Vardenafil Hydrochloride Tablets
80c4793d-e19e-10f0-e053-2991aa0a4620
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Effects on the Eye ( 5.4 ) 08/2017
Indications and Usage
Vardenafil hydrochloride tablets are indicated for the treatment of erectile dysfunction.
Dosage and Administration
Vardenafil hydrochloride tablets are taken as needed: For most patients, the starting dose is 10 mg, up to once daily. Increase to 20 mg or decrease to 5 mg based on efficacy/tolerability. ( 2.1 ) A starting dose of 5 mg vardenafil hydrochloride tablets should be considered in patients ≥ 65 years of age. ( 2.3 ) Vardenafil hydrochloride tablets are taken orally, approximately 60 minutes before sexual activity. ( 2.1 ) The maximum recommended dosing frequency is one tablet per day. ( 2.1 ) Vardenafil hydrochloride tablets may be taken with or without food. ( 2.2 ) If taking potent or moderate inhibitors of CYP3A4, the dose of vardenafil hydrochloride tablets should be adjusted as follows ( 2.4 , 5.2 , 7.2 ): Ritonavir: No more than 2.5 mg in a 72-hour period Indinavir, saquinavir, atazanavir, ketoconazole 400 mg daily, itraconazole 400 mg daily, clarithromycin: No more than 2.5 mg in a 24-hour period Ketoconazole 200 mg daily, itraconazole 200 mg daily, erythromycin: No more than 5 mg in a 24-hour period. In patients on stable alpha-blocker therapy the recommended starting dose of vardenafil hydrochloride tablets is 5 mg ( 2.4 , 5.6 ) The recommended starting dose of vardenafil hydrochloride tablets is 5 mg in patients with moderate hepatic impairment (Child-Pugh B). The maximum dose in patients with moderate hepatic impairment should not exceed 10 mg. ( 2.3 , 8.6 )
Warnings and Precautions
The evaluation of erectile dysfunction should include a medical assessment, a determination of potential underlying causes and the identification of appropriate treatment. Before prescribing vardenafil hydrochloride tablets, it is important to note the following:
Contraindications
Administration with nitrates and nitric oxide donors ( 2.4 , 4.1 ) Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 2.4 , 4.2 )
Adverse Reactions
The following serious adverse reactions with the use of vardenafil hydrochloride tablets (vardenafil) are discussed elsewhere in the labeling: Cardiovascular Effects [see Contraindications (4.1) and Warnings and Precautions (5.1) ] Priapism [see Warnings and Precautions (5.3) ] Effects on Eye [see Warnings and Precautions (5.4) ] Sudden Hearing Loss [see Warnings and Precautions (5.5) ] QT Prolongation [see Warnings and Precautions (5.7) ]
Drug Interactions
Concomitant administration with potent CYP3A4 inhibitors (such as ritonavir, indinavir, ketoconazole) or moderate CYP3A4 inhibitors (such as erythromycin) increases plasma concentrations of vardenafil. Dosage adjustment is necessary when vardenafil hydrochloride tablets are administered with certain CYP3A4 inhibitors [see Dosage and Administration (2.4) , Drug Interactions (7.2) ]. Long-term safety information is not available on the concomitant administration of vardenafil with HIV protease inhibitors.
Storage and Handling
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side equivalent to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively Package Strength NDC Code Bottles of 30 10 mg 42291-847-30 20 mg 42291-848-30
How Supplied
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side equivalent to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively Package Strength NDC Code Bottles of 30 10 mg 42291-847-30 20 mg 42291-848-30
Medication Information
Warnings and Precautions
The evaluation of erectile dysfunction should include a medical assessment, a determination of potential underlying causes and the identification of appropriate treatment. Before prescribing vardenafil hydrochloride tablets, it is important to note the following:
Indications and Usage
Vardenafil hydrochloride tablets are indicated for the treatment of erectile dysfunction.
Dosage and Administration
Vardenafil hydrochloride tablets are taken as needed: For most patients, the starting dose is 10 mg, up to once daily. Increase to 20 mg or decrease to 5 mg based on efficacy/tolerability. ( 2.1 ) A starting dose of 5 mg vardenafil hydrochloride tablets should be considered in patients ≥ 65 years of age. ( 2.3 ) Vardenafil hydrochloride tablets are taken orally, approximately 60 minutes before sexual activity. ( 2.1 ) The maximum recommended dosing frequency is one tablet per day. ( 2.1 ) Vardenafil hydrochloride tablets may be taken with or without food. ( 2.2 ) If taking potent or moderate inhibitors of CYP3A4, the dose of vardenafil hydrochloride tablets should be adjusted as follows ( 2.4 , 5.2 , 7.2 ): Ritonavir: No more than 2.5 mg in a 72-hour period Indinavir, saquinavir, atazanavir, ketoconazole 400 mg daily, itraconazole 400 mg daily, clarithromycin: No more than 2.5 mg in a 24-hour period Ketoconazole 200 mg daily, itraconazole 200 mg daily, erythromycin: No more than 5 mg in a 24-hour period. In patients on stable alpha-blocker therapy the recommended starting dose of vardenafil hydrochloride tablets is 5 mg ( 2.4 , 5.6 ) The recommended starting dose of vardenafil hydrochloride tablets is 5 mg in patients with moderate hepatic impairment (Child-Pugh B). The maximum dose in patients with moderate hepatic impairment should not exceed 10 mg. ( 2.3 , 8.6 )
Contraindications
Administration with nitrates and nitric oxide donors ( 2.4 , 4.1 ) Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 2.4 , 4.2 )
Adverse Reactions
The following serious adverse reactions with the use of vardenafil hydrochloride tablets (vardenafil) are discussed elsewhere in the labeling: Cardiovascular Effects [see Contraindications (4.1) and Warnings and Precautions (5.1) ] Priapism [see Warnings and Precautions (5.3) ] Effects on Eye [see Warnings and Precautions (5.4) ] Sudden Hearing Loss [see Warnings and Precautions (5.5) ] QT Prolongation [see Warnings and Precautions (5.7) ]
Drug Interactions
Concomitant administration with potent CYP3A4 inhibitors (such as ritonavir, indinavir, ketoconazole) or moderate CYP3A4 inhibitors (such as erythromycin) increases plasma concentrations of vardenafil. Dosage adjustment is necessary when vardenafil hydrochloride tablets are administered with certain CYP3A4 inhibitors [see Dosage and Administration (2.4) , Drug Interactions (7.2) ]. Long-term safety information is not available on the concomitant administration of vardenafil with HIV protease inhibitors.
Storage and Handling
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side equivalent to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively Package Strength NDC Code Bottles of 30 10 mg 42291-847-30 20 mg 42291-848-30
How Supplied
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side equivalent to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively Package Strength NDC Code Bottles of 30 10 mg 42291-847-30 20 mg 42291-848-30
Description
Warnings and Precautions, Effects on the Eye ( 5.4 ) 08/2017
Section 34083-6
Carcinogenesis
Vardenafil was not carcinogenic in rats and mice when administered daily for 24 months. In these studies systemic drug exposures (AUCs) for unbound (free) vardenafil and its major metabolite were approximately 400- and 170-fold for male and female rats, respectively, and 21- and 37-fold for male and female mice, respectively, the exposures observed in human males given the Maximum Recommended Human Dose (MRHD) of 20 mg.
Section 42229-5
Geriatrics: A starting dose of 5 mg vardenafil hydrochloride tablets should be considered in patients ≥65 years of age [see Use in Specific Populations (8.5)].
Section 42230-3
FDA-approved patient labeling
VARDENAFIL HYDROCHLORIDE
(var-DEN-a-fil HYE-droe-KLOR-ide)
TABLETS
Read the Patient Information about vardenafil hydrochloride tablets before you start taking them and again each time you get a refill. There may be new information. You may also find it helpful to share this information with your partner. This leaflet does not take the place of talking with your doctor. You and your doctor should talk about vardenafil hydrochloride tablets when you start taking it and at regular checkups. If you do not understand the information, or have questions, talk with your doctor or pharmacist.
WHAT IMPORTANT INFORMATION SHOULD YOU KNOW ABOUT VARDENAFIL HYDROCHLORIDE TABLETS?
Vardenafil hydrochloride tablets can cause your blood pressure to drop suddenly to an unsafe level if it is taken with certain other medicines. With a sudden drop in blood pressure, you could get dizzy, faint, or have a heart attack or stroke.
Do not take vardenafil hydrochloride tablets if you:
- Take any medicines called "nitrates" (often used to control chest pain, also known as angina).
- Use recreational drugs called "poppers" like amyl nitrate and butyl nitrate.
- Take riociguat (Adempas ®), a guanylate cyclase stimulator, a medicine that treats pulmonary arterial hypertension and chronic-thromboembolic pulmonary hypertension.
(See " Who Should Not Take Vardenafil Hydrochloride Tablets?")
Tell all your healthcare providers that you take vardenafil hydrochloride tablets. If you need emergency medical care for a heart problem, it will be important for your healthcare provider to know when you last took vardenafil hydrochloride tablets.
WHAT ARE VARDENAFIL HYDROCHLORIDE TABLETS?
Vardenafil hydrochloride tablets are a prescription medicine taken by mouth for the treatment of erectile dysfunction (ED) in men.
ED is a condition where the penis does not harden and expand when a man is sexually excited, or when he cannot keep an erection. A man who has trouble getting or keeping an erection should see his doctor for help if the condition bothers him. Vardenafil hydrochloride tablets may help a man with ED get and keep an erection when he is sexually excited.
Vardenafil hydrochloride tablets do not:
- Cure ED
- Increase a man's sexual desire
- Protect a man or his partner from sexually transmitted diseases, including HIV. Speak to your doctor about ways to guard against sexually transmitted diseases.
- Serve as a male form of birth control.
Vardenafil hydrochloride tablets are only for men with ED. Vardenafil hydrochloride tablets are not for women or children. Vardenafil hydrochloride tablets must be used only under a doctor's care.
HOW DO VARDENAFIL HYDROCHLORIDE TABLETS WORK?
When a man is sexually stimulated, his body's normal physical response is to increase blood flow to his penis. This results in an erection. Vardenafil hydrochloride tablets help increase blood flow to the penis and may help men with ED get and keep an erection satisfactory for sexual activity. Once a man has completed sexual activity, blood flow to his penis decreases, and his erection goes away.
WHO CAN TAKE VARDENAFIL HYDROCHLORIDE TABLETS?
Talk to your doctor to decide if vardenafil hydrochloride tablets are right for you.
Vardenafil hydrochloride tablets have been shown to be effective in men over the age of 18 years who have erectile dysfunction, including men with diabetes or who have undergone prostatectomy.
