These Highlights Do Not Include All The Information Needed To Use Prezista Safely And Effectively. See Full Prescribing Information For Prezista.
814301f9-c990-46a5-b481-2879a521a16f
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Contraindications ( 4 ) 4/2022
Indications and Usage
PREZISTA, co-administered with ritonavir (PREZISTA/ritonavir), in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus (HIV-1) infection in adult and pediatric patients 3 years of age and older [see Use in Specific Populations (8.4) and Clinical Studies (14) ] .
Dosage and Administration
Testing: In treatment-experienced patients, treatment history genotypic and/or phenotypic testing is recommended prior to initiation of therapy with PREZISTA/ritonavir to assess drug susceptibility of the HIV-1 virus ( 2.1 , 12.4 ) Monitor serum liver chemistry tests before and during therapy with PREZISTA/ritonavir. ( 2.1 , 2.2 , 5.2 ) Treatment-naïve adult patients and treatment-experienced adult patients with no darunavir resistance associated substitutions: 800 mg (one 800 mg tablet) taken with ritonavir 100 mg once daily and with food. ( 2.3 ) Treatment-experienced adult patients with at least one darunavir resistance associated substitution: 600 mg (one 600 mg tablet) taken with ritonavir 100 mg twice daily and with food. ( 2.3 ) Pregnant patients: 600 mg (one 600 mg tablet) taken with ritonavir 100 mg twice daily and with food. ( 2.4 ) Pediatric patients (3 to less than 18 years of age and weighing at least 10 kg): dosage of PREZISTA and ritonavir is based on body weight and should not exceed the adult dose. PREZISTA should be taken with ritonavir and with food. ( 2.5 ) PREZISTA/ritonavir is not recommended for use in patients with severe hepatic impairment. ( 2.6 )
Warnings and Precautions
Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) has been reported with PREZISTA/ritonavir. Monitor liver function before and during therapy, especially in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases. Post-marketing cases of liver injury, including some fatalities, have been reported. ( 5.2 ) Skin reactions ranging from mild to severe, including Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms and acute generalized exanthematous pustulosis, have been reported. Discontinue treatment if severe reaction develops. ( 5.3 ) Use with caution in patients with a known sulfonamide allergy. ( 5.4 ) Patients may develop new onset diabetes mellitus or hyperglycemia. Initiation or dose adjustments of insulin or oral hypoglycemic agents may be required. ( 5.6 ) Patients may develop redistribution/accumulation of body fat or immune reconstitution syndrome. ( 5.7 , 5.8 ) Patients with hemophilia may develop increased bleeding events. ( 5.9 ) PREZISTA/ritonavir is not recommended in pediatric patients below 3 years of age in view of toxicity and mortality observed in juvenile rats dosed with darunavir up to days 23 to 26 of age. ( 5.10 )
Contraindications
Co-administration of PREZISTA/ritonavir is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Examples of these drugs and other contraindicated drugs (which may lead to reduced efficacy of darunavir) are listed below [see Drug Interactions (7.3) ] . Due to the need for co-administration of PREZISTA with ritonavir, please refer to ritonavir prescribing information for a description of ritonavir contraindications. Alpha 1-adrenoreceptor antagonist: alfuzosin Anti-gout: colchicine, in patients with renal and/or hepatic impairment Antimycobacterial: rifampin Antipsychotics: lurasidone, pimozide Cardiac Disorders: dronedarone, ivabradine, ranolazine Ergot derivatives, e.g. dihydroergotamine, ergotamine, methylergonovine Herbal product: St. John's wort ( Hypericum perforatum ) Hepatitis C direct acting antiviral: elbasvir/grazoprevir Lipid modifying agents: lomitapide, lovastatin, simvastatin Opioid Antagonist: naloxegol PDE-5 inhibitor: sildenafil when used for treatment of pulmonary arterial hypertension Sedatives/hypnotics: orally administered midazolam, triazolam
Adverse Reactions
The following adverse reactions are discussed in other sections of labeling: Hepatotoxicity [see Warnings and Precautions (5.2) ] Severe Skin Reactions [see Warnings and Precautions (5.3) ] Diabetes Mellitus/Hyperglycemia [see Warnings and Precautions (5.6) ] Fat Redistribution [see Warnings and Precautions (5.7) ] Immune Reconstitution Syndrome [see Warnings and Precautions (5.8) ] Hemophilia [see Warnings and Precautions (5.9) ] Due to the need for co-administration of PREZISTA with ritonavir, please refer to ritonavir prescribing information for ritonavir-associated adverse reactions.
Drug Interactions
Co-administration of PREZISTA/ritonavir with other drugs can alter the concentrations of other drugs and other drugs may alter the concentrations of darunavir. The potential drug-drug interactions must be considered prior to and during therapy. ( 4 , 5.5 , 7 , 12.3 )
Storage and Handling
PREZISTA ® (darunavir) 100 mg per mL oral suspension is a white to off-white opaque liquid supplied in amber-colored multiple-dose bottles containing 100 mg of darunavir per mL packaged with a 6 mL oral dosing syringe with 0.2 mL gradations. PREZISTA ® (darunavir) 75 mg tablets are supplied as white, caplet-shaped, film-coated tablets debossed with "75" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 150 mg tablets are supplied as white, oval-shaped, film-coated tablets debossed with "150" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 600 mg tablets are supplied as orange, oval-shaped, film-coated tablets debossed with "600MG" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 800 mg tablets are supplied as dark red, oval-shaped, film-coated tablets debossed with "800" on one side and "T" on the other side. PREZISTA is packaged in bottles in the following configuration: 100 mg/mL oral suspension – 200 mL bottles (NDC 59676-565-01) 75 mg tablets — bottles of 480 (NDC 59676-563-01) 150 mg tablets — bottles of 240 (NDC 59676-564-01) 600 mg tablets — bottles of 60 (NDC 59676-562-01) 800 mg tablets — bottles of 30 (NDC 59676-566-30)
How Supplied
PREZISTA ® (darunavir) 100 mg per mL oral suspension is a white to off-white opaque liquid supplied in amber-colored multiple-dose bottles containing 100 mg of darunavir per mL packaged with a 6 mL oral dosing syringe with 0.2 mL gradations. PREZISTA ® (darunavir) 75 mg tablets are supplied as white, caplet-shaped, film-coated tablets debossed with "75" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 150 mg tablets are supplied as white, oval-shaped, film-coated tablets debossed with "150" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 600 mg tablets are supplied as orange, oval-shaped, film-coated tablets debossed with "600MG" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 800 mg tablets are supplied as dark red, oval-shaped, film-coated tablets debossed with "800" on one side and "T" on the other side. PREZISTA is packaged in bottles in the following configuration: 100 mg/mL oral suspension – 200 mL bottles (NDC 59676-565-01) 75 mg tablets — bottles of 480 (NDC 59676-563-01) 150 mg tablets — bottles of 240 (NDC 59676-564-01) 600 mg tablets — bottles of 60 (NDC 59676-562-01) 800 mg tablets — bottles of 30 (NDC 59676-566-30)
Medication Information
Warnings and Precautions
Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) has been reported with PREZISTA/ritonavir. Monitor liver function before and during therapy, especially in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases. Post-marketing cases of liver injury, including some fatalities, have been reported. ( 5.2 ) Skin reactions ranging from mild to severe, including Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms and acute generalized exanthematous pustulosis, have been reported. Discontinue treatment if severe reaction develops. ( 5.3 ) Use with caution in patients with a known sulfonamide allergy. ( 5.4 ) Patients may develop new onset diabetes mellitus or hyperglycemia. Initiation or dose adjustments of insulin or oral hypoglycemic agents may be required. ( 5.6 ) Patients may develop redistribution/accumulation of body fat or immune reconstitution syndrome. ( 5.7 , 5.8 ) Patients with hemophilia may develop increased bleeding events. ( 5.9 ) PREZISTA/ritonavir is not recommended in pediatric patients below 3 years of age in view of toxicity and mortality observed in juvenile rats dosed with darunavir up to days 23 to 26 of age. ( 5.10 )
Indications and Usage
PREZISTA, co-administered with ritonavir (PREZISTA/ritonavir), in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus (HIV-1) infection in adult and pediatric patients 3 years of age and older [see Use in Specific Populations (8.4) and Clinical Studies (14) ] .
Dosage and Administration
Testing: In treatment-experienced patients, treatment history genotypic and/or phenotypic testing is recommended prior to initiation of therapy with PREZISTA/ritonavir to assess drug susceptibility of the HIV-1 virus ( 2.1 , 12.4 ) Monitor serum liver chemistry tests before and during therapy with PREZISTA/ritonavir. ( 2.1 , 2.2 , 5.2 ) Treatment-naïve adult patients and treatment-experienced adult patients with no darunavir resistance associated substitutions: 800 mg (one 800 mg tablet) taken with ritonavir 100 mg once daily and with food. ( 2.3 ) Treatment-experienced adult patients with at least one darunavir resistance associated substitution: 600 mg (one 600 mg tablet) taken with ritonavir 100 mg twice daily and with food. ( 2.3 ) Pregnant patients: 600 mg (one 600 mg tablet) taken with ritonavir 100 mg twice daily and with food. ( 2.4 ) Pediatric patients (3 to less than 18 years of age and weighing at least 10 kg): dosage of PREZISTA and ritonavir is based on body weight and should not exceed the adult dose. PREZISTA should be taken with ritonavir and with food. ( 2.5 ) PREZISTA/ritonavir is not recommended for use in patients with severe hepatic impairment. ( 2.6 )
Contraindications
Co-administration of PREZISTA/ritonavir is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Examples of these drugs and other contraindicated drugs (which may lead to reduced efficacy of darunavir) are listed below [see Drug Interactions (7.3) ] . Due to the need for co-administration of PREZISTA with ritonavir, please refer to ritonavir prescribing information for a description of ritonavir contraindications. Alpha 1-adrenoreceptor antagonist: alfuzosin Anti-gout: colchicine, in patients with renal and/or hepatic impairment Antimycobacterial: rifampin Antipsychotics: lurasidone, pimozide Cardiac Disorders: dronedarone, ivabradine, ranolazine Ergot derivatives, e.g. dihydroergotamine, ergotamine, methylergonovine Herbal product: St. John's wort ( Hypericum perforatum ) Hepatitis C direct acting antiviral: elbasvir/grazoprevir Lipid modifying agents: lomitapide, lovastatin, simvastatin Opioid Antagonist: naloxegol PDE-5 inhibitor: sildenafil when used for treatment of pulmonary arterial hypertension Sedatives/hypnotics: orally administered midazolam, triazolam
Adverse Reactions
The following adverse reactions are discussed in other sections of labeling: Hepatotoxicity [see Warnings and Precautions (5.2) ] Severe Skin Reactions [see Warnings and Precautions (5.3) ] Diabetes Mellitus/Hyperglycemia [see Warnings and Precautions (5.6) ] Fat Redistribution [see Warnings and Precautions (5.7) ] Immune Reconstitution Syndrome [see Warnings and Precautions (5.8) ] Hemophilia [see Warnings and Precautions (5.9) ] Due to the need for co-administration of PREZISTA with ritonavir, please refer to ritonavir prescribing information for ritonavir-associated adverse reactions.
Drug Interactions
Co-administration of PREZISTA/ritonavir with other drugs can alter the concentrations of other drugs and other drugs may alter the concentrations of darunavir. The potential drug-drug interactions must be considered prior to and during therapy. ( 4 , 5.5 , 7 , 12.3 )
Storage and Handling
PREZISTA ® (darunavir) 100 mg per mL oral suspension is a white to off-white opaque liquid supplied in amber-colored multiple-dose bottles containing 100 mg of darunavir per mL packaged with a 6 mL oral dosing syringe with 0.2 mL gradations. PREZISTA ® (darunavir) 75 mg tablets are supplied as white, caplet-shaped, film-coated tablets debossed with "75" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 150 mg tablets are supplied as white, oval-shaped, film-coated tablets debossed with "150" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 600 mg tablets are supplied as orange, oval-shaped, film-coated tablets debossed with "600MG" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 800 mg tablets are supplied as dark red, oval-shaped, film-coated tablets debossed with "800" on one side and "T" on the other side. PREZISTA is packaged in bottles in the following configuration: 100 mg/mL oral suspension – 200 mL bottles (NDC 59676-565-01) 75 mg tablets — bottles of 480 (NDC 59676-563-01) 150 mg tablets — bottles of 240 (NDC 59676-564-01) 600 mg tablets — bottles of 60 (NDC 59676-562-01) 800 mg tablets — bottles of 30 (NDC 59676-566-30)
How Supplied
PREZISTA ® (darunavir) 100 mg per mL oral suspension is a white to off-white opaque liquid supplied in amber-colored multiple-dose bottles containing 100 mg of darunavir per mL packaged with a 6 mL oral dosing syringe with 0.2 mL gradations. PREZISTA ® (darunavir) 75 mg tablets are supplied as white, caplet-shaped, film-coated tablets debossed with "75" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 150 mg tablets are supplied as white, oval-shaped, film-coated tablets debossed with "150" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 600 mg tablets are supplied as orange, oval-shaped, film-coated tablets debossed with "600MG" on one side and "TMC" on the other side. PREZISTA ® (darunavir) 800 mg tablets are supplied as dark red, oval-shaped, film-coated tablets debossed with "800" on one side and "T" on the other side. PREZISTA is packaged in bottles in the following configuration: 100 mg/mL oral suspension – 200 mL bottles (NDC 59676-565-01) 75 mg tablets — bottles of 480 (NDC 59676-563-01) 150 mg tablets — bottles of 240 (NDC 59676-564-01) 600 mg tablets — bottles of 60 (NDC 59676-562-01) 800 mg tablets — bottles of 30 (NDC 59676-566-30)
Description
Contraindications ( 4 ) 4/2022
Section 42229-5
Treatment-Naïve Adult Patients
The recommended oral dose of PREZISTA is 800 mg (one 800 mg tablet or 8 mL of the oral suspension) taken with ritonavir 100 mg (one 100 mg tablet or capsule or 1.25 mL of a 80 mg per mL ritonavir oral solution) once daily and with food. An 8 mL PREZISTA dose should be taken as two 4 mL administrations with the included oral dosing syringe.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 03/2023 | |
| PATIENT INFORMATION | ||
|
PREZISTA
®
(pre-ZIS-ta)
(darunavir) oral suspension |
PREZISTA
®
(pre-ZIS-ta)
(darunavir) tablet |
|
| Read this Patient Information before you start taking PREZISTA and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Also read the Patient Information leaflet for ritonavir. |
||
What is the most important information I should know about PREZISTA?
|
||
|
|
|
|
||
|
|
|
| Rash occurred more often in people taking PREZISTA and raltegravir together than with either drug separately, but was generally mild.
