These Highlights Do Not Include All The Information Needed To Use Nexviazyme®
80eb3c9c-01d3-44fe-90ef-81df101b954d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: SEVERE HYPERSENSITIVITY REACTIONS, INFUSION-ASSOCIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE IN SUSCEPTIBLE PATIENTS See full prescribing information for complete boxed warning. Hypersensitivity Reactions Including Anaphylaxis Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available. If a severe hypersensitivity reaction occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. ( 5.1 ) Infusion-Associated Reactions (IARs) If severe IARs occur, consider immediate discontinuation and initiation of appropriate medical treatment. ( 5.2 ) Risk of Acute Cardiorespiratory Failure in Susceptible Patients Patients susceptible to fluid volume overload, or those with acute underlying respiratory illness or compromised cardiac or respiratory function, may be at risk of serious exacerbation of their cardiac or respiratory status during NEXVIAZYME infusion. ( 5.3 )
Indications and Usage
NEXVIAZYME is indicated for the treatment of patients 1 year of age and older with late-onset Pompe disease (lysosomal acid alpha-glucosidase [GAA] deficiency).
Dosage and Administration
NEXVIAZYME is administered as intravenous infusion. For patients weighing: 30 kg or more- the recommended dosage is 20 mg/kg (of actual body weight) every two weeks [see Dosage and Administration (2.6) ] Less than 30 kg- the recommended dosage is 40 mg/kg (of actual body weight) every two weeks [see Dosage and Administration (2.6) ] The initial recommended infusion rate is 1 mg/kg/hour. Gradually increase the infusion rate every 30 minutes if there are no signs of infusion-associated reactions (IARs) [see Dosage and Administration (2.6) ] . If one or more doses are missed, restart NEXVIAZYME treatment as soon as possible, maintaining the 2 week interval between infusions thereafter.
Warnings and Precautions
See boxed warning . ( 5.1 , 5.2 , 5.3)
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1) ] Infusion-Associated Reactions (IARs) [see Warnings and Precautions (5.2) ]
Medication Information
Warnings and Precautions
See boxed warning . ( 5.1 , 5.2 , 5.3)
Indications and Usage
NEXVIAZYME is indicated for the treatment of patients 1 year of age and older with late-onset Pompe disease (lysosomal acid alpha-glucosidase [GAA] deficiency).
Dosage and Administration
NEXVIAZYME is administered as intravenous infusion. For patients weighing: 30 kg or more- the recommended dosage is 20 mg/kg (of actual body weight) every two weeks [see Dosage and Administration (2.6) ] Less than 30 kg- the recommended dosage is 40 mg/kg (of actual body weight) every two weeks [see Dosage and Administration (2.6) ] The initial recommended infusion rate is 1 mg/kg/hour. Gradually increase the infusion rate every 30 minutes if there are no signs of infusion-associated reactions (IARs) [see Dosage and Administration (2.6) ] . If one or more doses are missed, restart NEXVIAZYME treatment as soon as possible, maintaining the 2 week interval between infusions thereafter.
Contraindications
None.
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling: Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1) ] Infusion-Associated Reactions (IARs) [see Warnings and Precautions (5.2) ]
Description
WARNING: SEVERE HYPERSENSITIVITY REACTIONS, INFUSION-ASSOCIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE IN SUSCEPTIBLE PATIENTS See full prescribing information for complete boxed warning. Hypersensitivity Reactions Including Anaphylaxis Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available. If a severe hypersensitivity reaction occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. ( 5.1 ) Infusion-Associated Reactions (IARs) If severe IARs occur, consider immediate discontinuation and initiation of appropriate medical treatment. ( 5.2 ) Risk of Acute Cardiorespiratory Failure in Susceptible Patients Patients susceptible to fluid volume overload, or those with acute underlying respiratory illness or compromised cardiac or respiratory function, may be at risk of serious exacerbation of their cardiac or respiratory status during NEXVIAZYME infusion. ( 5.3 )
Section 42229-5
Hypersensitivity Reactions Including Anaphylaxis
Patients treated with NEXVIAZYME have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available during NEXVIAZYME administration. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. In patients with severe hypersensitivity reactions, a desensitization procedure to NEXVIAZYME may be considered [see Warnings and Precautions (5.1)].
Section 43683-2
Section 44425-7
Storage and Handling
Store refrigerated at 2°C to 8°C (36°F to 46°F). Do not use NEXVIAZYME after the expiration date on the vial.
11 Description
Avalglucosidase alfa-ngpt is a hydrolytic lysosomal glycogen-specific recombinant human α-glucosidase enzyme conjugated with multiple synthetic bis-mannose-6-phosphate (bis-M6P)-tetra-mannose glycans resulting in approximately 15 moles of M6P per mole of enzyme (15 M6P) and is produced in Chinese hamster ovary cells (CHO). Avalglucosidase alfa-ngpt has a molecular weight of approximately 124 kDa.
NEXVIAZYME (avalglucosidase alfa-ngpt) for injection is a sterile white to pale-yellow lyophilized powder for intravenous use after reconstitution and dilution. Each single-dose vial contains 100 mg of avalglucosidase alfa-ngpt, glycine (200 mg), L-Histidine (10.7 mg), L-Histidine HCl monohydrate (6.5 mg), mannitol (200 mg), and polysorbate 80 (1 mg). After reconstitution with 10 mL of Sterile Water for Injection, USP, the resultant concentration is 100 mg/10 mL (10 mg/mL) with a pH of approximately 6.2.
