These Highlights Do Not Include All The Information Needed To Use Tepmetko Safely And Effectively. See Full Prescribing Information For Tepmetko.
80a0f1b9-071a-47f5-9e67-32d638a669dc
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Indication and Usage ( 1 ) 02/2024 Dosage and Administration ( 2.3 , 2.4 ) 02/2024 Warnings and Precautions ( 5.3 ) 02/2024
Indications and Usage
TEPMETKO is indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) harboring mesenchymal-epithelial transition ( MET ) exon 14 skipping alterations.
Dosage and Administration
Select patients for treatment with TEPMETKO on the presence of MET ex14 skipping. ( 2.1 , 14 ) Recommended dosage : 450 mg orally once daily with food until disease progression or unacceptable toxicity. ( 2.2 )
Warnings and Precautions
Interstitial Lung Disease (ILD)/Pneumonitis : Immediately withhold TEPMETKO in patients with suspected ILD/pneumonitis. Permanently discontinue TEPMETKO in patients diagnosed with ILD/pneumonitis of any severity. ( 2.4 , 5.1 ) Hepatotoxicity : Monitor liver function tests. Withhold, dose reduce, or permanently discontinue TEPMETKO based on severity. ( 5.2 ) Pancreatic Toxicity : Monitor amylase and lipase. Withhold, dose reduce, or permanently discontinue TEPMETKO based on severity. ( 5.3 ) Embryo-fetal toxicity : TEPMETKO can cause fetal harm. Advise of potential risk to a fetus and use of effective contraception. ( 5.4 , 8.1 , 8.3 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are described in greater detail elsewhere in the labeling: Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1) ] Hepatotoxicity [see Warnings and Precautions (5.2) ] Pancreatic Toxicity [see Warnings and Precautions (5.3) ]
Drug Interactions
Certain P-gp substrates : Avoid coadministration of TEPMETKO with P-gp substrates where minimal concentration changes may lead to serious or life-threatening toxicities. ( 7.1 )
Storage and Handling
TEPMETKO (tepotinib) tablets: 225 mg tepotinib, white-pink, oval, biconvex film-coated tablet with embossment "M" on one side and plain on the other side. NDC number Size 44087-5000-3 Box of 30 tablets: 3 blister cards each containing 10 tablets 44087-5000-6 Box of 60 tablets: 6 blister cards each containing 10 tablets The blister cards consist of a child-resistant blister foil.
How Supplied
TEPMETKO (tepotinib) tablets: 225 mg tepotinib, white-pink, oval, biconvex film-coated tablet with embossment "M" on one side and plain on the other side. NDC number Size 44087-5000-3 Box of 30 tablets: 3 blister cards each containing 10 tablets 44087-5000-6 Box of 60 tablets: 6 blister cards each containing 10 tablets The blister cards consist of a child-resistant blister foil.
Medication Information
Warnings and Precautions
Interstitial Lung Disease (ILD)/Pneumonitis : Immediately withhold TEPMETKO in patients with suspected ILD/pneumonitis. Permanently discontinue TEPMETKO in patients diagnosed with ILD/pneumonitis of any severity. ( 2.4 , 5.1 ) Hepatotoxicity : Monitor liver function tests. Withhold, dose reduce, or permanently discontinue TEPMETKO based on severity. ( 5.2 ) Pancreatic Toxicity : Monitor amylase and lipase. Withhold, dose reduce, or permanently discontinue TEPMETKO based on severity. ( 5.3 ) Embryo-fetal toxicity : TEPMETKO can cause fetal harm. Advise of potential risk to a fetus and use of effective contraception. ( 5.4 , 8.1 , 8.3 )
Indications and Usage
TEPMETKO is indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) harboring mesenchymal-epithelial transition ( MET ) exon 14 skipping alterations.
Dosage and Administration
Select patients for treatment with TEPMETKO on the presence of MET ex14 skipping. ( 2.1 , 14 ) Recommended dosage : 450 mg orally once daily with food until disease progression or unacceptable toxicity. ( 2.2 )
Contraindications
None.
Adverse Reactions
The following adverse reactions are described in greater detail elsewhere in the labeling: Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1) ] Hepatotoxicity [see Warnings and Precautions (5.2) ] Pancreatic Toxicity [see Warnings and Precautions (5.3) ]
Drug Interactions
Certain P-gp substrates : Avoid coadministration of TEPMETKO with P-gp substrates where minimal concentration changes may lead to serious or life-threatening toxicities. ( 7.1 )
Storage and Handling
TEPMETKO (tepotinib) tablets: 225 mg tepotinib, white-pink, oval, biconvex film-coated tablet with embossment "M" on one side and plain on the other side. NDC number Size 44087-5000-3 Box of 30 tablets: 3 blister cards each containing 10 tablets 44087-5000-6 Box of 60 tablets: 6 blister cards each containing 10 tablets The blister cards consist of a child-resistant blister foil.
How Supplied
TEPMETKO (tepotinib) tablets: 225 mg tepotinib, white-pink, oval, biconvex film-coated tablet with embossment "M" on one side and plain on the other side. NDC number Size 44087-5000-3 Box of 30 tablets: 3 blister cards each containing 10 tablets 44087-5000-6 Box of 60 tablets: 6 blister cards each containing 10 tablets The blister cards consist of a child-resistant blister foil.
Description
Indication and Usage ( 1 ) 02/2024 Dosage and Administration ( 2.3 , 2.4 ) 02/2024 Warnings and Precautions ( 5.3 ) 02/2024
Section 42229-5
Increased Creatinine
A median increase in serum creatinine of 30% was observed 21 days after initiation of treatment with TEPMETKO. The serum creatinine increases persisted throughout treatment and were reversible upon treatment completion.
Section 42230-3
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised 01/2025 | |
|
PATIENT INFORMATION
TEPMETKO® (tep-MET-co) (tepotinib) tablets, for oral use |
||
|
What is the most important information I should know about TEPMETKO?
|
||
|
What is TEPMETKO?
TEPMETKO is a prescription medicine used to treat adults with non-small cell lung cancer (NSCLC) that:
|
||
Before you take TEPMETKO, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I take TEPMETKO?
|
||
|
What are the possible side effects of TEPMETKO?
|
||
|
|
|
|
||
|
|
|
| The most common side effects of TEPMETKO include: | ||
|
|
|
| Your healthcare provider may change your dose, temporarily stop, or permanently stop treatment with TEPMETKO if you develop serious side effects during treatment. These are not all of the possible side effects of TEPMETKO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store TEPMETKO?
|
||
|
General information about the safe and effective use of TEPMETKO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use TEPMETKO for a condition for which it was not prescribed. Do not give TEPMETKO to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about TEPMETKO that is written for health professionals. |
||
|
What are the ingredients in TEPMETKO?
