Xolair
7f6a2191-adfb-48b9-9bfa-0d9920479f0d
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
XOLAIR is an anti-IgE antibody indicated for: Moderate to severe persistent asthma in adults and pediatric patients 6 years of age and older with a positive skin test or in vitro reactivity to a perennial aeroallergen and symptoms that are inadequately controlled with inhaled corticosteroids ( 1.1 ) Chronic rhinosinusitis with nasal polyps (CRSwNP) in adult patients 18 years of age and older with inadequate response to nasal corticosteroids, as add-on maintenance treatment ( 1.2 ) IgE-mediated food allergy in adult and pediatric patients aged 1 year and older for the reduction of allergic reactions (Type I), including anaphylaxis, that may occur with accidental exposure to one or more foods. To be used in conjunction with food allergen avoidance ( 1.3 ) Chronic spontaneous urticaria (CSU) in adults and adolescents 12 years of age and older who remain symptomatic despite H1 antihistamine treatment ( 1.4 ) Limitations of Use : Not indicated for acute bronchospasm or status asthmaticus. ( 1.1 , 5.3 ) Not indicated for the emergency treatment of allergic reactions, including anaphylaxis ( 1.3 ) Not indicated for other forms of urticaria. ( 1.4 )
Dosage and Administration
For subcutaneous (SC) administration only. ( 2.2 , 2.3 , 2.4 , 2.5 ) See full prescribing information for administration instructions ( 2.6 , 2.7 , 2.8 ). Asthma : XOLAIR 75 to 375 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination charts. ( 2.2 ) Chronic Rhinosinusitis with Nasal Polyps : XOLAIR 75 to 600 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination charts. ( 2.3 ) IgE-Mediated Food Allergy : XOLAIR 75 mg to 600 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination chart. ( 2.4 ) Chronic Spontaneous Urticaria : XOLAIR 150 or 300 mg SC every 4 weeks. Dosing in CSU is not dependent on serum IgE level or body weight. ( 2.5 )
Contraindications
XOLAIR is contraindicated in patients with severe hypersensitivity reaction to XOLAIR or any ingredient of XOLAIR [see Warnings and Precautions (5.1) ] .
Warnings and Precautions
Anaphylaxis: Initiate XOLAIR therapy in a healthcare setting prepared to manage anaphylaxis which can be life-threatening and observe patients for an appropriate period of time after administration. ( 5.1 ) Malignancy: Malignancies have been observed in clinical studies. ( 5.2 ) Acute Asthma Symptoms: Do not use for the treatment of acute bronchospasm or status asthmaticus. ( 5.3 ) Corticosteroid Reduction: Do not abruptly discontinue corticosteroids upon initiation of XOLAIR therapy. ( 5.4 ) Eosinophilic Conditions: Be alert to eosinophilia, vasculitic rash, worsening pulmonary symptoms, cardiac complications, and/or neuropathy, especially upon reduction of oral corticosteroids. ( 5.5 ) Fever, Arthralgia, and Rash: Stop XOLAIR if patients develop signs and symptoms similar to serum sickness. ( 5.6 ) Potential Medication Error Related to Emergency Treatment of Anaphylaxis: XOLAIR should not be used for emergency treatment of allergic reactions, including anaphylaxis. ( 5.9 )
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Anaphylaxis [see Boxed Warning and Warnings and Precautions (5.1) ] Malignancies [see Warnings and Precautions (5.2) ]
Drug Interactions
No formal drug interaction studies have been performed with XOLAIR. In patients with asthma, CRSwNP, and IgE-mediated food allergy the concomitant use of XOLAIR and allergen immunotherapy has not been evaluated . In patients with CSU, the use of XOLAIR in combination with immunosuppressive therapies has not been studied.