8.4 Pediatric Use
The safety and effectiveness of NEXVIAZYME for the treatment of late-onset Pompe disease have been established in pediatric patients aged 1 year and older. Use of NEXVIAZYME for this indication is supported by evidence from two clinical studies which included adults with LOPD, and 1 pediatric patient with LOPD (16 years of age) and from safety experience in 19 pediatric patients with infantile-onset Pompe disease (IOPD) (1 to 12 years of age) treated with NEXVIAZYME [see Clinical Studies (14.1)]. NEXVIAZYME is not approved for the treatment of IOPD.
The safety profile of NEXVIAZYME in pediatric patients 1 to 12 years old with Pompe disease was similar to the safety profile of NEXVIAZYME in older pediatric and adult patients with LOPD. The safety and effectiveness of NEXVIAZYME have not been established in pediatric patients younger than 1 year of age.
8.5 Geriatric Use
Clinical studies with NEXVIAZYME included 13 patients 65 to 74 years of age and 4 patients 75 years of age and older. The recommended dosage in geriatric patients is the same as the recommended dosage in younger adult patients [see Adverse Reactions (6.1)].
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADAs in the studies described below with the incidence of ADAs in other studies, including those of NEXVIAZYME or of other avalglucosidase alfa products.
Table 5 presents the incidence of anti-avalglucosidase alfa-ngpt antibodies (referred to as ADA) in NEXVIAZYME-treated patients with Pompe disease [see Clinical Studies (14.1)]. In ERT-naïve LOPD patients who received NEXVIAZYME 20 mg/kg every two weeks for up to 436.2 weeks (with mean of 204.7 weeks), 95% (59/62) of patients developed ADA. The median time to seroconversion was 8 weeks.
ADA cross-reactivity studies showed that antibodies to avalglucosidase alfa-ngpt were cross-reactive to alglucosidase alfa.
4 Contraindications
None.
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1)]
- Infusion-Associated Reactions (IARs) [see Warnings and Precautions (5.2)]
12.2 Pharmacodynamics
In patients with Pompe disease, excess of glycogen is degraded to hexose tetrasaccharide (Hex4) which is then excreted in urine. The urinary Hex4 assay measures the major component, glucose tetrasaccharide (Glc4). Treatment with NEXVIAZYME resulted in reductions of urinary Glc4 concentrations (normalized by urine creatinine and reported as mmol Glc4/mol creatinine) in patients with Pompe disease.
In ERT-naïve LOPD patients in Study 1, the baseline mean (SD) urinary Glc4 concentration was 12.7 mmol/mol (10.10) and 8.7 mmol/mol (5.04) in NEXVIAZYME and alglucosidase alfa treatment groups, respectively [see Clinical Studies (14.1)]. At Week 145, the mean urinary Glc4 concentration was 4.32 mmol/mol (4.28) in patients who continued with NEXVIAZYME and 5.25 mmol/mol (7.48) in patients who switched from alglucosidase alfa to NEXVIAZYME.
For patients who started on NEXVIAZYME, the mean percentage (SD) change in urinary Glc4 concentration from baseline was -54% (24), at Week 49 and -53% (73) at Week 145. For patients who started on alglucosidase alfa and switched to NEXVIAZYME at Week 49, the mean percentage (SD) change in urinary Glc4 concentration from baseline was -11% (32) at Week 49, and -48% (42) at Week 145.
12.3 Pharmacokinetics
The avalglucosidase alfa-ngpt exposure increases in an approximately proportional manner with increasing doses over a range from 5 to 20 mg/kg (0.25 to 1 time the approved recommended dosage in LOPD patients weighing greater than or equal to 30 kg or 0.125 to 0.5 times the approved recommended dosage in LOPD patients weighing less than 30 kg). No accumulation was observed following every two weeks dosing. Following intravenous infusion of 20 mg/kg of NEXVIAZYME every two weeks in LOPD patients weighing greater than or equal to 30 kg, the mean ± SD plasma Cmax of avalglucosidase alfa-ngpt at Week 1 and Week 49 was 259 ± 72 µg/mL and 242 ± 81 µg/mL, respectively; the mean ± SD plasma AUC of avalglucosidase alfa-ngpt at Week 1 and Week 49 was 1,290 ± 420 µg∙h/mL and 1,250 ± 433 µg∙h/mL, respectively. Patients weighing less than 30 kg are expected to have similar AUC following intravenous infusion of 40 mg/kg of NEXVIAZYME every two weeks.
1 Indications and Usage
NEXVIAZYME is indicated for the treatment of patients 1 year of age and older with late-onset Pompe disease (lysosomal acid alpha-glucosidase [GAA] deficiency).
12.1 Mechanism of Action
Pompe disease (also known as glycogen storage disease type II, acid maltase deficiency, and glycogenosis type II) is an inherited disorder of glycogen metabolism caused by a deficiency of the lysosomal enzyme acid α-glucosidase (GAA) that degrades glycogen to glucose in the lysosome. GAA deficiency results in intralysosomal accumulation of glycogen in various tissues.
Avalglucosidase alfa-ngpt provides an exogenous source of GAA. The M6P on avalglucosidase alfa-ngpt mediates binding to M6P receptors on the cell surface with high affinity. After binding, it is internalized and transported into lysosomes where it undergoes proteolytic cleavage that results in increased GAA enzymatic activity. Avalglucosidase alfa-ngpt then exerts enzymatic activity in cleaving glycogen.
5 Warnings and Precautions
See boxed warning. (5.1, 5.2, 5.3)
2 Dosage and Administration
- Consider administering antihistamines, antipyretics, and/or corticosteroids prior to NEXVIAZYME administration to reduce the risk of IARs. (2.1)
- NEXVIAZYME is administered as intravenous infusion. For patients weighing (2.2):
- ≥30 kg, the recommended dosage is 20 mg/kg (of actual body weight) every two weeks.