WHO SHOULD NOT TAKE VARDENAFIL HYDROCHLORIDE TABLETS?
Do not take vardenafil hydrochloride tablets if you:
-
Take any medicines called "nitrates" (See "
What important information should you know about vardenafil hydrochloride tablets?
"). Nitrates are commonly used to treat angina. Angina is a symptom of heart disease and can cause pain in your chest, jaw, or down your arm.
Medicines called nitrates include nitroglycerin that is found in tablets, sprays, ointments, pastes, or patches. Nitrates can also be found in other medicines such as isosorbide dinitrate or isosorbide mononitrate. Some recreational drugs called "poppers" also contain nitrates, such as amyl nitrate and butyl nitrate. Do not use vardenafil hydrochloride tablets if you are using these drugs. Ask your doctor or pharmacist if you are not sure if any of your medicines are nitrates. - Take riociguat, a guanylate cyclase stimulator, a medicine that treats pulmonary arterial hypertension and chronic-throembolic pulmonary hypertension.
- Have been told by your healthcare provider to not have sexual activity because of health problems. Sexual activity can put an extra strain on your heart, especially if your heart is already weak from a heart attack or heart disease.
WHAT SHOULD YOU DISCUSS WITH YOUR DOCTOR BEFORE TAKING VARDENAFIL HYDROCHLORIDE TABLETS?
Before taking vardenafil hydrochloride tablets, tell your doctor about all your medical problems, including if you:
- Have heart problems such as angina, heart failure, irregular heartbeats, or have had a heart attack. Ask your doctor if it is safe for you to have sexual activity.
- Have low blood pressure or have high blood pressure that is not controlled.
- Have pulmonary hypertension.
- Have had a stroke.
- Have had a seizure.
- Or any family members have a rare heart condition known as prolongation of the QT interval (long QT syndrome).
- Have liver problems.
- Have kidney problems and require dialysis.
- Have retinitis pigmentosa, a rare genetic (runs in families) eye disease
- Have ever had severe vision loss, or if you have an eye condition called non-arteritic anterior ischemic optic neuropathy (NAION).
- Have stomach ulcers.
- Have a bleeding problem.
- Have a deformed penis shape or Peyronie's disease.
- Have had an erection that lasted more than 4 hours.
- Have blood cell problems such as sickle cell anemia, multiple myeloma, or leukemia.
- Have hearing problems.
CAN OTHER MEDICATIONS AFFECT VARDENAFIL HYDROCHLORIDE TABLETS?
Tell your doctor about all the medicines you take including prescription and non-prescription medicines, vitamins, and herbal supplements. Vardenafil hydrochloride tablets and other medicines may affect each other. Always check with your doctor before starting or stopping any medicines. Especially tell your doctor if you take any of the following:
- Medicines called nitrates (see " What important information should you know about vardenafil hydrochloride tablets?").
- Ketoconazole or itraconazole (such as Nizoral ® or Sporanox ®).
- Ritonavir (Norvir ®) or indinavir sulfate (Crixivan ®) saquinavir (Fortavase ® or Invirase ®) or atazanavir (Reyataz ®).
- Erythromycin or clarithromycin.
- Medicines called alpha-blockers. These include Hytrin ® (terazosin HCl), Flomax ® (tamsulosin HCl), Cardura ® (doxazosin mesylate), Minipress ® (prazosin HCl), Rapaflo ® (silodosin) or Uroxatral ® (alfuzosin HCl). Alpha-blockers are sometimes prescribed for prostate problems or high blood pressure. In some patients the use of PDE5 inhibitor drugs, including vardenafil hydrochloride tablets, with alpha-blockers can lower blood pressure significantly leading to fainting.
- You should contact the prescribing physician if alpha-blockers or other drugs that lower blood pressure are prescribed by another healthcare provider.
- Medicines that treat abnormal heartbeat. These include quinidine, procainamide, amiodarone and sotalol.
- Other medicines or treatments for ED.
HOW SHOULD YOU TAKE VARDENAFIL HYDROCHLORIDE TABLETS?
Take vardenafil hydrochloride tablets exactly as your doctor prescribes. Do not take more than one vardenafil hydrochloride tablet a day. Doses should be taken at least 24 hours apart. Some men can only take a low dose of vardenafil hydrochloride tablets because of medical conditions or medicines they take. Your doctor will prescribe the dose that is right for you.
- If you are older than 65 or have liver problems, your doctor may start you on a lower dose of vardenafil hydrochloride tablets.
- If you have prostate problems or high blood pressure, for which you take medicines called alpha-blockers, your doctor may start you on a lower dose of vardenafil hydrochloride tablets.
- If you are taking certain other medicines your doctor may prescribe a lower starting dose and limit you to one dose of vardenafil hydrochloride tablets in a 72-hour (3 days) period.
Take 1 vardenafil hydrochloride tablet about 1 hour (60 minutes) before sexual activity. Some form of sexual stimulation is needed for an erection to happen with vardenafil hydrochloride tablet. Vardenafil hydrochloride tablets may be taken with or without meals.
Do not change your dose of vardenafil hydrochloride tablets without talking to your doctor. Your doctor may lower your dose or raise your dose, depending on how your body reacts to vardenafil hydrochloride tablets.
Call your doctor or emergency room immediately if you accidentally took more vardenafil hydrochloride tablets than prescribed.
WHAT ARE THE POSSIBLE SIDE EFFECTS OF VARDENAFIL HYDROCHLORIDE TABLETS?
The most common side effects with vardenafil hydrochloride tablets are headache, flushing, stuffy or runny nose, indigestion, upset stomach, dizziness or back pain. These side effects usually go away after a few hours. Call your doctor if you get a side effect that bothers you or one that will not go away.
Vardenafil hydrochloride tablets may uncommonly cause:
- An erection that won't go away (priapism). If you get an erection that lasts more than 4 hours, get medical help right away. Priapism must be treated as soon as possible or lasting damage can happen to your penis including the inability to have erections.
- Color vision changes, such as seeing a blue tinge to objects or having difficulty telling the difference between the colors blue and green.
In rare instances, men taking PDE5 inhibitors (oral erectile dysfunction medicines, including vardenafil hydrochloride tablets) reported a sudden decrease or loss of vision in one or both eyes. It is uncertain whether PDE5 inhibitors directly cause the vision loss. If you experience sudden decrease or loss of vision, stop taking PDE5 inhibitors, including vardenafil hydrochloride tablets, and call a doctor right away.
Sudden loss or decrease in hearing, sometimes with ringing in the ears and dizziness, has been rarely reported in people taking PDE5 inhibitors, including vardenafil hydrochloride tablets. It is not possible to determine whether these events are related directly to the PDE5 inhibitors, to other diseases or medications, to other factors, or to a combination of factors. If you experience these symptoms, stop taking vardenafil hydrochloride tablets and contact a doctor right away.
These are not all the side effects of vardenafil hydrochloride tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
HOW SHOULD VARDENAFIL HYDROCHLORIDE TABLETS BE STORED?
- Store vardenafil hydrochloride tablets at room temperature between 59–86° F (15–30° C).
- Keep vardenafil hydrochloride tablets and all medicines out of the reach of children.
GENERAL INFORMATION ABOUT VARDENAFIL HYDROCHLORIDE TABLETS
Medicines are sometimes prescribed for conditions other than those described in patient information leaflets. Do not use vardenafil hydrochloride tablets for a condition for which it was not prescribed. Do not give vardenafil hydrochloride tablets to other people, even if they have the same symptoms that you have. It may harm them.
This leaflet summarizes the most important information about vardenafil hydrochloride tablets. If you would like more information, talk with your healthcare provider. You can ask your doctor or pharmacist for information about vardenafil hydrochloride tablets that is written for health professionals.
WHAT ARE THE INGREDIENTS OF VARDENAFIL HYDROCHLORIDE TABLETS?
Active Ingredient: vardenafil hydrochloride
Inactive Ingredients: microcrystalline cellulose, crospovidone, colloidal silicon dioxide, magnesium stearate, aspartame, titanium dioxide, ferric oxide red, and ferric oxide yellow.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Products cited in vardenafil hydrochloride tablets USPI
Norvir (ritonavir) is a trademark of Abbott Laboratories
Crixivan (indinavir sulfate) is a trademark of Merck & Co., Inc.
Invirase or Fortavase (saquinavir mesylate) is a trademark of Roche Laboratories Inc.
Reyataz (atazanavir sulfate) is a trademark of Bristol-Myers Squibb Company
Nizoral (ketoconazole) is a trademark of Johnson & Johnson
Sporanox (itraconazole) is a trademark of Johnson & Johnson
Hytrin (terazosin HCl) is a trademark of Abbott Laboratories
Flomax (tamsulosin HCl) is a trademark of Yamanouchi Pharmaceutical Co., Ltd.
Cardura (doxazosin mesylate) is a trademark of Pfizer Inc.
Minipress (prazosin HCl) is a trademark of Pfizer Inc.
Rapaflo (silodosin) is a trademark of Watson Pharma Inc.
Uroxatral (alfuzosin HCl) is a trademark of Sanofi-Synthelabo
Manufactured For:
AvKARE
Pulaski, TN 38478
Mfg. Rev. 10/18
AV Rev. 04/2020 (P)
Section 43683-2
| Warnings and Precautions, Effects on the Eye ( 5.4) | 08/2017 |
Section 44425-7
Store at 25°C (77°F); excursions permitted within15-30°C (59-86°F) [see USP Controlled Room Temperature].
4.1 Nitrates
Administration of vardenafil hydrochloride tablets with nitrates (either regularly and/or intermittently) and nitric oxide donors is contraindicated [see Clinical Pharmacology (12.2)] . Consistent with the effects of PDE5 inhibition on the nitric oxide/cyclic guanosine monophosphate pathway, PDE5 inhibitors, including vardenafil hydrochloride tablets, may potentiate the hypotensive effects of nitrates. A suitable time interval following dosing of vardenafil hydrochloride tablets for the safe administration of nitrates or nitric oxide donors has not been determined.
10 Overdosage
The maximum dose of vardenafil hydrochloride tablets for which human data are available is a single 120 mg dose administered to healthy male volunteers. The majority of these subjects experienced reversible back pain/myalgia and/or "abnormal vision." Single doses up to 80 mg vardenafil and multiple doses up to 40 mg vardenafil administered once daily over 4 weeks were tolerated without producing serious adverse side effects.
When 40 mg of vardenafil was administered twice daily, cases of severe back pain were observed. No muscle or neurological toxicity was identified.
In cases of overdose, standard supportive measures should be taken as required. Renal dialysis is not expected to accelerate clearance as vardenafil is highly bound to plasma proteins and not significantly eliminated in the urine.
