | 84 mg twice daily |
| IMs | |
| PMs | 84 mg once daily |
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsules: 84 mg of eliglustat is in a capsule with a pearl blue-green opaque cap and pearl white opaque body imprinted with "GZ02" in black.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions to CERDELGA (occurring in ≥10% of the 126 GD1 patients treated with CERDELGA across Trials 1 and 2) were fatigue, headache, nausea, diarrhea, back pain, pain in extremities, and upper abdominal pain.
The adverse reaction profile of CERDELGA is based on two controlled studies, Trials 1 and 2 [see Clinical Studies (14.1, 14.2)]. Table 3 presents the profile from the 9-month double-blind, randomized, placebo-controlled trial of 40 treatment-naive patients (Trial 1). Patients were between the ages of 16 and 63 on the date of the first dose of study drug, and included 20 males and 20 females.
| CERDELGA (N=20) |
Placebo (N=20) |
|
|---|---|---|
| Adverse Reaction | Patients n (%) |
Patients n (%) |
| Arthralgia | 9 (45) | 2 (10) |
| Headache | 8 (40) | 6 (30) |
| Migraine | 2 (10) | 0 (0) |
| Flatulence | 2 (10) | 1 (5) |
| Nausea | 2 (10) | 1 (5) |
| Oropharyngeal pain | 2 (10) | 1 (5) |
Table 4 presents the profile from the 12-month open-label, randomized, imiglucerase-controlled trial of 159 treated patients switching from enzyme replacement therapy (ERT) (Trial 2). Patients were between the ages of 18 and 69 on the date of the first dose of CERDELGA, and included 87 females and 72 males.
| CERDELGA (N=106) |
Imiglucerase (N=53) |
|
|---|---|---|
| Adverse Reaction | Patients n (%) |
Patients n (%) |
| Fatigue | 15 (14) | 1 (2) |
| Headache | 14 (13) | 1 (2) |
| Nausea | 13 (12) | 0 (0) |
| Diarrhea | 13 (12) | 2 (4) |
| Back pain | 13 (12) | 3 (6) |
| Pain in extremity | 12 (11) | 1 (2) |
| Upper abdominal pain | 11 (10) | 0 (0) |
| Dizziness | 9 (8) | 0 (0) |
| Asthenia | 9 (8) | 0 (0) |
| Cough | 7 (7) | 2 (4) |
| Dyspepsia | 7 (7) | 1 (2) |
| Gastroesophageal reflux disease | 7 (7) | 0 (0) |
| Constipation | 5 (5) | 0 (0) |
| Palpitations | 5 (5) | 0 (0) |
| Rash | 5 (5) | 0 (0) |
In a separate uncontrolled study, with up to 4 years of treatment in 26 naive GD1 patients, the types and incidences of adverse reactions were similar to Trials 1 and 2.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
CERDELGA is supplied as 84 mg eliglustat in a capsule with a pearl blue-green opaque cap and pearl white opaque body imprinted with "GZ02" in black.
CERDELGA 84 mg capsules are supplied as:
NDC 58468-0220-1 – Carton containing 4 packs of capsules (56 capsules total). Each pack is composed of 1 blister card of 14 capsules and a cardboard wallet.
NDC 58468-0220-2 – Carton containing 1 pack of capsules (14 capsules total). Each pack is comprised of 1 blister card of 14 capsules and a cardboard wallet.
7.1 Effect of Other Drugs On Cerdelga (7.1 Effect of Other Drugs on CERDELGA)
Coadministration of CERDELGA with:
- CYP2D6 or CYP3A inhibitors may increase eliglustat concentrations which may increase the risk of cardiac arrhythmias from prolongation of the PR, QTc, and/or QRS cardiac interval [see Warnings and Precautions (5.1), Clinical Pharmacology (12.3)].
- strong CYP3A inducers decrease eliglustat concentrations which may reduce CERDELGA efficacy [see Clinical Pharmacology (12.3)].
See Table 5 for prevention and management of interactions with drugs affecting CERDELGA. Use of CERDELGA is contraindicated, to be avoided, or may require dosage adjustment depending on the concomitant drug and CYP2D6 metabolizer status [see Dosage and Administration (2.2, 2.3), Contraindications (4), Drug Interactions (7.1)].
| Concomitant Drug(s) | CYP2D6 Metabolizer Status | |
|---|---|---|
| EMs | IMs | PMs |
| CYP2D6 Inhibitor | ||
| Strong | Reduce frequency of CERDELGA 84 mg to once daily | Continue CERDELGA 84 mg once daily No effect of CYP2D6 inhibitor due to little or no CYP2D6 activity in CYP2D6 PMs.