Active ingredient: tepotinib Inactive ingredients: mannitol, microcrystalline cellulose, crospovidone, magnesium stearate, and colloidal silicon dioxide. Tablet coating: hypromellose, titanium dioxide, lactose monohydrate, polyethylene glycol, triacetin, and red iron oxides. Manufactured for: EMD Serono, Inc., Boston, MA 02210, U.S.A. TEPMETKO is a trademark of Merck KGaA, Darmstadt, Germany. Product of Germany. For more information, call toll-free 1-844-662-3631 or go to www.TEPMETKO.com. |
Section 43683-2
Section 44425-7
Store TEPMETKO at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30°C (59°F-86°F) [see USP-NF Controlled Room Temperature]. Store in original package.
11 Description
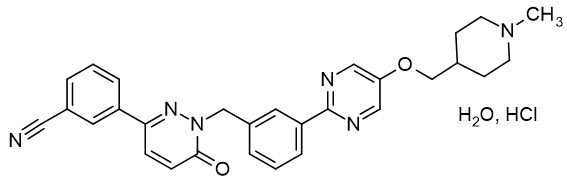
Tepotinib is a kinase inhibitor. TEPMETKO (tepotinib) tablets for oral use are formulated with tepotinib hydrochloride hydrate. The chemical name for tepotinib hydrochloride hydrate is 3-{1-[(3-{5-[(1-methylpiperidin-4-yl)methoxy]pyrimidin-2-yl}phenyl)methyl]-6-oxo-1,6-dihydropyridazin-3-yl}benzonitrile hydrochloride hydrate. The molecular formula is C29H28N6O2∙HCl∙H2O and the molecular weight is 547.05 g/mol for tepotinib hydrochloride hydrate and 492.58 g/mol for tepotinib (free base). The chemical structure is shown below:
Tepotinib hydrochloride hydrate is a white to off-white powder with a pKa of 9.5.
TEPMETKO is supplied as film-coated tablets containing 225 mg of tepotinib (equivalent to 250 mg tepotinib hydrochloride hydrate). Inactive ingredients in the tablet core are mannitol, microcrystalline cellulose, crospovidone, magnesium stearate, and colloidal silicon dioxide. The tablet coating consists of hypromellose, titanium dioxide, lactose monohydrate, polyethylene glycol, triacetin, and red iron oxides.
8.4 Pediatric Use
The safety and efficacy of TEPMETKO in pediatric patients have not been established.
8.5 Geriatric Use
Of 313 patients with NSCLC positive for METex14 skipping alterations in VISION who received 450 mg TEPMETKO once daily, 79% were 65 years or older, and 41% were 75 years or older. No clinically important differences in safety or efficacy were observed between patients aged 65 years or older and younger patients.
5.2 Hepatotoxicity
Hepatotoxicity occurred in patients treated with TEPMETKO [see Adverse Reactions (6.1)]. Increased alanine aminotransferase (ALT)/increased aspartate aminotransferase (AST) occurred in 18% of patients treated with TEPMETKO. Grade 3 or 4 increased ALT/AST occurred in 4.7% of patients. A fatal adverse reaction of hepatic failure occurred in one patient (0.2%). Four patients (0.8%) discontinued TEPMETKO due to increased ALT/AST. The median time-to-onset of Grade 3 or higher increased ALT/AST was 47 days (range 1 to 262).
Monitor liver function tests (including ALT, AST, and total bilirubin) prior to the start of TEPMETKO, every 2 weeks during the first 3 months of treatment, then once a month or as clinically indicated, with more frequent testing in patients who develop increased transaminases or bilirubin. Based on the severity of the adverse reaction, withhold, dose reduce, or permanently discontinue TEPMETKO [see Dosage and Administration (2.4)].
14 Clinical Studies
The efficacy of TEPMETKO was evaluated in a single-arm, open-label, multicenter, non-randomized, multicohort study (VISION, NCT02864992). Eligible patients were required to have advanced or metastatic NSCLC harboring METex14 skipping alterations, epidermal growth factor receptor (EGFR) wild-type and anaplastic lymphoma kinase (ALK) negative status, at least one measurable lesion as defined by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1, and Eastern Cooperative Oncology Group (ECOG) Performance Status of 0 to 1. Patients with symptomatic CNS metastases, clinically significant uncontrolled cardiac disease, or who received treatment with any MET or hepatocyte growth factor (HGF) inhibitor were not eligible for the study.
Identification of METex14 skipping alterations was prospectively determined using central laboratories employing either a PCR-based or next-generation sequencing-based clinical trial assay using tissue (66%) and/or plasma (57%) samples.
Patients received TEPMETKO 450 mg once daily until disease progression or unacceptable toxicity. The major efficacy outcome measure was confirmed overall response rate (ORR) according to Response Evaluation Criteria in Solid Tumors (RECIST v1.1) as evaluated by a Blinded Independent Review Committee (BIRC). An additional efficacy outcome measure was duration of response (DOR) by BIRC.
The efficacy population included 164 treatment naïve patients and 149 previously treated patients. The median age was 72 years (range 41 to 94 years); 51% female; 62% White, 34% Asian; 26% had Eastern Cooperative Oncology Group (ECOG) Performance Status (PS) 0 and 74% had ECOG PS 1; 49% never smoked; 81% had adenocarcinoma; 94% had metastatic disease; and 13% had CNS metastases. Amongst previously treated patients, 84% received prior platinum-based chemotherapy.
Efficacy results are presented in Table 4.
| Efficacy parameter | Treatment-Naïve (N=164) |
Previously Treated (N=149) |
|---|---|---|
| CI: Confidence interval, + denotes ongoing response. | ||
| Overall response rate (95% CI) Blinded Independent Review Committee (BIRC) review.
,
Confirmed responses.
|
57% (49, 65) | 45% (37, 53) |
| Duration of Response | ||
| Range in months Based on observed duration of response.
|
1.3+, 56.6+ | 1.4+, 67.6+ |
| Patients with DOR ≥ 6 months | 66% | 66% |
| Patients with DOR ≥ 12 months | 40% | 36% |
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions are described in greater detail elsewhere in the labeling:
- Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1)]
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Pancreatic Toxicity [see Warnings and Precautions (5.3)]
7 Drug Interactions
- Certain P-gp substrates: Avoid coadministration of TEPMETKO with P-gp substrates where minimal concentration changes may lead to serious or life-threatening toxicities. (7.1)
8.6 Renal Impairment
No dosage modification is recommended in patients with mild or moderate renal impairment (creatinine clearance [CLcr] 30 to 89 mL/min, estimated by Cockcroft-Gault). The recommended dosage has not been established for patients with severe renal impairment (CLcr < 30 mL/min) [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of tepotinib were evaluated in patients with cancer administered 450 mg once daily unless otherwise specified. Tepotinib exposure (AUC0-12h and Cmax) increases dose-proportionally over the dose range of 27 mg (0.06 times the recommended daily dosage) to 450 mg. At the recommended dosage, the geometric mean (coefficient of variation [CV] %) steady state Cmax was 1,291 ng/mL (48.1%) and the AUC0-24h was 27,438 ng∙h/mL (51.7%). The oral clearance of tepotinib did not change with respect to time. The median accumulation was 2.5-fold for Cmax and 3.3-fold for AUC0-24h after multiple daily doses of tepotinib.