See " What are the possible side effects of PREZISTA?" for more information about side effects. |
||
|
What is PREZISTA?
PREZISTA is a prescription HIV-1 (Human Immunodeficiency Virus-type 1) medicine used with ritonavir and other antiretroviral medicines to treat HIV-1 infection in adults and children 3 years of age and older. HIV is the virus that causes AIDS (Acquired Immune Deficiency Syndrome). PREZISTA should not be used in children under 3 years of age. When used with other antiretroviral medicines to treat HIV-1 infection, PREZISTA may help:
Reducing the amount of HIV-1 and increasing the CD4+ (T) cells in your blood may improve your immune system. This may reduce your risk of death or getting infections that can happen when your immune system is weak (opportunistic infections).
Ask your healthcare provider if you have any questions on how to prevent passing HIV to other people. |
||
|
Who should not take PREZISTA?
Do not take PREZISTA with any medicine that contains:
Serious problems can happen if you or your child take any of these medicines with PREZISTA. This is not a complete list of medicines. Therefore, tell your healthcare provider about allmedicines you take. |
||
|
What should I tell my healthcare provider before taking PREZISTA?
Before taking PREZISTA, tell your healthcare provider if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, topical creams, vitamins, and herbal supplements. Some medicines interact with PREZISTA. Keep a list of your medicines to show your healthcare provider and pharmacist.
|
||
How should I take PREZISTA?
|
||
|
What are the possible side effects of PREZISTA?
PREZISTA may cause serious side effects, including:
The most common side effects of PREZISTA include: |
||
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all of the possible side effects of PREZISTA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store PREZISTA?
Keep PREZISTA and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of PREZISTA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use PREZISTA for a condition for which it was not prescribed. Do not give PREZISTA to other people even if they have the same condition you have. It may harm them. This leaflet summarizes the most important information about PREZISTA. If you would like more information, talk to your healthcare provider. You can ask your healthcare provider or pharmacist for information about PREZISTA that is written for health professionals. For more information, call 1-800-526-7736. |
||
|
What are the ingredients in PREZISTA?
Active ingredient: darunavir Inactive ingredients: PREZISTA oral suspension:citric acid monohydrate, hydrochloric acid (for pH adjustment), hydroxypropyl cellulose, masking flavor, methylparaben sodium, microcrystalline cellulose, purified water, sodium carboxymethylcellulose, strawberry cream flavor, and sucralose. PREZISTA 75 mg and 150 mg tablets:colloidal silicon dioxide, crospovidone, magnesium stearate, microcrystalline cellulose. The film coating contains: OPADRY ®White (polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, titanium dioxide). PREZISTA 600 mg tablets:colloidal silicon dioxide, crospovidone, magnesium stearate, microcrystalline cellulose. The film coating contains: OPADRY ®Orange (FD&C Yellow No. 6, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, titanium dioxide). PREZISTA 800 mg tablets:colloidal silicon dioxide, crospovidone, magnesium stearate, microcrystalline cellulose, hypromellose. The film coating contains: OPADRY ®Dark Red (iron oxide red, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, titanium dioxide). Manufactured for: Janssen Products, LP, Horsham PA 19044, USA PREZISTA ®is a registered trademark of Janssen Pharmaceuticals © 2006 Janssen Pharmaceutical Companies |
Section 43679-0
Mechanism of Action
Darunavir is an inhibitor of the HIV-1 protease. It selectively inhibits the cleavage of HIV-1 encoded Gag-Pol polyproteins in infected cells, thereby preventing the formation of mature virus particles.
Section 43683-2
| Contraindications ( 4) | 4/2022 |
Section 44425-7
Storage
Section 59845-8
Instructions for Use
Advise patients to take PREZISTA and ritonavir with food every day on a regular dosing schedule, as missed doses can result in development of resistance. PREZISTA must always be used with ritonavir in combination with other antiretroviral drugs. Advise patients not to alter the dose of either PREZISTA or ritonavir, discontinue ritonavir, or discontinue therapy with PREZISTA without consulting their physician [see Dosage and Administration (2)] .
10 Overdosage
Human experience of acute overdose with PREZISTA/ritonavir is limited. No specific antidote is available for overdose with PREZISTA. Treatment of overdose with PREZISTA consists of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. Since PREZISTA is highly protein bound, dialysis is unlikely to be beneficial in significant removal of the active substance.
11 Description
PREZISTA (darunavir) is an inhibitor of the human immunodeficiency virus (HIV-1) protease.
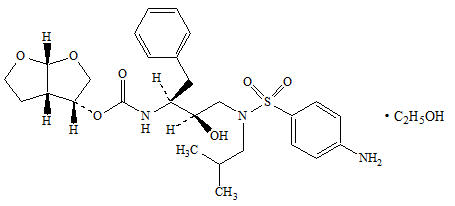
PREZISTA tablets and oral suspension contain the active ingredient darunavir, (present as darunavir ethanolate) which has the following chemical name: [(1S,2R)-3-[[(4-aminophenyl)sulfonyl](2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]-carbamic acid (3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl ester monoethanolate. Its molecular formula is C 27H 37N 3O 7S ∙ C 2H 5OH and its molecular weight is 593.73. Darunavir ethanolate has the following structural formula:
Darunavir ethanolate is a white to off-white powder with a solubility of approximately 0.15 mg per mL in water at 20°C.
PREZISTA ®100 mg per mL oral suspension is available as a white to off-white opaque suspension for oral administration.
Each mL of the oral suspension contains darunavir 100 mg (present as darunavir ethanolate). In addition, each mL contains the inactive ingredients citric acid monohydrate, hydrochloric acid (for pH adjustment), hydroxypropyl cellulose, masking flavor, methylparaben sodium, microcrystalline cellulose, purified water, sodium carboxymethylcellulose, strawberry cream flavor and sucralose.
PREZISTA ®75 mg tablets are available as white, caplet-shaped, film-coated tablets for oral administration. Each 75 mg tablet contains darunavir 75 mg (present as darunavir ethanolate).
PREZISTA ®150 mg tablets are available as white, oval-shaped, film-coated tablets for oral administration. Each 150 mg tablet contains darunavir 150 mg (present as darunavir ethanolate).
PREZISTA ®600 mg tablets are available as orange, oval-shaped, film-coated tablets for oral administration. Each 600 mg tablet contains darunavir 600 mg (present as darunavir ethanolate).
PREZISTA ®800 mg tablets are available as dark red, oval-shaped, film-coated tablets for oral administration. Each 800 mg tablet contains darunavir 800 mg (present as darunavir ethanolate).
During storage, partial conversion from ethanolate to hydrate may occur; however, this does not affect product quality or performance. Each tablet also contains the inactive ingredients colloidal silicon dioxide, crospovidone, magnesium stearate, and microcrystalline cellulose. The 800 mg tablet also contains hypromellose. The 75 and 150 mg tablet film coating, OPADRY ®White, contains polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, and titanium dioxide. The 600 mg tablet film coating, OPADRY ®Orange, contains FD&C Yellow No. 6, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, and titanium dioxide. The 800 mg tablet film coating, OPADRY ®Dark Red, contains iron oxide red, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, and titanium dioxide.
All strengths for PREZISTA are expressed in terms of the free form of darunavir.
5.9 Hemophilia
There have been reports of increased bleeding, including spontaneous skin hematomas and hemarthrosis in patients with hemophilia type A and B treated with PIs. In some patients, additional factor VIII was given. In more than half of the reported cases, treatment with PIs was continued or reintroduced if treatment had been discontinued. A causal relationship between PI therapy and these episodes has not been established.
5.4 Sulfa Allergy
Darunavir contains a sulfonamide moiety. PREZISTA should be used with caution in patients with a known sulfonamide allergy. In clinical studies with PREZISTA/ritonavir, the incidence and severity of rash were similar in subjects with or without a history of sulfonamide allergy.
8.4 Pediatric Use
PREZISTA/ritonavir is not recommended in pediatric patients below 3 years of age because of toxicity and mortality observed in juvenile rats dosed with darunavir (from 20 mg/kg to 1000 mg/kg) up to days 23 to 26 of age [see Warnings and Precautions (5.10) , Use in Specific Populations (8.1)and Clinical Pharmacology (12.3)] .
The safety, pharmacokinetic profile, and virologic and immunologic responses of PREZISTA/ritonavir administered twice daily were evaluated in treatment-experienced HIV-1-infected pediatric subjects 3 to less than 18 years of age and weighting at least 10 kg. These subjects were evaluated in clinical trials TMC114-C212 (80 subjects, 6 to less than 18 years of age) and TMC114-228 (21 subjects, 3 to less than 6 years of age) [see Adverse Reactions (6.1) , Clinical Pharmacology (12.3)and Clinical Studies (14.4)] . Frequency, type, and severity of adverse drug reactions in pediatric subjects were comparable to those observed in adults [see Adverse Reactions (6.1)] . Refer to Dosage and Administration (2.5)for twice-daily dosing recommendations for pediatric subjects 3 to less than 18 years of age and weighing at least 10 kg.
In clinical trial TMC114-C230, the safety, pharmacokinetic profile and virologic and immunologic responses of PREZISTA/ritonavir administered once daily were evaluated in treatment-naïve HIV-1 infected pediatric subjects 12 to less than 18 years of age (12 subjects) [see Adverse Reactions (6.1) , Clinical Pharmacology (12.3)and Clinical Studies (14.4)] . Frequency, type, and severity of adverse drug reactions in pediatric subjects were comparable to those observed in adults [see Adverse Reactions (6.1)] . Once daily dosing recommendations for pediatric patients 3 to less than 12 years of age were derived using population pharmacokinetic modeling and simulation. Although a PREZISTA/ritonavir once daily dosing pediatric trial was not conducted in children less than 12 years of age, there is sufficient clinical safety data to support the predicted PREZISTA exposures for the dosing recommendations in this age group [see Clinical Pharmacology (12.3)] . Please see Dosage and Administration (2.5) for once-daily dosing recommendations for pediatric subjects 3 to less than 18 years of age and weighing at least 10 kg.
8.5 Geriatric Use
Clinical studies of PREZISTA did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently from younger patients. In general, caution should be exercised in the administration and monitoring of PREZISTA in elderly patients, reflecting the greater frequency of decreased hepatic function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
5.2 Hepatotoxicity
Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) has been reported with PREZISTA/ritonavir. During the clinical development program (N=3063), hepatitis was reported in 0.5% of patients receiving combination therapy with PREZISTA/ritonavir. Patients with pre-existing liver dysfunction, including chronic active hepatitis B or C, have an increased risk for liver function abnormalities including severe hepatic adverse events.
Post-marketing cases of liver injury, including some fatalities, have been reported. These have generally occurred in patients with advanced HIV-1 disease taking multiple concomitant medications, having co-morbidities including hepatitis B or C co-infection, and/or developing immune reconstitution syndrome. A causal relationship with PREZISTA/ritonavir therapy has not been established.
Appropriate laboratory testing should be conducted prior to initiating therapy with PREZISTA/ritonavir and patients should be monitored during treatment. Increased AST/ALT monitoring should be considered in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases, especially during the first several months of PREZISTA/ritonavir treatment.
Evidence of new or worsening liver dysfunction (including clinically significant elevation of liver enzymes and/or symptoms such as fatigue, anorexia, nausea, jaundice, dark urine, liver tenderness, hepatomegaly) in patients on PREZISTA/ritonavir should prompt consideration of interruption or discontinuation of treatment.
4 Contraindications
Co-administration of PREZISTA/ritonavir is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Examples of these drugs and other contraindicated drugs (which may lead to reduced efficacy of darunavir) are listed below [see Drug Interactions (7.3)] . Due to the need for co-administration of PREZISTA with ritonavir, please refer to ritonavir prescribing information for a description of ritonavir contraindications.
- Alpha 1-adrenoreceptor antagonist: alfuzosin
- Anti-gout: colchicine, in patients with renal and/or hepatic impairment
- Antimycobacterial: rifampin
- Antipsychotics: lurasidone, pimozide
- Cardiac Disorders: dronedarone, ivabradine, ranolazine
- Ergot derivatives, e.g. dihydroergotamine, ergotamine, methylergonovine
- Herbal product: St. John's wort ( Hypericum perforatum)
- Hepatitis C direct acting antiviral: elbasvir/grazoprevir
- Lipid modifying agents: lomitapide, lovastatin, simvastatin
- Opioid Antagonist: naloxegol
- PDE-5 inhibitor: sildenafil when used for treatment of pulmonary arterial hypertension
- Sedatives/hypnotics: orally administered midazolam, triazolam
6 Adverse Reactions
The following adverse reactions are discussed in other sections of labeling:
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Severe Skin Reactions [see Warnings and Precautions (5.3)]
- Diabetes Mellitus/Hyperglycemia [see Warnings and Precautions (5.6)]
- Fat Redistribution [see Warnings and Precautions (5.7)]
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.8)]
- Hemophilia [see Warnings and Precautions (5.9)]
Due to the need for co-administration of PREZISTA with ritonavir, please refer to ritonavir prescribing information for ritonavir-associated adverse reactions.
7 Drug Interactions
8.7 Renal Impairment
Population pharmacokinetic analysis showed that the pharmacokinetics of darunavir were not significantly affected in HIV-infected subjects with moderate renal impairment (CrCL between 30–60 mL/min, n=20). No pharmacokinetic data are available in HIV-1-infected patients with severe renal impairment or end stage renal disease; however, because the renal clearance of darunavir is limited, a decrease in total body clearance is not expected in patients with renal impairment. As darunavir and ritonavir are highly bound to plasma proteins, it is unlikely that they will be significantly removed by hemodialysis or peritoneal dialysis [see Clinical Pharmacology (12.3)] .
Instructions for Use
Be sure that you read, understand, and follow these Instructions for Use so that you measure and take PREZISTA oral suspension correctly. Ask your healthcare provider if you are not sure.
Each PREZISTA oral suspension carton contains:
|
Important information for use:
- Shake PREZISTA oral suspension well before each use.