|
| Moderate | ||
| Weak | Continue CERDELGA 84 mg twice daily | |
| CYP3A Inhibitor | ||
| Strong | Reduce frequency of CERDELGA 84 mg to once daily | Contraindicated |
| Moderate | Avoid coadministration | |
| Weak | Continue CERDELGA 84 mg twice daily | Avoid coadministration |
| CYP2D6 Inhibitor Concomitantly with a strong CYP3A Inhibitor | ||
| Strong | Contraindicated | |
| Moderate | ||
| CYP2D6 Inhibitor Concomitantly with a moderate CYP3A Inhibitor | ||
| Strong | Contraindicated | Avoid coadministration |
| Moderate | ||
| CYP3A Inducer | ||
| Strong | Avoid coadministration |
7.2 Effect of Cerdelga On Other Drugs (7.2 Effect of CERDELGA on Other Drugs)
See Table 6 for clinically relevant interactions affecting P-gp or CYP2D6 substrates when coadministered with CERDELGA.
| Substrates for P-gp or CYP2D6 | ||
|---|---|---|
| Clinical Impact | Coadministration of CERDELGA may increase concentrations of drugs that are substrates for P-gp or CYP2D6 [see Clinical Pharmacology (12.3)] and may increase the risk of toxicity of these drugs. | |
| Prevention or Management | Digoxin | Monitor serum digoxin concentrations before initiating CERDELGA. Reduce digoxin dose by 30% and continue monitoring. |
| Other P-gp substrates or CYP2D6 substrates | Monitor therapeutic drug concentrations, as indicated, or consider reducing the dosage of the concomitant drug and titrate to clinical effect. |
2.4 Important Administration Instructions
- Swallow capsules whole, preferably with water, and do not crush, dissolve, or open the capsules.
- CERDELGA can be taken with or without food.
- Avoid the consumption of grapefruit or grapefruit juice (strong CYP3A inhibitors) with CERDELGA [see Drug Interactions (7.1)].
- If a dose of CERDELGA is missed, take the prescribed dose at the next scheduled time; do not double the next dose.
- For patients currently treated with imiglucerase, velaglucerase alfa, or taliglucerase alfa, CERDELGA may be administered 24 hours after the last dose of the previous enzyme replacement therapy (ERT).
Principal Display Panel 4 Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 4 Blister Pack Carton)
NDC 58468-0220-1
Cerdelga®
(eliglustat) capsules
84 mg*
*Each capsule contains 84 mg of eliglustat which is
equivalent to 100 mg of eliglustat tartrate
Dispense the enclosed Medication Guide to each patient
Four cartons of 14 capsules each
Rx Only
5.1 Ecg Changes and Potential for Cardiac Arrhythmias (5.1 ECG Changes and Potential for Cardiac Arrhythmias)
CERDELGA is predicted to cause increases in ECG intervals (PR, QTc, and QRS) at substantially elevated eliglustat plasma concentrations and may increase the risk of cardiac arrhythmias.
- Use of CERDELGA is contraindicated, to be avoided, or requires dosage adjustment in patients taking CYP2D6 or CYP3A inhibitors, depending on CYP2D6 metabolizer status, type of inhibitor, or degree of hepatic impairment [see Dosage and Administration (2.3), Contraindications (4), Drug Interactions (7.1)].
Use of CERDELGA in patients with pre-existing cardiac conditions has not been studied during clinical trials. Avoid use of CERDELGA in patients with:
- pre-existing cardiac disease (congestive heart failure, recent acute myocardial infarction, bradycardia, heart block, ventricular arrhythmia)
- long QT syndrome
- in combination with Class IA (e.g., quinidine, procainamide) or Class III (e.g., amiodarone, sotalol) antiarrhythmic medications [see Clinical Pharmacology (12.2)]
14.1 Cerdelga in Treatment Naive Gd1 Patients – Trial 1 (14.1 CERDELGA in Treatment-Naive GD1 Patients – Trial 1)
Trial 1 (NCT00891202) was a randomized, double-blind, placebo-controlled, multicenter clinical study evaluating the efficacy and safety of CERDELGA in 40 treatment- naive GD1 patients 16 years of age or older (median age 30.4 years) with pre-existing splenomegaly and hematological abnormalities. Patients were required to have received no treatment with substrate reduction therapy within 6 months or ERT within 9 months prior to randomization; all but 5 patients in the study had no prior therapy. Patients were stratified according to baseline spleen volume (≤20 or >20 multiples of normal [MN]) and randomized in a 1:1 ratio to receive CERDELGA or placebo for the duration of the 9-month blinded primary analysis period. The CERDELGA treatment group was comprised of IM (5%), EM (90%) and URM (5%) patients. Patients randomized to CERDELGA treatment received a starting dose of 42 mg twice daily, with a dose increase to 84 mg twice daily possible at Week 4 based on the plasma trough concentration at Week 2. The majority of patients (17 [85%]) received a dose escalation to 84 mg twice daily at Week 4, and 3 (15%) continued to receive 42 mg twice daily for the duration of the 9-month blinded primary analysis period.