2.2 Recommended Dosage
The recommended dosage of TEPMETKO is 450 mg orally once daily with food [see Clinical Pharmacology (12.3)] until disease progression or unacceptable toxicity.
Instruct patients to take their dose of TEPMETKO at approximately the same time every day and to swallow tablets whole. Do not chew, crush or split tablets. Patients who have difficulty swallowing solids can disperse tablets in water [see Dosage and Administration (2.3)].
Advise patients not to make up a missed dose within 8 hours of the next scheduled dose.
If vomiting occurs after taking a dose of TEPMETKO, advise patients to take the next dose at the scheduled time.
8.7 Hepatic Impairment
No dosage modification is recommended in patients with mild (Child Pugh Class A) or moderate (Child Pugh Class B) hepatic impairment. The pharmacokinetics and safety of tepotinib in patients with severe hepatic impairment (Child Pugh Class C) have not been studied [see Clinical Pharmacology (12.3)].
1 Indications and Usage
TEPMETKO is indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) harboring mesenchymal-epithelial transition (MET) exon 14 skipping alterations.
5.3 Pancreatic Toxicity
Elevations in amylase and lipase levels occurred in patients treated with TEPMETKO [see Adverse Reactions (6.1)]. Increased amylase and/or lipase occurred in 13% of patients treated with TEPMETKO. Grade 3 and 4 increased amylase and/or lipase occurred in 5% and 1.2% of patients, respectively. Monitor amylase and lipase at baseline and regularly during treatment with TEPMETKO. Based on the severity of the adverse drug reaction, temporarily withhold, dose reduce, or permanently discontinue TEPMETKO [see Dosage and Administration (2.4)].
12.1 Mechanism of Action
Tepotinib is a kinase inhibitor that targets MET, including variants with exon 14 skipping alterations. Tepotinib inhibits hepatocyte growth factor (HGF)-dependent and -independent MET phosphorylation and MET-dependent downstream signaling pathways. Tepotinib also inhibited melatonin 2 and imidazoline 1 receptors at clinically achievable concentrations.
In vitro, tepotinib inhibited tumor cell proliferation, anchorage-independent growth, and migration of MET-dependent tumor cells. In mice implanted with tumor cell lines with oncogenic activation of MET, including METex14 skipping alterations, tepotinib inhibited tumor growth, led to sustained inhibition of MET phosphorylation, and, in one model, decreased the formation of metastases.
5.4 Embryo Fetal Toxicity
Based on findings in animal studies and its mechanism of action TEPMETKO can cause fetal harm when administered to a pregnant woman. Oral administration of tepotinib to pregnant rabbits during the period of organogenesis resulted in malformations (teratogenicity) and anomalies at exposures less than the human exposure based on area under the curve (AUC) at the 450 mg daily clinical dose. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential or males with female partners of reproductive potential to use effective contraception during treatment with TEPMETKO and for one week after the last dose. [See Use in Specific Populations (8.1, 8.3)]
5 Warnings and Precautions
- Interstitial Lung Disease (ILD)/Pneumonitis: Immediately withhold TEPMETKO in patients with suspected ILD/pneumonitis. Permanently discontinue TEPMETKO in patients diagnosed with ILD/pneumonitis of any severity. (2.4, 5.1)
- Hepatotoxicity: Monitor liver function tests. Withhold, dose reduce, or permanently discontinue TEPMETKO based on severity. (5.2)
- Pancreatic Toxicity: Monitor amylase and lipase. Withhold, dose reduce, or permanently discontinue TEPMETKO based on severity. (5.3)
- Embryo-fetal toxicity: TEPMETKO can cause fetal harm. Advise of potential risk to a fetus and use of effective contraception. (5.4, 8.1, 8.3)
2 Dosage and Administration
3 Dosage Forms and Strengths
Tablets: 225 mg, white-pink, oval, biconvex film-coated tablets with embossment "M" on one side and plain on the other side.
8 Use in Specific Populations
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety population described in the WARNINGS AND PRECAUTIONS reflect exposure to TEPMETKO in 506 patients with solid tumors enrolled in five open-label, single-arm studies receiving TEPMETKO as single agent at a dose of 450 mg once daily. This included 313 patients with NSCLC positive for METex14 skipping alterations, who received TEPMETKO in VISION. Among 506 patients who received TEPMETKO, 44% were exposed for 6 months or longer, and 22% were exposed for more than one year.
The data described below reflect exposure to TEPMETKO 450 mg once daily in 313 patients with metastatic non-small cell lung cancer (NSCLC) with METex14 skipping alterations in VISION [see Clinical Studies (14)].
Serious adverse reactions occurred in 51% of patients who received TEPMETKO. Serious adverse reactions in > 2% of patients included pleural effusion (6%), pneumonia (6%), edema (5%), general health deterioration (3.8%), dyspnea (3.5%), musculoskeletal pain (2.9%), and pulmonary embolism (2.2%). Fatal adverse reactions occurred in 1.9% of patients who received TEPMETKO, including pneumonitis (0.3%), hepatic failure (0.3%), dyspnea from fluid overload (0.3%), pneumonia (0.3%), sepsis (0.3%), and death of unknown cause (0.3%).
Permanent discontinuation due to an adverse reaction occurred in 25% of patients who received TEPMETKO. The most frequent adverse reactions (> 1%) leading to permanent discontinuations of TEPMETKO were edema (8%), pleural effusion (1.6%), and general health deterioration (1.6%).
Dosage interruptions due to an adverse reaction occurred in 53% of patients who received TEPMETKO. Adverse reactions which required dosage interruption in > 2% of patients who received TEPMETKO included edema (28%), increased blood creatinine (6%), pleural effusion (3.5%), nausea (3.2%), increased ALT (2.9%), pneumonia (2.6%), decreased appetite (2.2%), and dyspnea (2.2%).
Dose reductions due to an adverse reaction occurred in 36% of patients who received TEPMETKO. Adverse reactions which required dose reductions in > 2% of patients who received TEPMETKO included edema (22%), increased blood creatinine (2.9%), fatigue (2.2%), and pleural effusion (2.2%).
The most common adverse reactions (≥ 20%) in patients who received TEPMETKO were edema, nausea, fatigue, musculoskeletal pain, diarrhea, dyspnea, decreased appetite, and rash. The most common Grade 3 to 4 laboratory abnormalities (≥ 2%) were decreased lymphocytes, decreased albumin, decreased sodium, increased gamma-glutamyltransferase, increased amylase, increased lipase, increased ALT, increased AST, and decreased hemoglobin.
Table 2 summarizes the adverse reactions in VISION.
| Adverse Reactions | TEPMETKO (N=313) |
|
|---|---|---|
| All Grades Severity as defined by National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 4.03.