- PREZISTA oral suspension should be given with the oral dosing syringe provided to make sure you measure the right amount. The oral dosing syringe provided with your PREZISTA oral suspension should not be used for any other medicines.
|
Figure A: Shake the bottle
|
|
|
Figure B: Opening the bottle
|
|
|
Figure C: Inserting the adapter
|
|
|
Figure D: Inserting the syringe
|
|
|
Figure E: Withdrawing the Oral Suspension
|
|
|
Figure F: Removing the syringe
|
|
If you or your child's dose is more than 6 mL you will need to divide the dose. Follow the instructions given to you by your healthcare provider or pharmacist about how to divide the dose, and repeat steps 4 through 7. |
Figure G: Taking the dose of PREZISTA
|
|
|
Figure H: Closing the bottle
|
|
|
Figure I: Removing the plunger from the barrel
|
|
|
Figure J: Putting the syringe back together
|
How should I store PREZISTA?
- Store PREZISTA oral suspension and the oral dosing syringe at room temperature 77°F (25°C).
- Do not refrigerate or freeze PREZISTA oral suspension.
- Keep PREZISTA oral suspension away from high heat.
- Store PREZISTA oral suspension in the original container.
Keep PREZISTA and all medicines out of the reach of children.
This Instruction for Use has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Janssen Products, LP, Horsham PA 19044
Revised: May 2022
©2006 Janssen Pharmaceutical Companies
5.7 Fat Redistribution
Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
8.6 Hepatic Impairment
No dosage adjustment of PREZISTA/ritonavir is necessary for patients with either mild or moderate hepatic impairment. No pharmacokinetic or safety data are available regarding the use of PREZISTA/ritonavir in subjects with severe hepatic impairment. Therefore, PREZISTA/ritonavir is not recommended for use in patients with severe hepatic impairment [see Dosage and Administration (2.6)and Clinical Pharmacology (12.3)] .
1 Indications and Usage
PREZISTA, co-administered with ritonavir (PREZISTA/ritonavir), in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus (HIV-1) infection in adult and pediatric patients 3 years of age and older [see Use in Specific Populations (8.4)and Clinical Studies (14)] .
14.4 Pediatric Patients
The pharmacokinetic profile, safety and antiviral activity of PREZISTA/ritonavir were evaluated in 3 randomized, open-label, multicenter studies.
12.1 Mechanism of Action
Darunavir is an HIV-1 antiviral drug [see Microbiology (12.4)] .
5.3 Severe Skin Reactions
During the clinical development program (n=3063), severe skin reactions, accompanied by fever and/or elevations of transaminases in some cases, have been reported in 0.4% of subjects. Stevens-Johnson Syndrome was rarely (less than 0.1%) reported during the clinical development program. During post-marketing experience toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis have been reported. Discontinue PREZISTA/ritonavir immediately if signs or symptoms of severe skin reactions develop. These can include but are not limited to severe rash or rash accompanied with fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, hepatitis and/or eosinophilia.
Rash (all grades, regardless of causality) occurred in 10.3% of subjects treated with PREZISTA/ritonavir [see Adverse Reactions (6)] . Rash was mostly mild-to-moderate, often occurring within the first four weeks of treatment and resolving with continued dosing. The discontinuation rate due to rash in subjects using PREZISTA/ritonavir was 0.5%.
Rash occurred more commonly in treatment-experienced subjects receiving regimens containing PREZISTA/ritonavir + raltegravir compared to subjects receiving PREZISTA/ritonavir without raltegravir or raltegravir without PREZISTA/ritonavir. However, rash that was considered drug related occurred at similar rates for all three groups. These rashes were mild to moderate in severity and did not limit therapy; there were no discontinuations due to rash.
5 Warnings and Precautions
- Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) has been reported with PREZISTA/ritonavir. Monitor liver function before and during therapy, especially in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases. Post-marketing cases of liver injury, including some fatalities, have been reported. ( 5.2)
- Skin reactions ranging from mild to severe, including Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms and acute generalized exanthematous pustulosis, have been reported. Discontinue treatment if severe reaction develops. ( 5.3)
- Use with caution in patients with a known sulfonamide allergy. ( 5.4)
- Patients may develop new onset diabetes mellitus or hyperglycemia. Initiation or dose adjustments of insulin or oral hypoglycemic agents may be required. ( 5.6)
- Patients may develop redistribution/accumulation of body fat or immune reconstitution syndrome. ( 5.7, 5.8)
- Patients with hemophilia may develop increased bleeding events. ( 5.9)
- PREZISTA/ritonavir is not recommended in pediatric patients below 3 years of age in view of toxicity and mortality observed in juvenile rats dosed with darunavir up to days 23 to 26 of age. ( 5.10)
2 Dosage and Administration
- Testing:
- In treatment-experienced patients, treatment history genotypic and/or phenotypic testing is recommended prior to initiation of therapy with PREZISTA/ritonavir to assess drug susceptibility of the HIV-1 virus ( 2.1, 12.4)
- Monitor serum liver chemistry tests before and during therapy with PREZISTA/ritonavir. ( 2.1, 2.2, 5.2)
- Treatment-naïve adult patients and treatment-experienced adult patients with no darunavir resistance associated substitutions: 800 mg (one 800 mg tablet) taken with ritonavir 100 mg once daily and with food. ( 2.3)
- Treatment-experienced adult patients with at least one darunavir resistance associated substitution: 600 mg (one 600 mg tablet) taken with ritonavir 100 mg twice daily and with food. ( 2.3)
- Pregnant patients: 600 mg (one 600 mg tablet) taken with ritonavir 100 mg twice daily and with food. ( 2.4)
- Pediatric patients (3 to less than 18 years of age and weighing at least 10 kg): dosage of PREZISTA and ritonavir is based on body weight and should not exceed the adult dose. PREZISTA should be taken with ritonavir and with food. ( 2.5)
- PREZISTA/ritonavir is not recommended for use in patients with severe hepatic impairment. ( 2.6)
3 Dosage Forms and Strengths
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of PREZISTA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Metabolism and Nutrition Disorders:Redistribution of body fat
Musculoskeletal and Connective Tissue Disorders:Rhabdomyolysis (associated with co-administration with HMG-CoA reductase inhibitors and PREZISTA/ritonavir)
Skin and Subcutaneous Tissue Disorders:Toxic epidermal necrolysis, acute generalized exanthematous pustulosis, drug rash with eosinophilia and systemic symptoms [see Warnings and Precautions (5.3)]
Renal and Urinary Disorders:Crystal nephropathy, crystalluria
8 Use in Specific Populations
- Pregnancy: Total darunavir exposures were generally lower during pregnancy compared to postpartum period. The reduction in darunavir exposures during pregnancy were greater for once daily dosing compared to the twice daily dosing regimen. ( 8.1, 12.3)
- Lactation: Women infected with HIV should be instructed not to breastfeed due to the potential for HIV transmission. ( 8.2)
- Pediatrics: Not recommended for patients less than 3 years of age. ( 8.4)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instruction for Use).
5.8 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including PREZISTA. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium aviuminfection, cytomegalovirus, Pneumocystis jiroveciipneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of antiretroviral treatment.
5.6 Diabetes Mellitus/hyperglycemia
New onset diabetes mellitus, exacerbation of pre-existing diabetes mellitus, and hyperglycemia have been reported during postmarketing surveillance in HIV-infected patients receiving protease inhibitor (PI) therapy. Some patients required either initiation or dose adjustments of insulin or oral hypoglycemic agents for treatment of these events. In some cases, diabetic ketoacidosis has occurred. In those patients who discontinued PI therapy, hyperglycemia persisted in some cases. Because these events have been reported voluntarily during clinical practice, estimates of frequency cannot be made and causal relationships between PI therapy and these events have not been established.
16 How Supplied/storage and Handling
PREZISTA ®(darunavir) 100 mg per mL oral suspension is a white to off-white opaque liquid supplied in amber-colored multiple-dose bottles containing 100 mg of darunavir per mL packaged with a 6 mL oral dosing syringe with 0.2 mL gradations.
PREZISTA ®(darunavir) 75 mg tablets are supplied as white, caplet-shaped, film-coated tablets debossed with "75" on one side and "TMC" on the other side.
PREZISTA ®(darunavir) 150 mg tablets are supplied as white, oval-shaped, film-coated tablets debossed with "150" on one side and "TMC" on the other side.
PREZISTA ®(darunavir) 600 mg tablets are supplied as orange, oval-shaped, film-coated tablets debossed with "600MG" on one side and "TMC" on the other side.
PREZISTA ®(darunavir) 800 mg tablets are supplied as dark red, oval-shaped, film-coated tablets debossed with "800" on one side and "T" on the other side.
PREZISTA is packaged in bottles in the following configuration:
- 100 mg/mL oral suspension – 200 mL bottles (NDC 59676-565-01)
- 75 mg tablets — bottles of 480 (NDC 59676-563-01)
- 150 mg tablets — bottles of 240 (NDC 59676-564-01)
- 600 mg tablets — bottles of 60 (NDC 59676-562-01)
- 800 mg tablets — bottles of 30 (NDC 59676-566-30)
2.4 Recommended Dosage During Pregnancy
The recommended dosage in pregnant patients is PREZISTA 600 mg taken with ritonavir 100 mg twice daily with food.
PREZISTA 800 mg taken with ritonavir 100 mg once daily should only be considered in certain pregnant patients who are already on a stable PREZISTA 800 mg with ritonavir 100 mg once daily regimen prior to pregnancy, are virologically suppressed (HIV-1 RNA less than 50 copies per mL), and in whom a change to twice daily PREZISTA 600 mg with ritonavir 100 mg may compromise tolerability or compliance.
2.3 Recommended Dosage in Adult Patients
PREZISTA must be co-administered with ritonavir to exert its therapeutic effect. Failure to correctly co-administer PREZISTA with ritonavir will result in plasma levels of darunavir that will be insufficient to achieve the desired antiviral effect and will alter some drug interactions.
Patients who have difficulty swallowing PREZISTA tablets can use the 100 mg per mL PREZISTA oral suspension.
14.1 Description of Adult Clinical Trials
The evidence of efficacy of PREZISTA/ritonavir is based on the analyses of 192-week data from a randomized, controlled open-label Phase 3 trial in treatment-naïve (TMC114-C211) HIV-1-infected adult subjects and 96-week data from a randomized, controlled, open-label Phase 3 trial in antiretroviral treatment-experienced (TMC114-C214) HIV-1-infected adult subjects. In addition, 96-week data are included from 2 randomized, controlled Phase 2b trials, TMC114-C213 and TMC114-C202, in antiretroviral treatment-experienced HIV-1-infected adult subjects.
Principal Display Panel 200 Ml Bottle Carton
NDC 59676- 565-01
PREZISTA
®
(darunavir) Oral Suspension
100 mg per mL
DISPENSING:For information concerning
proper usage of the oral dosing syringe,
see accompanying PATIENT INFORMATION
and INSTRUCTIONS FOR USE.
ALERT
Find out about medicines
that should NOT be taken
with PREZISTA.
Rx only
200 mL
Johnson
&Johnson
7.2 Potential for Other Drugs to Affect Darunavir
Darunavir and ritonavir are metabolized by CYP3A. In vitrodata indicate that darunavir may be a P-gp substrate. Drugs that induce CYP3A activity would be expected to increase the clearance of darunavir and ritonavir, resulting in lowered plasma concentrations of darunavir and ritonavir. Co-administration of darunavir and ritonavir and other drugs that inhibit CYP3A, or P-gp may decrease the clearance of darunavir and ritonavir and may result in increased plasma concentrations of darunavir and ritonavir (see Table 10).
5.1 Importance of Co Administration With Ritonavir
PREZISTA must be co-administered with ritonavir and food to achieve the desired antiviral effect. Failure to administer PREZISTA with ritonavir and food may result in a loss of efficacy of darunavir.
Please refer to ritonavir prescribing information for additional information on precautionary measures.
Principal Display Panel 75 Mg Tablet Bottle Label
NDC 59676- 563-01
PREZISTA
®
(darunavir) tablets
75 mg
ALERT
Find out about medicines that
should NOT be taken with PREZISTA
Rx only
480 Tablets
Johnson
&Johnson
Principal Display Panel 150 Mg Tablet Bottle Label
NDC 59676- 564-01
PREZISTA
®
(darunavir) tablets
150 mg
ALERT
Find out about medicines that
should NOT be taken with PREZISTA.
Rx only
240 Tablets
Johnson
&Johnson
Principal Display Panel 600 Mg Tablet Bottle Label
NDC 59676- 562-01
PREZISTA
®
(darunavir) tablets
600 mg
ALERT
Find out about medicines that
should NOT be taken with PREZISTA.
Rx only
60 Tablets
Johnson
&Johnson
Principal Display Panel 800 Mg Tablet Bottle Label
NDC 59676- 566-30
PREZISTA
®
(darunavir) tablets
800 mg
ALERT
Find out about medicines that
should NOT be taken with PREZISTA.
Rx only
30 Tablets
Johnson
&Johnson
2.1 Testing Prior to Initiation of Prezista/ritonavir
In treatment-experienced patients, treatment history, genotypic and/or phenotypic testing is recommended to assess drug susceptibility of the HIV-1 virus [see Microbiology (12.4)]. Refer to Dosage and Administration (2.3), (2.4)and (2.5) for dosing recommendations.
Appropriate laboratory testing such as serum liver biochemistries should be conducted prior to initiating therapy with PREZISTA/ritonavir [see Warnings and Precautions (5.2)] .
2.2 Monitoring During Treatment With Prezista/ritonavir
Patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases should be monitored for elevation in serum liver biochemistries, especially during the first several months of PREZISTA/ritonavir treatment [see Warnings and Precautions (5.2)] .
7.1 Potential for Prezista/ritonavir to Affect Other Drugs
PREZISTA co-administered with ritonavir is an inhibitor of CYP3A, CYP2D6, and P-gp. Co-administration of PREZISTA and ritonavir with drugs that are primarily metabolized by CYP3A and CYP2D6 or are transported by P-gp may result in increased plasma concentrations of such drugs, which could increase or prolong their therapeutic effect and adverse events. PREZISTA co-administered with ritonavir with drugs that have active metabolite(s) formed by CYP3A may result in reduced plasma concentrations of these active metabolite(s), potentially leading to loss of their therapeutic effect (see Table 10).
2.6 Not Recommended in Patients With Severe Hepatic Impairment
No dosage adjustment is required in patients with mild or moderate hepatic impairment. No data are available regarding the use of PREZISTA/ritonavir when co-administered to subjects with severe hepatic impairment; therefore, PREZISTA/ritonavir is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)] .
