7fd350ec-af99-4eb2-8750-226edc7f24b0
7fd350ec-af99-4eb2-8750-226edc7f24b0
34390-5
HUMAN OTC DRUG LABEL
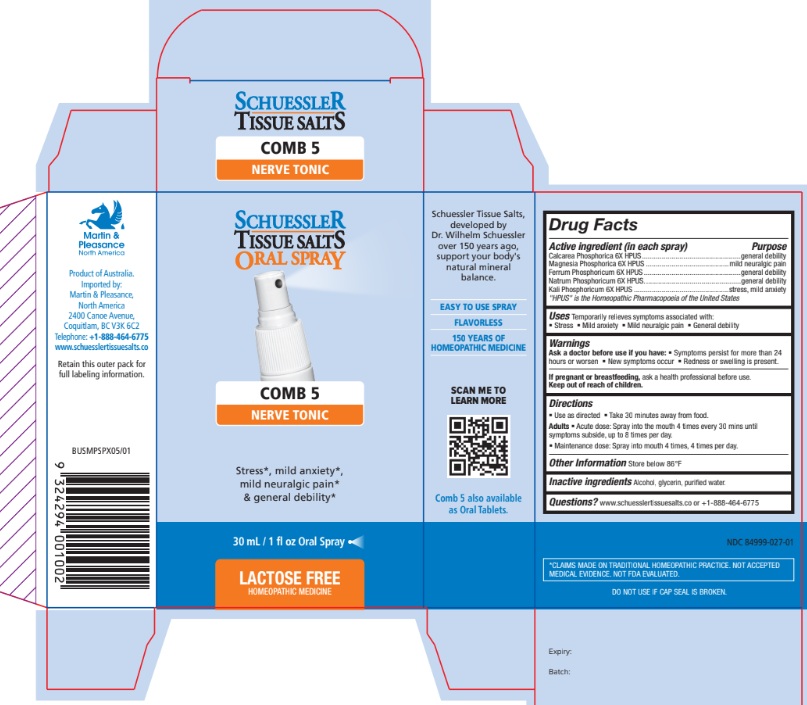
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient (in each spray) Purpose Calcarea Phosphorica 6X HPUS .................................................. general debility Magnesia Phosphorica 6X HPUS .......................................... mild neuralgic pain Ferrum Phosphoricum 6X HPUS ................................................. general debility Natrum Phosphoricum 6X HPUS ................................................. general debility Kali Phosphoricum 6X HPUS ................................................ stress, mild anxiety "HPUS" is the Homeopathic Pharmacopoeia of the United States
Medication Information
Warnings
Ask a doctor before use if you have:
- Symptoms persist for more than 24 hours or worsen
- New symptoms occur
- Redness or swelling is present.
If pregnant or breastfeeding, ask a health professional before use. Keep out of reach of children.
Active Ingredient
Active ingredient (in each spray) Purpose
Calcarea Phosphorica 6X HPUS .................................................. general debility
Magnesia Phosphorica 6X HPUS .......................................... mild neuralgic pain
Ferrum Phosphoricum 6X HPUS ................................................. general debility
Natrum Phosphoricum 6X HPUS ................................................. general debility
Kali Phosphoricum 6X HPUS ................................................ stress, mild anxiety
"HPUS" is the Homeopathic Pharmacopoeia of the United States
Uses
Temporarily relieves symptoms associated with:
- Stress
- Mild anxiety
- Mild neuralgic pain
- General debility
Directions
- Use as directed
- Take 30 minutes away from food.
Adults
- Acute dose: Spray into the mouth 4 times every 30 mins until symptoms subside, up to 8 times per day.
- Maintenance dose: Spray into mouth 4 times, 4 times per day.
Other Information
Store below 86°F
Inactive Ingredients
Alcohol, Glycerin, Purified Water
Description
Active ingredient (in each spray) Purpose Calcarea Phosphorica 6X HPUS .................................................. general debility Magnesia Phosphorica 6X HPUS .......................................... mild neuralgic pain Ferrum Phosphoricum 6X HPUS ................................................. general debility Natrum Phosphoricum 6X HPUS ................................................. general debility Kali Phosphoricum 6X HPUS ................................................ stress, mild anxiety "HPUS" is the Homeopathic Pharmacopoeia of the United States
Section 42229-5
Questions? www.schuesslertissuesalts.co or + 1-888-464-6775
Section 50565-1
Section 55105-1
Product Label
Structured Label Content
Warnings
Ask a doctor before use if you have:
- Symptoms persist for more than 24 hours or worsen
- New symptoms occur
- Redness or swelling is present.
If pregnant or breastfeeding, ask a health professional before use. Keep out of reach of children.
Active Ingredient (55106-9)
Active ingredient (in each spray) Purpose
Calcarea Phosphorica 6X HPUS .................................................. general debility
Magnesia Phosphorica 6X HPUS .......................................... mild neuralgic pain
Ferrum Phosphoricum 6X HPUS ................................................. general debility
Natrum Phosphoricum 6X HPUS ................................................. general debility
Kali Phosphoricum 6X HPUS ................................................ stress, mild anxiety
"HPUS" is the Homeopathic Pharmacopoeia of the United States
Uses
Temporarily relieves symptoms associated with:
- Stress
- Mild anxiety
- Mild neuralgic pain
- General debility
Directions
- Use as directed
- Take 30 minutes away from food.
Adults
- Acute dose: Spray into the mouth 4 times every 30 mins until symptoms subside, up to 8 times per day.
- Maintenance dose: Spray into mouth 4 times, 4 times per day.
Other Information
Store below 86°F
Inactive Ingredients
Alcohol, Glycerin, Purified Water
Section 42229-5 (42229-5)
Questions? www.schuesslertissuesalts.co or + 1-888-464-6775
Section 50565-1 (50565-1)
Section 55105-1 (55105-1)
Product Label (Product label)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:12:05.533946 · Updated: 2026-03-14T23:17:28.220549