11 Description
Vardenafil hydrochloride tablets (vardenafil hydrochloride) are administered orally for the treatment of erectile dysfunction. This monohydrochloride salt of vardenafil is a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5).
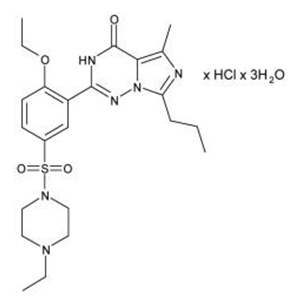
Vardenafil HCl is designated chemically as piperazine, 1-[[3-(1,4-dihydro-5-methyl-4-oxo-7-propylimidazo[5,1- f][1,2,4]triazin-2-yl)-4-ethoxyphenyl]sulfonyl]-4-ethyl-, monohydrochloride and has the following structural formula:
Vardenafil HCl is a nearly colorless, solid substance with a molecular weight of 579.1 g/mol and a solubility of 0.11 mg/mL in water.
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side corresponding to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively. In addition to the active ingredient, vardenafil HCl, each tablet contains microcrystalline cellulose, crospovidone, colloidal silicon dioxide, magnesium stearate, aspartame, titanium dioxide, ferric oxide red, and ferric oxide yellow.
2.2 Use With Food
Vardenafil hydrochloride tablets can be taken with or without food.
8.4 Pediatric Use
Vardenafil hydrochloride tablets are not indicated for use in pediatric patients. Safety and efficacy have not been established in this population.
8.5 Geriatric Use
Elderly males 65 years of age and older have higher vardenafil plasma concentrations than younger males (18 – 45 years), mean C max and AUC were 34% and 52% higher, respectively. Phase 3 clinical trials included more than 834 elderly patients, and no differences in safety or effectiveness of vardenafil hydrochloride tablets 5, 10, or 20 mg were noted when these elderly patients were compared to younger patients. However, due to increased vardenafil concentrations in the elderly, a starting dose of 5 mg vardenafil hydrochloride tablets should be considered in patients ≥65 years of age [see Clinical Pharmacology (12.3)].
5.6 Alpha Blockers
Caution is advised when PDE5 inhibitors are co-administered with alpha-blockers. PDE5 inhibitors, including vardenafil hydrochloride tablets, and alpha-adrenergic blocking agents are both vasodilators with blood-pressure lowering effects. When vasodilators are used in combination, an additive effect on blood pressure may be anticipated. In some patients, concomitant use of these two drug classes can lower blood pressure significantly leading to symptomatic hypotension (for example, fainting) [see Drug Interactions (7.1) and Clinical Pharmacology (12.2)] . Consideration should be given to the following:
- Patients should be stable on alpha-blocker therapy prior to initiating a PDE5 inhibitor. Patients who demonstrate hemodynamic instability on alpha-blocker therapy alone are at increased risk of symptomatic hypotension with concomitant use of PDE5 inhibitors.
- In those patients who are stable on alpha-blocker therapy, PDE5 inhibitors should be initiated at the lowest recommended starting dose [see Dosage and Administration (2.4)] .
- In those patients already taking an optimized dose of PDE5 inhibitor, alpha-blocker therapy should be initiated at the lowest dose. Stepwise increase in alpha-blocker dose may be associated with further lowering of blood pressure in patients taking a PDE5 inhibitor.
- Safety of combined use of PDE5 inhibitors and alpha-blockers may be affected by other variables, including intravascular volume depletion and other anti-hypertensive drugs.
14 Clinical Studies
Vardenafil hydrochloride tablets were evaluated in four major double-blind, randomized, placebo-controlled, fixed-dose, parallel design, multicenter trials in 2431 men aged 20-83 (mean age 57 years; 78% White, 7% Black, 2% Asian, 3% Hispanic and 10% Other/Unknown). The doses of vardenafil hydrochloride tablets in these studies were 5 mg, 10 mg, and 20 mg. Two of these trials were conducted in the general erectile dysfunction (ED) population and two in special ED populations (one in patients with diabetes mellitus and one in post-prostatectomy patients). Vardenafil hydrochloride tablets were dosed without regard to meals on an as needed basis in men with ED, many of whom had multiple other medical conditions. The primary endpoints were assessed at 3 months.
Primary efficacy assessment in all four major trials was by means of the Erectile Function (EF) Domain score of the validated International Index of Erectile Function (IIEF) Questionnaire and two questions from the Sexual Encounter Profile (SEP) dealing with the ability to achieve vaginal penetration (SEP2), and the ability to maintain an erection long enough for successful intercourse (SEP3).
In all four fixed-dose efficacy trials, vardenafil hydrochloride tablets showed clinically meaningful and statistically significant improvement in the EF Domain, SEP2, and SEP3 scores compared to placebo. The mean baseline EF Domain score in these trials was 11.8 (scores range from 0-30 where lower scores represent more severe disease). Vardenafil hydrochloride tablets (5 mg, 10 mg, and 20 mg) were effective in all age categories (<45, 45 to <65, and ≥65 years) and was also effective regardless of race (White, Black, Other).
4 Contraindications
6 Adverse Reactions
The following serious adverse reactions with the use of vardenafil hydrochloride tablets (vardenafil) are discussed elsewhere in the labeling:
- Cardiovascular Effects [see Contraindications (4.1) and Warnings and Precautions (5.1)]
- Priapism [see Warnings and Precautions (5.3)]
- Effects on Eye [see Warnings and Precautions (5.4)]
- Sudden Hearing Loss [see Warnings and Precautions (5.5)]
- QT Prolongation [see Warnings and Precautions (5.7)]
7 Drug Interactions
- Vardenafil hydrochloride tablets can potentiate the hypotensive effects of nitrates, alpha- blockers, and antihypertensives. ( 7.1)
8.3 Nursing Mothers
Vardenafil hydrochloride tablets are not indicated for use in women. It is not known if vardenafil is excreted in human breast milk .
Vardenafil was secreted into the milk of lactating rats at concentrations approximately 10-fold greater than found in the plasma. Following a single oral dose of 3 mg/kg, 3.3% of the administered dose was excreted into the milk within 24 hours.
5.3 Risk of Priapism
There have been rare reports of prolonged erections greater than 4 hours and priapism (painful erections greater than 6 hours in duration) for this class of compounds, including vardenafil. In the event that an erection persists longer than 4 hours, the patient should seek immediate medical assistance. If priapism is not treated immediately, penile tissue damage and permanent loss of potency may result.
Vardenafil hydrochloride tablets should be used with caution by patients with anatomical deformation of the penis (such as angulation, cavernosal fibrosis, or Peyronie's disease) or by patients who have conditions that may predispose them to priapism (such as sickle cell anemia, multiple myeloma, or leukemia).
5.9 Renal Impairment
Do not use vardenafil hydrochloride tablets in patients on renal dialysis, as vardenafil has not been evaluated in this population [see Dosage and Administration (2.3) and Use in Specific Populations (8.7)].
8.7 Renal Impairment
Do not use vardenafil hydrochloride tablets in patients on renal dialysis as vardenafil has not been evaluated in such patients.
No dosage adjustment is necessary in patients with creatinine clearance (CLcr) of 30–80 mL/min. In male volunteers with CLcr = 50-80 ml/min, the pharmacokinetics of vardenafil were similar to those observed in a control group with CLcr >80 mL/min. In male volunteers with CLcr = 30-50 mL/min or CLcr <30 mL/min, the AUC of vardenafil was 20–30% higher compared to that observed in a control group with CLcr >80 mL/min. [See Dosage and Administration (2.3) and Warnings and Precautions (5.9).]
12.3 Pharmacokinetics
The pharmacokinetics of vardenafil are approximately dose proportional over the recommended dose range.
5.4 Effects On the Eye
Physicians should advise patients to stop use of all phosphodiesterase type 5 (PDE5) inhibitors, including vardenafil hydrochloride tablets, and seek medical attention in the event of sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a rare condition and a cause of decreased vision, including permanent loss of vision, that has been reported rarely postmarketing in temporal association with the use of all PDE5 inhibitors. Based on published literature, the annual incidence of NAION is 2.5–11.8 cases per 100,000 in males aged ≥50.
An observational case-crossover study evaluated the risk of NAION when PDE5 inhibitor use, as a class, occurred immediately before NAION onset (within 5 half-lives), compared to PDE5 inhibitor use in a prior time period. The results suggest an approximate 2-fold increase in the risk of NAION, with a risk estimate of 2.15 (95% CI 1.06, 4.34). A similar study reported a consistent result, with a risk estimate of 2.27 (95% CI 0.99, 5.20). Other risk factors for NAION, such as the presence of “crowded” optic disc, may have contributed to the occurrence of NAION in these studies.
Neither the rare postmarketing reports, nor the association of PDE5 inhibitor use and NAION in the observational studies, substantiate a causal relationship between PDE5 inhibitor use and NAION [see Adverse Reactions (6.2)] .
Physicians should consider whether their patients with underlying NAION risk factors could be adversely affected by use of PDE5 inhibitors. Individuals who have already experienced NAION are at increased risk of NAION recurrence. Therefore, PDE5 inhibitors, including vardenafil hydrochloride tablets, should be used with caution in these patients and only when the anticipated benefits outweigh the risks. Individuals with "crowded" optic disc are also considered at greater risk for NAION compared to the general population, however, evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including vardenafil hydrochloride tablets, for this uncommon condition.
Vardenafil hydrochloride tablets have not been evaluated in patients with known hereditary degenerative retinal disorders, including retinitis pigmentosa, therefore its use is not recommended until further information is available in those patients.
5.8 Hepatic Impairment
Dosage adjustment is necessary in patients with moderate hepatic impairment (Child-Pugh B). Do not use vardenafil hydrochloride tablets in patients with severe (Child-Pugh C) hepatic impairment. [See Dosage and Administration (2.3) Clinical Pharmacology (12.3)] and Use in Specific Populations (8.6)].
8.6 Hepatic Impairment
Dosage adjustment is necessary in patients with moderate hepatic impairment.
Do not use vardenafil hydrochloride tablets in patients with severe hepatic impairment (Child-Pugh C). Vardenafil has not been evaluated in this patient population.
A starting dose of 5 mg is recommended in patients with moderate hepatic impairment (Child-Pugh B) and the maximum dose should not exceed 10 mg. In volunteers with moderate hepatic impairment, the C max and AUC following a 10 mg vardenafil dose were increased by 130% and 160%, respectively, compared to healthy control subjects. [See Warnings and Precautions (5.8) and Dosage and Administration (2.3).]
In volunteers with mild hepatic impairment (Child-Pugh A), the C max and AUC following a 10 mg vardenafil dose were increased by 22% and 17%, respectively, compared to healthy control subjects. No dosage adjustment is necessary in patients with mild hepatic impairment.