The primary endpoint was the percentage change in spleen volume (in MN) from baseline to 9 months as compared to placebo. Secondary endpoints were absolute change in hemoglobin level, percentage change in liver volume (in MN), and percentage change in platelet count from baseline to 9 months compared to placebo.
At baseline, mean spleen volumes were 12.5 and 13.9 MN in the placebo and CERDELGA groups, respectively, and mean liver volumes were 1.4 MN for both groups. Mean hemoglobin levels were 12.8 and 12.1 g/dL, and platelet counts were 78.5 and 75.1 × 109/L, respectively.
During the 9-month primary analysis period, CERDELGA demonstrated statistically significant improvements in all primary and secondary endpoints compared to placebo, as shown in Table 10.
| Placebo (n=20) |
CERDELGA (n=20) | Difference (CERDELGA – Placebo) [95% CI] |
p value Estimates and p-value are based on ANCOVA model that includes treatment group, baseline spleen severity group (≤20 MN, >20 MN) and baseline parameter value.
|
|
|---|---|---|---|---|
| MN = Multiples of Normal, CI = confidence interval, NA = Not applicable | ||||
| Percentage Change in Spleen Volume MN (%) | 2.3 | -27.8 | -30.0 [-36.8, -23.2] |
<0.0001 |
| Absolute Change in Spleen Volume (MN) | 0.3 | -3.7 | -4.1 [-5.3, -2.9] |
NA |
| Absolute Change in Hemoglobin Level (g/dL) | -0.5 | 0.7 | 1.2 [0.6, 1.9] |
0.0006 |
| Percentage Change in Liver Volume MN (%) | 1.4 | -5.2 | -6.6 [-11.4, -1.9] |
0.0072 |
| Absolute Change in Liver Volume (MN) | 0.0 | -0.1 | -0.1 [-0.2, 0.0] |
NA |
| Percentage Change in Platelet Count (%) | -9.1 | 32.0 | 41.1 [24.0, 58.2] |
<0.0001 |
| Absolute Change in Platelet Count (× 109/L) | -7.2 | 24.1 | 31.3 [18.8, 43.8] |
NA |
In the open-label extension phase of Trial 1 in naive GD1 patients, 38 of 40 patients who continued treatment with CERDELGA for 2 years demonstrated the following changes in clinical parameters from baseline to 2 years: mean (SD) percent change in spleen volume (MN) -51.1% (10.7); mean (SD) percent change in liver volume (MN) -16.1% (11.3); mean (SD) absolute change in hemoglobin level (g/dL) 1.3 (1.2), and mean (SD) percent change in platelet count (mm3) 65.3% (40.9).
In a separate uncontrolled study (NCT00358150) of treatment-naive GD1 patients, improvements in spleen and liver volume, hemoglobin level, and platelet count continued through the 4-year treatment period.
14.2 Patients Switching From Enzyme Replacement Therapy to Cerdelga – Trial 2 (14.2 Patients Switching from Enzyme Replacement Therapy to CERDELGA – Trial 2)
Trial 2 (NCT00943111) was a randomized, open-label, active-controlled, non- inferiority, multicenter clinical study evaluating the efficacy and safety of CERDELGA compared with imiglucerase in 159 treated GD1 patients (median age 37.4 years) previously treated with enzyme replacement therapy (≥3 years of enzyme replacement therapy, dosed at 30–130 U/kg/month in at least 6 of the prior 9 months) who met pre-specified therapeutic goals at baseline. Pre-specified baseline therapeutic goals included: no bone crisis and free of symptomatic bone disease within the last year; mean hemoglobin level of ≥11 g/dL in females and ≥12 g/dL in males; mean platelet count ≥100,000/mm3; spleen volume <10 times normal and liver volume <1.5 times normal.
Patients were randomized 2:1 to receive CERDELGA or imiglucerase for the duration of the 12-month primary analysis period. Seventy-five percent of patients randomized to CERDELGA were previously treated with imiglucerase; 21% with velaglucerase alfa and 4% were unreported. Patients randomized to CERDELGA treatment received a starting dose of 42 mg twice daily, with dose increases to 84 mg twice daily and 127 mg twice daily possible at Weeks 4 and 8 based on plasma trough concentrations of CERDELGA at Weeks 2 and 6, respectively. The percentage of patients receiving the 3 possible CERDELGA doses was: 42 mg twice daily (20%), 84 mg twice daily (32%) and 127 mg twice daily (48%). The CERDELGA treatment group was comprised of PM (4%), IM (10%), EM (80%) and URM (4%) patients.