(%) |
Grades 3 to 4
(%) |
|
| General disorders and administration-site conditions | ||
| Edema Edema includes eye edema, face edema, generalized edema, localized edema, edema, genital edema, peripheral edema, peripheral swelling, periorbital edema, and scrotal edema.
|
81 | 16 |
| Fatigue Fatigue includes asthenia and fatigue.
|
30 | 1.9 |
| Gastrointestinal disorders | ||
| Nausea | 31 | 1.3 |
| Diarrhea | 29 | 0.6 |
| Abdominal pain Abdominal pain includes abdominal discomfort, abdominal pain, abdominal pain lower, abdominal pain upper, gastrointestinal pain, and hepatic pain.
|
19 | 0.6 |
| Constipation | 19 | 0.3 |
| Vomiting Vomiting includes retching and vomiting.
|
15 | 1 |
| Musculoskeletal and Connective Tissue Disorders | ||
| Musculoskeletal pain Musculoskeletal pain includes arthralgia, arthritis, back pain, bone pain, musculoskeletal chest pain, musculoskeletal pain, myalgia, non-cardiac chest pain, pain in extremity, and spinal pain.
|
30 | 3.2 |
| Respiratory, thoracic, and mediastinal disorders | ||
| Dyspnea Dyspnea includes dyspnea, dyspnea at rest, and dyspnea exertional.
|
24 | 2.6 |
| Cough Cough includes cough, and productive cough.
|
18 | 0.3 |
| Pleural effusion | 14 | 4.2 |
| Metabolism and nutrition disorders | ||
| Decreased appetite | 21 | 1.9 |
| Skin and Subcutaneous Tissue Disorders | ||
| Rash Rash includes rash, palmar-plantar erythrodysesthesia syndrome, rash maculo-papular, eczema, exfoliative rash, rash erythematous, rash pustular, skin exfoliation, dermatitis acneiform, drug eruption, dermatitis, rash pruritic, dermatitis bullous, toxic skin eruption.
|
21 | 1.3 |
| Infections and Infestations | ||
| Pneumonia Pneumonia includes pneumonia, pneumonia aspiration, and pneumonia bacterial.
|
12 | 3.8 |
Clinically relevant adverse reactions in < 10% of patients who received TEPMETKO included ILD/pneumonitis, fever, dizziness, pruritus, and headache.
Table 3 summarizes the laboratory abnormalities observed in VISION.
| Laboratory Abnormalities | TEPMETKO The denominator used to calculate the rate varied from 268 to 309 based on the number of patients with a baseline value and at least one post-treatment value.
|
|
|---|---|---|
| Grades 1 to 4 Severity as defined by National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 4.03.
(%) |
Grades 3 to 4
(%) |
|
| Chemistry | ||
| Decreased albumin | 81 | 9 |
| Increased creatinine | 60 | 1 |
| Increased alkaline phosphatase aminotransferase | 52 | 1.6 |
| Increased alanine aminotransferase | 50 | 4.9 |
| Increased aspartate aminotransferase | 40 | 3.6 |
| Decreased sodium | 36 | 9 |
| Increased gamma-glutamyltransferase | 29 | 6 |
| Increased potassium | 26 | 1.9 |
| Increased amylase | 25 | 5 |
| Increased lipase | 21 | 5 |
| Hematology | ||
| Decreased lymphocytes | 57 | 15 |
| Decreased hemoglobin | 31 | 3.6 |
| Decreased leukocytes | 25 | 1.9 |
| Decreased platelets | 24 | 0.6 |
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling
TEPMETKO (tepotinib) tablets: 225 mg tepotinib, white-pink, oval, biconvex film-coated tablet with embossment "M" on one side and plain on the other side.
| NDC number | Size |
|---|---|
| 44087-5000-3 | Box of 30 tablets: 3 blister cards each containing 10 tablets |
| 44087-5000-6 | Box of 60 tablets: 6 blister cards each containing 10 tablets |
The blister cards consist of a child-resistant blister foil.
2.4 Dose Modifications for Adverse Reactions
The recommended dose reduction of TEPMETKO for the management of adverse reactions is 225 mg orally once daily.
Permanently discontinue TEPMETKO in patients who are unable to tolerate 225 mg orally once daily.
The recommended dosage modifications of TEPMETKO for adverse reactions are provided in Table 1.
| Adverse Reaction | Severity Severity as defined by National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) Version 5.
|
Dose Modification |
|---|---|---|
| Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.1)] | Any Grade | Withhold TEPMETKO if ILD is suspected. Permanently discontinue TEPMETKO if ILD is confirmed. |
| Increased ALT and/or AST without increased total bilirubin [see Warnings and Precautions (5.2)] | Grade 3 | Withhold TEPMETKO until recovery to baseline ALT/AST. If recovered to baseline within 7 days, then resume TEPMETKO at the same dose; otherwise resume TEPMETKO at a reduced dose. |
| Grade 4 | Permanently discontinue TEPMETKO. | |
| Increased ALT and/or AST with increased total bilirubin in the absence of cholestasis or hemolysis [see Warnings and Precautions (5.2)] | ALT and/or AST greater than 3 times ULN with total bilirubin greater than 2 times ULN | Permanently discontinue TEPMETKO. |
| Increased total bilirubin without concurrent increased ALT and/or AST [see Warnings and Precautions (5.2)] | Grade 3 | Withhold TEPMETKO until recovery to baseline bilirubin. If recovered to baseline within 7 days, then resume TEPMETKO at a reduced dose; otherwise permanently discontinue. |
| Grade 4 | Permanently discontinue TEPMETKO. | |
| Increased lipase or amylase [see Warnings and Precautions (5.3)] | Grade 3 | Withhold TEPMETKO until ≤ Grade 2 or baseline. If recovered to baseline or ≤ Grade 2 within 14 days, resume TEPMETKO at a reduced dose; otherwise permanently discontinue TEPMETKO. |
| Grade 4 | Permanently discontinue TEPMETKO. | |
| Pancreatitis [see Warnings and Precautions (5.3)] | Grade 3 or 4 | Permanently discontinue TEPMETKO. |
| Other adverse reactions [see Adverse Reactions (6.1)] | Grade 2 | Maintain dose level. If intolerable, consider withholding TEPMETKO until resolved, then resume TEPMETKO at a reduced dose. |
| Grade 3 | Withhold TEPMETKO until resolved, then resume TEPMETKO at a reduced dose. | |
| Grade 4 | Permanently discontinue TEPMETKO. |
5.1 Interstitial Lung Disease (ild)/pneumonitis
ILD/pneumonitis, which can be fatal, occurred in patients treated with TEPMETKO [see Adverse Reactions (6.1)]. ILD/pneumonitis occurred in 2% patients treated with TEPMETKO, with one patient experiencing a Grade 3 or higher event; this event resulted in death. Five patients (1%) discontinued TEPMETKO due to ILD/pneumonitis.
Monitor patients for new or worsening pulmonary symptoms indicative of ILD/pneumonitis (e.g., dyspnea, cough, fever). Immediately withhold TEPMETKO in patients with suspected ILD/pneumonitis and permanently discontinue if no other potential causes of ILD/pneumonitis are identified [see Dosage and Administration (2.4)].