- <30 kg, the recommended dosage is 40 mg/kg (of actual body weight) every two weeks.
- See the full prescribing information for dosage modifications due to hypersensitivity reactions or IARs. (2.3)
- Must be reconstituted and diluted prior to use. (2.4)
- For instructions on storage and administration, see full prescribing information. (2.5, 2.6)
3 Dosage Forms and Strengths
For injection: 100 mg of avalglucosidase alfa-ngpt as a white to pale-yellow lyophilized powder in a single-dose vial for reconstitution.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.6 Administration Instructions
- If the diluted solution was refrigerated, allow solution to equilibrate to room temperature for 30 minutes prior to infusion.
- It is recommended to use an in-line, low protein-binding, 0.2 micron filter during administration.
- Administer the infusion incrementally, as determined by the patient's response and comfort.
When the recommended dose is 20 mg/kg- Initial and Subsequent Infusions: The initial recommended infusion rate is 1 mg/kg/hour (see Table 2). If there are no signs of hypersensitivity or infusion-associated reactions (IARs), gradually increase the infusion rate every 30 minutes in each of the following three steps: 3 mg/kg/hour, 5 mg/kg/hour, and then 7 mg/kg/hour; then, maintain the infusion rate at 7 mg/kg/hour until the infusion is complete. The approximate total infusion duration is 4 to 5 hours.
Table 2: Recommended Infusion Rates at 20 mg/kg DoseDose Step 1 Step 2 Step 3 Step 4 Step 5 Start infusion at step 1 and in absence of infusion-associated reaction increase infusion rate sequentially per the steps of infusion every 30 minutes until completion (total time approximately 4 to 5 hours). 20 mg/kg 1 mg/kg/hour 3 mg/kg/hour 5 mg/kg/hour 7 mg/kg/hour Continue 7 mg/kg/hour
When the recommended dose is 40 mg/kg- Initial Infusion: The initial recommended infusion rate is 1 mg/kg/hour (see Table 3). If there are no signs of hypersensitivity or IARs, gradually increase the infusion rate every 30 minutes in each of the following three steps: 3 mg/kg/hour, 5 mg/kg/hour, and then 7 mg/kg/hour; then, maintain the infusion rate at 7 mg/kg/hour until the infusion is complete (4-step process). The approximate total infusion duration is 7 hours.
- Subsequent Infusions: The initial recommended infusion rate is 1 mg/kg/hour (see Table 3) with gradual increase in infusion rate every 30 minutes if there are no signs of hypersensitivity or IARs. The process may use either the above 4-step process or the following 5-step process: 3 mg/kg/hour, 6 mg/kg/hour, 8 mg/kg/hour, and then 10 mg/kg/hour; then, maintain the infusion rate at 10 mg/kg/hour until the infusion is complete. The approximate total 5-step infusion duration is 5 hours.
Table 3: Recommended Infusion Rates at 40 mg/kg DoseDose Step 1 Step 2 Step 3 Step 4 Step 5 Start infusion at step 1 and in absence of infusion-associated reaction increase infusion rate sequentially per the steps of infusion every 30 minutes until completion. Total time for initial infusion approximately 7 hours and can optionally increase rate of subsequent infusions to decrease total duration to 5 hours. 40 mg/kg Initial infusion rate 1 mg/kg/hour 3 mg/kg/hour 5 mg/kg/hour 7 mg/kg/hour Continue 7 mg/kg/hour Subsequent infusions (optional) 1 mg/kg/hour 3 mg/kg/hour 6 mg/kg/hour 8 mg/kg/hour Continue 10 mg/kg/hour - After the infusion is complete, flush the intravenous line with 5% Dextrose Injection.
- Do not infuse NEXVIAZYME in the same intravenous line with other products.
5.2 Infusion Associated Reactions
In clinical studies, IARs were reported to occur at any time during and/or within a few hours after the NEXVIAZYME infusion and were more likely to occur with higher infusion rates. IARs were reported in 48 (34%) NEXVIAZYME-treated patients in all clinical studies. In these studies, 5 (4%) NEXVIAZYME-treated patients reported 10 severe IARs including symptoms of chest discomfort, decreased or increased blood pressure, dysphagia, erythema, generalized edema, hypoxia, nausea, respiratory distress, tongue edema, and urticaria. The majority of IARs were assessed as mild to moderate. IARs that led to treatment discontinuation were chest discomfort, cough, dizziness, erythema, flushing, nausea, ocular hyperemia, respiratory distress, and urticaria. Increased incidence of IARs was observed in patients with higher ADA titers [see Adverse Reactions (6.1)].
Prior to NEXVIAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids to reduce the risk of IARs. However, IARs may still occur in patients after receiving pretreatment.
If a severe IAR occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. Consider the benefits and risks of readministering NEXVIAZYME following a severe IAR. Patients may be rechallenged using slower infusion rates at a dosage lower than the recommended dosage. Once the patient tolerates the infusion, the dosage (dose and/or the rate) may be increased to reach the recommended dosage.
If a mild or moderate IAR occurs, consider temporarily holding the infusion or slowing the infusion rate [see Dosage and Administration (2.3)].
Patients with an acute underlying illness at the time of NEXVIAZYME infusion appear to be at greater risk for IARs. Patients with advanced Pompe disease may have compromised cardiac and respiratory function, which may predispose them to a higher risk of severe complications from IARs.