Description
Anaphylaxis presenting as bronchospasm, hypotension, syncope, urticaria, and/or angioedema of the throat or tongue, has been reported to occur after administration of XOLAIR. Anaphylaxis has occurred as early as after the first dose of XOLAIR, but also has occurred beyond 1 year after beginning regularly administered treatment. Because of the risk of anaphylaxis, initiate XOLAIR therapy in a healthcare setting and closely observe patients for an appropriate period of time after XOLAIR administration. Health care providers administering XOLAIR should be prepared to manage anaphylaxis which can be life-threatening. Inform patients of the signs and symptoms of anaphylaxis and instruct them to seek immediate medical care should symptoms occur. Selection of patients for self-administration of XOLAIR should be based on criteria to mitigate risk from anaphylaxis [see Dosage and Administration (2.6) , Warnings and Precautions (5.1) and Adverse Reactions (6.1 , 6.2) ] .
Medication Information
Warnings and Precautions
Anaphylaxis: Initiate XOLAIR therapy in a healthcare setting prepared to manage anaphylaxis which can be life-threatening and observe patients for an appropriate period of time after administration. ( 5.1 ) Malignancy: Malignancies have been observed in clinical studies. ( 5.2 ) Acute Asthma Symptoms: Do not use for the treatment of acute bronchospasm or status asthmaticus. ( 5.3 ) Corticosteroid Reduction: Do not abruptly discontinue corticosteroids upon initiation of XOLAIR therapy. ( 5.4 ) Eosinophilic Conditions: Be alert to eosinophilia, vasculitic rash, worsening pulmonary symptoms, cardiac complications, and/or neuropathy, especially upon reduction of oral corticosteroids. ( 5.5 ) Fever, Arthralgia, and Rash: Stop XOLAIR if patients develop signs and symptoms similar to serum sickness. ( 5.6 ) Potential Medication Error Related to Emergency Treatment of Anaphylaxis: XOLAIR should not be used for emergency treatment of allergic reactions, including anaphylaxis. ( 5.9 )
Indications and Usage
XOLAIR is an anti-IgE antibody indicated for: Moderate to severe persistent asthma in adults and pediatric patients 6 years of age and older with a positive skin test or in vitro reactivity to a perennial aeroallergen and symptoms that are inadequately controlled with inhaled corticosteroids ( 1.1 ) Chronic rhinosinusitis with nasal polyps (CRSwNP) in adult patients 18 years of age and older with inadequate response to nasal corticosteroids, as add-on maintenance treatment ( 1.2 ) IgE-mediated food allergy in adult and pediatric patients aged 1 year and older for the reduction of allergic reactions (Type I), including anaphylaxis, that may occur with accidental exposure to one or more foods. To be used in conjunction with food allergen avoidance ( 1.3 ) Chronic spontaneous urticaria (CSU) in adults and adolescents 12 years of age and older who remain symptomatic despite H1 antihistamine treatment ( 1.4 ) Limitations of Use : Not indicated for acute bronchospasm or status asthmaticus. ( 1.1 , 5.3 ) Not indicated for the emergency treatment of allergic reactions, including anaphylaxis ( 1.3 ) Not indicated for other forms of urticaria. ( 1.4 )
Dosage and Administration
For subcutaneous (SC) administration only. ( 2.2 , 2.3 , 2.4 , 2.5 ) See full prescribing information for administration instructions ( 2.6 , 2.7 , 2.8 ). Asthma : XOLAIR 75 to 375 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination charts. ( 2.2 ) Chronic Rhinosinusitis with Nasal Polyps : XOLAIR 75 to 600 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination charts. ( 2.3 ) IgE-Mediated Food Allergy : XOLAIR 75 mg to 600 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination chart. ( 2.4 ) Chronic Spontaneous Urticaria : XOLAIR 150 or 300 mg SC every 4 weeks. Dosing in CSU is not dependent on serum IgE level or body weight. ( 2.5 )
Contraindications
XOLAIR is contraindicated in patients with severe hypersensitivity reaction to XOLAIR or any ingredient of XOLAIR [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Anaphylaxis [see Boxed Warning and Warnings and Precautions (5.1) ] Malignancies [see Warnings and Precautions (5.2) ]
Drug Interactions
No formal drug interaction studies have been performed with XOLAIR. In patients with asthma, CRSwNP, and IgE-mediated food allergy the concomitant use of XOLAIR and allergen immunotherapy has not been evaluated . In patients with CSU, the use of XOLAIR in combination with immunosuppressive therapies has not been studied.