8.3 Females and Males of Reproductive Potential
Based on animal data, TEPMETKO can cause malformations at doses less than the human exposure based on AUC at the 450 mg clinical dose [see Use in Specific Populations (8.1)].
2.1 Patient Selection for Metex14 Skipping Alterations
Select patients for treatment with TEPMETKO based on the presence of MET exon 14 skipping alterations in plasma or tumor specimens. Testing for the presence of MET exon 14 skipping alterations in plasma specimens is recommended only in patients for whom a tumor biopsy cannot be obtained. If an alteration is not detected in a plasma specimen, re-evaluate the feasibility of biopsy for tumor tissue testing. An FDA-approved test for detection of MET exon 14 skipping alterations in NSCLC for selecting patients for treatment with TEPMETKO is not available.
Principal Display Panel 30 Tablet Blister Pack Carton
NDC 44087-5000-3
TEPMETKO®
(tepotinib) tablets
225 mg per tablet
Rx Only
Each tablet contains 225 mg of tepotinib
(equivalent to 250 mg tepotinib hydrochloride hydrate)
Each carton contains 3 child resistant blister
cards of 10 tablets each
30 tablets
EMD
SERONO
Principal Display Panel 60 Tablet Blister Pack Carton
NDC 44087-5000-6
TEPMETKO®
(tepotinib) tablets
225 mg per tablet
Rx Only
Each tablet contains 225 mg of tepotinib
(equivalent to 250 mg tepotinib hydrochloride hydrate)
Each carton contains 6 child resistant blister
cards of 10 tablets each
60 tablets
EMD
SERONO
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been performed with tepotinib. Tepotinib and its major circulating metabolite were not mutagenic in vitro in the bacterial reverse mutation (Ames) assay, or a mouse lymphoma assay. In vivo, tepotinib was not genotoxic in a rat micronucleus test.
Fertility studies of tepotinib have not been performed. There were no morphological changes in male or female reproductive organs in repeat-dose toxicity studies in dogs.
2.3 Administration to Patients Who Have Difficulty Swallowing Solids
Place TEPMETKO tablet(s) in a glass containing 30 mL (1 ounce) of non-carbonated water. No other liquids should be used or added. Stir, without crushing, until the tablet(s) is dispersed into small pieces (tablets will not completely dissolve) and drink immediately or within 1 hour. Swallow the tablet dispersion. Do not chew pieces of the tablet. Rinse the glass with an additional 30 mL and drink immediately ensuring no residue remains in the glass and the full dose is administered.
If an administration via a naso-gastric tube (with at least 8 French gauge) is required, disperse the tablet(s) in 30 mL of non-carbonated water as described above. Administer the 30 mL of liquid immediately or within 1 hour as per naso-gastric tube manufacturer's instructions. Immediately rinse twice with 30 mL each time to ensure that no residue remains in the glass or syringe and the full dose is administered.
Structured Label Content
Section 42229-5 (42229-5)
Increased Creatinine
A median increase in serum creatinine of 30% was observed 21 days after initiation of treatment with TEPMETKO. The serum creatinine increases persisted throughout treatment and were reversible upon treatment completion.
Section 42230-3 (42230-3)
| This Patient Information has been approved by the U.S. Food and Drug Administration. | Revised 01/2025 | |
|
PATIENT INFORMATION
TEPMETKO® (tep-MET-co) (tepotinib) tablets, for oral use |
||
|
What is the most important information I should know about TEPMETKO?
|
||
|
What is TEPMETKO?
TEPMETKO is a prescription medicine used to treat adults with non-small cell lung cancer (NSCLC) that:
|
||
Before you take TEPMETKO, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I take TEPMETKO?
|
||
|
What are the possible side effects of TEPMETKO?
|
||
|
|
|
|
||
|
|
|
| The most common side effects of TEPMETKO include: | ||
|
|
|
| Your healthcare provider may change your dose, temporarily stop, or permanently stop treatment with TEPMETKO if you develop serious side effects during treatment. These are not all of the possible side effects of TEPMETKO. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||
How should I store TEPMETKO?
|
||
|
General information about the safe and effective use of TEPMETKO.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use TEPMETKO for a condition for which it was not prescribed. Do not give TEPMETKO to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about TEPMETKO that is written for health professionals. |
||
|
What are the ingredients in TEPMETKO?
Active ingredient: tepotinib Inactive ingredients: mannitol, microcrystalline cellulose, crospovidone, magnesium stearate, and colloidal silicon dioxide. Tablet coating: hypromellose, titanium dioxide, lactose monohydrate, polyethylene glycol, triacetin, and red iron oxides. Manufactured for: EMD Serono, Inc., Boston, MA 02210, U.S.A. TEPMETKO is a trademark of Merck KGaA, Darmstadt, Germany. Product of Germany. For more information, call toll-free 1-844-662-3631 or go to www.TEPMETKO.com. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store TEPMETKO at 20°C-25°C (68°F-77°F); excursions permitted to 15°C-30°C (59°F-86°F) [see USP-NF Controlled Room Temperature]. Store in original package.
11 Description (11 DESCRIPTION)
Tepotinib is a kinase inhibitor. TEPMETKO (tepotinib) tablets for oral use are formulated with tepotinib hydrochloride hydrate. The chemical name for tepotinib hydrochloride hydrate is 3-{1-[(3-{5-[(1-methylpiperidin-4-yl)methoxy]pyrimidin-2-yl}phenyl)methyl]-6-oxo-1,6-dihydropyridazin-3-yl}benzonitrile hydrochloride hydrate. The molecular formula is C29H28N6O2∙HCl∙H2O and the molecular weight is 547.05 g/mol for tepotinib hydrochloride hydrate and 492.58 g/mol for tepotinib (free base). The chemical structure is shown below:
Tepotinib hydrochloride hydrate is a white to off-white powder with a pKa of 9.5.
TEPMETKO is supplied as film-coated tablets containing 225 mg of tepotinib (equivalent to 250 mg tepotinib hydrochloride hydrate). Inactive ingredients in the tablet core are mannitol, microcrystalline cellulose, crospovidone, magnesium stearate, and colloidal silicon dioxide. The tablet coating consists of hypromellose, titanium dioxide, lactose monohydrate, polyethylene glycol, triacetin, and red iron oxides.
8.4 Pediatric Use
The safety and efficacy of TEPMETKO in pediatric patients have not been established.
8.5 Geriatric Use
Of 313 patients with NSCLC positive for METex14 skipping alterations in VISION who received 450 mg TEPMETKO once daily, 79% were 65 years or older, and 41% were 75 years or older. No clinically important differences in safety or efficacy were observed between patients aged 65 years or older and younger patients.
5.2 Hepatotoxicity
Hepatotoxicity occurred in patients treated with TEPMETKO [see Adverse Reactions (6.1)]. Increased alanine aminotransferase (ALT)/increased aspartate aminotransferase (AST) occurred in 18% of patients treated with TEPMETKO. Grade 3 or 4 increased ALT/AST occurred in 4.7% of patients. A fatal adverse reaction of hepatic failure occurred in one patient (0.2%). Four patients (0.8%) discontinued TEPMETKO due to increased ALT/AST. The median time-to-onset of Grade 3 or higher increased ALT/AST was 47 days (range 1 to 262).