2.2 Recommended Dosage and Administration
- NEXVIAZYME is administered as intravenous infusion. For patients weighing:
- 30 kg or more- the recommended dosage is 20 mg/kg (of actual body weight) every two weeks [see Dosage and Administration (2.6)]
- Less than 30 kg- the recommended dosage is 40 mg/kg (of actual body weight) every two weeks [see Dosage and Administration (2.6)]
- The initial recommended infusion rate is 1 mg/kg/hour. Gradually increase the infusion rate every 30 minutes if there are no signs of infusion-associated reactions (IARs) [see Dosage and Administration (2.6)]. If one or more doses are missed, restart NEXVIAZYME treatment as soon as possible, maintaining the 2 week interval between infusions thereafter.
2.4 Reconstitution and Dilution Instructions
Reconstitute and dilute NEXVIAZYME in the following manner. Use aseptic technique during preparation.
Principal Display Panel 100 Mg Vial Carton
NDC 58468-0426-1
Rx only
Nexviazyme®
(avalglucosidase alfa-ngpt)
for Injection
100 mg per vial
For Intravenous Infusion
after Reconstitution
and Dilution
One single-dose vial
Discard unused portion
2.1 Recommendations Prior to Nexviazyme Treatment
- Prior to NEXVIAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids [see Warnings and Precautions (5.1, 5.2)].
- NEXVIAZYME must be reconstituted and diluted prior to use [see Dosage and Administration (2.4)].
- Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available during NEXVIAZYME administration.
5.1 Hypersensitivity Reactions Including Anaphylaxis
Life-threatening hypersensitivity reactions, including anaphylaxis, have been reported in NEXVIAZYME-treated patients. In NEXVIAZYME clinical studies, 67 (48%) NEXVIAZYME-treated patients experienced hypersensitivity reactions, including 6 (4%) patients who reported severe hypersensitivity reactions and 3 (2%) patients who experienced anaphylaxis; 2 (1%) patients who experienced anaphylaxis discontinued from the study. Some of the hypersensitivity reactions were IgE mediated. Symptoms of severe hypersensitivity reactions (e.g., anaphylaxis) included chest discomfort, erythema, generalized edema, hypotension, hypoxia, rash, respiratory distress, tongue edema, and urticaria.
Prior to NEXVIAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids. Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available during NEXVIAZYME administration.
- If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. Consider the risks and benefits of re-administering NEXVIAZYME following severe hypersensitivity reactions (including anaphylaxis). Patients may be rechallenged using slower infusion rates at a dosage lower than the recommended dosage. In patients with severe hypersensitivity reaction, desensitization measures to NEXVIAZYME may be considered. If the decision is made to readminister NEXVIAZYME, ensure the patient tolerates the infusion. Once the patient tolerates the infusion, the dosage (dose and/or the rate) may be increased to reach the recommended dosage.
- If a mild or moderate hypersensitivity reaction occurs, consider temporarily holding the infusion or slowing the infusion rate [see Dosage and Administration (2.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate the carcinogenic potential or studies to evaluate mutagenic potential have not been performed with avalglucosidase alfa-ngpt.
Intravenous administration of avalglucosidase alfa-ngpt every other day at doses up to 50 mg/kg (exposure not evaluated) had no adverse effects on fertility in male or female mice.
14.1 Clinical Trial in Patients With Late Onset Pompe Disease
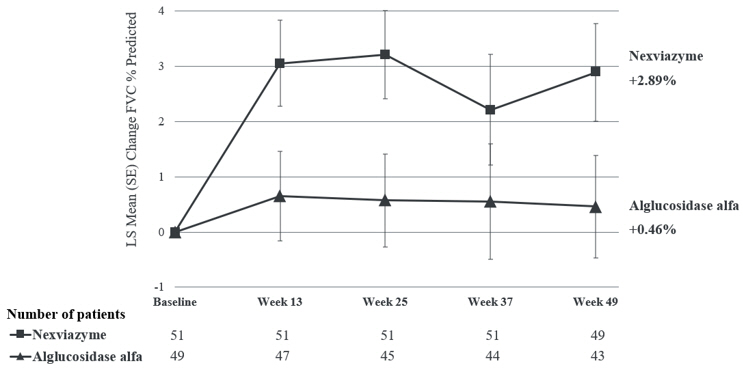
Study 1 (NCT02782741) was a randomized, double-blinded, multinational, multicenter trial comparing the efficacy and safety of NEXVIAZYME to alglucosidase alfa in treatment-naïve patients with LOPD. One hundred patients (51 in NEXVIAZYME and 49 in alglucosidase alfa) were randomized in a 1:1 ratio based on baseline forced vital capacity ([FVC] % predicted; <55% or ≥55%), sex, age (<18 years or ≥18 years), and country (Japan or not-Japan) to receive 20 mg/kg of NEXVIAZYME or alglucosidase alfa administered intravenously once every two weeks for 49 weeks. After 49 weeks, all randomized patients in Study 1 had the option to enter an open-label extension treatment period to receive NEXVIAZYME and continue treatment up to at least Week 145.
5.3 Risk of Acute Cardiorespiratory Failure in Susceptible Patients
Patients susceptible to fluid volume overload, or those with acute underlying respiratory illness or compromised cardiac or respiratory function for whom fluid restriction is indicated may be at risk of serious exacerbation of their cardiac or respiratory status during the NEXVIAZYME infusion. More frequent monitoring of vitals should be performed during NEXVIAZYME infusion in these patients. Some patients may require prolonged observation times.
2.3 Administration Modifications Due to Hypersensitivity Reactions And/or Infusion Associated Reactions
- In the event of a severe hypersensitivity reaction (e.g., anaphylaxis) or severe infusion-associated reaction (IAR), immediately discontinue NEXVIAZYME administration and initiate appropriate medical treatment. For additional recommendations in the event of a severe hypersensitivity reaction or IAR, [see Warnings and Precautions (5.1, 5.2)].