Description
Anaphylaxis presenting as bronchospasm, hypotension, syncope, urticaria, and/or angioedema of the throat or tongue, has been reported to occur after administration of XOLAIR. Anaphylaxis has occurred as early as after the first dose of XOLAIR, but also has occurred beyond 1 year after beginning regularly administered treatment. Because of the risk of anaphylaxis, initiate XOLAIR therapy in a healthcare setting and closely observe patients for an appropriate period of time after XOLAIR administration. Health care providers administering XOLAIR should be prepared to manage anaphylaxis which can be life-threatening. Inform patients of the signs and symptoms of anaphylaxis and instruct them to seek immediate medical care should symptoms occur. Selection of patients for self-administration of XOLAIR should be based on criteria to mitigate risk from anaphylaxis [see Dosage and Administration (2.6) , Warnings and Precautions (5.1) and Adverse Reactions (6.1 , 6.2) ] .
Section 42229-5
Limitations of Use:
XOLAIR is not indicated for the relief of acute bronchospasm or status asthmaticus.
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised:2/2024 | |
| MEDICATION GUIDE | ||
|
XOLAIR® (ZOHL-air)
(omalizumab) injection, for subcutaneous use |
XOLAIR® (ZOHL-air)
(omalizumab) for injection, for subcutaneous use |
|
|
What is the most important information I should know about XOLAIR? XOLAIR may cause serious side effects, including: Severe allergic reaction. A severe allergic reaction called anaphylaxis can happen when you receive XOLAIR. The reaction can occur after the first dose, or after many doses. It may also occur right after a XOLAIR injection or days later. Anaphylaxis is a life-threatening condition and can lead to death. Go to the nearest emergency room right away if you have any of these symptoms of an allergic reaction:
|
||
|
What is XOLAIR?
XOLAIR is an injectable prescription medicine used to treat:
|
||
|
Who should not receive and use XOLAIR? Do not receive and use XOLAIR if you:
|
||
|
What should I tell my healthcare provider before receiving XOLAIR? Before receiving XOLAIR, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I receive and use XOLAIR?
|
||
|
What are the possible side effects of XOLAIR? XOLAIR may cause serious side effects, including:
|
||
|
|
|
|
||
How should I store XOLAIR?
|
||
|
General information about the safe and effective use of XOLAIR.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use XOLAIR for a condition for which it was not prescribed. Do not give XOLAIR to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about XOLAIR that is written for health professionals. For more information, go to www.xolair.com or call 1-866-4XOLAIR (1-866-496-5247). |
||
|
What are the ingredients in XOLAIR? Active ingredient: omalizumab Inactive ingredients: Prefilled syringe or Autoinjector: arginine hydrochloride, histidine, L-histidine hydrochloride monohydrate, and polysorbate 20 Vial: histidine, L-histidine hydrochloride monohydrate, polysorbate 20 and sucrose Manufactured by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990. U.S. License No.: 1048 Jointly marketed by: Genentech USA, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 Novartis Pharmaceuticals Corporation, One Health Plaza, East Hanover, NJ 07936-1080 XOLAIR® is a registered trademark of Novartis AG. ©2024 Genentech USA, Inc. |
Section 43683-2
Section 44425-7
Storage
XOLAIR prefilled syringe and autoinjector should be shipped and stored under refrigerated conditions 2°C to 8°C (36°F to 46°F) in the original carton. Protect from direct sunlight. XOLAIR prefilled syringe and autoinjector can be removed from and placed back in the refrigerator if needed. The total combined time out of the refrigerator may not be more than 2 days. Do not use if prefilled syringe or autoinjector is left at temperatures above 25°C (77°F).