Monitor liver function tests (including ALT, AST, and total bilirubin) prior to the start of TEPMETKO, every 2 weeks during the first 3 months of treatment, then once a month or as clinically indicated, with more frequent testing in patients who develop increased transaminases or bilirubin. Based on the severity of the adverse reaction, withhold, dose reduce, or permanently discontinue TEPMETKO [see Dosage and Administration (2.4)].
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of TEPMETKO was evaluated in a single-arm, open-label, multicenter, non-randomized, multicohort study (VISION, NCT02864992). Eligible patients were required to have advanced or metastatic NSCLC harboring METex14 skipping alterations, epidermal growth factor receptor (EGFR) wild-type and anaplastic lymphoma kinase (ALK) negative status, at least one measurable lesion as defined by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1, and Eastern Cooperative Oncology Group (ECOG) Performance Status of 0 to 1. Patients with symptomatic CNS metastases, clinically significant uncontrolled cardiac disease, or who received treatment with any MET or hepatocyte growth factor (HGF) inhibitor were not eligible for the study.
Identification of METex14 skipping alterations was prospectively determined using central laboratories employing either a PCR-based or next-generation sequencing-based clinical trial assay using tissue (66%) and/or plasma (57%) samples.
Patients received TEPMETKO 450 mg once daily until disease progression or unacceptable toxicity. The major efficacy outcome measure was confirmed overall response rate (ORR) according to Response Evaluation Criteria in Solid Tumors (RECIST v1.1) as evaluated by a Blinded Independent Review Committee (BIRC). An additional efficacy outcome measure was duration of response (DOR) by BIRC.
The efficacy population included 164 treatment naïve patients and 149 previously treated patients. The median age was 72 years (range 41 to 94 years); 51% female; 62% White, 34% Asian; 26% had Eastern Cooperative Oncology Group (ECOG) Performance Status (PS) 0 and 74% had ECOG PS 1; 49% never smoked; 81% had adenocarcinoma; 94% had metastatic disease; and 13% had CNS metastases. Amongst previously treated patients, 84% received prior platinum-based chemotherapy.
Efficacy results are presented in Table 4.
| Efficacy parameter | Treatment-Naïve (N=164) |
Previously Treated (N=149) |
|---|---|---|
| CI: Confidence interval, + denotes ongoing response. | ||
| Overall response rate (95% CI) Blinded Independent Review Committee (BIRC) review.
,
Confirmed responses.
|
57% (49, 65) | 45% (37, 53) |
| Duration of Response | ||
| Range in months Based on observed duration of response.
|
1.3+, 56.6+ | 1.4+, 67.6+ |
| Patients with DOR ≥ 6 months | 66% | 66% |
| Patients with DOR ≥ 12 months | 40% | 36% |
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described in greater detail elsewhere in the labeling:
- Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1)]
- Hepatotoxicity [see Warnings and Precautions (5.2)]
- Pancreatic Toxicity [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Certain P-gp substrates: Avoid coadministration of TEPMETKO with P-gp substrates where minimal concentration changes may lead to serious or life-threatening toxicities. (7.1)
8.6 Renal Impairment
No dosage modification is recommended in patients with mild or moderate renal impairment (creatinine clearance [CLcr] 30 to 89 mL/min, estimated by Cockcroft-Gault). The recommended dosage has not been established for patients with severe renal impairment (CLcr < 30 mL/min) [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of tepotinib were evaluated in patients with cancer administered 450 mg once daily unless otherwise specified. Tepotinib exposure (AUC0-12h and Cmax) increases dose-proportionally over the dose range of 27 mg (0.06 times the recommended daily dosage) to 450 mg. At the recommended dosage, the geometric mean (coefficient of variation [CV] %) steady state Cmax was 1,291 ng/mL (48.1%) and the AUC0-24h was 27,438 ng∙h/mL (51.7%). The oral clearance of tepotinib did not change with respect to time. The median accumulation was 2.5-fold for Cmax and 3.3-fold for AUC0-24h after multiple daily doses of tepotinib.
2.2 Recommended Dosage
The recommended dosage of TEPMETKO is 450 mg orally once daily with food [see Clinical Pharmacology (12.3)] until disease progression or unacceptable toxicity.
Instruct patients to take their dose of TEPMETKO at approximately the same time every day and to swallow tablets whole. Do not chew, crush or split tablets. Patients who have difficulty swallowing solids can disperse tablets in water [see Dosage and Administration (2.3)].
Advise patients not to make up a missed dose within 8 hours of the next scheduled dose.
If vomiting occurs after taking a dose of TEPMETKO, advise patients to take the next dose at the scheduled time.
8.7 Hepatic Impairment
No dosage modification is recommended in patients with mild (Child Pugh Class A) or moderate (Child Pugh Class B) hepatic impairment. The pharmacokinetics and safety of tepotinib in patients with severe hepatic impairment (Child Pugh Class C) have not been studied [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
TEPMETKO is indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) harboring mesenchymal-epithelial transition (MET) exon 14 skipping alterations.
5.3 Pancreatic Toxicity
Elevations in amylase and lipase levels occurred in patients treated with TEPMETKO [see Adverse Reactions (6.1)]. Increased amylase and/or lipase occurred in 13% of patients treated with TEPMETKO. Grade 3 and 4 increased amylase and/or lipase occurred in 5% and 1.2% of patients, respectively. Monitor amylase and lipase at baseline and regularly during treatment with TEPMETKO. Based on the severity of the adverse drug reaction, temporarily withhold, dose reduce, or permanently discontinue TEPMETKO [see Dosage and Administration (2.4)].
12.1 Mechanism of Action
Tepotinib is a kinase inhibitor that targets MET, including variants with exon 14 skipping alterations. Tepotinib inhibits hepatocyte growth factor (HGF)-dependent and -independent MET phosphorylation and MET-dependent downstream signaling pathways. Tepotinib also inhibited melatonin 2 and imidazoline 1 receptors at clinically achievable concentrations.
In vitro, tepotinib inhibited tumor cell proliferation, anchorage-independent growth, and migration of MET-dependent tumor cells. In mice implanted with tumor cell lines with oncogenic activation of MET, including METex14 skipping alterations, tepotinib inhibited tumor growth, led to sustained inhibition of MET phosphorylation, and, in one model, decreased the formation of metastases.