1 Indications and Usage
Vardenafil hydrochloride tablets are indicated for the treatment of erectile dysfunction.
5.5 Sudden Hearing Loss
Physicians should advise patients to stop taking all PDE5 inhibitors, including vardenafil hydrochloride tablets, and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including vardenafil. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions (6.2)].
12.1 Mechanism of Action
Penile erection is a hemodynamic process initiated by the relaxation of smooth muscle in the corpus cavernosum and its associated arterioles. During sexual stimulation, nitric oxide is released from nerve endings and endothelial cells in the corpus cavernosum. Nitric oxide activates the enzyme guanylate cyclase resulting in increased synthesis of cyclic guanosine monophosphate (cGMP) in the smooth muscle cells of the corpus cavernosum. The cGMP in turn triggers smooth muscle relaxation, allowing increased blood flow into the penis, resulting in erection. The tissue concentration of cGMP is regulated by both the rates of synthesis and degradation via phosphodiesterases (PDEs). The most abundant PDE in the human corpus cavernosum is the cGMP-specific phosphodiesterase type 5 (PDE5); therefore, the inhibition of PDE5 enhances erectile function by increasing the amount of cGMP. Because sexual stimulation is required to initiate the local release of nitric oxide, the inhibition of PDE5 has no effect in the absence of sexual stimulation.
In vitro studies have shown that vardenafil is a selective inhibitor of PDE5. The inhibitory effect of vardenafil is more selective on PDE5 than for other known phosphodiesterases (>15-fold relative to PDE6, >130-fold relative to PDE1, >300-fold relative to PDE11, and >1,000-fold relative to PDE2, 3, 4, 7, 8, 9, and 10).
5.11 Effects On Bleeding
In humans, vardenafil alone in doses up to 20 mg does not prolong the bleeding time. There is no clinical evidence of any additive prolongation of the bleeding time when vardenafil is administered with aspirin. Vardenafil hydrochloride tablets have not been administered to patients with bleeding disorders or significant active peptic ulceration. Therefore vardenafil hydrochloride tablets should be administered to these patients after careful benefit-risk assessment.
5 Warnings and Precautions
The evaluation of erectile dysfunction should include a medical assessment, a determination of potential underlying causes and the identification of appropriate treatment.
Before prescribing vardenafil hydrochloride tablets, it is important to note the following:
2 Dosage and Administration
- Vardenafil hydrochloride tablets are taken as needed: For most patients, the starting dose is 10 mg, up to once daily. Increase to 20 mg or decrease to 5 mg based on efficacy/tolerability. ( 2.1)
- A starting dose of 5 mg vardenafil hydrochloride tablets should be considered in patients ≥ 65 years of age. ( 2.3)
- Vardenafil hydrochloride tablets are taken orally, approximately 60 minutes before sexual activity. ( 2.1)
- The maximum recommended dosing frequency is one tablet per day. ( 2.1)
- Vardenafil hydrochloride tablets may be taken with or without food. ( 2.2)
- If taking potent or moderate inhibitors of CYP3A4, the dose of vardenafil hydrochloride tablets should be adjusted as follows (
2.4,
5.2,
7.2):
- Ritonavir: No more than 2.5 mg in a 72-hour period
- Indinavir, saquinavir, atazanavir, ketoconazole 400 mg daily, itraconazole 400 mg daily, clarithromycin: No more than 2.5 mg in a 24-hour period
- Ketoconazole 200 mg daily, itraconazole 200 mg daily, erythromycin: No more than 5 mg in a 24-hour period.
- In patients on stable alpha-blocker therapy the recommended starting dose of vardenafil hydrochloride tablets is 5 mg ( 2.4, 5.6)
- The recommended starting dose of vardenafil hydrochloride tablets is 5 mg in patients with moderate hepatic impairment (Child-Pugh B). The maximum dose in patients with moderate hepatic impairment should not exceed 10 mg. ( 2.3, 8.6)
2.1 General Dose Information
For most patients, the recommended starting dose of vardenafil hydrochloride tablets is 10 mg, taken orally, as needed, approximately 60 minutes before sexual activity. The dose may be increased to a maximum recommended dose of 20 mg or decreased to 5 mg based on efficacy and side effects. The maximum recommended dosing frequency is once per day. Sexual stimulation is required for a response to treatment.
3 Dosage Forms and Strengths
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side corresponding to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of vardenafil hydrochloride tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure.
To report SUSPECTED ADVERSE REACTIONS, contact AvKARE at 1-855-361-3993; email [email protected]; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
8 Use in Specific Populations
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Vardenafil hydrochloride tablets were administered to over 4430 men (mean age 56, range 18-89 years; 81% White, 6% Black, 2% Asian, 2% Hispanic and 9% Other) during controlled and uncontrolled clinical trials worldwide. Over 2200 patients were treated for 6 months or longer and 880 patients were treated for at least 1 year.
In placebo-controlled clinical trials, the discontinuation rate due to adverse events was 3.4% for vardenafil hydrochloride tablets compared to 1.1% for placebo.
When vardenafil hydrochloride tablets were taken as recommended in placebo-controlled clinical trials, the following adverse reactions were reported (see Table 1).
| Adverse Reaction | Percentage of Patients Reporting Reactions | |
|---|---|---|
| Placebo
N = 1199 |
Vardenafil Hydrochloride Tablets
N = 2203 |
|
| Headache | 4% | 15% |
| Flushing | 1% | 11% |
| Rhinitis | 3% | 9% |
| Dyspepsia | 1% | 4% |
| Accidental Injury
All the events listed in the above table were deemed to be adverse drug reactions with the exception of accidental injury.
|
2% | 3% |
| Sinusitis | 1% | 3% |
| Flu Syndrome | 2% | 3% |
| Dizziness | 1% | 2% |
| Increased Creatine Kinase | 1% | 2% |
| Nausea | 1% | 2% |
Back pain was reported in 2.0% of patients treated with vardenafil hydrochloride tablets and 1.7% of patients on placebo.
Placebo-controlled trials suggested a dose effect in the incidence of some adverse reactions (headache, flushing, dyspepsia, nausea, and rhinitis) over the 5 mg, 10 mg, and 20 mg doses of vardenafil hydrochloride tablets.
17 Patient Counseling Information
5.12 Sexually Transmitted Disease
The use of vardenafil hydrochloride tablets offers no protection against sexually transmitted diseases. Counseling of patients about protective measures necessary to guard against sexually transmitted diseases, including the Human Immunodeficiency Virus (HIV), should be considered.
16 How Supplied/storage and Handling
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side equivalent to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively
| Package | Strength | NDC Code | |
|---|---|---|---|
| Bottles of 30 | 10 mg | 42291-847-30 | |
| 20 mg | 42291-848-30 |
4.2 Guanylate Cyclase (gc) Stimulators
Do not use vardenafil hydrochloride tablets in patients who are using a GC stimulator, such as riociguat. PDE5 inhibitors, including vardenafil hydrochloride tablets may potentiate the hypotensive effects of GC stimulators.
5.7 Congenital Or Acquired Qt Prolongation
In a study of the effect of vardenafil hydrochloride tablets on QT interval in 59 healthy males [see Clinical Pharmacology (12.2)] , therapeutic (10 mg) and supratherapeutic (80 mg) doses of vardenafil and the active control moxifloxacin (400 mg) produced similar increases in QTc interval. A postmarketing study evaluating the effect of combining vardenafil hydrochloride tablets with another drug of comparable QT effect showed an additive QT effect when compared with either drug alone [see Clinical Pharmacology (12.2)] . These observations should be considered in clinical decisions when prescribing vardenafil hydrochloride tablets to patients with known history of QT prolongation or patients who are taking medications known to prolong the QT interval.
Patients taking Class 1A (for example. quinidine, procainamide) or Class III (for example, amiodarone, sotalol) antiarrhythmic medications or those with congenital QT prolongation, should avoid using vardenafil hydrochloride tablets.
5.13 risks in Patients With Phenylketonuria
Phenylalanine can be harmful to patients with phenylketonuria (PKU). Vardenafil hydrochloride tablets contain phenylalanine, a component of aspartame. Each 2.5 mg vardenafil hydrochloride tablet contains 0.7 mg phenylalanine; each 5 mg tablet contains 1.4 mg phenylalanine; each 10 mg tablet contains 2.8 mg phenylalanine; each 20 mg tablet contains 5.6 mg phenylalanine. Before prescribing vardenafil hydrochloride tablets in a patient with PKU, consider the combined daily amount of phenylalanine from all sources, including vardenafil hydrochloride tablets.ardenafil hydrochloride tablets contain aspartame, a source of phenylalanine. Each 2.5 mg vardenafil hydrochloride tablet contains 0.7 mg phenylalanine; each 5 mg tablet contains 1.4 mg phenylalanine; each 10 mg tablet contains 2.8 mg phenylalanine; each 20 mg tablet contains 5.6 mg phenylalanine.
Principal Display Panel 10 Mg Tablet Bottle Label
Principal Display Panel 20 Mg Tablet Bottle Label
14.2 Trial in Patients With Ed and Diabetes Mellitus
Vardenafil hydrochloride tablets demonstrated clinically meaningful and statistically significant improvement in erectile function in a prospective, fixed-dose (10 and 20 mg vardenafil hydrochloride tablets), double-blind, placebo-controlled trial of patients with diabetes mellitus (n=439; mean age 57 years, range 33-81; 80% White, 9% Black, 8% Hispanic, and 3% Other).
Significant improvements in the EF Domain were shown in this study (EF Domain scores of 17 on 10 mg vardenafil hydrochloride tablets and 19 on 20 mg vardenafil hydrochloride tablets compared to 13 on placebo; p <0.0001).
Vardenafil hydrochloride tablets significantly improved the overall per-patient rate of achieving an erection sufficient for penetration (SEP2) (61% on 10 mg and 64% on 20 mg vardenafil hydrochloride tablets compared to 36% on placebo; p <0.0001).
Vardenafil hydrochloride tablets demonstrated a clinically meaningful and statistically significant increase in the overall per-patient rate of maintenance of erection to successful intercourse (SEP3) (49% on 10 mg, 54% on 20 mg vardenafil hydrochloride tablets compared to 23% on placebo; p <0.0001).
14.1 Trials in A General Erectile Dysfunction Population
In the major North American fixed-dose trial, 762 patients (mean age 57, range 20-83 years; 79% White, 13% Black, 4% Hispanic, 2% Asian and 2% Other) were evaluated. The mean baseline EF Domain scores were 13, 13, 13, 14 for the vardenafil hydrochloride tablets 5 mg, 10 mg, 20 mg and placebo groups, respectively. There was significant improvement (p <0.0001) at 3 months with vardenafil hydrochloride tablets (EF Domain scores of 18, 21, 21, for the 5 mg, 10 mg, and 20 mg dose groups, respectively) compared to the placebo group (EF Domain score of 15). The European trial (total N=803) confirmed these results. The improvement in mean score was maintained at all doses at 6 months in the North American trial.
