Qalsody ™
81356b45-1cb7-4eef-88ea-e44cc18b47c5
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
QALSODY is indicated for the treatment of amyotrophic lateral sclerosis (ALS) in adults who have a mutation in the superoxide dismutase 1 ( SOD1 ) gene. This indication is approved under accelerated approval based on reduction in plasma neurofilament light chain (NfL) observed in patients treated with QALSODY [see Clinical Studies ( 14 )] . Continued approval for this indication may be contingent upon verification of clinical benefit in confirmatory trial(s).
Dosage and Administration
QALSODY is administered intrathecally ( 2.1 ) Dosing Information ( 2.1 ) Recommended dose: 100 milligrams (15 mL) per administration Initiate QALSODY treatment with 3 loading doses administered at 14day intervals. A maintenance dose should be administered once every 28 days thereafter. Preparation and Administration Instructions ( 2.2 ) Allow to warm to room temperature prior to administration Administer within 4 hours of removal from vial Prior to administration, remove approximately 10 mL of cerebrospinal fluid Administer as an intrathecal bolus injection over 1 to 3 minutes
Contraindications
None.
Warnings and Precautions
Myelitis and/ or Radiculitis: Serious events of myelitis and radiculitis have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to the standard of care. ( 5.1 ) Papilledema and Elevated Intracranial Pressure: Serious events of papilledema and elevated intracranial pressure have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to standard of care. ( 5.2 ) Aseptic Meningitis: Serious events of aseptic meningitis have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to standard of care. ( 5.3 )
Adverse Reactions
The following clinically significant adverse reactions are discussed elsewhere in the labeling: Myelitis and/or Radiculitis [see Warnings and Precautions ( 5.1 )] Papilledema and Elevated Intracranial Pressure [see Warnings and Precautions ( 5.2 )] Aseptic Meningitis [see Warnings and Precautions ( 5.3 )]
How Supplied
QALSODY injection is a sterile, clear and colorless to slightly yellow solution supplied as 100 mg/15 mL (6.7 mg/mL) solution in a single-dose glass vial free of preservatives. The NDC is 64406-109-01.
Storage and Handling
Store refrigerated between 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze. If no refrigeration is available, QALSODY may be stored in its original carton, protected from light at or below 30°C (86°F) for up to 14 days. If removed from the original carton, unopened vials of QALSODY can be removed from and returned to the refrigerator, if necessary, for not more than 6 hours per day at or below 30°C (86°F) for a maximum of 6 days (36 hours).
Description
QALSODY is indicated for the treatment of amyotrophic lateral sclerosis (ALS) in adults who have a mutation in the superoxide dismutase 1 ( SOD1 ) gene. This indication is approved under accelerated approval based on reduction in plasma neurofilament light chain (NfL) observed in patients treated with QALSODY [see Clinical Studies ( 14 )] . Continued approval for this indication may be contingent upon verification of clinical benefit in confirmatory trial(s).
Medication Information
Warnings and Precautions
Myelitis and/ or Radiculitis: Serious events of myelitis and radiculitis have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to the standard of care. ( 5.1 ) Papilledema and Elevated Intracranial Pressure: Serious events of papilledema and elevated intracranial pressure have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to standard of care. ( 5.2 ) Aseptic Meningitis: Serious events of aseptic meningitis have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to standard of care. ( 5.3 )
Indications and Usage
QALSODY is indicated for the treatment of amyotrophic lateral sclerosis (ALS) in adults who have a mutation in the superoxide dismutase 1 ( SOD1 ) gene. This indication is approved under accelerated approval based on reduction in plasma neurofilament light chain (NfL) observed in patients treated with QALSODY [see Clinical Studies ( 14 )] . Continued approval for this indication may be contingent upon verification of clinical benefit in confirmatory trial(s).
Dosage and Administration
QALSODY is administered intrathecally ( 2.1 ) Dosing Information ( 2.1 ) Recommended dose: 100 milligrams (15 mL) per administration Initiate QALSODY treatment with 3 loading doses administered at 14day intervals. A maintenance dose should be administered once every 28 days thereafter. Preparation and Administration Instructions ( 2.2 ) Allow to warm to room temperature prior to administration Administer within 4 hours of removal from vial Prior to administration, remove approximately 10 mL of cerebrospinal fluid Administer as an intrathecal bolus injection over 1 to 3 minutes
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are discussed elsewhere in the labeling: Myelitis and/or Radiculitis [see Warnings and Precautions ( 5.1 )] Papilledema and Elevated Intracranial Pressure [see Warnings and Precautions ( 5.2 )] Aseptic Meningitis [see Warnings and Precautions ( 5.3 )]
Storage and Handling
Store refrigerated between 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze. If no refrigeration is available, QALSODY may be stored in its original carton, protected from light at or below 30°C (86°F) for up to 14 days. If removed from the original carton, unopened vials of QALSODY can be removed from and returned to the refrigerator, if necessary, for not more than 6 hours per day at or below 30°C (86°F) for a maximum of 6 days (36 hours).