- In the event of a mild to moderate hypersensitivity reaction or a mild to moderate IAR, consider temporarily holding the infusion for 30 minutes or slowing the infusion rate by 50% [see Dosage and Administration (2.6)] and initiating appropriate medical treatment [see Warnings and Precautions (5.1, 5.2)].
- If symptoms persist for longer than 30 minutes despite holding or slowing the infusion, stop the infusion and monitor the patient. Consider re-initiating the infusion on the same day when symptoms subside at 50% of the rate at which the reaction occurred with appropriate pretreatment.
- If symptoms subside after holding the infusion, resume infusion at 50% of the rate at which the reaction occurred, and subsequently increase the infusion rate every 15 to 30 minutes by 50% as tolerated. Alternatively, if symptoms subside after slowing the infusion, complete the infusion at the reduced rate as tolerated.
- Starting with the next infusion, increase the infusion rate until the infusion rate at which the reaction occurred is reached. Consider continuing to increase the infusion rate in a stepwise manner until reaching the recommended infusion rate. Closely monitor the patient.
Warning: Severe Hypersensitivity Reactions, Infusion Associated Reactions, and Risk of Acute Cardiorespiratory Failure in Susceptible Patients
WARNING: SEVERE HYPERSENSITIVITY REACTIONS, INFUSION-ASSOCIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE IN SUSCEPTIBLE PATIENTS
See full prescribing information for complete boxed warning.
Hypersensitivity Reactions Including Anaphylaxis
- Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available. If a severe hypersensitivity reaction occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. (5.1)
Infusion-Associated Reactions (IARs)
- If severe IARs occur, consider immediate discontinuation and initiation of appropriate medical treatment. (5.2)
Risk of Acute Cardiorespiratory Failure in Susceptible Patients
- Patients susceptible to fluid volume overload, or those with acute underlying respiratory illness or compromised cardiac or respiratory function, may be at risk of serious exacerbation of their cardiac or respiratory status during NEXVIAZYME infusion. (5.3)
Structured Label Content
Section 42229-5 (42229-5)
Hypersensitivity Reactions Including Anaphylaxis
Patients treated with NEXVIAZYME have experienced life-threatening hypersensitivity reactions, including anaphylaxis. Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available during NEXVIAZYME administration. If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. In patients with severe hypersensitivity reactions, a desensitization procedure to NEXVIAZYME may be considered [see Warnings and Precautions (5.1)].
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage and Handling
Store refrigerated at 2°C to 8°C (36°F to 46°F). Do not use NEXVIAZYME after the expiration date on the vial.
11 Description (11 DESCRIPTION)
Avalglucosidase alfa-ngpt is a hydrolytic lysosomal glycogen-specific recombinant human α-glucosidase enzyme conjugated with multiple synthetic bis-mannose-6-phosphate (bis-M6P)-tetra-mannose glycans resulting in approximately 15 moles of M6P per mole of enzyme (15 M6P) and is produced in Chinese hamster ovary cells (CHO). Avalglucosidase alfa-ngpt has a molecular weight of approximately 124 kDa.
NEXVIAZYME (avalglucosidase alfa-ngpt) for injection is a sterile white to pale-yellow lyophilized powder for intravenous use after reconstitution and dilution. Each single-dose vial contains 100 mg of avalglucosidase alfa-ngpt, glycine (200 mg), L-Histidine (10.7 mg), L-Histidine HCl monohydrate (6.5 mg), mannitol (200 mg), and polysorbate 80 (1 mg). After reconstitution with 10 mL of Sterile Water for Injection, USP, the resultant concentration is 100 mg/10 mL (10 mg/mL) with a pH of approximately 6.2.
8.4 Pediatric Use
The safety and effectiveness of NEXVIAZYME for the treatment of late-onset Pompe disease have been established in pediatric patients aged 1 year and older. Use of NEXVIAZYME for this indication is supported by evidence from two clinical studies which included adults with LOPD, and 1 pediatric patient with LOPD (16 years of age) and from safety experience in 19 pediatric patients with infantile-onset Pompe disease (IOPD) (1 to 12 years of age) treated with NEXVIAZYME [see Clinical Studies (14.1)]. NEXVIAZYME is not approved for the treatment of IOPD.
The safety profile of NEXVIAZYME in pediatric patients 1 to 12 years old with Pompe disease was similar to the safety profile of NEXVIAZYME in older pediatric and adult patients with LOPD. The safety and effectiveness of NEXVIAZYME have not been established in pediatric patients younger than 1 year of age.
8.5 Geriatric Use
Clinical studies with NEXVIAZYME included 13 patients 65 to 74 years of age and 4 patients 75 years of age and older. The recommended dosage in geriatric patients is the same as the recommended dosage in younger adult patients [see Adverse Reactions (6.1)].
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADAs in the studies described below with the incidence of ADAs in other studies, including those of NEXVIAZYME or of other avalglucosidase alfa products.
Table 5 presents the incidence of anti-avalglucosidase alfa-ngpt antibodies (referred to as ADA) in NEXVIAZYME-treated patients with Pompe disease [see Clinical Studies (14.1)]. In ERT-naïve LOPD patients who received NEXVIAZYME 20 mg/kg every two weeks for up to 436.2 weeks (with mean of 204.7 weeks), 95% (59/62) of patients developed ADA. The median time to seroconversion was 8 weeks.
ADA cross-reactivity studies showed that antibodies to avalglucosidase alfa-ngpt were cross-reactive to alglucosidase alfa.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections of the labeling:
- Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.1)]
- Infusion-Associated Reactions (IARs) [see Warnings and Precautions (5.2)]
12.2 Pharmacodynamics
In patients with Pompe disease, excess of glycogen is degraded to hexose tetrasaccharide (Hex4) which is then excreted in urine. The urinary Hex4 assay measures the major component, glucose tetrasaccharide (Glc4). Treatment with NEXVIAZYME resulted in reductions of urinary Glc4 concentrations (normalized by urine creatinine and reported as mmol Glc4/mol creatinine) in patients with Pompe disease.