In the North American trial, vardenafil hydrochloride tablets significantly improved the rates of achieving an erection sufficient for penetration (SEP2) at doses of 5 mg, 10 mg, and 20 mg compared to placebo (65%, 75%, and 80%, respectively, compared to a 52% response in the placebo group at 3 months; p <0.0001). The European trial confirmed these results.
Vardenafil hydrochloride tablets demonstrated a clinically meaningful and statistically significant increase in the overall per-patient rate of maintenance of erection to successful intercourse (SEP3) (51% on 5 mg, 64% on 10 mg, and 65% on 20 mg, respectively, compared to 32% on placebo; p <0.0001) at 3 months in the North American trial. The European trial showed comparable efficacy. This improvement in mean score was maintained at all doses at 6 months in the North American trial.
14.3 Trial in Patients With Ed After Radical Prostatectomy
Vardenafil hydrochloride tablets demonstrated clinically meaningful and statistically significant improvement in erectile function in a prospective, fixed-dose (10 and 20 mg vardenafil hydrochloride tablets), double-blind, placebo-controlled trial in post-prostatectomy patients (n=427, mean age 60, range 44-77 years; 93% White, 5% Black, 2% Other).
Significant improvements in the EF Domain were shown in this study (EF Domain scores of 15 on 10 mg vardenafil hydrochloride tablets and 15 on 20 mg vardenafil hydrochloride tablets compared to 9 on placebo; p <0.0001).
Vardenafil hydrochloride tablets significantly improved the overall per-patient rate of achieving an erection sufficient for penetration (SEP2) (47% on 10 mg and 48% on 20 mg vardenafil hydrochloride tablets compared to 22% on placebo; p <0.0001).
Vardenafil hydrochloride tablets demonstrated a clinically meaningful and statistically significant increase in the overall per-patient rate of maintenance of erection to successful intercourse (SEP3) (37% on 10 mg, 34% on 20 mg vardenafil hydrochloride tablets compared to 10% on placebo; p <0.0001).
5.10 Combination With Other Erectile Dysfunction Therapies
The safety and efficacy of vardenafil hydrochloride tablets used in combination with other treatments for erectile dysfunction have not been studied. Therefore, the use of such combinations is not recommended.
5.2 Potential for Drug Interactions With Potent Or Moderate Cyp3a4 Inhibitors
Concomitant administration with potent CYP3A4 inhibitors (such as ritonavir, indinavir, ketoconazole) or moderate CYP3A4 inhibitors (such as erythromycin) increases plasma concentrations of vardenafil. Dosage adjustment is necessary when vardenafil hydrochloride tablets are administered with certain CYP3A4 inhibitors [see Dosage and Administration (2.4), Drug Interactions (7.2)].
Long-term safety information is not available on the concomitant administration of vardenafil with HIV protease inhibitors.
Structured Label Content
Section 34083-6 (34083-6)
Carcinogenesis
Vardenafil was not carcinogenic in rats and mice when administered daily for 24 months. In these studies systemic drug exposures (AUCs) for unbound (free) vardenafil and its major metabolite were approximately 400- and 170-fold for male and female rats, respectively, and 21- and 37-fold for male and female mice, respectively, the exposures observed in human males given the Maximum Recommended Human Dose (MRHD) of 20 mg.
Section 42229-5 (42229-5)
Geriatrics: A starting dose of 5 mg vardenafil hydrochloride tablets should be considered in patients ≥65 years of age [see Use in Specific Populations (8.5)].
Section 42230-3 (42230-3)
FDA-approved patient labeling
VARDENAFIL HYDROCHLORIDE
(var-DEN-a-fil HYE-droe-KLOR-ide)
TABLETS
Read the Patient Information about vardenafil hydrochloride tablets before you start taking them and again each time you get a refill. There may be new information. You may also find it helpful to share this information with your partner. This leaflet does not take the place of talking with your doctor. You and your doctor should talk about vardenafil hydrochloride tablets when you start taking it and at regular checkups. If you do not understand the information, or have questions, talk with your doctor or pharmacist.
WHAT IMPORTANT INFORMATION SHOULD YOU KNOW ABOUT VARDENAFIL HYDROCHLORIDE TABLETS?
Vardenafil hydrochloride tablets can cause your blood pressure to drop suddenly to an unsafe level if it is taken with certain other medicines. With a sudden drop in blood pressure, you could get dizzy, faint, or have a heart attack or stroke.
Do not take vardenafil hydrochloride tablets if you:
- Take any medicines called "nitrates" (often used to control chest pain, also known as angina).
- Use recreational drugs called "poppers" like amyl nitrate and butyl nitrate.
- Take riociguat (Adempas ®), a guanylate cyclase stimulator, a medicine that treats pulmonary arterial hypertension and chronic-thromboembolic pulmonary hypertension.
(See " Who Should Not Take Vardenafil Hydrochloride Tablets?")
Tell all your healthcare providers that you take vardenafil hydrochloride tablets. If you need emergency medical care for a heart problem, it will be important for your healthcare provider to know when you last took vardenafil hydrochloride tablets.
WHAT ARE VARDENAFIL HYDROCHLORIDE TABLETS?
Vardenafil hydrochloride tablets are a prescription medicine taken by mouth for the treatment of erectile dysfunction (ED) in men.
ED is a condition where the penis does not harden and expand when a man is sexually excited, or when he cannot keep an erection. A man who has trouble getting or keeping an erection should see his doctor for help if the condition bothers him. Vardenafil hydrochloride tablets may help a man with ED get and keep an erection when he is sexually excited.
Vardenafil hydrochloride tablets do not:
- Cure ED
- Increase a man's sexual desire
- Protect a man or his partner from sexually transmitted diseases, including HIV. Speak to your doctor about ways to guard against sexually transmitted diseases.
- Serve as a male form of birth control.
Vardenafil hydrochloride tablets are only for men with ED. Vardenafil hydrochloride tablets are not for women or children. Vardenafil hydrochloride tablets must be used only under a doctor's care.
HOW DO VARDENAFIL HYDROCHLORIDE TABLETS WORK?
When a man is sexually stimulated, his body's normal physical response is to increase blood flow to his penis. This results in an erection. Vardenafil hydrochloride tablets help increase blood flow to the penis and may help men with ED get and keep an erection satisfactory for sexual activity. Once a man has completed sexual activity, blood flow to his penis decreases, and his erection goes away.
WHO CAN TAKE VARDENAFIL HYDROCHLORIDE TABLETS?
Talk to your doctor to decide if vardenafil hydrochloride tablets are right for you.
Vardenafil hydrochloride tablets have been shown to be effective in men over the age of 18 years who have erectile dysfunction, including men with diabetes or who have undergone prostatectomy.
WHO SHOULD NOT TAKE VARDENAFIL HYDROCHLORIDE TABLETS?
Do not take vardenafil hydrochloride tablets if you:
-
Take any medicines called "nitrates" (See "
What important information should you know about vardenafil hydrochloride tablets?
"). Nitrates are commonly used to treat angina. Angina is a symptom of heart disease and can cause pain in your chest, jaw, or down your arm.
Medicines called nitrates include nitroglycerin that is found in tablets, sprays, ointments, pastes, or patches. Nitrates can also be found in other medicines such as isosorbide dinitrate or isosorbide mononitrate. Some recreational drugs called "poppers" also contain nitrates, such as amyl nitrate and butyl nitrate. Do not use vardenafil hydrochloride tablets if you are using these drugs. Ask your doctor or pharmacist if you are not sure if any of your medicines are nitrates. - Take riociguat, a guanylate cyclase stimulator, a medicine that treats pulmonary arterial hypertension and chronic-throembolic pulmonary hypertension.
- Have been told by your healthcare provider to not have sexual activity because of health problems. Sexual activity can put an extra strain on your heart, especially if your heart is already weak from a heart attack or heart disease.
WHAT SHOULD YOU DISCUSS WITH YOUR DOCTOR BEFORE TAKING VARDENAFIL HYDROCHLORIDE TABLETS?
Before taking vardenafil hydrochloride tablets, tell your doctor about all your medical problems, including if you:
- Have heart problems such as angina, heart failure, irregular heartbeats, or have had a heart attack. Ask your doctor if it is safe for you to have sexual activity.
- Have low blood pressure or have high blood pressure that is not controlled.
- Have pulmonary hypertension.
- Have had a stroke.
- Have had a seizure.
- Or any family members have a rare heart condition known as prolongation of the QT interval (long QT syndrome).
- Have liver problems.
- Have kidney problems and require dialysis.
- Have retinitis pigmentosa, a rare genetic (runs in families) eye disease
- Have ever had severe vision loss, or if you have an eye condition called non-arteritic anterior ischemic optic neuropathy (NAION).
- Have stomach ulcers.
- Have a bleeding problem.
- Have a deformed penis shape or Peyronie's disease.
- Have had an erection that lasted more than 4 hours.
- Have blood cell problems such as sickle cell anemia, multiple myeloma, or leukemia.
- Have hearing problems.
CAN OTHER MEDICATIONS AFFECT VARDENAFIL HYDROCHLORIDE TABLETS?
Tell your doctor about all the medicines you take including prescription and non-prescription medicines, vitamins, and herbal supplements. Vardenafil hydrochloride tablets and other medicines may affect each other. Always check with your doctor before starting or stopping any medicines. Especially tell your doctor if you take any of the following:
- Medicines called nitrates (see " What important information should you know about vardenafil hydrochloride tablets?").
- Ketoconazole or itraconazole (such as Nizoral ® or Sporanox ®).
- Ritonavir (Norvir ®) or indinavir sulfate (Crixivan ®) saquinavir (Fortavase ® or Invirase ®) or atazanavir (Reyataz ®).
- Erythromycin or clarithromycin.
- Medicines called alpha-blockers. These include Hytrin ® (terazosin HCl), Flomax ® (tamsulosin HCl), Cardura ® (doxazosin mesylate), Minipress ® (prazosin HCl), Rapaflo ® (silodosin) or Uroxatral ® (alfuzosin HCl). Alpha-blockers are sometimes prescribed for prostate problems or high blood pressure. In some patients the use of PDE5 inhibitor drugs, including vardenafil hydrochloride tablets, with alpha-blockers can lower blood pressure significantly leading to fainting.
- You should contact the prescribing physician if alpha-blockers or other drugs that lower blood pressure are prescribed by another healthcare provider.