How Supplied
QALSODY injection is a sterile, clear and colorless to slightly yellow solution supplied as 100 mg/15 mL (6.7 mg/mL) solution in a single-dose glass vial free of preservatives. The NDC is 64406-109-01.
Description
QALSODY is indicated for the treatment of amyotrophic lateral sclerosis (ALS) in adults who have a mutation in the superoxide dismutase 1 ( SOD1 ) gene. This indication is approved under accelerated approval based on reduction in plasma neurofilament light chain (NfL) observed in patients treated with QALSODY [see Clinical Studies ( 14 )] . Continued approval for this indication may be contingent upon verification of clinical benefit in confirmatory trial(s).
Section 42229-5
Recommended Dosage
Administer QALSODY intrathecally using a lumbar puncture by, or under the direction of, healthcare professionals experienced in performing lumbar punctures.
The recommended dosage is 100 mg (15 mL) of QALSODY per administration.
Initiate QALSODY treatment with three (3) loading doses administered at 14-day intervals.
Administer a maintenance dose every 28 days thereafter.
Section 51945-4
Principal Display Panel - Carton Label
1 vial
NDC: 64406-109-01
QALSODY™
(tofersen) Injection
100 mg/15 mL
(6.7 mg/mL)
Sterile solution for
Intrathecal Injection Only
Rx only
Biogen ®
11 Description
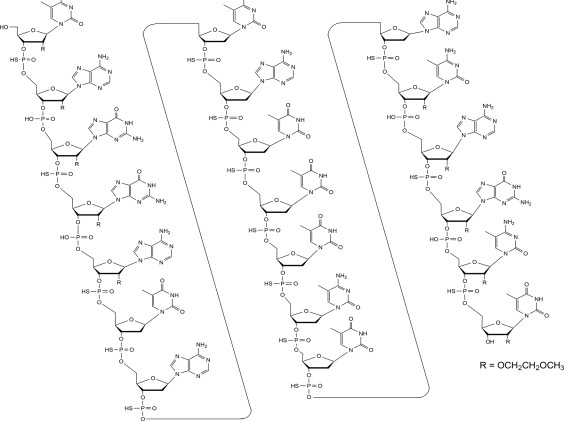
Tofersen, an antisense oligonucleotide, is a 20-base residue (20-mer) 5-10-5 MOE gapmer mixed backbone oligonucleotide. Of the nineteen internucleotide linkages, fifteen are 3′-O to 5′-O phosphorothioate diesters, and four are 3′-O to 5′-O phosphate diesters. Ten of the twenty sugar residues are 2-deoxy-D-ribose and the remainder are 2′-O-(2-methoxyethyl)-D-ribose (MOE). The residues are arranged so that there are five MOE nucleosides at the 5′ and 3′-ends of the molecule flanking a gap of ten 2′-deoxynucleosides. The cytosine and uridine bases are methylated at the 5-position. The structural formula is:
Figure 1: Structural Formula for Tofersen
The molecular formula is C230 H317 N72 O123 P19 S15 and the molecular weight is 7127.86 atomic mass units (amu).
QALSODY is supplied as a sterile, preservative-free, clear, and colorless to slightly yellow solution in a Type I glass vial to be administered by intrathecal administration. Each vial of drug product contains a single dose of 100 mg tofersen at a concentration of 6.7 mg/mL in a formulation containing 0.21 mg/mL calcium chloride dihydrate, 0.11 mg/mL dibasic sodium phosphate, 0.16 mg/mL magnesium chloride hexahydrate, 0.03 mg/mL monobasic sodium phosphate, 0.22 mg/mL potassium chloride, 8.77 mg/mL sodium chloride, and water for injection. The pH of QALSODY is approximately 7.2 (range 6.7 to 7.7).
16.1. How Supplied
QALSODY injection is a sterile, clear and colorless to slightly yellow solution supplied as 100 mg/15 mL (6.7 mg/mL) solution in a single-dose glass vial free of preservatives.
The NDC is 64406-109-01.