Do not freeze. Do not use if the prefilled syringe or autoinjector has been frozen.
Section 59845-8
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: 2/2024 |
|
Instructions for Use Xolair® (ZOHL-air) (omalizumab) injection, for subcutaneous use Prefilled Syringe |
|
| Read this Instructions for Use before you start using the XOLAIR prefilled syringe and each time you get a refill. Before you use the XOLAIR prefilled syringe for the first time, make sure your healthcare provider shows you the right way to use it. Contact your healthcare provider if you have any questions. | |
| Do not use XOLAIR for the emergency treatment of any allergic reactions, including anaphylaxis, hives, or sudden breathing problems. | |
|
|
|
Supplies needed to give your injection
|
|
|
Choose the correct prefilled syringe or combination of prefilled syringes
|
|
|
|
|
|
|
|
How should I store XOLAIR?
|
|
Important Information
|
|
| Preparing for the Injection | |
| 1 Take the carton containing the prefilled syringe out of the refrigerator. | |
|
|
| 2 Check the expiration date on the XOLAIR carton. | |
|
|
|
| 3 Place the carton on a clean, flat surface. | |
|
|
| 4 Open the carton. | |
|
|
|
|
|
|
|
|
5 Inspect the prefilled syringe closely.
|
|
| 6 Choose an injection site. | |
|
|
| 7 Wipe the injection site with an alcohol swab in a circular motion and let it air dry for 10 seconds. | |
|
|
| Giving the Injection | |
| 8 Hold the prefilled syringe firmly by the center with 1 hand and pull the needle cap straight off with your other hand. | |
|
|
|
|
|
| 9 Use your other hand and gently pinch the area of skin that was cleaned. Hold the pinched skin tight. | |
|
|
| 10 Continue holding the prefilled syringe by the center and use a quick, dart-like motion to insert the needle all the way into the pinched skin at an angle between 45 degrees to 90 degrees as shown. | |
|
|
| 11 Slowly inject all of the medicine by gently pushing the plunger all the way down until the needle-shield wings are pushed apart. | |
|
|
| 12 Release the plunger and allow the needle to be covered by the needle-shield. | |
|
|
| 13 There may be a little bleeding at the injection site. You can press a cotton ball or gauze over the injection site. | |
|
|
| If your prescribed dose requires more than 1 injection: | |
|
|
| After the Injection | |
|
14 Throw away (dispose of) your used XOLAIR prefilled syringes in an FDA-cleared sharps disposal container right away after use. |
|
|
|
| If you do not have an FDA-cleared sharps disposal container, you may use a household container that is: | |
|
|
| Manufactured by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 U.S. License No.: 1048 |
|
| Jointly marketed by: Genentech USA, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 Novartis Pharmaceuticals Corporation, One Health Plaza, East Hanover, NJ 07936-1080 |
|
| XOLAIR® is a registered trademark of Novartis AG. ©2024 Genentech USA, Inc. |
1.1 Asthma
XOLAIR is indicated for adults and pediatric patients 6 years of age and older with moderate to severe persistent asthma who have a positive skin test or in vitro reactivity to a perennial aeroallergen and whose symptoms are inadequately controlled with inhaled corticosteroids.
11 Description
Omalizumab is a recombinant DNA-derived humanized IgG1κ monoclonal antibody that selectively binds to human immunoglobulin E (IgE). The antibody has a molecular weight of approximately 149 kiloDaltons. XOLAIR is produced by a Chinese hamster ovary cell suspension culture.
XOLAIR (omalizumab) is administered as a subcutaneous (SC) injection and is available in prefilled syringe, autoinjector and in vials.
5.2 Malignancy
Malignant neoplasms were observed in 20 of 4127 (0.5%) XOLAIR-treated patients compared with 5 of 2236 (0.2%) control patients in clinical studies of adults and adolescents ≥12 years of age with asthma and other allergic disorders. The observed malignancies in XOLAIR-treated patients were a variety of types, with breast, non-melanoma skin, prostate, melanoma, and parotid occurring more than once, and five other types occurring once each. The majority of patients were observed for less than 1 year. The impact of longer exposure to XOLAIR or use in patients at higher risk for malignancy (e.g., elderly, current smokers) is not known.