5.5 Risk of Serious Adverse Reactions Due to Drug Interactions
Initiation of PREZISTA/ritonavir, a CYP3A inhibitor, in patients receiving medications metabolized by CYP3A or initiation of medications metabolized by CYP3A in patients already receiving PREZISTA/ritonavir, may increase plasma concentrations of medications metabolized by CYP3A and reduce plasma concentrations of active metabolite(s) formed by CYP3A.
Initiation of medications that inhibit or induce CYP3A may increase or decrease concentrations of PREZISTA/ritonavir, respectively.
These interactions may lead to:
- Clinically significant adverse reactions, potentially leading to severe, life threatening, or fatal events from greater exposures of concomitant medications.
- Clinically significant adverse reactions from greater exposures of PREZISTA/ritonavir.
- Loss of therapeutic effect of the concomitant medications from lower exposures of active metabolite(s).
- Loss of therapeutic effect of PREZISTA/ritonavir and possible development of resistance from lower exposures of PREZISTA/ritonavir.
See Table 10for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations [see Drug Interactions (7)] . Consider the potential for drug interactions prior to and during PREZISTA/ritonavir therapy; review concomitant medications during PREZISTA/ritonavir therapy; and monitor for the adverse reactions associated with the concomitant drugs [see Contraindications (4)and Drug Interactions (7)] .
5.10 Not Recommended in Pediatric Patients Below 3 Years of Age
PREZISTA/ritonavir in pediatric patients below 3 years of age is not recommended in view of toxicity and mortality observed in juvenile rats dosed with darunavir (from 20 mg/kg to 1000 mg/kg) up to days 23 to 26 of age [see Use in Specific Populations (8.1and 8.4)and Clinical Pharmacology (12.3)] .
7.3 Established and Other Potentially Significant Drug Interactions
Table 10 provides dosing recommendations as a result of drug interactions with PREZISTA/ritonavir. These recommendations are based on either drug interaction studies or predicted interactions due to the expected magnitude of interaction and potential for serious adverse events or loss of efficacy. The table includes examples of potentially significant interactions but is not all inclusive [see Contraindications (4)and Clinical Pharmacology (12.3)] , and therefore the label of each drug that is co-administered with PREZISTA/ritonavir should be consulted for information related to the route of metabolism, interaction pathways, potential risks, and specific actions to be taken with regard to co-administration.
| Concomitant Drug Class
Drug Name Examples |
Effect on Concentration of Darunavir Or Concomitant Drug | Clinical Comment |
|---|---|---|
| HIV-1-Antiviral Agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs) | ||
| didanosine | ↔ darunavir
↔ didanosine |
Didanosine should be administered one hour before or two hours after PREZISTA/ritonavir (which are administered with food). |
| HIV-1-Antiviral Agents: HIV-Protease Inhibitors (PIs) | ||
| indinavir | ↑ darunavir
↑ indinavir |
The appropriate dose of indinavir in combination with PREZISTA/ritonavir has not been established. |
| (The reference regimen for indinavir was indinavir/ritonavir 800/100 mg twice daily.) | ||
| lopinavir/ritonavir | ↓ darunavir
↔ lopinavir |
Appropriate doses of the combination have not been established. Hence, it is not recommended to co-administer lopinavir/ritonavir and PREZISTA, with or without ritonavir. |
| saquinavir | ↓ darunavir
↔ saquinavir |
Appropriate doses of the combination have not been established. Hence, it is not recommended to co-administer saquinavir and PREZISTA, with or without ritonavir. |
| Other HIV protease inhibitors, except atazanavir [see Drug Interactions (7.4)] | As co-administration with PREZISTA/ritonavir has not been studied, co-administration is not recommended. | |
| HIV-1-Antiviral Agents: CCR5 co-receptor antagonists | ||
| maraviroc | ↑ maraviroc | When used in combination with PREZISTA/ritonavir, the dose of maraviroc should be 150 mg twice daily. |
| Other Agents | ||
| Alpha 1-adrenoreceptor antagonist: | ||
| alfuzosin | ↑ alfuzosin | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as hypotension. |
|
Antibacterial:
clarithromycin |
↔ darunavir ↑ clarithromycin |
No dose adjustment of the combination is required for patients with normal renal function. For co-administration of clarithromycin and PREZISTA/ritonavir in patients with renal impairment, the following dose adjustments should be considered:
|
|
Anticoagulants:
Direct Oral Anticoagulants (DOACs) |
||
| apixaban | ↑ apixaban | Due to potentially increased bleeding risk, dosing recommendations for co-administration of apixaban with PREZISTA/ritonavir depend on the apixaban dose. Refer to apixaban dosing instructions for co-administration with P-gp and strong CYP3A inhibitors in apixaban prescribing information. |
| rivaroxaban | ↑ rivaroxaban | Co-administration of PREZISTA/ritonavir and rivaroxaban is not recommended because it may lead to an increased bleeding risk. |
| dabigatran etexilate
edoxaban |
↑ dabigatran
↑ edoxaban |
Refer to the dabigatran etexilate or edoxaban prescribing information for recommendations regarding co-administration. The specific recommendations are based on indication, renal function, and effect of the co-administered P-gp inhibitors on the concentration of dabigatran or edoxaban. Clinical monitoring is recommended when a DOAC not affected by CYP3A4 but transported by P-gp, including dabigatran etexilate and edoxaban, is co-administered with PREZISTA/ritonavir. |
| Other Anticoagulants | ||
| warfarin | ↓ warfarin
↔ darunavir |
Warfarin concentrations are decreased when co-administered with PREZISTA/ritonavir. It is recommended that the international normalized ratio (INR) be monitored when warfarin is combined with PREZISTA/ritonavir. |
|
Anticonvulsants:
carbamazepine |
↔ darunavir ↑ carbamazepine |
The dose of either PREZISTA/ritonavir or carbamazepine does not need to be adjusted when initiating co-administration with PREZISTA/ritonavir and carbamazepine. Clinical monitoring of carbamazepine concentrations and its dose titration is recommended to achieve the desired clinical response. |
| clonazepam | ↑ clonazepam | Clinical monitoring of anticonvulsants that are metabolized by CYP3A is recommended. |
| phenobarbital, phenytoin | ↔ darunavir
↓ phenytoin ↓ phenobarbital |
Phenytoin and phenobarbital levels should be monitored when co-administering with PREZISTA/ritonavir. |
|
Antidepressants:
Selective Serotonin Reuptake Inhibitors (SSRIs): |
||
| paroxetine, sertraline | ↓ paroxetine
↓ sertraline |
If either sertraline or paroxetine is initiated in patients receiving PREZISTA/ritonavir, dose titrating the SSRI based on a clinical assessment of antidepressant response is recommended. Monitor for antidepressant response in patients on a stable dose of sertraline or paroxetine who start treatment with PREZISTA/ritonavir. |
| Tricyclic Antidepressants (TCAs): | ||
| amitriptyline, desipramine, imipramine, nortriptyline | ↑ amitriptyline
↑ desipramine ↑ imipramine ↑ nortriptyline |
Use a lower dose of the tricyclic antidepressants and trazodone due to potential increased adverse events such as nausea, dizziness, hypotension and syncope. |
| Other:trazodone | ↑ trazodone | |
|
Antifungals:
itraconazole, isavuconazole, ketoconazole, posaconazole |
↑ darunavir ↑ itraconazole ↑ isavuconazole ↑ ketoconazole ↔ posaconazole |
Monitor for increased PREZISTA/ritonavir and/or antifungal adverse events with concomitant use of these antifungals. When co-administration is required, the daily dose of ketoconazole or itraconazole should not exceed 200 mg with monitoring for increased antifungal adverse events. |
| voriconazole | ↓ voriconazole | Voriconazole is not recommended for patients receiving PREZISTA/ritonavir unless an assessment comparing predicted benefit to risk ratio justifies the use of voriconazole. |
|
Anti-gout:
colchicine |
↑ colchicine |
Co-administration is contraindicated in patients with renal and/or hepatic impairment due to potential for serious and/or life-threatening reactions. For patients without renal or hepatic impairment:
|
|
Antimalarial:
artemether/lumefantrine |
↓ artemether ↓ dihydroartemisinin ↑ lumefantrine ↔ darunavir |
The combination of PREZISTA/ritonavir and artemether/lumefantrine can be used without dose adjustments. However, the combination should be used with caution as increased lumefantrine exposure may increase the risk of QT prolongation. |
| Antimycobacterials: | ||
| rifampin | ↓ darunavir | Co-administration is contraindicated due to potential for loss of therapeutic effect and development of resistance. |
| rifabutin
(The reference regimen for rifabutin was 300 mg once daily.) |
↑ darunavir
↑ rifabutin ↑ 25- O-desacetylrifabutin |
Dose reduction of rifabutin by at least 75% of the usual dose (300 mg once daily) is recommended (i.e., a maximum dose of 150 mg every other day). Increased monitoring for adverse events is warranted in patients receiving this combination and further dose reduction of rifabutin may be necessary. |
| rifapentine | ↓ darunavir | Co-administration of PREZISTA/ritonavir with rifapentine is not recommended. |
|
Antineoplastics:
dasatinib, nilotinib |
↑ antineoplastics |
A decrease in the dosage or an adjustment of the dosing interval of dasatinib and nilotinib may be necessary for patients. Please refer to the dasatinib and nilotinib prescribing information for dosing instructions. |
| vinblastine, vincristine | For vincristine and vinblastine, consideration should be given to temporarily withholding the ritonavir-containing antiretroviral regimen in patients who develop significant hematologic or gastrointestinal side effects when PREZISTA/ritonavir is administered concurrently with vincristine or vinblastine. If the antiretroviral regimen must be withheld for a prolonged period, consideration should be given to initiating a revised regimen that does not include a CYP3A or P-gp inhibitor. | |
| Antipsychotics: | ||
| lurasidone | ↑ lurasidone | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. |
| pimozide | ↑ pimozide | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. |
| quetiapine | ↑ quetiapine |
Initiation of PREZISTA with ritonavir in patients taking quetiapine:
Consider alternative antiretroviral therapy to avoid increases in quetiapine exposures. If co-administration is necessary, reduce the quetiapine dose to 1/6 of the current dose and monitor for quetiapine-associated adverse reactions. Refer to the quetiapine prescribing information for recommendations on adverse reaction monitoring. Initiation of quetiapine in patients taking PREZISTA with ritonavir: Refer to the quetiapine prescribing information for initial dosing and titration of quetiapine. |
| e.g. perphenazine, risperidone, thioridazine | ↑ antipsychotics | A decrease in the dose of antipsychotics that are metabolized by CYP3A or CYP2D6 may be needed when co-administered with PREZISTA/ritonavir. |
|
β-Blockers:
e.g. carvedilol, metoprolol, timolol |
↑ beta-blockers |
Clinical monitoring of patients is recommended. A dose decrease may be needed for these drugs when co-administered with PREZISTA/ritonavir and a lower dose of the beta blocker should be considered. |
|
Calcium Channel Blockers:
amlodipine, diltiazem, felodipine, nicardipine, nifedipine, verapamil |
↑ calcium channel blockers |
Clinical monitoring of patients is recommended. |
| Cardiac Disorders: | ||
| ranolazine, ivabradine | ↑ ranolazine
↑ ivabradine |
Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. |
| dronedarone | ↑ dronedarone | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. |
|
Other antiarrhythmics
e.g. amiodarone, bepridil, disopyramide, flecainide, lidocaine (systemic), mexiletine, propafenone, quinidine |
↑ antiarrhythmics | Therapeutic concentration monitoring, if available, is recommended for antiarrhythmics when co-administered with PREZISTA/ritonavir. |
| digoxin | ↑ digoxin | The lowest dose of digoxin should initially be prescribed. The serum digoxin concentrations should be monitored and used for titration of digoxin dose to obtain the desired clinical effect. |
| Corticosteroids: | ||
| dexamethasone (systemic) | ↓ darunavir | Co-administration of PREZISTA/ritonavir with systemic dexamethasone or other systemic corticosteroids that induce CYP3A may result in loss of therapeutic effect and development of resistance to darunavir. Consider alternative corticosteroids. |
| Corticosteroids primarily metabolized by CYP3A:
e.g. betamethasone budesonide ciclesonide fluticasone methylprednisolone mometasone triamcinolone |
↑ corticosteroids | Co-administration with corticosteroids (all routes of administration) of which exposures are significantly increased by strong CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression.
Alternative corticosteroids including beclomethasone, prednisone, and prednisolone (for which PK and/or PD are less affected by strong CYP3A inhibitors relative to other steroids) should be considered, particularly for long term use. |
| Endothelin receptor antagonist: | ||
| bosentan | ↑ bosentan |
Co-administration of bosentan in patients on PREZISTA/ritonavir:
Co-administration of PREZISTA/ritonavir in patients on bosentan:
|
| Ergot derivatives: | ||
| e.g. dihydroergotamine, ergotamine, methylergonovine | ↑ ergot derivatives | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. |
| Hepatitis C virus (HCV): | ||
|
Direct-Acting Antivirals:
elbasvir/grazoprevir |
↑ elbasvir/grazoprevir | Co-administration is contraindicated due to potential for the increased risk of alanine transaminase (ALT) elevations. |
| glecaprevir/pibrentasvir | ↑ glecaprevir
↑ pibrentasvir |
Co-administration of PREZISTA/ritonavir with glecaprevir/pibrentasvir is not recommended. |
| Herbal product: | ||
| St. John's wort ( Hypericum perforatum) | ↓ darunavir | Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. |
| Hormonal contraceptives: | Effective alternative (non-hormonal) contraceptive method or a barrier method of contraception is recommended [see Use in Specific Populations (8.3)] . | |
| ethinyl estradiol, norethindrone, drospirenone | ↓ ethinyl estradiol
↓ norethindrone drospirenone: effects unknown |
For co-administration with drospirenone, clinical monitoring is recommended due to the potential for hyperkalemia.