8.4. Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5. Geriatric Use
A total of 13.5% (22/162) patients were 65 years of age and older and 1.2% (2/162) patients were 75 years of age and older at initiation of treatment in clinical studies for ALS in patients who have a mutation in the superoxide dismutase 1 (SOD1) gene [see Clinical Studies (14)]. No overall differences in safety or effectiveness were observed between these patients and younger patients, but a greater sensitivity of some older individuals cannot be ruled out. There is no evidence for special dosage considerations based on age when QALSODY is administered.
14 Clinical Studies
The efficacy of QALSODY was assessed in a 28-week randomized, double-blind, placebocontrolled clinical study in patients 23 to 78 years of age with weakness attributable to ALS and a SOD1 mutation confirmed by a central laboratory (Study 1 Part C, NCT02623699). One hundred eight (108) patients were randomized 2:1 to receive treatment with either QALSODY 100 mg (n = 72) or placebo (n = 36) for 24 weeks (3 loading doses followed by 5 maintenance doses). Concomitant riluzole and/or edaravone use was permitted for patients.
The prespecified primary analysis population (n = 60, modified intent to treat [mITT]) had a slow vital capacity (SVC) ≥ 65% of predicted value and met prognostic enrichment criteria for rapid disease progression, defined based on their pre-randomization ALS Functional Rating Scale–Revised (ALSFRS-R) decline slope and SOD1 mutation type.
The non-mITT population (n = 48) had a slow vital capacity (SVC) ≥ 50% of predicted value and did not meet the enrichment criteria for rapid disease progression.
Baseline disease characteristics in the overall intent-to-treat (ITT) population (combined mITT and non-mITT population) were generally similar in patients treated with QALSODY and patients who received placebo, with slightly shorter time from symptom onset and higher plasma NfL at baseline in the QALSODY group. At baseline, 62% of patients were taking riluzole, and 8% of patients were taking edaravone. Mean baseline ALSFRS-R score was 36.9 (5.9) in the QALSODY treatment group and 37.3 (5.8) in the placebo group. Median time from symptom onset was 11.4 months in the QALSODY treatment group and 14.6 months in the placebo group.
The primary efficacy analysis was the change from baseline to Week 28 in the ALSFRS-R total score in the mITT population, analyzed using the joint rank test to account for mortality in conjunction with multiple imputation (MI) to account for missing data for withdrawals other than death. Patients treated with QALSODY experienced less decline from baseline in the ALSFRS-R compared to placebo, but the results were not statistically significant (QALSODY-placebo adjusted mean difference [95% CI]: 1.2 [-3.2, 5.5]). Other clinical secondary outcomes also did not reach statistical significance.
Secondary endpoints of change from baseline at Week 28 in plasma NfL and CSF SOD1 protein were nominally statistically significant (see Table 2). NfL reduction was consistently observed for all subgroups based on sex, disease duration since symptom onset, site of onset, and riluzole/edaravone use.
|
Note 1: N is the number of patients with baseline value. |
||
|
Note 2: MI was used for missing data. Model included treatment, use of riluzole or edaravone, relevant baseline score and postbaseline values (natural log transformed data). Separate models for mITT and nonmITT were used and combined for ITT analyses. |
||
|
Note 3: Adjusted geometric mean ratios to baseline, treatment differences in adjusted geometric mean ratios to baseline and corresponding 95% CIs and nominal p-values were obtained from the ANCOVA model for change from baseline including treatment as a fixed effect and adjusting for the following covariates: baseline disease duration since symptom onset, relevant baseline score, and use of riluzole or edaravone. The analysis was based on natural log transformed data. |
||
| Biomarker Endpoints | QALSODY | Placebo |
| Plasma NfL | ||
| ITT population | N=72 | N=36 |
| Adjusted geometric mean ratio to baseline | 0.45 | 1.12 |
| QALSODY to placebo difference in geometric mean ratio (95% CI) |
0.40 (0.33, 0.49) |
|
| Nominal p-value (ANCOVA+MI) | <0.0001 | |
| mITT population | N=39 | N=21 |
| Adjusted geometric mean ratio to baseline | 0.40 | 1.20 |
| QALSODY to placebo difference in geometric mean ratio (95% CI) |
0.33 (0.25, 0.45) |
|
| Nominal p-value (ANCOVA+MI) | <0.0001 | |
| CSF SOD1 Protein | ||
| ITT population | N=72 | N=36 |
| Adjusted geometric mean ratio to baseline | 0.65 | 0.98 |
| QALSODY to placebo difference in geometric mean ratio (95% CI) Nominal p-value (ANCOVA+MI) |
0.66 (0.57, 0.77) <0.0001 |
|
| mITT population | N=39 | N=21 |
| Adjusted geometric mean ratio to baseline | 0.71 | 1.16 |
| QALSODY to placebo difference in geometric mean ratio (95% CI) |
0.62 (0.49, 0.78) |
|
| Nominal p-value (ANCOVA+MI) | <0.0001 |
Figure 2: Plasma NfL Adjusted Geometric Mean Ratio to Baseline Values in Study 1 Part C by Study Week for the ITT Population
After completion of Study 1, patients had the option to enroll in an open-label extension study. At an interim analysis at 52 weeks, reductions in NfL were seen in patients previously receiving placebo who initiated QALSODY in the open-label extension study, similar to the reductions seen in patients treated with QALSODY in Study 1. Earlier initiation of QALSODY compared to placebo/delayed initiation of QALSODY was associated with trends for reduction in decline on ALSFRS-R, SVC percent-predicted, and hand-held dynamometry (HHD) megascore that were not statistically significant. Through all open-label follow-up at the time of the interim analysis, earlier initiation of QALSODY was also associated with a trend towards reduction of the risk of death or permanent ventilation, although it was not statistically significant. These exploratory analyses should be interpreted with caution given the limitations of data collected outside of a controlled study, which may be subject to confounding.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are discussed elsewhere in the labeling:
12.6. Immunogenicity
As with all therapeutic oligonucleotides, there is a potential for immunogenicity.