In a subsequent observational study of 5007 XOLAIR-treated and 2829 non-XOLAIR-treated adolescent and adult patients with moderate to severe persistent asthma and a positive skin test reaction or in vitro reactivity to a perennial aeroallergen, patients were followed for up to 5 years. In this study, the incidence rates of primary malignancies (per 1000 patient years) were similar among XOLAIR-treated (12.3) and non-XOLAIR-treated patients (13.0) [see Adverse Reactions (6.1)]. However, study limitations preclude definitively ruling out a malignancy risk with XOLAIR. Study limitations include: the observational study design, the bias introduced by allowing enrollment of patients previously exposed to XOLAIR (88%), enrollment of patients (56%) while a history of cancer or a premalignant condition were study exclusion criteria, and the high study discontinuation rate (44%).
5.1 Anaphylaxis
Anaphylaxis has been reported to occur after administration of XOLAIR in premarketing clinical trials and in postmarketing spontaneous reports [see Boxed Warning and Adverse Reactions (6.2)]. Signs and symptoms in these reported cases have included bronchospasm, hypotension, syncope, urticaria, and/or angioedema of the throat or tongue. Some of these events have been life-threatening. In premarketing clinical trials in patients with asthma, anaphylaxis was reported in 3 of 3507 (0.1%) patients. Anaphylaxis occurred with the first dose of XOLAIR in two patients and with the fourth dose in one patient. The time to onset of anaphylaxis was 90 minutes after administration in two patients and 2 hours after administration in one patient.
A case-control study in asthma patients showed that, among XOLAIR users, patients with a history of anaphylaxis to foods, medications, or other causes were at increased risk of anaphylaxis associated with XOLAIR, compared to those with no prior history of anaphylaxis [see Adverse Reactions (6.1)].
In postmarketing spontaneous reports, the frequency of anaphylaxis attributed to XOLAIR use was estimated to be at least 0.2% of patients based on an estimated exposure of about 57,300 patients from June 2003 through December 2006. Anaphylaxis has occurred as early as after the first dose of XOLAIR, but also has occurred beyond one year after beginning regularly scheduled treatment. Approximately 60% to 70% of anaphylaxis cases have been reported to occur within the first three doses of XOLAIR, with additional cases occurring sporadically beyond the third dose.
Initiate XOLAIR only in a healthcare setting equipped to manage anaphylaxis, which can be life-threatening. Observe patients closely for an appropriate period of time after administration of XOLAIR, taking into account the time to onset of anaphylaxis seen in premarketing clinical trials and postmarketing spontaneous reports [see Adverse Reactions (6.1, 6.2)]. Inform patients of the signs and symptoms of anaphylaxis, and instruct them to seek immediate medical care should signs or symptoms occur.
Once XOLAIR therapy has been established, administration of XOLAIR prefilled syringe or autoinjector outside of a healthcare setting by a patient or a caregiver may be appropriate for selected patients. Patient selection, determined by the healthcare provider in consultation with the patient, should take into account the pattern of anaphylaxis events seen in premarketing clinical trials and postmarketing spontaneous reports, as well as individual patient risk factors (e.g., prior history of anaphylaxis), ability to recognize signs and symptoms of anaphylaxis, and ability to perform subcutaneous injections with XOLAIR prefilled syringe or autoinjector with proper technique according to the prescribed dosing regimen and Instructions for Use [see Dosage and Administration (2.6), Adverse Reactions (6.1, 6.2)].
Discontinue XOLAIR in patients who experience a severe hypersensitivity reaction [see Contraindications (4)].
8.5 Geriatric Use
In clinical studies, 134 asthma patients, 20 CRSwNP patients, 37 CSU patients and no IgE-mediated food allergy patients 65 years of age or older were treated with XOLAIR. Although there were no apparent age-related differences observed in these studies, the number of patients aged 65 and over is not sufficient to determine whether they respond differently from younger patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of XOLAIR or of other omalizumab products.