5.4 Embryo Fetal Toxicity (5.4 Embryo-Fetal Toxicity)
Based on findings in animal studies and its mechanism of action TEPMETKO can cause fetal harm when administered to a pregnant woman. Oral administration of tepotinib to pregnant rabbits during the period of organogenesis resulted in malformations (teratogenicity) and anomalies at exposures less than the human exposure based on area under the curve (AUC) at the 450 mg daily clinical dose. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential or males with female partners of reproductive potential to use effective contraception during treatment with TEPMETKO and for one week after the last dose. [See Use in Specific Populations (8.1, 8.3)]
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Interstitial Lung Disease (ILD)/Pneumonitis: Immediately withhold TEPMETKO in patients with suspected ILD/pneumonitis. Permanently discontinue TEPMETKO in patients diagnosed with ILD/pneumonitis of any severity. (2.4, 5.1)
- Hepatotoxicity: Monitor liver function tests. Withhold, dose reduce, or permanently discontinue TEPMETKO based on severity. (5.2)
- Pancreatic Toxicity: Monitor amylase and lipase. Withhold, dose reduce, or permanently discontinue TEPMETKO based on severity. (5.3)
- Embryo-fetal toxicity: TEPMETKO can cause fetal harm. Advise of potential risk to a fetus and use of effective contraception. (5.4, 8.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 225 mg, white-pink, oval, biconvex film-coated tablets with embossment "M" on one side and plain on the other side.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety population described in the WARNINGS AND PRECAUTIONS reflect exposure to TEPMETKO in 506 patients with solid tumors enrolled in five open-label, single-arm studies receiving TEPMETKO as single agent at a dose of 450 mg once daily. This included 313 patients with NSCLC positive for METex14 skipping alterations, who received TEPMETKO in VISION. Among 506 patients who received TEPMETKO, 44% were exposed for 6 months or longer, and 22% were exposed for more than one year.
The data described below reflect exposure to TEPMETKO 450 mg once daily in 313 patients with metastatic non-small cell lung cancer (NSCLC) with METex14 skipping alterations in VISION [see Clinical Studies (14)].
Serious adverse reactions occurred in 51% of patients who received TEPMETKO. Serious adverse reactions in > 2% of patients included pleural effusion (6%), pneumonia (6%), edema (5%), general health deterioration (3.8%), dyspnea (3.5%), musculoskeletal pain (2.9%), and pulmonary embolism (2.2%). Fatal adverse reactions occurred in 1.9% of patients who received TEPMETKO, including pneumonitis (0.3%), hepatic failure (0.3%), dyspnea from fluid overload (0.3%), pneumonia (0.3%), sepsis (0.3%), and death of unknown cause (0.3%).
Permanent discontinuation due to an adverse reaction occurred in 25% of patients who received TEPMETKO. The most frequent adverse reactions (> 1%) leading to permanent discontinuations of TEPMETKO were edema (8%), pleural effusion (1.6%), and general health deterioration (1.6%).
Dosage interruptions due to an adverse reaction occurred in 53% of patients who received TEPMETKO. Adverse reactions which required dosage interruption in > 2% of patients who received TEPMETKO included edema (28%), increased blood creatinine (6%), pleural effusion (3.5%), nausea (3.2%), increased ALT (2.9%), pneumonia (2.6%), decreased appetite (2.2%), and dyspnea (2.2%).
Dose reductions due to an adverse reaction occurred in 36% of patients who received TEPMETKO. Adverse reactions which required dose reductions in > 2% of patients who received TEPMETKO included edema (22%), increased blood creatinine (2.9%), fatigue (2.2%), and pleural effusion (2.2%).
The most common adverse reactions (≥ 20%) in patients who received TEPMETKO were edema, nausea, fatigue, musculoskeletal pain, diarrhea, dyspnea, decreased appetite, and rash. The most common Grade 3 to 4 laboratory abnormalities (≥ 2%) were decreased lymphocytes, decreased albumin, decreased sodium, increased gamma-glutamyltransferase, increased amylase, increased lipase, increased ALT, increased AST, and decreased hemoglobin.
Table 2 summarizes the adverse reactions in VISION.
| Adverse Reactions | TEPMETKO (N=313) |
|
|---|---|---|
| All Grades Severity as defined by National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 4.03.
(%) |
Grades 3 to 4
(%) |
|
| General disorders and administration-site conditions | ||
| Edema Edema includes eye edema, face edema, generalized edema, localized edema, edema, genital edema, peripheral edema, peripheral swelling, periorbital edema, and scrotal edema.
|
81 | 16 |
| Fatigue Fatigue includes asthenia and fatigue.
|
30 | 1.9 |
| Gastrointestinal disorders | ||
| Nausea | 31 | 1.3 |
| Diarrhea | 29 | 0.6 |
| Abdominal pain Abdominal pain includes abdominal discomfort, abdominal pain, abdominal pain lower, abdominal pain upper, gastrointestinal pain, and hepatic pain.
|
19 | 0.6 |
| Constipation | 19 | 0.3 |
| Vomiting Vomiting includes retching and vomiting.
|
15 | 1 |
| Musculoskeletal and Connective Tissue Disorders | ||
| Musculoskeletal pain Musculoskeletal pain includes arthralgia, arthritis, back pain, bone pain, musculoskeletal chest pain, musculoskeletal pain, myalgia, non-cardiac chest pain, pain in extremity, and spinal pain.
|
30 | 3.2 |
| Respiratory, thoracic, and mediastinal disorders | ||
| Dyspnea Dyspnea includes dyspnea, dyspnea at rest, and dyspnea exertional.
|
24 | 2.6 |
| Cough Cough includes cough, and productive cough.
|
18 | 0.3 |
| Pleural effusion | 14 | 4.2 |
| Metabolism and nutrition disorders | ||
| Decreased appetite | 21 | 1.9 |
| Skin and Subcutaneous Tissue Disorders | ||
| Rash Rash includes rash, palmar-plantar erythrodysesthesia syndrome, rash maculo-papular, eczema, exfoliative rash, rash erythematous, rash pustular, skin exfoliation, dermatitis acneiform, drug eruption, dermatitis, rash pruritic, dermatitis bullous, toxic skin eruption.
|
21 | 1.3 |
| Infections and Infestations | ||
| Pneumonia Pneumonia includes pneumonia, pneumonia aspiration, and pneumonia bacterial.
|
12 | 3.8 |
Clinically relevant adverse reactions in < 10% of patients who received TEPMETKO included ILD/pneumonitis, fever, dizziness, pruritus, and headache.
Table 3 summarizes the laboratory abnormalities observed in VISION.
| Laboratory Abnormalities | TEPMETKO The denominator used to calculate the rate varied from 268 to 309 based on the number of patients with a baseline value and at least one post-treatment value.
|
|
|---|---|---|
| Grades 1 to 4 Severity as defined by National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 4.03.
(%) |
Grades 3 to 4
(%) |
|
| Chemistry | ||
| Decreased albumin | 81 | 9 |
| Increased creatinine | 60 | 1 |
| Increased alkaline phosphatase aminotransferase | 52 | 1.6 |
| Increased alanine aminotransferase | 50 | 4.9 |
| Increased aspartate aminotransferase | 40 | 3.6 |
| Decreased sodium | 36 | 9 |
| Increased gamma-glutamyltransferase | 29 | 6 |
| Increased potassium | 26 | 1.9 |
| Increased amylase | 25 | 5 |
| Increased lipase | 21 | 5 |
| Hematology | ||
| Decreased lymphocytes | 57 | 15 |
| Decreased hemoglobin | 31 | 3.6 |
| Decreased leukocytes | 25 | 1.9 |
| Decreased platelets | 24 | 0.6 |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
TEPMETKO (tepotinib) tablets: 225 mg tepotinib, white-pink, oval, biconvex film-coated tablet with embossment "M" on one side and plain on the other side.
| NDC number | Size |
|---|---|
| 44087-5000-3 | Box of 30 tablets: 3 blister cards each containing 10 tablets |
| 44087-5000-6 | Box of 60 tablets: 6 blister cards each containing 10 tablets |
The blister cards consist of a child-resistant blister foil.