Immunogenicity assay results are highly dependent on the sensitivity and specificity of the assay and may be influenced by several factors such as: assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to tofersen with the incidence of antibodies to other products may be misleading.
The immunogenic response to QALSODY was evaluated in 166 patients with post-baseline plasma samples for anti-drug antibodies (ADAs). Overall, 97 QALSODY-treated patients (58.4%) developed treatment-emergent ADAs, of which 14 were transient and 83 were persistent. The presence of anti-drug antibodies (ADA) appeared to decrease plasma tofersen clearance by 32%. Effects of ADA on CSF tofersen clearance is unknown. No discernible effects of ADAs on total SOD1 protein reduction or plasma NfL reduction have been observed. No discernible effects of ADAs on safety (incidence of AEs including hypersensitivity, anaphylactic reaction, and angioedema) have been observed. Medical review of individual cases of serious neurological events also showed no association with ADA status.
1 Indications and Usage
QALSODY is indicated for the treatment of amyotrophic lateral sclerosis (ALS) in adults who have a mutation in the superoxide dismutase 1 (SOD1) gene. This indication is approved under accelerated approval based on reduction in plasma neurofilament light chain (NfL) observed in patients treated with QALSODY [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification of clinical benefit in confirmatory trial(s).
5.3. Aseptic Meningitis
Serious adverse reactions of aseptic meningitis (also called chemical meningitis or drug-induced aseptic meningitis) have been reported in patients treated with QALSODY. One patient experienced a serious adverse reaction of chemical meningitis, which led to discontinuation of QALSODY. One patient experienced a serious adverse reaction of aseptic meningitis, which did not lead to discontinuation of QALSODY. In addition, nonserious adverse drug reactions of CSF white blood cell increased, and CSF protein increased have also been reported with QALSODY [see Adverse Reactions (6.1)]. If symptoms consistent with aseptic meningitis develop, diagnostic workup and treatment should be initiated according to the standard of care.
12.1. Mechanism of Action
Tofersen is an antisense oligonucleotide that causes degradation of SOD1 mRNA through binding to SOD1 mRNA, which results in a reduction of SOD1 protein synthesis.
16.2. Storage and Handling
Store refrigerated between 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze.
If no refrigeration is available, QALSODY may be stored in its original carton, protected from light at or below 30°C (86°F) for up to 14 days.
If removed from the original carton, unopened vials of QALSODY can be removed from and returned to the refrigerator, if necessary, for not more than 6 hours per day at or below 30°C (86°F) for a maximum of 6 days (36 hours).
5 Warnings and Precautions
- Myelitis and/ or Radiculitis: Serious events of myelitis and radiculitis have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to the standard of care. (5.1)
- Papilledema and Elevated Intracranial Pressure: Serious events of papilledema and elevated intracranial pressure have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to standard of care. (5.2)
- Aseptic Meningitis: Serious events of aseptic meningitis have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to standard of care. (5.3)
2 Dosage and Administration
QALSODY is administered intrathecally (2.1)
Dosing Information (2.1)
- Recommended dose: 100 milligrams (15 mL) per administration
- Initiate QALSODY treatment with 3 loading doses administered at 14day intervals. A maintenance dose should be administered once every 28 days thereafter.