- Medicines that treat abnormal heartbeat. These include quinidine, procainamide, amiodarone and sotalol.
- Other medicines or treatments for ED.
HOW SHOULD YOU TAKE VARDENAFIL HYDROCHLORIDE TABLETS?
Take vardenafil hydrochloride tablets exactly as your doctor prescribes. Do not take more than one vardenafil hydrochloride tablet a day. Doses should be taken at least 24 hours apart. Some men can only take a low dose of vardenafil hydrochloride tablets because of medical conditions or medicines they take. Your doctor will prescribe the dose that is right for you.
- If you are older than 65 or have liver problems, your doctor may start you on a lower dose of vardenafil hydrochloride tablets.
- If you have prostate problems or high blood pressure, for which you take medicines called alpha-blockers, your doctor may start you on a lower dose of vardenafil hydrochloride tablets.
- If you are taking certain other medicines your doctor may prescribe a lower starting dose and limit you to one dose of vardenafil hydrochloride tablets in a 72-hour (3 days) period.
Take 1 vardenafil hydrochloride tablet about 1 hour (60 minutes) before sexual activity. Some form of sexual stimulation is needed for an erection to happen with vardenafil hydrochloride tablet. Vardenafil hydrochloride tablets may be taken with or without meals.
Do not change your dose of vardenafil hydrochloride tablets without talking to your doctor. Your doctor may lower your dose or raise your dose, depending on how your body reacts to vardenafil hydrochloride tablets.
Call your doctor or emergency room immediately if you accidentally took more vardenafil hydrochloride tablets than prescribed.
WHAT ARE THE POSSIBLE SIDE EFFECTS OF VARDENAFIL HYDROCHLORIDE TABLETS?
The most common side effects with vardenafil hydrochloride tablets are headache, flushing, stuffy or runny nose, indigestion, upset stomach, dizziness or back pain. These side effects usually go away after a few hours. Call your doctor if you get a side effect that bothers you or one that will not go away.
Vardenafil hydrochloride tablets may uncommonly cause:
- An erection that won't go away (priapism). If you get an erection that lasts more than 4 hours, get medical help right away. Priapism must be treated as soon as possible or lasting damage can happen to your penis including the inability to have erections.
- Color vision changes, such as seeing a blue tinge to objects or having difficulty telling the difference between the colors blue and green.
In rare instances, men taking PDE5 inhibitors (oral erectile dysfunction medicines, including vardenafil hydrochloride tablets) reported a sudden decrease or loss of vision in one or both eyes. It is uncertain whether PDE5 inhibitors directly cause the vision loss. If you experience sudden decrease or loss of vision, stop taking PDE5 inhibitors, including vardenafil hydrochloride tablets, and call a doctor right away.
Sudden loss or decrease in hearing, sometimes with ringing in the ears and dizziness, has been rarely reported in people taking PDE5 inhibitors, including vardenafil hydrochloride tablets. It is not possible to determine whether these events are related directly to the PDE5 inhibitors, to other diseases or medications, to other factors, or to a combination of factors. If you experience these symptoms, stop taking vardenafil hydrochloride tablets and contact a doctor right away.
These are not all the side effects of vardenafil hydrochloride tablets. For more information, ask your doctor or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
HOW SHOULD VARDENAFIL HYDROCHLORIDE TABLETS BE STORED?
- Store vardenafil hydrochloride tablets at room temperature between 59–86° F (15–30° C).
- Keep vardenafil hydrochloride tablets and all medicines out of the reach of children.
GENERAL INFORMATION ABOUT VARDENAFIL HYDROCHLORIDE TABLETS
Medicines are sometimes prescribed for conditions other than those described in patient information leaflets. Do not use vardenafil hydrochloride tablets for a condition for which it was not prescribed. Do not give vardenafil hydrochloride tablets to other people, even if they have the same symptoms that you have. It may harm them.
This leaflet summarizes the most important information about vardenafil hydrochloride tablets. If you would like more information, talk with your healthcare provider. You can ask your doctor or pharmacist for information about vardenafil hydrochloride tablets that is written for health professionals.
WHAT ARE THE INGREDIENTS OF VARDENAFIL HYDROCHLORIDE TABLETS?
Active Ingredient: vardenafil hydrochloride
Inactive Ingredients: microcrystalline cellulose, crospovidone, colloidal silicon dioxide, magnesium stearate, aspartame, titanium dioxide, ferric oxide red, and ferric oxide yellow.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Products cited in vardenafil hydrochloride tablets USPI
Norvir (ritonavir) is a trademark of Abbott Laboratories
Crixivan (indinavir sulfate) is a trademark of Merck & Co., Inc.
Invirase or Fortavase (saquinavir mesylate) is a trademark of Roche Laboratories Inc.
Reyataz (atazanavir sulfate) is a trademark of Bristol-Myers Squibb Company
Nizoral (ketoconazole) is a trademark of Johnson & Johnson
Sporanox (itraconazole) is a trademark of Johnson & Johnson
Hytrin (terazosin HCl) is a trademark of Abbott Laboratories
Flomax (tamsulosin HCl) is a trademark of Yamanouchi Pharmaceutical Co., Ltd.
Cardura (doxazosin mesylate) is a trademark of Pfizer Inc.
Minipress (prazosin HCl) is a trademark of Pfizer Inc.
Rapaflo (silodosin) is a trademark of Watson Pharma Inc.
Uroxatral (alfuzosin HCl) is a trademark of Sanofi-Synthelabo
Manufactured For:
AvKARE
Pulaski, TN 38478
Mfg. Rev. 10/18
AV Rev. 04/2020 (P)
Section 43683-2 (43683-2)
| Warnings and Precautions, Effects on the Eye ( 5.4) | 08/2017 |
Section 44425-7 (44425-7)
Store at 25°C (77°F); excursions permitted within15-30°C (59-86°F) [see USP Controlled Room Temperature].
4.1 Nitrates
Administration of vardenafil hydrochloride tablets with nitrates (either regularly and/or intermittently) and nitric oxide donors is contraindicated [see Clinical Pharmacology (12.2)] . Consistent with the effects of PDE5 inhibition on the nitric oxide/cyclic guanosine monophosphate pathway, PDE5 inhibitors, including vardenafil hydrochloride tablets, may potentiate the hypotensive effects of nitrates. A suitable time interval following dosing of vardenafil hydrochloride tablets for the safe administration of nitrates or nitric oxide donors has not been determined.
10 Overdosage (10 OVERDOSAGE)
The maximum dose of vardenafil hydrochloride tablets for which human data are available is a single 120 mg dose administered to healthy male volunteers. The majority of these subjects experienced reversible back pain/myalgia and/or "abnormal vision." Single doses up to 80 mg vardenafil and multiple doses up to 40 mg vardenafil administered once daily over 4 weeks were tolerated without producing serious adverse side effects.
When 40 mg of vardenafil was administered twice daily, cases of severe back pain were observed. No muscle or neurological toxicity was identified.
In cases of overdose, standard supportive measures should be taken as required. Renal dialysis is not expected to accelerate clearance as vardenafil is highly bound to plasma proteins and not significantly eliminated in the urine.
11 Description (11 DESCRIPTION)
Vardenafil hydrochloride tablets (vardenafil hydrochloride) are administered orally for the treatment of erectile dysfunction. This monohydrochloride salt of vardenafil is a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5).
Vardenafil HCl is designated chemically as piperazine, 1-[[3-(1,4-dihydro-5-methyl-4-oxo-7-propylimidazo[5,1- f][1,2,4]triazin-2-yl)-4-ethoxyphenyl]sulfonyl]-4-ethyl-, monohydrochloride and has the following structural formula:
Vardenafil HCl is a nearly colorless, solid substance with a molecular weight of 579.1 g/mol and a solubility of 0.11 mg/mL in water.
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side corresponding to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively. In addition to the active ingredient, vardenafil HCl, each tablet contains microcrystalline cellulose, crospovidone, colloidal silicon dioxide, magnesium stearate, aspartame, titanium dioxide, ferric oxide red, and ferric oxide yellow.
2.2 Use With Food (2.2 Use with Food)
Vardenafil hydrochloride tablets can be taken with or without food.
8.4 Pediatric Use
Vardenafil hydrochloride tablets are not indicated for use in pediatric patients. Safety and efficacy have not been established in this population.
8.5 Geriatric Use
Elderly males 65 years of age and older have higher vardenafil plasma concentrations than younger males (18 – 45 years), mean C max and AUC were 34% and 52% higher, respectively. Phase 3 clinical trials included more than 834 elderly patients, and no differences in safety or effectiveness of vardenafil hydrochloride tablets 5, 10, or 20 mg were noted when these elderly patients were compared to younger patients. However, due to increased vardenafil concentrations in the elderly, a starting dose of 5 mg vardenafil hydrochloride tablets should be considered in patients ≥65 years of age [see Clinical Pharmacology (12.3)].
5.6 Alpha Blockers (5.6 Alpha-Blockers)
Caution is advised when PDE5 inhibitors are co-administered with alpha-blockers. PDE5 inhibitors, including vardenafil hydrochloride tablets, and alpha-adrenergic blocking agents are both vasodilators with blood-pressure lowering effects. When vasodilators are used in combination, an additive effect on blood pressure may be anticipated. In some patients, concomitant use of these two drug classes can lower blood pressure significantly leading to symptomatic hypotension (for example, fainting) [see Drug Interactions (7.1) and Clinical Pharmacology (12.2)] . Consideration should be given to the following:
- Patients should be stable on alpha-blocker therapy prior to initiating a PDE5 inhibitor. Patients who demonstrate hemodynamic instability on alpha-blocker therapy alone are at increased risk of symptomatic hypotension with concomitant use of PDE5 inhibitors.
- In those patients who are stable on alpha-blocker therapy, PDE5 inhibitors should be initiated at the lowest recommended starting dose [see Dosage and Administration (2.4)] .
- In those patients already taking an optimized dose of PDE5 inhibitor, alpha-blocker therapy should be initiated at the lowest dose. Stepwise increase in alpha-blocker dose may be associated with further lowering of blood pressure in patients taking a PDE5 inhibitor.
- Safety of combined use of PDE5 inhibitors and alpha-blockers may be affected by other variables, including intravascular volume depletion and other anti-hypertensive drugs.