In ERT-naïve LOPD patients in Study 1, the baseline mean (SD) urinary Glc4 concentration was 12.7 mmol/mol (10.10) and 8.7 mmol/mol (5.04) in NEXVIAZYME and alglucosidase alfa treatment groups, respectively [see Clinical Studies (14.1)]. At Week 145, the mean urinary Glc4 concentration was 4.32 mmol/mol (4.28) in patients who continued with NEXVIAZYME and 5.25 mmol/mol (7.48) in patients who switched from alglucosidase alfa to NEXVIAZYME.
For patients who started on NEXVIAZYME, the mean percentage (SD) change in urinary Glc4 concentration from baseline was -54% (24), at Week 49 and -53% (73) at Week 145. For patients who started on alglucosidase alfa and switched to NEXVIAZYME at Week 49, the mean percentage (SD) change in urinary Glc4 concentration from baseline was -11% (32) at Week 49, and -48% (42) at Week 145.
12.3 Pharmacokinetics
The avalglucosidase alfa-ngpt exposure increases in an approximately proportional manner with increasing doses over a range from 5 to 20 mg/kg (0.25 to 1 time the approved recommended dosage in LOPD patients weighing greater than or equal to 30 kg or 0.125 to 0.5 times the approved recommended dosage in LOPD patients weighing less than 30 kg). No accumulation was observed following every two weeks dosing. Following intravenous infusion of 20 mg/kg of NEXVIAZYME every two weeks in LOPD patients weighing greater than or equal to 30 kg, the mean ± SD plasma Cmax of avalglucosidase alfa-ngpt at Week 1 and Week 49 was 259 ± 72 µg/mL and 242 ± 81 µg/mL, respectively; the mean ± SD plasma AUC of avalglucosidase alfa-ngpt at Week 1 and Week 49 was 1,290 ± 420 µg∙h/mL and 1,250 ± 433 µg∙h/mL, respectively. Patients weighing less than 30 kg are expected to have similar AUC following intravenous infusion of 40 mg/kg of NEXVIAZYME every two weeks.
1 Indications and Usage (1 INDICATIONS AND USAGE)
NEXVIAZYME is indicated for the treatment of patients 1 year of age and older with late-onset Pompe disease (lysosomal acid alpha-glucosidase [GAA] deficiency).
12.1 Mechanism of Action
Pompe disease (also known as glycogen storage disease type II, acid maltase deficiency, and glycogenosis type II) is an inherited disorder of glycogen metabolism caused by a deficiency of the lysosomal enzyme acid α-glucosidase (GAA) that degrades glycogen to glucose in the lysosome. GAA deficiency results in intralysosomal accumulation of glycogen in various tissues.
Avalglucosidase alfa-ngpt provides an exogenous source of GAA. The M6P on avalglucosidase alfa-ngpt mediates binding to M6P receptors on the cell surface with high affinity. After binding, it is internalized and transported into lysosomes where it undergoes proteolytic cleavage that results in increased GAA enzymatic activity. Avalglucosidase alfa-ngpt then exerts enzymatic activity in cleaving glycogen.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
See boxed warning. (5.1, 5.2, 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Consider administering antihistamines, antipyretics, and/or corticosteroids prior to NEXVIAZYME administration to reduce the risk of IARs. (2.1)
- NEXVIAZYME is administered as intravenous infusion. For patients weighing (2.2):
- ≥30 kg, the recommended dosage is 20 mg/kg (of actual body weight) every two weeks.
- <30 kg, the recommended dosage is 40 mg/kg (of actual body weight) every two weeks.
- See the full prescribing information for dosage modifications due to hypersensitivity reactions or IARs. (2.3)
- Must be reconstituted and diluted prior to use. (2.4)
- For instructions on storage and administration, see full prescribing information. (2.5, 2.6)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 100 mg of avalglucosidase alfa-ngpt as a white to pale-yellow lyophilized powder in a single-dose vial for reconstitution.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
2.6 Administration Instructions
- If the diluted solution was refrigerated, allow solution to equilibrate to room temperature for 30 minutes prior to infusion.
- It is recommended to use an in-line, low protein-binding, 0.2 micron filter during administration.
- Administer the infusion incrementally, as determined by the patient's response and comfort.
When the recommended dose is 20 mg/kg- Initial and Subsequent Infusions: The initial recommended infusion rate is 1 mg/kg/hour (see Table 2). If there are no signs of hypersensitivity or infusion-associated reactions (IARs), gradually increase the infusion rate every 30 minutes in each of the following three steps: 3 mg/kg/hour, 5 mg/kg/hour, and then 7 mg/kg/hour; then, maintain the infusion rate at 7 mg/kg/hour until the infusion is complete. The approximate total infusion duration is 4 to 5 hours.
Table 2: Recommended Infusion Rates at 20 mg/kg DoseDose Step 1 Step 2 Step 3 Step 4 Step 5 Start infusion at step 1 and in absence of infusion-associated reaction increase infusion rate sequentially per the steps of infusion every 30 minutes until completion (total time approximately 4 to 5 hours). 20 mg/kg 1 mg/kg/hour 3 mg/kg/hour 5 mg/kg/hour 7 mg/kg/hour Continue 7 mg/kg/hour
When the recommended dose is 40 mg/kg- Initial Infusion: The initial recommended infusion rate is 1 mg/kg/hour (see Table 3). If there are no signs of hypersensitivity or IARs, gradually increase the infusion rate every 30 minutes in each of the following three steps: 3 mg/kg/hour, 5 mg/kg/hour, and then 7 mg/kg/hour; then, maintain the infusion rate at 7 mg/kg/hour until the infusion is complete (4-step process). The approximate total infusion duration is 7 hours.