Preparation and Administration Instructions (2.2)
- Allow to warm to room temperature prior to administration
- Administer within 4 hours of removal from vial
- Prior to administration, remove approximately 10 mL of cerebrospinal fluid
- Administer as an intrathecal bolus injection over 1 to 3 minutes
3 Dosage Forms and Strengths
Injection: 100 mg/15 mL (6.7 mg/mL) as a clear and colorless to slightly yellow solution in a single-dose vial.
6.1. Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of QALSODY cannot be directly compared to rates in clinical trials of other drugs and may not reflect the rates observed in practice.
The safety of QALSODY 100 mg was evaluated in 147 patients with SOD1-ALS. The median patient exposure was 119.4 weeks (range from 4 to 212 weeks). QALSODY was evaluated in the placebo-controlled Study 1 and in the open label extension Study 2. In Study 1 Part C, approximately 43% were female; 57% were male; 64% were White and 8% were Asian. The mean age at entry in Study 1 Part C was 49.8 years (range from 23 to 78 years).
The most common adverse reactions (≥ 10% of patients treated with QALSODY and greater than placebo) were pain, fatigue, arthralgia, CSF white blood cell increased, and myalgia. Table 1 shows the common adverse reactions that occurred in at least 5% of patients treated with QALSODY and at a 5% or higher frequency than placebo.
|
‡‡ Pain includes preferred terms of pain, back pain, and pain in extremity. |
||
|
* CSF white blood cell increased includes preferred terms of CSF white blood cell increased and pleocytosis. |
||
| Adverse Reaction | Study 1 Part C | |
|
QALSODY
100 mg (n = 72) % |
Placebo
(n = 36) % |
|
| Pain‡‡ | 42 | 22 |
| Fatigue | 17 | 6 |
| Arthralgia | 14 | 6 |
| CSF white blood cell increased* | 14 | 0 |
| Myalgia | 14 | 6 |
| CSF protein increased | 8 | 3 |
| Musculoskeletal stiffness | 6 | 0 |
| Neuralgia | 6 | 0 |
5.1. Myelitis And/or Radiculitis
Serious adverse reactions of myelitis and radiculitis have been reported in patients treated with QALSODY. Six patients treated with QALSODY experienced myelitis or radiculitis in the clinical studies. Two patients discontinued treatment with QALSODY and required symptomatic management with full resolution of symptoms. In the remaining 4 patients, symptoms resolved without discontinuation of QALSODY. If symptoms consistent with myelitis or radiculitis develop, diagnostic workup and treatment should be initiated according to the standard of care. Management may require interruption or discontinuation of QALSODY.
2.2. Preparation and Administration Instructions
Use aseptic technique when preparing and administering QALSODY intrathecally. Prepare and administer QALSODY according to the following steps:
5.2. Papilledema and Elevated Intracranial Pressure
Serious adverse reactions of papilledema and elevated intracranial pressure have been reported in patients treated with QALSODY. Four patients developed elevated intracranial pressure and/or papilledema. All patients received treatment with standard of care with resolution of symptoms, and no events led to discontinuation of QALSODY. If symptoms consistent with papilledema or elevated intracranial pressure develop, diagnostic workup and treatment should be initiated according to the standard of care.
Structured Label Content
Section 42229-5 (42229-5)
Recommended Dosage
Administer QALSODY intrathecally using a lumbar puncture by, or under the direction of, healthcare professionals experienced in performing lumbar punctures.
The recommended dosage is 100 mg (15 mL) of QALSODY per administration.
Initiate QALSODY treatment with three (3) loading doses administered at 14-day intervals.
Administer a maintenance dose every 28 days thereafter.
Section 51945-4 (51945-4)
Principal Display Panel - Carton Label
1 vial
NDC: 64406-109-01
QALSODY™
(tofersen) Injection
100 mg/15 mL
(6.7 mg/mL)
Sterile solution for
Intrathecal Injection Only
Rx only
Biogen ®
11 Description (11 DESCRIPTION)
Tofersen, an antisense oligonucleotide, is a 20-base residue (20-mer) 5-10-5 MOE gapmer mixed backbone oligonucleotide. Of the nineteen internucleotide linkages, fifteen are 3′-O to 5′-O phosphorothioate diesters, and four are 3′-O to 5′-O phosphate diesters. Ten of the twenty sugar residues are 2-deoxy-D-ribose and the remainder are 2′-O-(2-methoxyethyl)-D-ribose (MOE). The residues are arranged so that there are five MOE nucleosides at the 5′ and 3′-ends of the molecule flanking a gap of ten 2′-deoxynucleosides. The cytosine and uridine bases are methylated at the 5-position. The structural formula is:
Figure 1: Structural Formula for Tofersen
The molecular formula is C230 H317 N72 O123 P19 S15 and the molecular weight is 7127.86 atomic mass units (amu).