No data are available to make recommendations on co-administration with other hormonal contraceptives. |
|
Immunosuppressants:
e.g. cyclosporine, tacrolimus, sirolimus |
↑ immunosuppressants | Therapeutic concentration monitoring of the immunosuppressive agent is recommended when co-administered with PREZISTA/ritonavir. |
|
Immunosuppressant/neoplastic:
everolimus |
Co-administration of everolimus and PREZISTA/ritonavir is not recommended. | |
| irinotecan | Discontinue PREZISTA/ritonavir at least 1 week prior to starting irinotecan therapy. Do not administer PREZISTA/ritonavir with irinotecan unless there are no therapeutic alternatives. | |
| Inhaled beta agonist: | ||
| salmeterol | ↑ salmeterol | Co-administration of salmeterol and PREZISTA/ritonavir is not recommended. The combination may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations and sinus tachycardia. |
| Lipid Modifying Agents: | ||
| HMG-CoA reductase inhibitors: | ||
| lovastatin, simvastatin | ↑ lovastatin
↑ simvastatin |
Co-administration is contraindicated due to potential for serious reactions such as myopathy including rhabdomyolysis. |
| atorvastatin, pravastatin, rosuvastatin | ↑ HMG-CoA reductase inhibitors | Co-administration of PREZISTA/ritonavir with HMG-Co A reductase inhibitors may lead to adverse events such as myopathy. Titrate atorvastatin, pravastatin or rosuvastatin dose carefully and use the lowest necessary dose while monitoring for adverse events. Do not exceed atorvastatin 20 mg/day. |
| Other lipid modifying agents: | ||
| lomitapide | ↑ lomitapide | Co-administration is contraindicated due to potential for markedly increased transaminases. |
| Narcotic analgesics metabolized by CYP3A: | ||
| e.g. fentanyl, oxycodone | ↑ fentanyl
↑ oxycodone |
Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. |
| tramadol | ↑ tramadol | A dose decrease may be needed for tramadol with concomitant use. |
|
Narcotic analgesics/treatment of opioid dependence:
buprenorphine, buprenorphine/naloxone |
↔ buprenorphine, naloxone ↑ norbuprenorphine (metabolite) |
No dose adjustment for buprenorphine or buprenorphine/naloxone is required with concurrent administration of PREZISTA/ritonavir. Clinical monitoring is recommended if PREZISTA/ritonavir and buprenorphine or buprenorphine/naloxone are co-administered. |
| methadone | ↓ methadone | No adjustment of methadone dosage is required when initiating co-administration of PREZISTA/ritonavir. However, clinical monitoring is recommended as the dose of methadone during maintenance therapy may need to be adjusted in some patients. |
| Opioid Antagonist | ||
| naloxegol | ↑ naloxegol | Co-administration of PREZISTA/ritonavir and naloxegol is contraindicated due to potential for precipitating opioid withdrawal symptoms. |
|
PDE-5 inhibitors:
e.g. avanafil, sildenafil, tadalafil, vardenafil |
↑ PDE-5 inhibitors (only the use of sildenafil at doses used for treatment of erectile dysfunction has been studied with PREZISTA/ritonavir) |
Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Co-administration with sildenafil used for PAH is contraindicated due to potential for sildenafil associated adverse reactions (which include visual disturbances, hypotension, prolonged erection, and syncope).
Use of PDE-5 inhibitors for erectile dysfunction:
Co-administration of PREZISTA/ritonavir and avanafil is not recommended. |
|
Platelet aggregation inhibitor:
ticagrelor |
↑ ticagrelor |
Co-administration of PREZISTA/ritonavir and ticagrelor is not recommended. |
| clopidogrel | ↓ clopidogrel active metabolite | Co-administration of PREZISTA/ritonavir and clopidogrel is not recommended due to potential reduction of the antiplatelet activity of clopidogrel. |
| prasugrel | ↔ prasugrel active metabolite | No dose adjustment is needed when prasugrel is co-administered with PREZISTA/ritonavir. |
|
Proton pump inhibitor:
omeprazole |
↓ omeprazole ↔ darunavir |
When omeprazole is co-administered with PREZISTA/ritonavir, monitor patients for decreased efficacy of omeprazole. Consider increasing the omeprazole dose in patients whose symptoms are not well controlled; avoid use of more than 40 mg per day of omeprazole. |
| Sedatives/hypnotics: | ||
| orally administered midazolam, triazolam | ↑ midazolam
↑ triazolam |
Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as prolonged or increased sedation or respiratory depression. Triazolam and orally administered midazolam are extensively metabolized by CYP3A. Co-administration of triazolam or orally administered midazolam with PREZISTA may cause large increases in the concentrations of these benzodiazepines. |
| metabolized by CYP3A
e.g. buspirone, diazepam, estazolam, zolpidem |
↑ sedatives/hypnotics | Titration is recommended when co-administering PREZISTA/ritonavir with sedatives/hypnotics metabolized by CYP3A and a lower dose of the sedatives/hypnotics should be considered with monitoring for adverse events. |
| parenterally administered midazolam | Co-administration of parenteral midazolam should be done in a setting which ensures close clinical monitoring and appropriate medical management in case of respiratory depression and/or prolonged sedation. Dosage reduction for midazolam should be considered, especially if more than a single dose of midazolam is administered. | |
| Urinary antispasmodics | ||
| fesoterodine | ↑ fesoterodine | When fesoterodine is co-administered with PREZISTA/ritonavir, do not exceed a fesoterodine dose of 4 mg once daily. |
| solifenacin | ↑ solifenacin | When solifenacin is co-administered with PREZISTA/ritonavir, do not exceed a solifenacin dose of 5 mg once daily. |
7.4 Drugs Without Clinically Significant Interactions With Prezista
No dosage adjustments are recommended when PREZISTA/ritonavir is co-administered with the following medications: atazanavir, dolutegravir, efavirenz, etravirine, nevirapine, nucleoside reverse transcriptase inhibitors (abacavir, emtricitabine, emtricitabine/tenofovir alafenamide, lamivudine, stavudine, tenofovir disoproxil fumarate, zidovudine), pitavastatin, raltegravir, ranitidine, or rilpivirine.
2.5 Recommended Dosage in Pediatric Patients (age 3 to Less Than 18 Years)
Healthcare professionals should pay special attention to accurate dose selection of PREZISTA, transcription of the medication order, dispensing information and dosing instruction to minimize risk for medication errors, overdose, and underdose.
Prescribers should select the appropriate dose of PREZISTA/ritonavir for each individual child based on body weight (kg) and should not exceed the recommended dose for adults.
Before prescribing PREZISTA, children weighing greater than or equal to 15 kg should be assessed for the ability to swallow tablets. If a child is unable to reliably swallow a tablet, the use of PREZISTA oral suspension should be considered.
The recommended dose of PREZISTA/ritonavir for pediatric patients (3 to less than 18 years of age and weighing at least 10 kg is based on body weight (see Tables 2, 3, 4, and 5) and should not exceed the recommended adult dose. PREZISTA should be taken with ritonavir and with food.
The recommendations for the PREZISTA/ritonavir dosage regimens were based on pediatric clinical trial data and population pharmacokinetic modeling and simulation [see Use in Specific Populations (8.4)and Clinical Pharmacology (12.3)].
Structured Label Content
Section 42229-5 (42229-5)
Treatment-Naïve Adult Patients
The recommended oral dose of PREZISTA is 800 mg (one 800 mg tablet or 8 mL of the oral suspension) taken with ritonavir 100 mg (one 100 mg tablet or capsule or 1.25 mL of a 80 mg per mL ritonavir oral solution) once daily and with food. An 8 mL PREZISTA dose should be taken as two 4 mL administrations with the included oral dosing syringe.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised: 03/2023 | |
| PATIENT INFORMATION | ||
|
PREZISTA
®
(pre-ZIS-ta)
(darunavir) oral suspension |
PREZISTA
®
(pre-ZIS-ta)
(darunavir) tablet |
|
| Read this Patient Information before you start taking PREZISTA and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Also read the Patient Information leaflet for ritonavir. |
||
What is the most important information I should know about PREZISTA?
|
||
|
|
|
|
||
|
|
|
| Rash occurred more often in people taking PREZISTA and raltegravir together than with either drug separately, but was generally mild.
See " What are the possible side effects of PREZISTA?" for more information about side effects. |
||
|
What is PREZISTA?
PREZISTA is a prescription HIV-1 (Human Immunodeficiency Virus-type 1) medicine used with ritonavir and other antiretroviral medicines to treat HIV-1 infection in adults and children 3 years of age and older. HIV is the virus that causes AIDS (Acquired Immune Deficiency Syndrome). PREZISTA should not be used in children under 3 years of age. When used with other antiretroviral medicines to treat HIV-1 infection, PREZISTA may help:
Reducing the amount of HIV-1 and increasing the CD4+ (T) cells in your blood may improve your immune system. This may reduce your risk of death or getting infections that can happen when your immune system is weak (opportunistic infections).
Ask your healthcare provider if you have any questions on how to prevent passing HIV to other people. |
||
|
Who should not take PREZISTA?
Do not take PREZISTA with any medicine that contains:
Serious problems can happen if you or your child take any of these medicines with PREZISTA. This is not a complete list of medicines. Therefore, tell your healthcare provider about allmedicines you take. |
||
|
What should I tell my healthcare provider before taking PREZISTA?
Before taking PREZISTA, tell your healthcare provider if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, topical creams, vitamins, and herbal supplements. Some medicines interact with PREZISTA. Keep a list of your medicines to show your healthcare provider and pharmacist.
|
||
How should I take PREZISTA?
|
||
|
What are the possible side effects of PREZISTA?
PREZISTA may cause serious side effects, including:
The most common side effects of PREZISTA include: |
||
|
|
|
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all of the possible side effects of PREZISTA. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store PREZISTA?
Keep PREZISTA and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of PREZISTA.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use PREZISTA for a condition for which it was not prescribed. Do not give PREZISTA to other people even if they have the same condition you have. It may harm them. This leaflet summarizes the most important information about PREZISTA. If you would like more information, talk to your healthcare provider. You can ask your healthcare provider or pharmacist for information about PREZISTA that is written for health professionals. For more information, call 1-800-526-7736. |
||
|
What are the ingredients in PREZISTA?
Active ingredient: darunavir Inactive ingredients: PREZISTA oral suspension:citric acid monohydrate, hydrochloric acid (for pH adjustment), hydroxypropyl cellulose, masking flavor, methylparaben sodium, microcrystalline cellulose, purified water, sodium carboxymethylcellulose, strawberry cream flavor, and sucralose. PREZISTA 75 mg and 150 mg tablets:colloidal silicon dioxide, crospovidone, magnesium stearate, microcrystalline cellulose. The film coating contains: OPADRY ®White (polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, titanium dioxide). PREZISTA 600 mg tablets:colloidal silicon dioxide, crospovidone, magnesium stearate, microcrystalline cellulose. The film coating contains: OPADRY ®Orange (FD&C Yellow No. 6, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, titanium dioxide). PREZISTA 800 mg tablets:colloidal silicon dioxide, crospovidone, magnesium stearate, microcrystalline cellulose, hypromellose. The film coating contains: OPADRY ®Dark Red (iron oxide red, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, titanium dioxide). Manufactured for: Janssen Products, LP, Horsham PA 19044, USA PREZISTA ®is a registered trademark of Janssen Pharmaceuticals © 2006 Janssen Pharmaceutical Companies |
Section 43679-0 (43679-0)
Mechanism of Action
Darunavir is an inhibitor of the HIV-1 protease. It selectively inhibits the cleavage of HIV-1 encoded Gag-Pol polyproteins in infected cells, thereby preventing the formation of mature virus particles.
Section 43683-2 (43683-2)
| Contraindications ( 4) | 4/2022 |
Section 44425-7 (44425-7)
Storage
Section 59845-8 (59845-8)
Instructions for Use
Advise patients to take PREZISTA and ritonavir with food every day on a regular dosing schedule, as missed doses can result in development of resistance. PREZISTA must always be used with ritonavir in combination with other antiretroviral drugs. Advise patients not to alter the dose of either PREZISTA or ritonavir, discontinue ritonavir, or discontinue therapy with PREZISTA without consulting their physician [see Dosage and Administration (2)] .
10 Overdosage (10 OVERDOSAGE)
Human experience of acute overdose with PREZISTA/ritonavir is limited. No specific antidote is available for overdose with PREZISTA. Treatment of overdose with PREZISTA consists of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. Since PREZISTA is highly protein bound, dialysis is unlikely to be beneficial in significant removal of the active substance.
11 Description (11 DESCRIPTION)
PREZISTA (darunavir) is an inhibitor of the human immunodeficiency virus (HIV-1) protease.
PREZISTA tablets and oral suspension contain the active ingredient darunavir, (present as darunavir ethanolate) which has the following chemical name: [(1S,2R)-3-[[(4-aminophenyl)sulfonyl](2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]-carbamic acid (3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl ester monoethanolate. Its molecular formula is C 27H 37N 3O 7S ∙ C 2H 5OH and its molecular weight is 593.73. Darunavir ethanolate has the following structural formula:
Darunavir ethanolate is a white to off-white powder with a solubility of approximately 0.15 mg per mL in water at 20°C.
PREZISTA ®100 mg per mL oral suspension is available as a white to off-white opaque suspension for oral administration.
Each mL of the oral suspension contains darunavir 100 mg (present as darunavir ethanolate). In addition, each mL contains the inactive ingredients citric acid monohydrate, hydrochloric acid (for pH adjustment), hydroxypropyl cellulose, masking flavor, methylparaben sodium, microcrystalline cellulose, purified water, sodium carboxymethylcellulose, strawberry cream flavor and sucralose.
PREZISTA ®75 mg tablets are available as white, caplet-shaped, film-coated tablets for oral administration. Each 75 mg tablet contains darunavir 75 mg (present as darunavir ethanolate).
PREZISTA ®150 mg tablets are available as white, oval-shaped, film-coated tablets for oral administration. Each 150 mg tablet contains darunavir 150 mg (present as darunavir ethanolate).
PREZISTA ®600 mg tablets are available as orange, oval-shaped, film-coated tablets for oral administration. Each 600 mg tablet contains darunavir 600 mg (present as darunavir ethanolate).
PREZISTA ®800 mg tablets are available as dark red, oval-shaped, film-coated tablets for oral administration. Each 800 mg tablet contains darunavir 800 mg (present as darunavir ethanolate).
During storage, partial conversion from ethanolate to hydrate may occur; however, this does not affect product quality or performance. Each tablet also contains the inactive ingredients colloidal silicon dioxide, crospovidone, magnesium stearate, and microcrystalline cellulose. The 800 mg tablet also contains hypromellose. The 75 and 150 mg tablet film coating, OPADRY ®White, contains polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, and titanium dioxide. The 600 mg tablet film coating, OPADRY ®Orange, contains FD&C Yellow No. 6, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, and titanium dioxide. The 800 mg tablet film coating, OPADRY ®Dark Red, contains iron oxide red, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, and titanium dioxide.
All strengths for PREZISTA are expressed in terms of the free form of darunavir.