Antibodies to XOLAIR were detected in approximately 1/1723 (<0.1%) of patients treated with XOLAIR in the clinical studies evaluated for asthma in patients 12 years of age and older. In three pediatric studies, antibodies to XOLAIR were detected in one patient out of 581 patients 6 to <12 years of age treated with XOLAIR and evaluated for antibodies. There were no detectable antibodies in the patients treated in the CSU clinical trials, but due to levels of XOLAIR at the time of anti-therapeutic antibody sampling and missing samples for some patients, antibodies to XOLAIR could only have been determined in 88% of the 733 patients treated in these clinical studies. The data reflect the percentage of patients whose test results were considered positive for antibodies to XOLAIR in ELISA assays and are highly dependent on the sensitivity and specificity of the assays.
Anti-drug antibodies were not measured in the CRSwNP or IgE-mediated food allergy trials.
4 Contraindications
XOLAIR is contraindicated in patients with severe hypersensitivity reaction to XOLAIR or any ingredient of XOLAIR [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Anaphylaxis [see Boxed Warning and Warnings and Precautions (5.1)]
- Malignancies [see Warnings and Precautions (5.2)]
7 Drug Interactions
No formal drug interaction studies have been performed with XOLAIR.
In patients with asthma, CRSwNP, and IgE-mediated food allergy the concomitant use of XOLAIR and allergen immunotherapy has not been evaluated.
In patients with CSU, the use of XOLAIR in combination with immunosuppressive therapies has not been studied.
5.8 Laboratory Tests
Serum total IgE levels increase following administration of XOLAIR due to formation of XOLAIR:IgE complexes [see Clinical Pharmacology (12.2)]. Elevated serum total IgE levels may persist for up to 1 year following discontinuation of XOLAIR. Do not use serum total IgE levels obtained less than 1 year following discontinuation to reassess the dosing regimen for asthma, CRSwNP or IgE-mediated food allergy patients, because these levels may not reflect steady-state free IgE levels [see Dosage and Administration (2.2, 2.3, 2.4)].
Warning: Anaphylaxis
Anaphylaxis presenting as bronchospasm, hypotension, syncope, urticaria, and/or angioedema of the throat or tongue, has been reported to occur after administration of XOLAIR. Anaphylaxis has occurred as early as after the first dose of XOLAIR, but also has occurred beyond 1 year after beginning regularly administered treatment. Because of the risk of anaphylaxis, initiate XOLAIR therapy in a healthcare setting and closely observe patients for an appropriate period of time after XOLAIR administration. Health care providers administering XOLAIR should be prepared to manage anaphylaxis which can be life-threatening. Inform patients of the signs and symptoms of anaphylaxis and instruct them to seek immediate medical care should symptoms occur. Selection of patients for self-administration of XOLAIR should be based on criteria to mitigate risk from anaphylaxis [see Dosage and Administration (2.6), Warnings and Precautions (5.1) and Adverse Reactions (6.1, 6.2)].
12.3 Pharmacokinetics
After SC administration, omalizumab was absorbed with an average absolute bioavailability of 62%. Following a single SC dose in adult and adolescent patients with asthma, omalizumab was absorbed slowly, reaching peak serum concentrations after an average of 7–8 days. In patients with CSU, the peak serum concentration was reached at a similar time after a single SC dose. The pharmacokinetics of omalizumab was linear at doses greater than 0.5 mg/kg. In patients with asthma, following multiple doses of XOLAIR, areas under the serum concentration-time curve from Day 0 to Day 14 at steady state were up to 6-fold of those after the first dose. In patients with CSU, omalizumab exhibited linear pharmacokinetics across the dose range of 75 mg to 600 mg given as single subcutaneous dose. Following repeat dosing from 75 to 300 mg every 4 weeks, trough serum concentrations of omalizumab increased proportionally with the dose levels.