QALSODY is supplied as a sterile, preservative-free, clear, and colorless to slightly yellow solution in a Type I glass vial to be administered by intrathecal administration. Each vial of drug product contains a single dose of 100 mg tofersen at a concentration of 6.7 mg/mL in a formulation containing 0.21 mg/mL calcium chloride dihydrate, 0.11 mg/mL dibasic sodium phosphate, 0.16 mg/mL magnesium chloride hexahydrate, 0.03 mg/mL monobasic sodium phosphate, 0.22 mg/mL potassium chloride, 8.77 mg/mL sodium chloride, and water for injection. The pH of QALSODY is approximately 7.2 (range 6.7 to 7.7).
16.1. How Supplied
QALSODY injection is a sterile, clear and colorless to slightly yellow solution supplied as 100 mg/15 mL (6.7 mg/mL) solution in a single-dose glass vial free of preservatives.
The NDC is 64406-109-01.
8.4. Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5. Geriatric Use
A total of 13.5% (22/162) patients were 65 years of age and older and 1.2% (2/162) patients were 75 years of age and older at initiation of treatment in clinical studies for ALS in patients who have a mutation in the superoxide dismutase 1 (SOD1) gene [see Clinical Studies (14)]. No overall differences in safety or effectiveness were observed between these patients and younger patients, but a greater sensitivity of some older individuals cannot be ruled out. There is no evidence for special dosage considerations based on age when QALSODY is administered.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of QALSODY was assessed in a 28-week randomized, double-blind, placebocontrolled clinical study in patients 23 to 78 years of age with weakness attributable to ALS and a SOD1 mutation confirmed by a central laboratory (Study 1 Part C, NCT02623699). One hundred eight (108) patients were randomized 2:1 to receive treatment with either QALSODY 100 mg (n = 72) or placebo (n = 36) for 24 weeks (3 loading doses followed by 5 maintenance doses). Concomitant riluzole and/or edaravone use was permitted for patients.
The prespecified primary analysis population (n = 60, modified intent to treat [mITT]) had a slow vital capacity (SVC) ≥ 65% of predicted value and met prognostic enrichment criteria for rapid disease progression, defined based on their pre-randomization ALS Functional Rating Scale–Revised (ALSFRS-R) decline slope and SOD1 mutation type.
The non-mITT population (n = 48) had a slow vital capacity (SVC) ≥ 50% of predicted value and did not meet the enrichment criteria for rapid disease progression.
Baseline disease characteristics in the overall intent-to-treat (ITT) population (combined mITT and non-mITT population) were generally similar in patients treated with QALSODY and patients who received placebo, with slightly shorter time from symptom onset and higher plasma NfL at baseline in the QALSODY group. At baseline, 62% of patients were taking riluzole, and 8% of patients were taking edaravone. Mean baseline ALSFRS-R score was 36.9 (5.9) in the QALSODY treatment group and 37.3 (5.8) in the placebo group. Median time from symptom onset was 11.4 months in the QALSODY treatment group and 14.6 months in the placebo group.
The primary efficacy analysis was the change from baseline to Week 28 in the ALSFRS-R total score in the mITT population, analyzed using the joint rank test to account for mortality in conjunction with multiple imputation (MI) to account for missing data for withdrawals other than death. Patients treated with QALSODY experienced less decline from baseline in the ALSFRS-R compared to placebo, but the results were not statistically significant (QALSODY-placebo adjusted mean difference [95% CI]: 1.2 [-3.2, 5.5]). Other clinical secondary outcomes also did not reach statistical significance.
Secondary endpoints of change from baseline at Week 28 in plasma NfL and CSF SOD1 protein were nominally statistically significant (see Table 2). NfL reduction was consistently observed for all subgroups based on sex, disease duration since symptom onset, site of onset, and riluzole/edaravone use.