14 Clinical Studies (14 CLINICAL STUDIES)
Vardenafil hydrochloride tablets were evaluated in four major double-blind, randomized, placebo-controlled, fixed-dose, parallel design, multicenter trials in 2431 men aged 20-83 (mean age 57 years; 78% White, 7% Black, 2% Asian, 3% Hispanic and 10% Other/Unknown). The doses of vardenafil hydrochloride tablets in these studies were 5 mg, 10 mg, and 20 mg. Two of these trials were conducted in the general erectile dysfunction (ED) population and two in special ED populations (one in patients with diabetes mellitus and one in post-prostatectomy patients). Vardenafil hydrochloride tablets were dosed without regard to meals on an as needed basis in men with ED, many of whom had multiple other medical conditions. The primary endpoints were assessed at 3 months.
Primary efficacy assessment in all four major trials was by means of the Erectile Function (EF) Domain score of the validated International Index of Erectile Function (IIEF) Questionnaire and two questions from the Sexual Encounter Profile (SEP) dealing with the ability to achieve vaginal penetration (SEP2), and the ability to maintain an erection long enough for successful intercourse (SEP3).
In all four fixed-dose efficacy trials, vardenafil hydrochloride tablets showed clinically meaningful and statistically significant improvement in the EF Domain, SEP2, and SEP3 scores compared to placebo. The mean baseline EF Domain score in these trials was 11.8 (scores range from 0-30 where lower scores represent more severe disease). Vardenafil hydrochloride tablets (5 mg, 10 mg, and 20 mg) were effective in all age categories (<45, 45 to <65, and ≥65 years) and was also effective regardless of race (White, Black, Other).
4 Contraindications (4 CONTRAINDICATIONS)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions with the use of vardenafil hydrochloride tablets (vardenafil) are discussed elsewhere in the labeling:
- Cardiovascular Effects [see Contraindications (4.1) and Warnings and Precautions (5.1)]
- Priapism [see Warnings and Precautions (5.3)]
- Effects on Eye [see Warnings and Precautions (5.4)]
- Sudden Hearing Loss [see Warnings and Precautions (5.5)]
- QT Prolongation [see Warnings and Precautions (5.7)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Vardenafil hydrochloride tablets can potentiate the hypotensive effects of nitrates, alpha- blockers, and antihypertensives. ( 7.1)
8.3 Nursing Mothers
Vardenafil hydrochloride tablets are not indicated for use in women. It is not known if vardenafil is excreted in human breast milk .
Vardenafil was secreted into the milk of lactating rats at concentrations approximately 10-fold greater than found in the plasma. Following a single oral dose of 3 mg/kg, 3.3% of the administered dose was excreted into the milk within 24 hours.
5.3 Risk of Priapism
There have been rare reports of prolonged erections greater than 4 hours and priapism (painful erections greater than 6 hours in duration) for this class of compounds, including vardenafil. In the event that an erection persists longer than 4 hours, the patient should seek immediate medical assistance. If priapism is not treated immediately, penile tissue damage and permanent loss of potency may result.
Vardenafil hydrochloride tablets should be used with caution by patients with anatomical deformation of the penis (such as angulation, cavernosal fibrosis, or Peyronie's disease) or by patients who have conditions that may predispose them to priapism (such as sickle cell anemia, multiple myeloma, or leukemia).
5.9 Renal Impairment
Do not use vardenafil hydrochloride tablets in patients on renal dialysis, as vardenafil has not been evaluated in this population [see Dosage and Administration (2.3) and Use in Specific Populations (8.7)].
8.7 Renal Impairment
Do not use vardenafil hydrochloride tablets in patients on renal dialysis as vardenafil has not been evaluated in such patients.
No dosage adjustment is necessary in patients with creatinine clearance (CLcr) of 30–80 mL/min. In male volunteers with CLcr = 50-80 ml/min, the pharmacokinetics of vardenafil were similar to those observed in a control group with CLcr >80 mL/min. In male volunteers with CLcr = 30-50 mL/min or CLcr <30 mL/min, the AUC of vardenafil was 20–30% higher compared to that observed in a control group with CLcr >80 mL/min. [See Dosage and Administration (2.3) and Warnings and Precautions (5.9).]
12.3 Pharmacokinetics
The pharmacokinetics of vardenafil are approximately dose proportional over the recommended dose range.
5.4 Effects On the Eye (5.4 Effects on the Eye)
Physicians should advise patients to stop use of all phosphodiesterase type 5 (PDE5) inhibitors, including vardenafil hydrochloride tablets, and seek medical attention in the event of sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a rare condition and a cause of decreased vision, including permanent loss of vision, that has been reported rarely postmarketing in temporal association with the use of all PDE5 inhibitors. Based on published literature, the annual incidence of NAION is 2.5–11.8 cases per 100,000 in males aged ≥50.
An observational case-crossover study evaluated the risk of NAION when PDE5 inhibitor use, as a class, occurred immediately before NAION onset (within 5 half-lives), compared to PDE5 inhibitor use in a prior time period. The results suggest an approximate 2-fold increase in the risk of NAION, with a risk estimate of 2.15 (95% CI 1.06, 4.34). A similar study reported a consistent result, with a risk estimate of 2.27 (95% CI 0.99, 5.20). Other risk factors for NAION, such as the presence of “crowded” optic disc, may have contributed to the occurrence of NAION in these studies.
Neither the rare postmarketing reports, nor the association of PDE5 inhibitor use and NAION in the observational studies, substantiate a causal relationship between PDE5 inhibitor use and NAION [see Adverse Reactions (6.2)] .
Physicians should consider whether their patients with underlying NAION risk factors could be adversely affected by use of PDE5 inhibitors. Individuals who have already experienced NAION are at increased risk of NAION recurrence. Therefore, PDE5 inhibitors, including vardenafil hydrochloride tablets, should be used with caution in these patients and only when the anticipated benefits outweigh the risks. Individuals with "crowded" optic disc are also considered at greater risk for NAION compared to the general population, however, evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including vardenafil hydrochloride tablets, for this uncommon condition.
Vardenafil hydrochloride tablets have not been evaluated in patients with known hereditary degenerative retinal disorders, including retinitis pigmentosa, therefore its use is not recommended until further information is available in those patients.
5.8 Hepatic Impairment
Dosage adjustment is necessary in patients with moderate hepatic impairment (Child-Pugh B). Do not use vardenafil hydrochloride tablets in patients with severe (Child-Pugh C) hepatic impairment. [See Dosage and Administration (2.3) Clinical Pharmacology (12.3)] and Use in Specific Populations (8.6)].
8.6 Hepatic Impairment
Dosage adjustment is necessary in patients with moderate hepatic impairment.
Do not use vardenafil hydrochloride tablets in patients with severe hepatic impairment (Child-Pugh C). Vardenafil has not been evaluated in this patient population.
A starting dose of 5 mg is recommended in patients with moderate hepatic impairment (Child-Pugh B) and the maximum dose should not exceed 10 mg. In volunteers with moderate hepatic impairment, the C max and AUC following a 10 mg vardenafil dose were increased by 130% and 160%, respectively, compared to healthy control subjects. [See Warnings and Precautions (5.8) and Dosage and Administration (2.3).]
In volunteers with mild hepatic impairment (Child-Pugh A), the C max and AUC following a 10 mg vardenafil dose were increased by 22% and 17%, respectively, compared to healthy control subjects. No dosage adjustment is necessary in patients with mild hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Vardenafil hydrochloride tablets are indicated for the treatment of erectile dysfunction.
5.5 Sudden Hearing Loss
Physicians should advise patients to stop taking all PDE5 inhibitors, including vardenafil hydrochloride tablets, and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including vardenafil. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions (6.2)].
12.1 Mechanism of Action
Penile erection is a hemodynamic process initiated by the relaxation of smooth muscle in the corpus cavernosum and its associated arterioles. During sexual stimulation, nitric oxide is released from nerve endings and endothelial cells in the corpus cavernosum. Nitric oxide activates the enzyme guanylate cyclase resulting in increased synthesis of cyclic guanosine monophosphate (cGMP) in the smooth muscle cells of the corpus cavernosum. The cGMP in turn triggers smooth muscle relaxation, allowing increased blood flow into the penis, resulting in erection. The tissue concentration of cGMP is regulated by both the rates of synthesis and degradation via phosphodiesterases (PDEs). The most abundant PDE in the human corpus cavernosum is the cGMP-specific phosphodiesterase type 5 (PDE5); therefore, the inhibition of PDE5 enhances erectile function by increasing the amount of cGMP. Because sexual stimulation is required to initiate the local release of nitric oxide, the inhibition of PDE5 has no effect in the absence of sexual stimulation.
In vitro studies have shown that vardenafil is a selective inhibitor of PDE5. The inhibitory effect of vardenafil is more selective on PDE5 than for other known phosphodiesterases (>15-fold relative to PDE6, >130-fold relative to PDE1, >300-fold relative to PDE11, and >1,000-fold relative to PDE2, 3, 4, 7, 8, 9, and 10).
5.11 Effects On Bleeding (5.11 Effects on Bleeding)
In humans, vardenafil alone in doses up to 20 mg does not prolong the bleeding time. There is no clinical evidence of any additive prolongation of the bleeding time when vardenafil is administered with aspirin. Vardenafil hydrochloride tablets have not been administered to patients with bleeding disorders or significant active peptic ulceration. Therefore vardenafil hydrochloride tablets should be administered to these patients after careful benefit-risk assessment.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
The evaluation of erectile dysfunction should include a medical assessment, a determination of potential underlying causes and the identification of appropriate treatment.
Before prescribing vardenafil hydrochloride tablets, it is important to note the following:
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Vardenafil hydrochloride tablets are taken as needed: For most patients, the starting dose is 10 mg, up to once daily. Increase to 20 mg or decrease to 5 mg based on efficacy/tolerability. ( 2.1)
- A starting dose of 5 mg vardenafil hydrochloride tablets should be considered in patients ≥ 65 years of age. ( 2.3)
- Vardenafil hydrochloride tablets are taken orally, approximately 60 minutes before sexual activity. ( 2.1)
- The maximum recommended dosing frequency is one tablet per day. ( 2.1)
- Vardenafil hydrochloride tablets may be taken with or without food. ( 2.2)
- If taking potent or moderate inhibitors of CYP3A4, the dose of vardenafil hydrochloride tablets should be adjusted as follows (
2.4,
5.2,
7.2):
- Ritonavir: No more than 2.5 mg in a 72-hour period
- Indinavir, saquinavir, atazanavir, ketoconazole 400 mg daily, itraconazole 400 mg daily, clarithromycin: No more than 2.5 mg in a 24-hour period
- Ketoconazole 200 mg daily, itraconazole 200 mg daily, erythromycin: No more than 5 mg in a 24-hour period.