- Subsequent Infusions: The initial recommended infusion rate is 1 mg/kg/hour (see Table 3) with gradual increase in infusion rate every 30 minutes if there are no signs of hypersensitivity or IARs. The process may use either the above 4-step process or the following 5-step process: 3 mg/kg/hour, 6 mg/kg/hour, 8 mg/kg/hour, and then 10 mg/kg/hour; then, maintain the infusion rate at 10 mg/kg/hour until the infusion is complete. The approximate total 5-step infusion duration is 5 hours.
Table 3: Recommended Infusion Rates at 40 mg/kg DoseDose Step 1 Step 2 Step 3 Step 4 Step 5 Start infusion at step 1 and in absence of infusion-associated reaction increase infusion rate sequentially per the steps of infusion every 30 minutes until completion. Total time for initial infusion approximately 7 hours and can optionally increase rate of subsequent infusions to decrease total duration to 5 hours. 40 mg/kg Initial infusion rate 1 mg/kg/hour 3 mg/kg/hour 5 mg/kg/hour 7 mg/kg/hour Continue 7 mg/kg/hour Subsequent infusions (optional) 1 mg/kg/hour 3 mg/kg/hour 6 mg/kg/hour 8 mg/kg/hour Continue 10 mg/kg/hour - After the infusion is complete, flush the intravenous line with 5% Dextrose Injection.
- Do not infuse NEXVIAZYME in the same intravenous line with other products.
5.2 Infusion Associated Reactions (5.2 Infusion-Associated Reactions)
In clinical studies, IARs were reported to occur at any time during and/or within a few hours after the NEXVIAZYME infusion and were more likely to occur with higher infusion rates. IARs were reported in 48 (34%) NEXVIAZYME-treated patients in all clinical studies. In these studies, 5 (4%) NEXVIAZYME-treated patients reported 10 severe IARs including symptoms of chest discomfort, decreased or increased blood pressure, dysphagia, erythema, generalized edema, hypoxia, nausea, respiratory distress, tongue edema, and urticaria. The majority of IARs were assessed as mild to moderate. IARs that led to treatment discontinuation were chest discomfort, cough, dizziness, erythema, flushing, nausea, ocular hyperemia, respiratory distress, and urticaria. Increased incidence of IARs was observed in patients with higher ADA titers [see Adverse Reactions (6.1)].
Prior to NEXVIAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids to reduce the risk of IARs. However, IARs may still occur in patients after receiving pretreatment.
If a severe IAR occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. Consider the benefits and risks of readministering NEXVIAZYME following a severe IAR. Patients may be rechallenged using slower infusion rates at a dosage lower than the recommended dosage. Once the patient tolerates the infusion, the dosage (dose and/or the rate) may be increased to reach the recommended dosage.
If a mild or moderate IAR occurs, consider temporarily holding the infusion or slowing the infusion rate [see Dosage and Administration (2.3)].
Patients with an acute underlying illness at the time of NEXVIAZYME infusion appear to be at greater risk for IARs. Patients with advanced Pompe disease may have compromised cardiac and respiratory function, which may predispose them to a higher risk of severe complications from IARs.
2.2 Recommended Dosage and Administration
- NEXVIAZYME is administered as intravenous infusion. For patients weighing:
- 30 kg or more- the recommended dosage is 20 mg/kg (of actual body weight) every two weeks [see Dosage and Administration (2.6)]
- Less than 30 kg- the recommended dosage is 40 mg/kg (of actual body weight) every two weeks [see Dosage and Administration (2.6)]
- The initial recommended infusion rate is 1 mg/kg/hour. Gradually increase the infusion rate every 30 minutes if there are no signs of infusion-associated reactions (IARs) [see Dosage and Administration (2.6)]. If one or more doses are missed, restart NEXVIAZYME treatment as soon as possible, maintaining the 2 week interval between infusions thereafter.
2.4 Reconstitution and Dilution Instructions
Reconstitute and dilute NEXVIAZYME in the following manner. Use aseptic technique during preparation.
Principal Display Panel 100 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 100 mg Vial Carton)
NDC 58468-0426-1
Rx only
Nexviazyme®
(avalglucosidase alfa-ngpt)
for Injection
100 mg per vial
For Intravenous Infusion
after Reconstitution
and Dilution
One single-dose vial
Discard unused portion
2.1 Recommendations Prior to Nexviazyme Treatment (2.1 Recommendations Prior to NEXVIAZYME Treatment)
- Prior to NEXVIAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids [see Warnings and Precautions (5.1, 5.2)].
- NEXVIAZYME must be reconstituted and diluted prior to use [see Dosage and Administration (2.4)].
- Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available during NEXVIAZYME administration.
5.1 Hypersensitivity Reactions Including Anaphylaxis
Life-threatening hypersensitivity reactions, including anaphylaxis, have been reported in NEXVIAZYME-treated patients. In NEXVIAZYME clinical studies, 67 (48%) NEXVIAZYME-treated patients experienced hypersensitivity reactions, including 6 (4%) patients who reported severe hypersensitivity reactions and 3 (2%) patients who experienced anaphylaxis; 2 (1%) patients who experienced anaphylaxis discontinued from the study. Some of the hypersensitivity reactions were IgE mediated. Symptoms of severe hypersensitivity reactions (e.g., anaphylaxis) included chest discomfort, erythema, generalized edema, hypotension, hypoxia, rash, respiratory distress, tongue edema, and urticaria.