|
Note 1: N is the number of patients with baseline value. |
||
|
Note 2: MI was used for missing data. Model included treatment, use of riluzole or edaravone, relevant baseline score and postbaseline values (natural log transformed data). Separate models for mITT and nonmITT were used and combined for ITT analyses. |
||
|
Note 3: Adjusted geometric mean ratios to baseline, treatment differences in adjusted geometric mean ratios to baseline and corresponding 95% CIs and nominal p-values were obtained from the ANCOVA model for change from baseline including treatment as a fixed effect and adjusting for the following covariates: baseline disease duration since symptom onset, relevant baseline score, and use of riluzole or edaravone. The analysis was based on natural log transformed data. |
||
| Biomarker Endpoints | QALSODY | Placebo |
| Plasma NfL | ||
| ITT population | N=72 | N=36 |
| Adjusted geometric mean ratio to baseline | 0.45 | 1.12 |
| QALSODY to placebo difference in geometric mean ratio (95% CI) |
0.40 (0.33, 0.49) |
|
| Nominal p-value (ANCOVA+MI) | <0.0001 | |
| mITT population | N=39 | N=21 |
| Adjusted geometric mean ratio to baseline | 0.40 | 1.20 |
| QALSODY to placebo difference in geometric mean ratio (95% CI) |
0.33 (0.25, 0.45) |
|
| Nominal p-value (ANCOVA+MI) | <0.0001 | |
| CSF SOD1 Protein | ||
| ITT population | N=72 | N=36 |
| Adjusted geometric mean ratio to baseline | 0.65 | 0.98 |
| QALSODY to placebo difference in geometric mean ratio (95% CI) Nominal p-value (ANCOVA+MI) |
0.66 (0.57, 0.77) <0.0001 |
|
| mITT population | N=39 | N=21 |
| Adjusted geometric mean ratio to baseline | 0.71 | 1.16 |
| QALSODY to placebo difference in geometric mean ratio (95% CI) |
0.62 (0.49, 0.78) |
|
| Nominal p-value (ANCOVA+MI) | <0.0001 |
Figure 2: Plasma NfL Adjusted Geometric Mean Ratio to Baseline Values in Study 1 Part C by Study Week for the ITT Population
After completion of Study 1, patients had the option to enroll in an open-label extension study. At an interim analysis at 52 weeks, reductions in NfL were seen in patients previously receiving placebo who initiated QALSODY in the open-label extension study, similar to the reductions seen in patients treated with QALSODY in Study 1. Earlier initiation of QALSODY compared to placebo/delayed initiation of QALSODY was associated with trends for reduction in decline on ALSFRS-R, SVC percent-predicted, and hand-held dynamometry (HHD) megascore that were not statistically significant. Through all open-label follow-up at the time of the interim analysis, earlier initiation of QALSODY was also associated with a trend towards reduction of the risk of death or permanent ventilation, although it was not statistically significant. These exploratory analyses should be interpreted with caution given the limitations of data collected outside of a controlled study, which may be subject to confounding.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed elsewhere in the labeling:
12.6. Immunogenicity
As with all therapeutic oligonucleotides, there is a potential for immunogenicity.
Immunogenicity assay results are highly dependent on the sensitivity and specificity of the assay and may be influenced by several factors such as: assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to tofersen with the incidence of antibodies to other products may be misleading.
The immunogenic response to QALSODY was evaluated in 166 patients with post-baseline plasma samples for anti-drug antibodies (ADAs). Overall, 97 QALSODY-treated patients (58.4%) developed treatment-emergent ADAs, of which 14 were transient and 83 were persistent. The presence of anti-drug antibodies (ADA) appeared to decrease plasma tofersen clearance by 32%. Effects of ADA on CSF tofersen clearance is unknown. No discernible effects of ADAs on total SOD1 protein reduction or plasma NfL reduction have been observed. No discernible effects of ADAs on safety (incidence of AEs including hypersensitivity, anaphylactic reaction, and angioedema) have been observed. Medical review of individual cases of serious neurological events also showed no association with ADA status.
1 Indications and Usage (1 INDICATIONS AND USAGE)
QALSODY is indicated for the treatment of amyotrophic lateral sclerosis (ALS) in adults who have a mutation in the superoxide dismutase 1 (SOD1) gene. This indication is approved under accelerated approval based on reduction in plasma neurofilament light chain (NfL) observed in patients treated with QALSODY [see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification of clinical benefit in confirmatory trial(s).
5.3. Aseptic Meningitis
Serious adverse reactions of aseptic meningitis (also called chemical meningitis or drug-induced aseptic meningitis) have been reported in patients treated with QALSODY. One patient experienced a serious adverse reaction of chemical meningitis, which led to discontinuation of QALSODY. One patient experienced a serious adverse reaction of aseptic meningitis, which did not lead to discontinuation of QALSODY. In addition, nonserious adverse drug reactions of CSF white blood cell increased, and CSF protein increased have also been reported with QALSODY [see Adverse Reactions (6.1)]. If symptoms consistent with aseptic meningitis develop, diagnostic workup and treatment should be initiated according to the standard of care.