5.9 Hemophilia
There have been reports of increased bleeding, including spontaneous skin hematomas and hemarthrosis in patients with hemophilia type A and B treated with PIs. In some patients, additional factor VIII was given. In more than half of the reported cases, treatment with PIs was continued or reintroduced if treatment had been discontinued. A causal relationship between PI therapy and these episodes has not been established.
5.4 Sulfa Allergy
Darunavir contains a sulfonamide moiety. PREZISTA should be used with caution in patients with a known sulfonamide allergy. In clinical studies with PREZISTA/ritonavir, the incidence and severity of rash were similar in subjects with or without a history of sulfonamide allergy.
8.4 Pediatric Use
PREZISTA/ritonavir is not recommended in pediatric patients below 3 years of age because of toxicity and mortality observed in juvenile rats dosed with darunavir (from 20 mg/kg to 1000 mg/kg) up to days 23 to 26 of age [see Warnings and Precautions (5.10) , Use in Specific Populations (8.1)and Clinical Pharmacology (12.3)] .
The safety, pharmacokinetic profile, and virologic and immunologic responses of PREZISTA/ritonavir administered twice daily were evaluated in treatment-experienced HIV-1-infected pediatric subjects 3 to less than 18 years of age and weighting at least 10 kg. These subjects were evaluated in clinical trials TMC114-C212 (80 subjects, 6 to less than 18 years of age) and TMC114-228 (21 subjects, 3 to less than 6 years of age) [see Adverse Reactions (6.1) , Clinical Pharmacology (12.3)and Clinical Studies (14.4)] . Frequency, type, and severity of adverse drug reactions in pediatric subjects were comparable to those observed in adults [see Adverse Reactions (6.1)] . Refer to Dosage and Administration (2.5)for twice-daily dosing recommendations for pediatric subjects 3 to less than 18 years of age and weighing at least 10 kg.
In clinical trial TMC114-C230, the safety, pharmacokinetic profile and virologic and immunologic responses of PREZISTA/ritonavir administered once daily were evaluated in treatment-naïve HIV-1 infected pediatric subjects 12 to less than 18 years of age (12 subjects) [see Adverse Reactions (6.1) , Clinical Pharmacology (12.3)and Clinical Studies (14.4)] . Frequency, type, and severity of adverse drug reactions in pediatric subjects were comparable to those observed in adults [see Adverse Reactions (6.1)] . Once daily dosing recommendations for pediatric patients 3 to less than 12 years of age were derived using population pharmacokinetic modeling and simulation. Although a PREZISTA/ritonavir once daily dosing pediatric trial was not conducted in children less than 12 years of age, there is sufficient clinical safety data to support the predicted PREZISTA exposures for the dosing recommendations in this age group [see Clinical Pharmacology (12.3)] . Please see Dosage and Administration (2.5) for once-daily dosing recommendations for pediatric subjects 3 to less than 18 years of age and weighing at least 10 kg.
8.5 Geriatric Use
Clinical studies of PREZISTA did not include sufficient numbers of patients aged 65 years and over to determine whether they respond differently from younger patients. In general, caution should be exercised in the administration and monitoring of PREZISTA in elderly patients, reflecting the greater frequency of decreased hepatic function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
5.2 Hepatotoxicity
Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) has been reported with PREZISTA/ritonavir. During the clinical development program (N=3063), hepatitis was reported in 0.5% of patients receiving combination therapy with PREZISTA/ritonavir. Patients with pre-existing liver dysfunction, including chronic active hepatitis B or C, have an increased risk for liver function abnormalities including severe hepatic adverse events.
Post-marketing cases of liver injury, including some fatalities, have been reported. These have generally occurred in patients with advanced HIV-1 disease taking multiple concomitant medications, having co-morbidities including hepatitis B or C co-infection, and/or developing immune reconstitution syndrome. A causal relationship with PREZISTA/ritonavir therapy has not been established.
Appropriate laboratory testing should be conducted prior to initiating therapy with PREZISTA/ritonavir and patients should be monitored during treatment. Increased AST/ALT monitoring should be considered in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases, especially during the first several months of PREZISTA/ritonavir treatment.
Evidence of new or worsening liver dysfunction (including clinically significant elevation of liver enzymes and/or symptoms such as fatigue, anorexia, nausea, jaundice, dark urine, liver tenderness, hepatomegaly) in patients on PREZISTA/ritonavir should prompt consideration of interruption or discontinuation of treatment.
4 Contraindications (4 CONTRAINDICATIONS)
Co-administration of PREZISTA/ritonavir is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Examples of these drugs and other contraindicated drugs (which may lead to reduced efficacy of darunavir) are listed below [see Drug Interactions (7.3)] . Due to the need for co-administration of PREZISTA with ritonavir, please refer to ritonavir prescribing information for a description of ritonavir contraindications.
- Alpha 1-adrenoreceptor antagonist: alfuzosin
- Anti-gout: colchicine, in patients with renal and/or hepatic impairment
- Antimycobacterial: rifampin
- Antipsychotics: lurasidone, pimozide
- Cardiac Disorders: dronedarone, ivabradine, ranolazine
- Ergot derivatives, e.g. dihydroergotamine, ergotamine, methylergonovine
- Herbal product: St. John's wort ( Hypericum perforatum)
- Hepatitis C direct acting antiviral: elbasvir/grazoprevir
- Lipid modifying agents: lomitapide, lovastatin, simvastatin
- Opioid Antagonist: naloxegol
- PDE-5 inhibitor: sildenafil when used for treatment of pulmonary arterial hypertension
- Sedatives/hypnotics: orally administered midazolam, triazolam
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are discussed in other sections of labeling:
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Severe Skin Reactions [see Warnings and Precautions (5.3)]
- Diabetes Mellitus/Hyperglycemia [see Warnings and Precautions (5.6)]
- Fat Redistribution [see Warnings and Precautions (5.7)]
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.8)]
- Hemophilia [see Warnings and Precautions (5.9)]
Due to the need for co-administration of PREZISTA with ritonavir, please refer to ritonavir prescribing information for ritonavir-associated adverse reactions.
7 Drug Interactions (7 DRUG INTERACTIONS)
8.7 Renal Impairment
Population pharmacokinetic analysis showed that the pharmacokinetics of darunavir were not significantly affected in HIV-infected subjects with moderate renal impairment (CrCL between 30–60 mL/min, n=20). No pharmacokinetic data are available in HIV-1-infected patients with severe renal impairment or end stage renal disease; however, because the renal clearance of darunavir is limited, a decrease in total body clearance is not expected in patients with renal impairment. As darunavir and ritonavir are highly bound to plasma proteins, it is unlikely that they will be significantly removed by hemodialysis or peritoneal dialysis [see Clinical Pharmacology (12.3)] .
Instructions for Use (INSTRUCTIONS FOR USE)
Be sure that you read, understand, and follow these Instructions for Use so that you measure and take PREZISTA oral suspension correctly. Ask your healthcare provider if you are not sure.
Each PREZISTA oral suspension carton contains:
|
Important information for use:
- Shake PREZISTA oral suspension well before each use.
- PREZISTA oral suspension should be given with the oral dosing syringe provided to make sure you measure the right amount. The oral dosing syringe provided with your PREZISTA oral suspension should not be used for any other medicines.
|
Figure A: Shake the bottle
|
|
|
Figure B: Opening the bottle
|
|
|
Figure C: Inserting the adapter
|
|
|
Figure D: Inserting the syringe
|
|
|
Figure E: Withdrawing the Oral Suspension
|
|
|
Figure F: Removing the syringe
|
|
If you or your child's dose is more than 6 mL you will need to divide the dose. Follow the instructions given to you by your healthcare provider or pharmacist about how to divide the dose, and repeat steps 4 through 7. |
Figure G: Taking the dose of PREZISTA
|
|
|
Figure H: Closing the bottle
|
|
|
Figure I: Removing the plunger from the barrel
|
|
|
Figure J: Putting the syringe back together
|
How should I store PREZISTA?
- Store PREZISTA oral suspension and the oral dosing syringe at room temperature 77°F (25°C).
- Do not refrigerate or freeze PREZISTA oral suspension.
- Keep PREZISTA oral suspension away from high heat.
- Store PREZISTA oral suspension in the original container.
Keep PREZISTA and all medicines out of the reach of children.
This Instruction for Use has been approved by the U.S. Food and Drug Administration.
Manufactured for:
Janssen Products, LP, Horsham PA 19044
Revised: May 2022
©2006 Janssen Pharmaceutical Companies
5.7 Fat Redistribution
Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
8.6 Hepatic Impairment
No dosage adjustment of PREZISTA/ritonavir is necessary for patients with either mild or moderate hepatic impairment. No pharmacokinetic or safety data are available regarding the use of PREZISTA/ritonavir in subjects with severe hepatic impairment. Therefore, PREZISTA/ritonavir is not recommended for use in patients with severe hepatic impairment [see Dosage and Administration (2.6)and Clinical Pharmacology (12.3)] .
1 Indications and Usage (1 INDICATIONS AND USAGE)
PREZISTA, co-administered with ritonavir (PREZISTA/ritonavir), in combination with other antiretroviral agents, is indicated for the treatment of human immunodeficiency virus (HIV-1) infection in adult and pediatric patients 3 years of age and older [see Use in Specific Populations (8.4)and Clinical Studies (14)] .
14.4 Pediatric Patients
The pharmacokinetic profile, safety and antiviral activity of PREZISTA/ritonavir were evaluated in 3 randomized, open-label, multicenter studies.
12.1 Mechanism of Action
Darunavir is an HIV-1 antiviral drug [see Microbiology (12.4)] .
5.3 Severe Skin Reactions
During the clinical development program (n=3063), severe skin reactions, accompanied by fever and/or elevations of transaminases in some cases, have been reported in 0.4% of subjects. Stevens-Johnson Syndrome was rarely (less than 0.1%) reported during the clinical development program. During post-marketing experience toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis have been reported. Discontinue PREZISTA/ritonavir immediately if signs or symptoms of severe skin reactions develop. These can include but are not limited to severe rash or rash accompanied with fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, hepatitis and/or eosinophilia.
Rash (all grades, regardless of causality) occurred in 10.3% of subjects treated with PREZISTA/ritonavir [see Adverse Reactions (6)] . Rash was mostly mild-to-moderate, often occurring within the first four weeks of treatment and resolving with continued dosing. The discontinuation rate due to rash in subjects using PREZISTA/ritonavir was 0.5%.
Rash occurred more commonly in treatment-experienced subjects receiving regimens containing PREZISTA/ritonavir + raltegravir compared to subjects receiving PREZISTA/ritonavir without raltegravir or raltegravir without PREZISTA/ritonavir. However, rash that was considered drug related occurred at similar rates for all three groups. These rashes were mild to moderate in severity and did not limit therapy; there were no discontinuations due to rash.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) has been reported with PREZISTA/ritonavir. Monitor liver function before and during therapy, especially in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases. Post-marketing cases of liver injury, including some fatalities, have been reported. ( 5.2)
- Skin reactions ranging from mild to severe, including Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms and acute generalized exanthematous pustulosis, have been reported. Discontinue treatment if severe reaction develops. ( 5.3)
- Use with caution in patients with a known sulfonamide allergy. ( 5.4)
- Patients may develop new onset diabetes mellitus or hyperglycemia. Initiation or dose adjustments of insulin or oral hypoglycemic agents may be required. ( 5.6)
- Patients may develop redistribution/accumulation of body fat or immune reconstitution syndrome. ( 5.7, 5.8)
- Patients with hemophilia may develop increased bleeding events. ( 5.9)
- PREZISTA/ritonavir is not recommended in pediatric patients below 3 years of age in view of toxicity and mortality observed in juvenile rats dosed with darunavir up to days 23 to 26 of age. ( 5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Testing:
- In treatment-experienced patients, treatment history genotypic and/or phenotypic testing is recommended prior to initiation of therapy with PREZISTA/ritonavir to assess drug susceptibility of the HIV-1 virus ( 2.1, 12.4)
- Monitor serum liver chemistry tests before and during therapy with PREZISTA/ritonavir. ( 2.1, 2.2, 5.2)
- Treatment-naïve adult patients and treatment-experienced adult patients with no darunavir resistance associated substitutions: 800 mg (one 800 mg tablet) taken with ritonavir 100 mg once daily and with food. ( 2.3)
- Treatment-experienced adult patients with at least one darunavir resistance associated substitution: 600 mg (one 600 mg tablet) taken with ritonavir 100 mg twice daily and with food. ( 2.3)
- Pregnant patients: 600 mg (one 600 mg tablet) taken with ritonavir 100 mg twice daily and with food. ( 2.4)
- Pediatric patients (3 to less than 18 years of age and weighing at least 10 kg): dosage of PREZISTA and ritonavir is based on body weight and should not exceed the adult dose. PREZISTA should be taken with ritonavir and with food. ( 2.5)
- PREZISTA/ritonavir is not recommended for use in patients with severe hepatic impairment. ( 2.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of PREZISTA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Metabolism and Nutrition Disorders:Redistribution of body fat
Musculoskeletal and Connective Tissue Disorders:Rhabdomyolysis (associated with co-administration with HMG-CoA reductase inhibitors and PREZISTA/ritonavir)
Skin and Subcutaneous Tissue Disorders:Toxic epidermal necrolysis, acute generalized exanthematous pustulosis, drug rash with eosinophilia and systemic symptoms [see Warnings and Precautions (5.3)]
Renal and Urinary Disorders:Crystal nephropathy, crystalluria
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Total darunavir exposures were generally lower during pregnancy compared to postpartum period. The reduction in darunavir exposures during pregnancy were greater for once daily dosing compared to the twice daily dosing regimen. ( 8.1, 12.3)
- Lactation: Women infected with HIV should be instructed not to breastfeed due to the potential for HIV transmission. ( 8.2)
- Pediatrics: Not recommended for patients less than 3 years of age. ( 8.4)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instruction for Use).
5.8 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including PREZISTA. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium aviuminfection, cytomegalovirus, Pneumocystis jiroveciipneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome, and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of antiretroviral treatment.