At baseline, mean spleen volumes were 2.6 and 3.2 MN in the imiglucerase and CERDELGA groups, respectively, and liver volumes were 0.9 MN in both groups. Mean hemoglobin levels were 13.8 and 13.6 g/dL, and platelet counts were 192 and 207 × 109/L, respectively.
The primary composite endpoint required stability in all four component domains (hemoglobin level, platelet count, liver volume, and spleen volume) based on changes between baseline and 12 months. Stability was defined by the following pre-specified thresholds of change: hemoglobin level <1.5 g/dL decrease, platelet count <25% decrease, liver volume <20% increase and spleen volume <25% increase. The percentages of patients meeting the criteria for stability in the individual components of the composite endpoint were assessed as secondary efficacy endpoints.
CERDELGA met the criteria to be declared non-inferior to imiglucerase in maintaining patient stability. After 12 months of treatment, the percentage of patients meeting the primary composite endpoint was 84.8% for the CERDELGA group compared to 93.6% for the imiglucerase group. The lower bound of the 95% CI of the 8.8% difference, -17.6%, was within the pre-specified non-inferiority margin of -25%. At Month 12, the percentages of CERDELGA and imiglucerase patients respectively, who met stability criteria for the individual components of the composite endpoint were: hemoglobin level, 94.9% and 100%; platelet count, 92.9% and 100%; spleen volume, 95.8% and 100%; and liver volume, 96.0% and 93.6%. Of the patients who did not meet stability criteria for the individual components, 12 of 15 CERDELGA patients and 3 of 3 imiglucerase patients remained within therapeutic goals for GD1.
Mean changes from baseline in the hematological and visceral parameters through 12 months of treatment are shown in Table 11. There were no clinically meaningful differences between groups for any of the four parameters.
| Imiglucerase (N=47) Mean [95% CI] |
CERDELGA (N=99) Mean [95% CI] |
|
|---|---|---|
| MN = Multiples of Normal, CI = confidence interval | ||
| Percentage Change in Spleen Volume MN (%) Excludes patients with a total splenectomy.
|
-3.0 [-6.4, 0.4] |
-6.2 [-9.5, -2.8] |
| Absolute Change in Spleen Volume (MN) | -0.1 [-0.2, 0.0] |
-0.2 [-0.3, -0.1] |
| Absolute Change in Hemoglobin Level (g/dL) | 0.0 [-0.2, 0.2] |
-0.2 [-0.4, -0.1] |
| Percentage Change in Liver Volume MN (%) | 3.6 [0.6, 6.6] |
1.8 [-0.2, 3.7] |
| Absolute Change in Liver Volume (MN) | 0.0 [0.0, 0.1] |
0.0 [0.0, 0.0] |
| Percentage Change in Platelet Count (%) | 2.9 [-0.6, 6.4] |
3.8 [0.0, 7.6] |
| Absolute Change in Platelet Count (× 109/L) | 6.0 [-0.9, 13.0] |
9.5 [1.4, 17.6] |
| Patients Stable for 52 Weeks, n (%) (Composite Primary Endpoint) |
44 (93.6) | 84 (84.8) |
In the open-label extension phase of Trial 2, 141 of 146 patients (42 patients previously treated with enzyme treatment therapy and 99 who continued treatment with CERDELGA) were evaluated for stability, as defined in the initial 12 months of the trial, in clinical parameters (composite of spleen and liver volume, hemoglobin level, and platelet count). Stability was shown in 120/141 (85%) patients at one year and 111/129 (86%) patients at 2 years of CERDELGA exposure.
2.3 Dosage Adjustment in Ems and Ims With Or Without Hepatic Impairment and Concomitant Use of Cyp2d6 Or Cyp3a Inhibitors (2.3 Dosage Adjustment in EMs and IMs With or Without Hepatic Impairment and Concomitant Use of CYP2D6 or CYP3A Inhibitors)
Reduce dosage frequency of CERDELGA 84 mg to once daily in CYP2D6 EMs and IMs with or without hepatic impairment taking CYP2D6 or CYP3A inhibitors, as shown in Table 2 [see Warnings and Precautions (5.1), Drug Interactions (7.1), Use in Specific Populations (8.7)].
| CYP2D6 Metabolizer Status | Hepatic Impairment Status | Concomitant CYP Inhibitor |
|---|---|---|
| EMs | Without Hepatic Impairment |
|
| Mild (Child-Pugh Class A) Hepatic Impairment |
|
|
| IMs | Without Hepatic Impairment |
|
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:40:30.351578 · Updated: 2026-03-14T22:02:25.018264