12.1. Mechanism of Action
Tofersen is an antisense oligonucleotide that causes degradation of SOD1 mRNA through binding to SOD1 mRNA, which results in a reduction of SOD1 protein synthesis.
16.2. Storage and Handling
Store refrigerated between 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Do not freeze.
If no refrigeration is available, QALSODY may be stored in its original carton, protected from light at or below 30°C (86°F) for up to 14 days.
If removed from the original carton, unopened vials of QALSODY can be removed from and returned to the refrigerator, if necessary, for not more than 6 hours per day at or below 30°C (86°F) for a maximum of 6 days (36 hours).
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Myelitis and/ or Radiculitis: Serious events of myelitis and radiculitis have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to the standard of care. (5.1)
- Papilledema and Elevated Intracranial Pressure: Serious events of papilledema and elevated intracranial pressure have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to standard of care. (5.2)
- Aseptic Meningitis: Serious events of aseptic meningitis have been reported. Monitor for symptoms; diagnostic workup and treatment should be initiated according to standard of care. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
QALSODY is administered intrathecally (2.1)
Dosing Information (2.1)
- Recommended dose: 100 milligrams (15 mL) per administration
- Initiate QALSODY treatment with 3 loading doses administered at 14day intervals. A maintenance dose should be administered once every 28 days thereafter.
Preparation and Administration Instructions (2.2)
- Allow to warm to room temperature prior to administration
- Administer within 4 hours of removal from vial
- Prior to administration, remove approximately 10 mL of cerebrospinal fluid
- Administer as an intrathecal bolus injection over 1 to 3 minutes
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 100 mg/15 mL (6.7 mg/mL) as a clear and colorless to slightly yellow solution in a single-dose vial.
6.1. Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of QALSODY cannot be directly compared to rates in clinical trials of other drugs and may not reflect the rates observed in practice.
The safety of QALSODY 100 mg was evaluated in 147 patients with SOD1-ALS. The median patient exposure was 119.4 weeks (range from 4 to 212 weeks). QALSODY was evaluated in the placebo-controlled Study 1 and in the open label extension Study 2. In Study 1 Part C, approximately 43% were female; 57% were male; 64% were White and 8% were Asian. The mean age at entry in Study 1 Part C was 49.8 years (range from 23 to 78 years).
The most common adverse reactions (≥ 10% of patients treated with QALSODY and greater than placebo) were pain, fatigue, arthralgia, CSF white blood cell increased, and myalgia. Table 1 shows the common adverse reactions that occurred in at least 5% of patients treated with QALSODY and at a 5% or higher frequency than placebo.
|
‡‡ Pain includes preferred terms of pain, back pain, and pain in extremity. |
||
|
* CSF white blood cell increased includes preferred terms of CSF white blood cell increased and pleocytosis. |
||
| Adverse Reaction | Study 1 Part C | |
|
QALSODY
100 mg (n = 72) % |
Placebo
(n = 36) % |
|
| Pain‡‡ | 42 | 22 |
| Fatigue | 17 | 6 |
| Arthralgia | 14 | 6 |
| CSF white blood cell increased* | 14 | 0 |
| Myalgia | 14 | 6 |
| CSF protein increased | 8 | 3 |
| Musculoskeletal stiffness | 6 | 0 |
| Neuralgia | 6 | 0 |
5.1. Myelitis And/or Radiculitis (5.1. Myelitis and/or Radiculitis)
Serious adverse reactions of myelitis and radiculitis have been reported in patients treated with QALSODY. Six patients treated with QALSODY experienced myelitis or radiculitis in the clinical studies. Two patients discontinued treatment with QALSODY and required symptomatic management with full resolution of symptoms. In the remaining 4 patients, symptoms resolved without discontinuation of QALSODY. If symptoms consistent with myelitis or radiculitis develop, diagnostic workup and treatment should be initiated according to the standard of care. Management may require interruption or discontinuation of QALSODY.
2.2. Preparation and Administration Instructions
Use aseptic technique when preparing and administering QALSODY intrathecally. Prepare and administer QALSODY according to the following steps:
5.2. Papilledema and Elevated Intracranial Pressure
Serious adverse reactions of papilledema and elevated intracranial pressure have been reported in patients treated with QALSODY. Four patients developed elevated intracranial pressure and/or papilledema. All patients received treatment with standard of care with resolution of symptoms, and no events led to discontinuation of QALSODY. If symptoms consistent with papilledema or elevated intracranial pressure develop, diagnostic workup and treatment should be initiated according to the standard of care.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:48.990674 · Updated: 2026-03-14T22:17:42.518224