5.6 Diabetes Mellitus/hyperglycemia (5.6 Diabetes Mellitus/Hyperglycemia)
New onset diabetes mellitus, exacerbation of pre-existing diabetes mellitus, and hyperglycemia have been reported during postmarketing surveillance in HIV-infected patients receiving protease inhibitor (PI) therapy. Some patients required either initiation or dose adjustments of insulin or oral hypoglycemic agents for treatment of these events. In some cases, diabetic ketoacidosis has occurred. In those patients who discontinued PI therapy, hyperglycemia persisted in some cases. Because these events have been reported voluntarily during clinical practice, estimates of frequency cannot be made and causal relationships between PI therapy and these events have not been established.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
PREZISTA ®(darunavir) 100 mg per mL oral suspension is a white to off-white opaque liquid supplied in amber-colored multiple-dose bottles containing 100 mg of darunavir per mL packaged with a 6 mL oral dosing syringe with 0.2 mL gradations.
PREZISTA ®(darunavir) 75 mg tablets are supplied as white, caplet-shaped, film-coated tablets debossed with "75" on one side and "TMC" on the other side.
PREZISTA ®(darunavir) 150 mg tablets are supplied as white, oval-shaped, film-coated tablets debossed with "150" on one side and "TMC" on the other side.
PREZISTA ®(darunavir) 600 mg tablets are supplied as orange, oval-shaped, film-coated tablets debossed with "600MG" on one side and "TMC" on the other side.
PREZISTA ®(darunavir) 800 mg tablets are supplied as dark red, oval-shaped, film-coated tablets debossed with "800" on one side and "T" on the other side.
PREZISTA is packaged in bottles in the following configuration:
- 100 mg/mL oral suspension – 200 mL bottles (NDC 59676-565-01)
- 75 mg tablets — bottles of 480 (NDC 59676-563-01)
- 150 mg tablets — bottles of 240 (NDC 59676-564-01)
- 600 mg tablets — bottles of 60 (NDC 59676-562-01)
- 800 mg tablets — bottles of 30 (NDC 59676-566-30)
2.4 Recommended Dosage During Pregnancy
The recommended dosage in pregnant patients is PREZISTA 600 mg taken with ritonavir 100 mg twice daily with food.
PREZISTA 800 mg taken with ritonavir 100 mg once daily should only be considered in certain pregnant patients who are already on a stable PREZISTA 800 mg with ritonavir 100 mg once daily regimen prior to pregnancy, are virologically suppressed (HIV-1 RNA less than 50 copies per mL), and in whom a change to twice daily PREZISTA 600 mg with ritonavir 100 mg may compromise tolerability or compliance.
2.3 Recommended Dosage in Adult Patients
PREZISTA must be co-administered with ritonavir to exert its therapeutic effect. Failure to correctly co-administer PREZISTA with ritonavir will result in plasma levels of darunavir that will be insufficient to achieve the desired antiviral effect and will alter some drug interactions.
Patients who have difficulty swallowing PREZISTA tablets can use the 100 mg per mL PREZISTA oral suspension.
14.1 Description of Adult Clinical Trials
The evidence of efficacy of PREZISTA/ritonavir is based on the analyses of 192-week data from a randomized, controlled open-label Phase 3 trial in treatment-naïve (TMC114-C211) HIV-1-infected adult subjects and 96-week data from a randomized, controlled, open-label Phase 3 trial in antiretroviral treatment-experienced (TMC114-C214) HIV-1-infected adult subjects. In addition, 96-week data are included from 2 randomized, controlled Phase 2b trials, TMC114-C213 and TMC114-C202, in antiretroviral treatment-experienced HIV-1-infected adult subjects.
Principal Display Panel 200 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 200 mL Bottle Carton)
NDC 59676- 565-01
PREZISTA
®
(darunavir) Oral Suspension
100 mg per mL
DISPENSING:For information concerning
proper usage of the oral dosing syringe,
see accompanying PATIENT INFORMATION
and INSTRUCTIONS FOR USE.
ALERT
Find out about medicines
that should NOT be taken
with PREZISTA.
Rx only
200 mL
Johnson
&Johnson
7.2 Potential for Other Drugs to Affect Darunavir
Darunavir and ritonavir are metabolized by CYP3A. In vitrodata indicate that darunavir may be a P-gp substrate. Drugs that induce CYP3A activity would be expected to increase the clearance of darunavir and ritonavir, resulting in lowered plasma concentrations of darunavir and ritonavir. Co-administration of darunavir and ritonavir and other drugs that inhibit CYP3A, or P-gp may decrease the clearance of darunavir and ritonavir and may result in increased plasma concentrations of darunavir and ritonavir (see Table 10).
5.1 Importance of Co Administration With Ritonavir (5.1 Importance of Co-administration with Ritonavir)
PREZISTA must be co-administered with ritonavir and food to achieve the desired antiviral effect. Failure to administer PREZISTA with ritonavir and food may result in a loss of efficacy of darunavir.
Please refer to ritonavir prescribing information for additional information on precautionary measures.
Principal Display Panel 75 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 75 mg Tablet Bottle Label)
NDC 59676- 563-01
PREZISTA
®
(darunavir) tablets
75 mg
ALERT
Find out about medicines that
should NOT be taken with PREZISTA
Rx only
480 Tablets
Johnson
&Johnson
Principal Display Panel 150 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 150 mg Tablet Bottle Label)
NDC 59676- 564-01
PREZISTA
®
(darunavir) tablets
150 mg
ALERT
Find out about medicines that
should NOT be taken with PREZISTA.
Rx only
240 Tablets
Johnson
&Johnson
Principal Display Panel 600 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 600 mg Tablet Bottle Label)
NDC 59676- 562-01
PREZISTA
®
(darunavir) tablets
600 mg
ALERT
Find out about medicines that
should NOT be taken with PREZISTA.
Rx only
60 Tablets
Johnson
&Johnson
Principal Display Panel 800 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 800 mg Tablet Bottle Label)
NDC 59676- 566-30
PREZISTA
®
(darunavir) tablets
800 mg
ALERT
Find out about medicines that
should NOT be taken with PREZISTA.
Rx only
30 Tablets
Johnson
&Johnson
2.1 Testing Prior to Initiation of Prezista/ritonavir (2.1 Testing Prior to Initiation of PREZISTA/ritonavir)
In treatment-experienced patients, treatment history, genotypic and/or phenotypic testing is recommended to assess drug susceptibility of the HIV-1 virus [see Microbiology (12.4)]. Refer to Dosage and Administration (2.3), (2.4)and (2.5) for dosing recommendations.
Appropriate laboratory testing such as serum liver biochemistries should be conducted prior to initiating therapy with PREZISTA/ritonavir [see Warnings and Precautions (5.2)] .
2.2 Monitoring During Treatment With Prezista/ritonavir (2.2 Monitoring During Treatment with PREZISTA/ritonavir)
Patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases should be monitored for elevation in serum liver biochemistries, especially during the first several months of PREZISTA/ritonavir treatment [see Warnings and Precautions (5.2)] .
7.1 Potential for Prezista/ritonavir to Affect Other Drugs (7.1 Potential for PREZISTA/ritonavir to Affect Other Drugs)
PREZISTA co-administered with ritonavir is an inhibitor of CYP3A, CYP2D6, and P-gp. Co-administration of PREZISTA and ritonavir with drugs that are primarily metabolized by CYP3A and CYP2D6 or are transported by P-gp may result in increased plasma concentrations of such drugs, which could increase or prolong their therapeutic effect and adverse events. PREZISTA co-administered with ritonavir with drugs that have active metabolite(s) formed by CYP3A may result in reduced plasma concentrations of these active metabolite(s), potentially leading to loss of their therapeutic effect (see Table 10).
2.6 Not Recommended in Patients With Severe Hepatic Impairment (2.6 Not Recommended in Patients with Severe Hepatic Impairment)
No dosage adjustment is required in patients with mild or moderate hepatic impairment. No data are available regarding the use of PREZISTA/ritonavir when co-administered to subjects with severe hepatic impairment; therefore, PREZISTA/ritonavir is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)] .
5.5 Risk of Serious Adverse Reactions Due to Drug Interactions (5.5 Risk of Serious Adverse Reactions due to Drug Interactions)
Initiation of PREZISTA/ritonavir, a CYP3A inhibitor, in patients receiving medications metabolized by CYP3A or initiation of medications metabolized by CYP3A in patients already receiving PREZISTA/ritonavir, may increase plasma concentrations of medications metabolized by CYP3A and reduce plasma concentrations of active metabolite(s) formed by CYP3A.
Initiation of medications that inhibit or induce CYP3A may increase or decrease concentrations of PREZISTA/ritonavir, respectively.
These interactions may lead to:
- Clinically significant adverse reactions, potentially leading to severe, life threatening, or fatal events from greater exposures of concomitant medications.
- Clinically significant adverse reactions from greater exposures of PREZISTA/ritonavir.
- Loss of therapeutic effect of the concomitant medications from lower exposures of active metabolite(s).
- Loss of therapeutic effect of PREZISTA/ritonavir and possible development of resistance from lower exposures of PREZISTA/ritonavir.
See Table 10for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations [see Drug Interactions (7)] . Consider the potential for drug interactions prior to and during PREZISTA/ritonavir therapy; review concomitant medications during PREZISTA/ritonavir therapy; and monitor for the adverse reactions associated with the concomitant drugs [see Contraindications (4)and Drug Interactions (7)] .
5.10 Not Recommended in Pediatric Patients Below 3 Years of Age
PREZISTA/ritonavir in pediatric patients below 3 years of age is not recommended in view of toxicity and mortality observed in juvenile rats dosed with darunavir (from 20 mg/kg to 1000 mg/kg) up to days 23 to 26 of age [see Use in Specific Populations (8.1and 8.4)and Clinical Pharmacology (12.3)] .
7.3 Established and Other Potentially Significant Drug Interactions
Table 10 provides dosing recommendations as a result of drug interactions with PREZISTA/ritonavir. These recommendations are based on either drug interaction studies or predicted interactions due to the expected magnitude of interaction and potential for serious adverse events or loss of efficacy. The table includes examples of potentially significant interactions but is not all inclusive [see Contraindications (4)and Clinical Pharmacology (12.3)] , and therefore the label of each drug that is co-administered with PREZISTA/ritonavir should be consulted for information related to the route of metabolism, interaction pathways, potential risks, and specific actions to be taken with regard to co-administration.
| Concomitant Drug Class
Drug Name Examples |
Effect on Concentration of Darunavir Or Concomitant Drug | Clinical Comment |
|---|---|---|
| HIV-1-Antiviral Agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs) | ||
| didanosine | ↔ darunavir
↔ didanosine |
Didanosine should be administered one hour before or two hours after PREZISTA/ritonavir (which are administered with food). |
| HIV-1-Antiviral Agents: HIV-Protease Inhibitors (PIs) | ||
| indinavir | ↑ darunavir
↑ indinavir |
The appropriate dose of indinavir in combination with PREZISTA/ritonavir has not been established. |
| (The reference regimen for indinavir was indinavir/ritonavir 800/100 mg twice daily.) | ||
| lopinavir/ritonavir | ↓ darunavir
↔ lopinavir |
Appropriate doses of the combination have not been established. Hence, it is not recommended to co-administer lopinavir/ritonavir and PREZISTA, with or without ritonavir. |
| saquinavir | ↓ darunavir
↔ saquinavir |
Appropriate doses of the combination have not been established. Hence, it is not recommended to co-administer saquinavir and PREZISTA, with or without ritonavir. |
| Other HIV protease inhibitors, except atazanavir [see Drug Interactions (7.4)] | As co-administration with PREZISTA/ritonavir has not been studied, co-administration is not recommended. | |
| HIV-1-Antiviral Agents: CCR5 co-receptor antagonists | ||
| maraviroc | ↑ maraviroc | When used in combination with PREZISTA/ritonavir, the dose of maraviroc should be 150 mg twice daily. |
| Other Agents | ||
| Alpha 1-adrenoreceptor antagonist: | ||
| alfuzosin | ↑ alfuzosin | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as hypotension. |
|
Antibacterial:
clarithromycin |
↔ darunavir ↑ clarithromycin |
No dose adjustment of the combination is required for patients with normal renal function. For co-administration of clarithromycin and PREZISTA/ritonavir in patients with renal impairment, the following dose adjustments should be considered:
|
|
Anticoagulants:
Direct Oral Anticoagulants (DOACs) |
||
| apixaban | ↑ apixaban | Due to potentially increased bleeding risk, dosing recommendations for co-administration of apixaban with PREZISTA/ritonavir depend on the apixaban dose. Refer to apixaban dosing instructions for co-administration with P-gp and strong CYP3A inhibitors in apixaban prescribing information. |
| rivaroxaban | ↑ rivaroxaban | Co-administration of PREZISTA/ritonavir and rivaroxaban is not recommended because it may lead to an increased bleeding risk. |
| dabigatran etexilate
edoxaban |
↑ dabigatran
↑ edoxaban |
Refer to the dabigatran etexilate or edoxaban prescribing information for recommendations regarding co-administration. The specific recommendations are based on indication, renal function, and effect of the co-administered P-gp inhibitors on the concentration of dabigatran or edoxaban. Clinical monitoring is recommended when a DOAC not affected by CYP3A4 but transported by P-gp, including dabigatran etexilate and edoxaban, is co-administered with PREZISTA/ritonavir. |
| Other Anticoagulants | ||
| warfarin | ↓ warfarin
↔ darunavir |
Warfarin concentrations are decreased when co-administered with PREZISTA/ritonavir. It is recommended that the international normalized ratio (INR) be monitored when warfarin is combined with PREZISTA/ritonavir. |
|
Anticonvulsants:
carbamazepine |
↔ darunavir ↑ carbamazepine |
The dose of either PREZISTA/ritonavir or carbamazepine does not need to be adjusted when initiating co-administration with PREZISTA/ritonavir and carbamazepine. Clinical monitoring of carbamazepine concentrations and its dose titration is recommended to achieve the desired clinical response. |
| clonazepam | ↑ clonazepam | Clinical monitoring of anticonvulsants that are metabolized by CYP3A is recommended. |
| phenobarbital, phenytoin | ↔ darunavir
↓ phenytoin ↓ phenobarbital |
Phenytoin and phenobarbital levels should be monitored when co-administering with PREZISTA/ritonavir. |
|
Antidepressants:
Selective Serotonin Reuptake Inhibitors (SSRIs): |
||
| paroxetine, sertraline | ↓ paroxetine
↓ sertraline |
If either sertraline or paroxetine is initiated in patients receiving PREZISTA/ritonavir, dose titrating the SSRI based on a clinical assessment of antidepressant response is recommended. Monitor for antidepressant response in patients on a stable dose of sertraline or paroxetine who start treatment with PREZISTA/ritonavir. |
