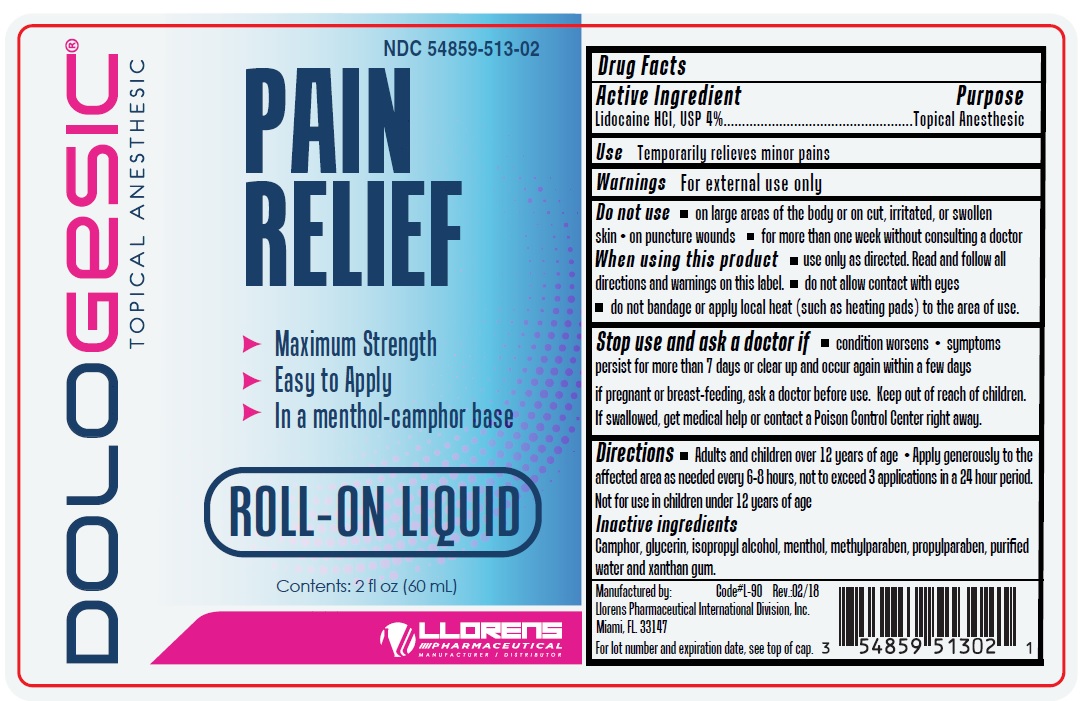
Dologesic Roll-on 513
81546810-85a8-45fe-b9e6-cdd02cc4c080
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient Lidocaine HCL, USP 4%
Medication Information
Warnings and Precautions
Warnings
For external use only
Do not use
- on large areas of the body or on cut, irritated or swollen skin
- on puncture wounds
- for more than one week without consulting a doctor
When using this product
- use only as directed. Read and follow all directions and warnings on this label.
- do not allow contact with eyes
- do not bandage or apply local heat (such as heating pads) to the area of use.
Stop use and ask doctor if
- condition worsens
- symptoms persist for more than 7 days or clear up and occur again within a few days
Indications and Usage
Use
Temporarily relieves minor pains
Dosage and Administration
Directions
- Adults and children over 12 years of age
- Apply generously to the affected area as needed every 6-8 hours, not to exceed 3 application in a 24 hour period.
- Not for use in children under 12 years of age.
Description
Active Ingredient Lidocaine HCL, USP 4%
Section 50565-1
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6
Inactive ingredients
camphor, glycerin, isopropyl alcohol, menthol, methylparaben, propylparaben, purified water and xantham gum.
Section 51945-4
Section 53413-1
Questions
Llorens Pharmaceutical International Division, Inc.
Miami Fl 33147
Section 53414-9
If pregnant or breast-feeding, ask a doctor before use.
Section 55105-1
Purpose
Topical Anesthetic
Section 55106-9
Active Ingredient
Lidocaine HCL, USP 4%
Structured Label Content
Indications and Usage (34067-9)
Use
Temporarily relieves minor pains
Dosage and Administration (34068-7)
Directions
- Adults and children over 12 years of age
- Apply generously to the affected area as needed every 6-8 hours, not to exceed 3 application in a 24 hour period.
- Not for use in children under 12 years of age.
Warnings and Precautions (34071-1)
Warnings
For external use only
Do not use
- on large areas of the body or on cut, irritated or swollen skin
- on puncture wounds
- for more than one week without consulting a doctor
When using this product
- use only as directed. Read and follow all directions and warnings on this label.
- do not allow contact with eyes
- do not bandage or apply local heat (such as heating pads) to the area of use.
Stop use and ask doctor if
- condition worsens
- symptoms persist for more than 7 days or clear up and occur again within a few days
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Section 51727-6 (51727-6)
Inactive ingredients
camphor, glycerin, isopropyl alcohol, menthol, methylparaben, propylparaben, purified water and xantham gum.
Section 51945-4 (51945-4)
Section 53413-1 (53413-1)
Questions
Llorens Pharmaceutical International Division, Inc.
Miami Fl 33147
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a doctor before use.
Section 55105-1 (55105-1)
Purpose
Topical Anesthetic
Section 55106-9 (55106-9)
Active Ingredient
Lidocaine HCL, USP 4%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:08:01.257088 · Updated: 2026-03-14T23:12:08.624404