- In patients on stable alpha-blocker therapy the recommended starting dose of vardenafil hydrochloride tablets is 5 mg ( 2.4, 5.6)
- The recommended starting dose of vardenafil hydrochloride tablets is 5 mg in patients with moderate hepatic impairment (Child-Pugh B). The maximum dose in patients with moderate hepatic impairment should not exceed 10 mg. ( 2.3, 8.6)
2.1 General Dose Information
For most patients, the recommended starting dose of vardenafil hydrochloride tablets is 10 mg, taken orally, as needed, approximately 60 minutes before sexual activity. The dose may be increased to a maximum recommended dose of 20 mg or decreased to 5 mg based on efficacy and side effects. The maximum recommended dosing frequency is once per day. Sexual stimulation is required for a response to treatment.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side corresponding to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of vardenafil hydrochloride tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate their frequency or establish a causal relationship to drug exposure.
To report SUSPECTED ADVERSE REACTIONS, contact AvKARE at 1-855-361-3993; email [email protected]; or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Vardenafil hydrochloride tablets were administered to over 4430 men (mean age 56, range 18-89 years; 81% White, 6% Black, 2% Asian, 2% Hispanic and 9% Other) during controlled and uncontrolled clinical trials worldwide. Over 2200 patients were treated for 6 months or longer and 880 patients were treated for at least 1 year.
In placebo-controlled clinical trials, the discontinuation rate due to adverse events was 3.4% for vardenafil hydrochloride tablets compared to 1.1% for placebo.
When vardenafil hydrochloride tablets were taken as recommended in placebo-controlled clinical trials, the following adverse reactions were reported (see Table 1).
| Adverse Reaction | Percentage of Patients Reporting Reactions | |
|---|---|---|
| Placebo
N = 1199 |
Vardenafil Hydrochloride Tablets
N = 2203 |
|
| Headache | 4% | 15% |
| Flushing | 1% | 11% |
| Rhinitis | 3% | 9% |
| Dyspepsia | 1% | 4% |
| Accidental Injury
All the events listed in the above table were deemed to be adverse drug reactions with the exception of accidental injury.
|
2% | 3% |
| Sinusitis | 1% | 3% |
| Flu Syndrome | 2% | 3% |
| Dizziness | 1% | 2% |
| Increased Creatine Kinase | 1% | 2% |
| Nausea | 1% | 2% |
Back pain was reported in 2.0% of patients treated with vardenafil hydrochloride tablets and 1.7% of patients on placebo.
Placebo-controlled trials suggested a dose effect in the incidence of some adverse reactions (headache, flushing, dyspepsia, nausea, and rhinitis) over the 5 mg, 10 mg, and 20 mg doses of vardenafil hydrochloride tablets.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
5.12 Sexually Transmitted Disease
The use of vardenafil hydrochloride tablets offers no protection against sexually transmitted diseases. Counseling of patients about protective measures necessary to guard against sexually transmitted diseases, including the Human Immunodeficiency Virus (HIV), should be considered.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Vardenafil hydrochloride tablets are formulated as orange, round, tablets embossed with "2.5", "5", "10" or "20" on one side equivalent to 2.5 mg, 5 mg, 10 mg, and 20 mg of vardenafil, respectively
| Package | Strength | NDC Code | |
|---|---|---|---|
| Bottles of 30 | 10 mg | 42291-847-30 | |
| 20 mg | 42291-848-30 |
4.2 Guanylate Cyclase (gc) Stimulators (4.2 Guanylate Cyclase (GC) Stimulators)
Do not use vardenafil hydrochloride tablets in patients who are using a GC stimulator, such as riociguat. PDE5 inhibitors, including vardenafil hydrochloride tablets may potentiate the hypotensive effects of GC stimulators.
5.7 Congenital Or Acquired Qt Prolongation (5.7 Congenital or Acquired QT Prolongation)
In a study of the effect of vardenafil hydrochloride tablets on QT interval in 59 healthy males [see Clinical Pharmacology (12.2)] , therapeutic (10 mg) and supratherapeutic (80 mg) doses of vardenafil and the active control moxifloxacin (400 mg) produced similar increases in QTc interval. A postmarketing study evaluating the effect of combining vardenafil hydrochloride tablets with another drug of comparable QT effect showed an additive QT effect when compared with either drug alone [see Clinical Pharmacology (12.2)] . These observations should be considered in clinical decisions when prescribing vardenafil hydrochloride tablets to patients with known history of QT prolongation or patients who are taking medications known to prolong the QT interval.
Patients taking Class 1A (for example. quinidine, procainamide) or Class III (for example, amiodarone, sotalol) antiarrhythmic medications or those with congenital QT prolongation, should avoid using vardenafil hydrochloride tablets.
5.13 risks in Patients With Phenylketonuria (5.13 Risks in Patients with Phenylketonuria)
Phenylalanine can be harmful to patients with phenylketonuria (PKU). Vardenafil hydrochloride tablets contain phenylalanine, a component of aspartame. Each 2.5 mg vardenafil hydrochloride tablet contains 0.7 mg phenylalanine; each 5 mg tablet contains 1.4 mg phenylalanine; each 10 mg tablet contains 2.8 mg phenylalanine; each 20 mg tablet contains 5.6 mg phenylalanine. Before prescribing vardenafil hydrochloride tablets in a patient with PKU, consider the combined daily amount of phenylalanine from all sources, including vardenafil hydrochloride tablets.ardenafil hydrochloride tablets contain aspartame, a source of phenylalanine. Each 2.5 mg vardenafil hydrochloride tablet contains 0.7 mg phenylalanine; each 5 mg tablet contains 1.4 mg phenylalanine; each 10 mg tablet contains 2.8 mg phenylalanine; each 20 mg tablet contains 5.6 mg phenylalanine.
Principal Display Panel 10 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label)
Principal Display Panel 20 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label)
14.2 Trial in Patients With Ed and Diabetes Mellitus (14.2 Trial in Patients with ED and Diabetes Mellitus)
Vardenafil hydrochloride tablets demonstrated clinically meaningful and statistically significant improvement in erectile function in a prospective, fixed-dose (10 and 20 mg vardenafil hydrochloride tablets), double-blind, placebo-controlled trial of patients with diabetes mellitus (n=439; mean age 57 years, range 33-81; 80% White, 9% Black, 8% Hispanic, and 3% Other).
Significant improvements in the EF Domain were shown in this study (EF Domain scores of 17 on 10 mg vardenafil hydrochloride tablets and 19 on 20 mg vardenafil hydrochloride tablets compared to 13 on placebo; p <0.0001).
Vardenafil hydrochloride tablets significantly improved the overall per-patient rate of achieving an erection sufficient for penetration (SEP2) (61% on 10 mg and 64% on 20 mg vardenafil hydrochloride tablets compared to 36% on placebo; p <0.0001).
Vardenafil hydrochloride tablets demonstrated a clinically meaningful and statistically significant increase in the overall per-patient rate of maintenance of erection to successful intercourse (SEP3) (49% on 10 mg, 54% on 20 mg vardenafil hydrochloride tablets compared to 23% on placebo; p <0.0001).
14.1 Trials in A General Erectile Dysfunction Population (14.1 Trials in a General Erectile Dysfunction Population)
In the major North American fixed-dose trial, 762 patients (mean age 57, range 20-83 years; 79% White, 13% Black, 4% Hispanic, 2% Asian and 2% Other) were evaluated. The mean baseline EF Domain scores were 13, 13, 13, 14 for the vardenafil hydrochloride tablets 5 mg, 10 mg, 20 mg and placebo groups, respectively. There was significant improvement (p <0.0001) at 3 months with vardenafil hydrochloride tablets (EF Domain scores of 18, 21, 21, for the 5 mg, 10 mg, and 20 mg dose groups, respectively) compared to the placebo group (EF Domain score of 15). The European trial (total N=803) confirmed these results. The improvement in mean score was maintained at all doses at 6 months in the North American trial.
In the North American trial, vardenafil hydrochloride tablets significantly improved the rates of achieving an erection sufficient for penetration (SEP2) at doses of 5 mg, 10 mg, and 20 mg compared to placebo (65%, 75%, and 80%, respectively, compared to a 52% response in the placebo group at 3 months; p <0.0001). The European trial confirmed these results.
Vardenafil hydrochloride tablets demonstrated a clinically meaningful and statistically significant increase in the overall per-patient rate of maintenance of erection to successful intercourse (SEP3) (51% on 5 mg, 64% on 10 mg, and 65% on 20 mg, respectively, compared to 32% on placebo; p <0.0001) at 3 months in the North American trial. The European trial showed comparable efficacy. This improvement in mean score was maintained at all doses at 6 months in the North American trial.
14.3 Trial in Patients With Ed After Radical Prostatectomy (14.3 Trial in Patients with ED after Radical Prostatectomy)
Vardenafil hydrochloride tablets demonstrated clinically meaningful and statistically significant improvement in erectile function in a prospective, fixed-dose (10 and 20 mg vardenafil hydrochloride tablets), double-blind, placebo-controlled trial in post-prostatectomy patients (n=427, mean age 60, range 44-77 years; 93% White, 5% Black, 2% Other).
Significant improvements in the EF Domain were shown in this study (EF Domain scores of 15 on 10 mg vardenafil hydrochloride tablets and 15 on 20 mg vardenafil hydrochloride tablets compared to 9 on placebo; p <0.0001).
Vardenafil hydrochloride tablets significantly improved the overall per-patient rate of achieving an erection sufficient for penetration (SEP2) (47% on 10 mg and 48% on 20 mg vardenafil hydrochloride tablets compared to 22% on placebo; p <0.0001).
Vardenafil hydrochloride tablets demonstrated a clinically meaningful and statistically significant increase in the overall per-patient rate of maintenance of erection to successful intercourse (SEP3) (37% on 10 mg, 34% on 20 mg vardenafil hydrochloride tablets compared to 10% on placebo; p <0.0001).
5.10 Combination With Other Erectile Dysfunction Therapies (5.10 Combination with Other Erectile Dysfunction Therapies)
The safety and efficacy of vardenafil hydrochloride tablets used in combination with other treatments for erectile dysfunction have not been studied. Therefore, the use of such combinations is not recommended.
5.2 Potential for Drug Interactions With Potent Or Moderate Cyp3a4 Inhibitors (5.2 Potential for Drug Interactions with Potent or Moderate CYP3A4 Inhibitors)
Concomitant administration with potent CYP3A4 inhibitors (such as ritonavir, indinavir, ketoconazole) or moderate CYP3A4 inhibitors (such as erythromycin) increases plasma concentrations of vardenafil. Dosage adjustment is necessary when vardenafil hydrochloride tablets are administered with certain CYP3A4 inhibitors [see Dosage and Administration (2.4), Drug Interactions (7.2)].
Long-term safety information is not available on the concomitant administration of vardenafil with HIV protease inhibitors.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:52:22.117969 · Updated: 2026-03-14T22:43:29.901552