Prior to NEXVIAZYME administration, consider pretreating with antihistamines, antipyretics, and/or corticosteroids. Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available during NEXVIAZYME administration.
- If a severe hypersensitivity reaction (e.g., anaphylaxis) occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. Consider the risks and benefits of re-administering NEXVIAZYME following severe hypersensitivity reactions (including anaphylaxis). Patients may be rechallenged using slower infusion rates at a dosage lower than the recommended dosage. In patients with severe hypersensitivity reaction, desensitization measures to NEXVIAZYME may be considered. If the decision is made to readminister NEXVIAZYME, ensure the patient tolerates the infusion. Once the patient tolerates the infusion, the dosage (dose and/or the rate) may be increased to reach the recommended dosage.
- If a mild or moderate hypersensitivity reaction occurs, consider temporarily holding the infusion or slowing the infusion rate [see Dosage and Administration (2.3)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals to evaluate the carcinogenic potential or studies to evaluate mutagenic potential have not been performed with avalglucosidase alfa-ngpt.
Intravenous administration of avalglucosidase alfa-ngpt every other day at doses up to 50 mg/kg (exposure not evaluated) had no adverse effects on fertility in male or female mice.
14.1 Clinical Trial in Patients With Late Onset Pompe Disease (14.1 Clinical Trial in Patients with Late-Onset Pompe Disease)
Study 1 (NCT02782741) was a randomized, double-blinded, multinational, multicenter trial comparing the efficacy and safety of NEXVIAZYME to alglucosidase alfa in treatment-naïve patients with LOPD. One hundred patients (51 in NEXVIAZYME and 49 in alglucosidase alfa) were randomized in a 1:1 ratio based on baseline forced vital capacity ([FVC] % predicted; <55% or ≥55%), sex, age (<18 years or ≥18 years), and country (Japan or not-Japan) to receive 20 mg/kg of NEXVIAZYME or alglucosidase alfa administered intravenously once every two weeks for 49 weeks. After 49 weeks, all randomized patients in Study 1 had the option to enter an open-label extension treatment period to receive NEXVIAZYME and continue treatment up to at least Week 145.
5.3 Risk of Acute Cardiorespiratory Failure in Susceptible Patients
Patients susceptible to fluid volume overload, or those with acute underlying respiratory illness or compromised cardiac or respiratory function for whom fluid restriction is indicated may be at risk of serious exacerbation of their cardiac or respiratory status during the NEXVIAZYME infusion. More frequent monitoring of vitals should be performed during NEXVIAZYME infusion in these patients. Some patients may require prolonged observation times.
2.3 Administration Modifications Due to Hypersensitivity Reactions And/or Infusion Associated Reactions (2.3 Administration Modifications Due to Hypersensitivity Reactions and/or Infusion-Associated Reactions)
- In the event of a severe hypersensitivity reaction (e.g., anaphylaxis) or severe infusion-associated reaction (IAR), immediately discontinue NEXVIAZYME administration and initiate appropriate medical treatment. For additional recommendations in the event of a severe hypersensitivity reaction or IAR, [see Warnings and Precautions (5.1, 5.2)].
- In the event of a mild to moderate hypersensitivity reaction or a mild to moderate IAR, consider temporarily holding the infusion for 30 minutes or slowing the infusion rate by 50% [see Dosage and Administration (2.6)] and initiating appropriate medical treatment [see Warnings and Precautions (5.1, 5.2)].
- If symptoms persist for longer than 30 minutes despite holding or slowing the infusion, stop the infusion and monitor the patient. Consider re-initiating the infusion on the same day when symptoms subside at 50% of the rate at which the reaction occurred with appropriate pretreatment.
- If symptoms subside after holding the infusion, resume infusion at 50% of the rate at which the reaction occurred, and subsequently increase the infusion rate every 15 to 30 minutes by 50% as tolerated. Alternatively, if symptoms subside after slowing the infusion, complete the infusion at the reduced rate as tolerated.
- Starting with the next infusion, increase the infusion rate until the infusion rate at which the reaction occurred is reached. Consider continuing to increase the infusion rate in a stepwise manner until reaching the recommended infusion rate. Closely monitor the patient.
Warning: Severe Hypersensitivity Reactions, Infusion Associated Reactions, and Risk of Acute Cardiorespiratory Failure in Susceptible Patients (WARNING: SEVERE HYPERSENSITIVITY REACTIONS, INFUSION-ASSOCIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE IN SUSCEPTIBLE PATIENTS)
WARNING: SEVERE HYPERSENSITIVITY REACTIONS, INFUSION-ASSOCIATED REACTIONS, and RISK OF ACUTE CARDIORESPIRATORY FAILURE IN SUSCEPTIBLE PATIENTS
See full prescribing information for complete boxed warning.
Hypersensitivity Reactions Including Anaphylaxis
- Appropriate medical monitoring and support measures, including cardiopulmonary resuscitation equipment, should be readily available. If a severe hypersensitivity reaction occurs, discontinue NEXVIAZYME immediately and initiate appropriate medical treatment. (5.1)
Infusion-Associated Reactions (IARs)
- If severe IARs occur, consider immediate discontinuation and initiation of appropriate medical treatment. (5.2)
Risk of Acute Cardiorespiratory Failure in Susceptible Patients
- Patients susceptible to fluid volume overload, or those with acute underlying respiratory illness or compromised cardiac or respiratory function, may be at risk of serious exacerbation of their cardiac or respiratory status during NEXVIAZYME infusion. (5.3)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:33.362591 · Updated: 2026-03-14T22:27:27.393048