| Tricyclic Antidepressants (TCAs): | ||
| amitriptyline, desipramine, imipramine, nortriptyline | ↑ amitriptyline
↑ desipramine ↑ imipramine ↑ nortriptyline |
Use a lower dose of the tricyclic antidepressants and trazodone due to potential increased adverse events such as nausea, dizziness, hypotension and syncope. |
| Other:trazodone | ↑ trazodone | |
|
Antifungals:
itraconazole, isavuconazole, ketoconazole, posaconazole |
↑ darunavir ↑ itraconazole ↑ isavuconazole ↑ ketoconazole ↔ posaconazole |
Monitor for increased PREZISTA/ritonavir and/or antifungal adverse events with concomitant use of these antifungals. When co-administration is required, the daily dose of ketoconazole or itraconazole should not exceed 200 mg with monitoring for increased antifungal adverse events. |
| voriconazole | ↓ voriconazole | Voriconazole is not recommended for patients receiving PREZISTA/ritonavir unless an assessment comparing predicted benefit to risk ratio justifies the use of voriconazole. |
|
Anti-gout:
colchicine |
↑ colchicine |
Co-administration is contraindicated in patients with renal and/or hepatic impairment due to potential for serious and/or life-threatening reactions. For patients without renal or hepatic impairment:
|
|
Antimalarial:
artemether/lumefantrine |
↓ artemether ↓ dihydroartemisinin ↑ lumefantrine ↔ darunavir |
The combination of PREZISTA/ritonavir and artemether/lumefantrine can be used without dose adjustments. However, the combination should be used with caution as increased lumefantrine exposure may increase the risk of QT prolongation. |
| Antimycobacterials: | ||
| rifampin | ↓ darunavir | Co-administration is contraindicated due to potential for loss of therapeutic effect and development of resistance. |
| rifabutin
(The reference regimen for rifabutin was 300 mg once daily.) |
↑ darunavir
↑ rifabutin ↑ 25- O-desacetylrifabutin |
Dose reduction of rifabutin by at least 75% of the usual dose (300 mg once daily) is recommended (i.e., a maximum dose of 150 mg every other day). Increased monitoring for adverse events is warranted in patients receiving this combination and further dose reduction of rifabutin may be necessary. |
| rifapentine | ↓ darunavir | Co-administration of PREZISTA/ritonavir with rifapentine is not recommended. |
|
Antineoplastics:
dasatinib, nilotinib |
↑ antineoplastics |
A decrease in the dosage or an adjustment of the dosing interval of dasatinib and nilotinib may be necessary for patients. Please refer to the dasatinib and nilotinib prescribing information for dosing instructions. |
| vinblastine, vincristine | For vincristine and vinblastine, consideration should be given to temporarily withholding the ritonavir-containing antiretroviral regimen in patients who develop significant hematologic or gastrointestinal side effects when PREZISTA/ritonavir is administered concurrently with vincristine or vinblastine. If the antiretroviral regimen must be withheld for a prolonged period, consideration should be given to initiating a revised regimen that does not include a CYP3A or P-gp inhibitor. | |
| Antipsychotics: | ||
| lurasidone | ↑ lurasidone | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. |
| pimozide | ↑ pimozide | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. |
| quetiapine | ↑ quetiapine |
Initiation of PREZISTA with ritonavir in patients taking quetiapine:
Consider alternative antiretroviral therapy to avoid increases in quetiapine exposures. If co-administration is necessary, reduce the quetiapine dose to 1/6 of the current dose and monitor for quetiapine-associated adverse reactions. Refer to the quetiapine prescribing information for recommendations on adverse reaction monitoring. Initiation of quetiapine in patients taking PREZISTA with ritonavir: Refer to the quetiapine prescribing information for initial dosing and titration of quetiapine. |
| e.g. perphenazine, risperidone, thioridazine | ↑ antipsychotics | A decrease in the dose of antipsychotics that are metabolized by CYP3A or CYP2D6 may be needed when co-administered with PREZISTA/ritonavir. |
|
β-Blockers:
e.g. carvedilol, metoprolol, timolol |
↑ beta-blockers |
Clinical monitoring of patients is recommended. A dose decrease may be needed for these drugs when co-administered with PREZISTA/ritonavir and a lower dose of the beta blocker should be considered. |
|
Calcium Channel Blockers:
amlodipine, diltiazem, felodipine, nicardipine, nifedipine, verapamil |
↑ calcium channel blockers |
Clinical monitoring of patients is recommended. |
| Cardiac Disorders: | ||
| ranolazine, ivabradine | ↑ ranolazine
↑ ivabradine |
Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. |
| dronedarone | ↑ dronedarone | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. |
|
Other antiarrhythmics
e.g. amiodarone, bepridil, disopyramide, flecainide, lidocaine (systemic), mexiletine, propafenone, quinidine |
↑ antiarrhythmics | Therapeutic concentration monitoring, if available, is recommended for antiarrhythmics when co-administered with PREZISTA/ritonavir. |
| digoxin | ↑ digoxin | The lowest dose of digoxin should initially be prescribed. The serum digoxin concentrations should be monitored and used for titration of digoxin dose to obtain the desired clinical effect. |
| Corticosteroids: | ||
| dexamethasone (systemic) | ↓ darunavir | Co-administration of PREZISTA/ritonavir with systemic dexamethasone or other systemic corticosteroids that induce CYP3A may result in loss of therapeutic effect and development of resistance to darunavir. Consider alternative corticosteroids. |
| Corticosteroids primarily metabolized by CYP3A:
e.g. betamethasone budesonide ciclesonide fluticasone methylprednisolone mometasone triamcinolone |
↑ corticosteroids | Co-administration with corticosteroids (all routes of administration) of which exposures are significantly increased by strong CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression.
Alternative corticosteroids including beclomethasone, prednisone, and prednisolone (for which PK and/or PD are less affected by strong CYP3A inhibitors relative to other steroids) should be considered, particularly for long term use. |
| Endothelin receptor antagonist: | ||
| bosentan | ↑ bosentan |
Co-administration of bosentan in patients on PREZISTA/ritonavir:
Co-administration of PREZISTA/ritonavir in patients on bosentan:
|
| Ergot derivatives: | ||
| e.g. dihydroergotamine, ergotamine, methylergonovine | ↑ ergot derivatives | Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. |
| Hepatitis C virus (HCV): | ||
|
Direct-Acting Antivirals:
elbasvir/grazoprevir |
↑ elbasvir/grazoprevir | Co-administration is contraindicated due to potential for the increased risk of alanine transaminase (ALT) elevations. |
| glecaprevir/pibrentasvir | ↑ glecaprevir
↑ pibrentasvir |
Co-administration of PREZISTA/ritonavir with glecaprevir/pibrentasvir is not recommended. |
| Herbal product: | ||
| St. John's wort ( Hypericum perforatum) | ↓ darunavir | Co-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. |
| Hormonal contraceptives: | Effective alternative (non-hormonal) contraceptive method or a barrier method of contraception is recommended [see Use in Specific Populations (8.3)] . | |
| ethinyl estradiol, norethindrone, drospirenone | ↓ ethinyl estradiol
↓ norethindrone drospirenone: effects unknown |
For co-administration with drospirenone, clinical monitoring is recommended due to the potential for hyperkalemia.
No data are available to make recommendations on co-administration with other hormonal contraceptives. |
|
Immunosuppressants:
e.g. cyclosporine, tacrolimus, sirolimus |
↑ immunosuppressants | Therapeutic concentration monitoring of the immunosuppressive agent is recommended when co-administered with PREZISTA/ritonavir. |
|
Immunosuppressant/neoplastic:
everolimus |
Co-administration of everolimus and PREZISTA/ritonavir is not recommended. | |
| irinotecan | Discontinue PREZISTA/ritonavir at least 1 week prior to starting irinotecan therapy. Do not administer PREZISTA/ritonavir with irinotecan unless there are no therapeutic alternatives. | |
| Inhaled beta agonist: | ||
| salmeterol | ↑ salmeterol | Co-administration of salmeterol and PREZISTA/ritonavir is not recommended. The combination may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations and sinus tachycardia. |
| Lipid Modifying Agents: | ||
| HMG-CoA reductase inhibitors: | ||
| lovastatin, simvastatin | ↑ lovastatin
↑ simvastatin |
Co-administration is contraindicated due to potential for serious reactions such as myopathy including rhabdomyolysis. |
| atorvastatin, pravastatin, rosuvastatin | ↑ HMG-CoA reductase inhibitors | Co-administration of PREZISTA/ritonavir with HMG-Co A reductase inhibitors may lead to adverse events such as myopathy. Titrate atorvastatin, pravastatin or rosuvastatin dose carefully and use the lowest necessary dose while monitoring for adverse events. Do not exceed atorvastatin 20 mg/day. |
| Other lipid modifying agents: | ||
| lomitapide | ↑ lomitapide | Co-administration is contraindicated due to potential for markedly increased transaminases. |
| Narcotic analgesics metabolized by CYP3A: | ||
| e.g. fentanyl, oxycodone | ↑ fentanyl
↑ oxycodone |
Careful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. |
| tramadol | ↑ tramadol | A dose decrease may be needed for tramadol with concomitant use. |
|
Narcotic analgesics/treatment of opioid dependence:
buprenorphine, buprenorphine/naloxone |
↔ buprenorphine, naloxone ↑ norbuprenorphine (metabolite) |
No dose adjustment for buprenorphine or buprenorphine/naloxone is required with concurrent administration of PREZISTA/ritonavir. Clinical monitoring is recommended if PREZISTA/ritonavir and buprenorphine or buprenorphine/naloxone are co-administered. |
| methadone | ↓ methadone | No adjustment of methadone dosage is required when initiating co-administration of PREZISTA/ritonavir. However, clinical monitoring is recommended as the dose of methadone during maintenance therapy may need to be adjusted in some patients. |
| Opioid Antagonist | ||
| naloxegol | ↑ naloxegol | Co-administration of PREZISTA/ritonavir and naloxegol is contraindicated due to potential for precipitating opioid withdrawal symptoms. |
|
PDE-5 inhibitors:
e.g. avanafil, sildenafil, tadalafil, vardenafil |
↑ PDE-5 inhibitors (only the use of sildenafil at doses used for treatment of erectile dysfunction has been studied with PREZISTA/ritonavir) |
Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH): Co-administration with sildenafil used for PAH is contraindicated due to potential for sildenafil associated adverse reactions (which include visual disturbances, hypotension, prolonged erection, and syncope).
Use of PDE-5 inhibitors for erectile dysfunction:
Co-administration of PREZISTA/ritonavir and avanafil is not recommended. |
|
Platelet aggregation inhibitor:
ticagrelor |
↑ ticagrelor |
Co-administration of PREZISTA/ritonavir and ticagrelor is not recommended. |
| clopidogrel | ↓ clopidogrel active metabolite | Co-administration of PREZISTA/ritonavir and clopidogrel is not recommended due to potential reduction of the antiplatelet activity of clopidogrel. |
| prasugrel | ↔ prasugrel active metabolite | No dose adjustment is needed when prasugrel is co-administered with PREZISTA/ritonavir. |
|
Proton pump inhibitor:
omeprazole |
↓ omeprazole ↔ darunavir |
When omeprazole is co-administered with PREZISTA/ritonavir, monitor patients for decreased efficacy of omeprazole. Consider increasing the omeprazole dose in patients whose symptoms are not well controlled; avoid use of more than 40 mg per day of omeprazole. |
| Sedatives/hypnotics: | ||
| orally administered midazolam, triazolam | ↑ midazolam
↑ triazolam |
Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as prolonged or increased sedation or respiratory depression. Triazolam and orally administered midazolam are extensively metabolized by CYP3A. Co-administration of triazolam or orally administered midazolam with PREZISTA may cause large increases in the concentrations of these benzodiazepines. |
| metabolized by CYP3A
e.g. buspirone, diazepam, estazolam, zolpidem |
↑ sedatives/hypnotics | Titration is recommended when co-administering PREZISTA/ritonavir with sedatives/hypnotics metabolized by CYP3A and a lower dose of the sedatives/hypnotics should be considered with monitoring for adverse events. |
| parenterally administered midazolam | Co-administration of parenteral midazolam should be done in a setting which ensures close clinical monitoring and appropriate medical management in case of respiratory depression and/or prolonged sedation. Dosage reduction for midazolam should be considered, especially if more than a single dose of midazolam is administered. | |
| Urinary antispasmodics | ||
| fesoterodine | ↑ fesoterodine | When fesoterodine is co-administered with PREZISTA/ritonavir, do not exceed a fesoterodine dose of 4 mg once daily. |
| solifenacin | ↑ solifenacin | When solifenacin is co-administered with PREZISTA/ritonavir, do not exceed a solifenacin dose of 5 mg once daily. |
7.4 Drugs Without Clinically Significant Interactions With Prezista (7.4 Drugs without Clinically Significant Interactions with PREZISTA)
No dosage adjustments are recommended when PREZISTA/ritonavir is co-administered with the following medications: atazanavir, dolutegravir, efavirenz, etravirine, nevirapine, nucleoside reverse transcriptase inhibitors (abacavir, emtricitabine, emtricitabine/tenofovir alafenamide, lamivudine, stavudine, tenofovir disoproxil fumarate, zidovudine), pitavastatin, raltegravir, ranitidine, or rilpivirine.
2.5 Recommended Dosage in Pediatric Patients (age 3 to Less Than 18 Years) (2.5 Recommended Dosage in Pediatric Patients (age 3 to less than 18 years))
Healthcare professionals should pay special attention to accurate dose selection of PREZISTA, transcription of the medication order, dispensing information and dosing instruction to minimize risk for medication errors, overdose, and underdose.
Prescribers should select the appropriate dose of PREZISTA/ritonavir for each individual child based on body weight (kg) and should not exceed the recommended dose for adults.
Before prescribing PREZISTA, children weighing greater than or equal to 15 kg should be assessed for the ability to swallow tablets. If a child is unable to reliably swallow a tablet, the use of PREZISTA oral suspension should be considered.
The recommended dose of PREZISTA/ritonavir for pediatric patients (3 to less than 18 years of age and weighing at least 10 kg is based on body weight (see Tables 2, 3, 4, and 5) and should not exceed the recommended adult dose. PREZISTA should be taken with ritonavir and with food.
The recommendations for the PREZISTA/ritonavir dosage regimens were based on pediatric clinical trial data and population pharmacokinetic modeling and simulation [see Use in Specific Populations (8.4)and Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:09.954863 · Updated: 2026-03-14T22:32:10.275892