2.4 Dose Modifications for Adverse Reactions
The recommended dose reduction of TEPMETKO for the management of adverse reactions is 225 mg orally once daily.
Permanently discontinue TEPMETKO in patients who are unable to tolerate 225 mg orally once daily.
The recommended dosage modifications of TEPMETKO for adverse reactions are provided in Table 1.
| Adverse Reaction | Severity Severity as defined by National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) Version 5.
|
Dose Modification |
|---|---|---|
| Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.1)] | Any Grade | Withhold TEPMETKO if ILD is suspected. Permanently discontinue TEPMETKO if ILD is confirmed. |
| Increased ALT and/or AST without increased total bilirubin [see Warnings and Precautions (5.2)] | Grade 3 | Withhold TEPMETKO until recovery to baseline ALT/AST. If recovered to baseline within 7 days, then resume TEPMETKO at the same dose; otherwise resume TEPMETKO at a reduced dose. |
| Grade 4 | Permanently discontinue TEPMETKO. | |
| Increased ALT and/or AST with increased total bilirubin in the absence of cholestasis or hemolysis [see Warnings and Precautions (5.2)] | ALT and/or AST greater than 3 times ULN with total bilirubin greater than 2 times ULN | Permanently discontinue TEPMETKO. |
| Increased total bilirubin without concurrent increased ALT and/or AST [see Warnings and Precautions (5.2)] | Grade 3 | Withhold TEPMETKO until recovery to baseline bilirubin. If recovered to baseline within 7 days, then resume TEPMETKO at a reduced dose; otherwise permanently discontinue. |
| Grade 4 | Permanently discontinue TEPMETKO. | |
| Increased lipase or amylase [see Warnings and Precautions (5.3)] | Grade 3 | Withhold TEPMETKO until ≤ Grade 2 or baseline. If recovered to baseline or ≤ Grade 2 within 14 days, resume TEPMETKO at a reduced dose; otherwise permanently discontinue TEPMETKO. |
| Grade 4 | Permanently discontinue TEPMETKO. | |
| Pancreatitis [see Warnings and Precautions (5.3)] | Grade 3 or 4 | Permanently discontinue TEPMETKO. |
| Other adverse reactions [see Adverse Reactions (6.1)] | Grade 2 | Maintain dose level. If intolerable, consider withholding TEPMETKO until resolved, then resume TEPMETKO at a reduced dose. |
| Grade 3 | Withhold TEPMETKO until resolved, then resume TEPMETKO at a reduced dose. | |
| Grade 4 | Permanently discontinue TEPMETKO. |
5.1 Interstitial Lung Disease (ild)/pneumonitis (5.1 Interstitial Lung Disease (ILD)/Pneumonitis)
ILD/pneumonitis, which can be fatal, occurred in patients treated with TEPMETKO [see Adverse Reactions (6.1)]. ILD/pneumonitis occurred in 2% patients treated with TEPMETKO, with one patient experiencing a Grade 3 or higher event; this event resulted in death. Five patients (1%) discontinued TEPMETKO due to ILD/pneumonitis.
Monitor patients for new or worsening pulmonary symptoms indicative of ILD/pneumonitis (e.g., dyspnea, cough, fever). Immediately withhold TEPMETKO in patients with suspected ILD/pneumonitis and permanently discontinue if no other potential causes of ILD/pneumonitis are identified [see Dosage and Administration (2.4)].
8.3 Females and Males of Reproductive Potential
Based on animal data, TEPMETKO can cause malformations at doses less than the human exposure based on AUC at the 450 mg clinical dose [see Use in Specific Populations (8.1)].
2.1 Patient Selection for Metex14 Skipping Alterations (2.1 Patient Selection for METex14 Skipping Alterations)
Select patients for treatment with TEPMETKO based on the presence of MET exon 14 skipping alterations in plasma or tumor specimens. Testing for the presence of MET exon 14 skipping alterations in plasma specimens is recommended only in patients for whom a tumor biopsy cannot be obtained. If an alteration is not detected in a plasma specimen, re-evaluate the feasibility of biopsy for tumor tissue testing. An FDA-approved test for detection of MET exon 14 skipping alterations in NSCLC for selecting patients for treatment with TEPMETKO is not available.
Principal Display Panel 30 Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 30 Tablet Blister Pack Carton)
NDC 44087-5000-3
TEPMETKO®
(tepotinib) tablets
225 mg per tablet
Rx Only
Each tablet contains 225 mg of tepotinib
(equivalent to 250 mg tepotinib hydrochloride hydrate)
Each carton contains 3 child resistant blister
cards of 10 tablets each
30 tablets
EMD
SERONO
Principal Display Panel 60 Tablet Blister Pack Carton (PRINCIPAL DISPLAY PANEL - 60 Tablet Blister Pack Carton)
NDC 44087-5000-6
TEPMETKO®
(tepotinib) tablets
225 mg per tablet
Rx Only
Each tablet contains 225 mg of tepotinib
(equivalent to 250 mg tepotinib hydrochloride hydrate)
Each carton contains 6 child resistant blister
cards of 10 tablets each
60 tablets
EMD
SERONO
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been performed with tepotinib. Tepotinib and its major circulating metabolite were not mutagenic in vitro in the bacterial reverse mutation (Ames) assay, or a mouse lymphoma assay. In vivo, tepotinib was not genotoxic in a rat micronucleus test.
Fertility studies of tepotinib have not been performed. There were no morphological changes in male or female reproductive organs in repeat-dose toxicity studies in dogs.
2.3 Administration to Patients Who Have Difficulty Swallowing Solids
Place TEPMETKO tablet(s) in a glass containing 30 mL (1 ounce) of non-carbonated water. No other liquids should be used or added. Stir, without crushing, until the tablet(s) is dispersed into small pieces (tablets will not completely dissolve) and drink immediately or within 1 hour. Swallow the tablet dispersion. Do not chew pieces of the tablet. Rinse the glass with an additional 30 mL and drink immediately ensuring no residue remains in the glass and the full dose is administered.
If an administration via a naso-gastric tube (with at least 8 French gauge) is required, disperse the tablet(s) in 30 mL of non-carbonated water as described above. Administer the 30 mL of liquid immediately or within 1 hour as per naso-gastric tube manufacturer's instructions. Immediately rinse twice with 30 mL each time to ensure that no residue remains in the glass or syringe and the full dose is administered.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:46.032065 · Updated: 2026-03-14T22:25:35.146821