In vitro, omalizumab formed complexes of limited size with IgE. Precipitating complexes and complexes larger than 1 million daltons in molecular weight were not observed in vitro or in vivo. Tissue distribution studies in Cynomolgus monkeys showed no specific uptake of 125I-omalizumab by any organ or tissue. The apparent volume of distribution of omalizumab in patients with asthma following SC administration was 78 ± 32 mL/kg. In patients with CSU, based on population pharmacokinetics, distribution of omalizumab was similar to that in patients with asthma.
Clearance of omalizumab involved IgG clearance processes as well as clearance via specific binding and complex formation with its target ligand, IgE. Liver elimination of IgG included degradation in the liver reticuloendothelial system (RES) and endothelial cells. Intact IgG was also excreted in bile. In studies with mice and monkeys, omalizumab:IgE complexes were eliminated by interactions with Fcγ receptors within the RES at rates that were generally faster than IgG clearance. In asthma patients omalizumab serum elimination half-life averaged 26 days, with apparent clearance averaging 2.4 ± 1.1 mL/kg/day. Doubling body weight approximately doubled apparent clearance. In CSU patients, at steady state, based on population pharmacokinetics, omalizumab serum elimination half-life averaged 24 days and apparent clearance averaged 240 mL/day (corresponding to 3.0 mL/kg/day for an 80 kg patient).
1 Indications and Usage
XOLAIR is an anti-IgE antibody indicated for:
- Moderate to severe persistent asthma in adults and pediatric patients 6 years of age and older with a positive skin test or in vitro reactivity to a perennial aeroallergen and symptoms that are inadequately controlled with inhaled corticosteroids (1.1)
- Chronic rhinosinusitis with nasal polyps (CRSwNP) in adult patients 18 years of age and older with inadequate response to nasal corticosteroids, as add-on maintenance treatment (1.2)
- IgE-mediated food allergy in adult and pediatric patients aged 1 year and older for the reduction of allergic reactions (Type I), including anaphylaxis, that may occur with accidental exposure to one or more foods. To be used in conjunction with food allergen avoidance (1.3)
- Chronic spontaneous urticaria (CSU) in adults and adolescents 12 years of age and older who remain symptomatic despite H1 antihistamine treatment (1.4)
Limitations of Use:
5 Warnings and Precautions
- Anaphylaxis: Initiate XOLAIR therapy in a healthcare setting prepared to manage anaphylaxis which can be life-threatening and observe patients for an appropriate period of time after administration. (5.1)
- Malignancy: Malignancies have been observed in clinical studies. (5.2)
- Acute Asthma Symptoms: Do not use for the treatment of acute bronchospasm or status asthmaticus. (5.3)
- Corticosteroid Reduction: Do not abruptly discontinue corticosteroids upon initiation of XOLAIR therapy. (5.4)
- Eosinophilic Conditions: Be alert to eosinophilia, vasculitic rash, worsening pulmonary symptoms, cardiac complications, and/or neuropathy, especially upon reduction of oral corticosteroids. (5.5)
- Fever, Arthralgia, and Rash: Stop XOLAIR if patients develop signs and symptoms similar to serum sickness. (5.6)
- Potential Medication Error Related to Emergency Treatment of Anaphylaxis: XOLAIR should not be used for emergency treatment of allergic reactions, including anaphylaxis. (5.9)
2 Dosage and Administration
For subcutaneous (SC) administration only. (2.2, 2.3, 2.4, 2.5)
See full prescribing information for administration instructions (2.6, 2.7, 2.8).
- Asthma: XOLAIR 75 to 375 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination charts. (2.2)
- Chronic Rhinosinusitis with Nasal Polyps: XOLAIR 75 to 600 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination charts. (2.3)
- IgE-Mediated Food Allergy: XOLAIR 75 mg to 600 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination chart. (2.4)
- Chronic Spontaneous Urticaria: XOLAIR 150 or 300 mg SC every 4 weeks. Dosing in CSU is not dependent on serum IgE level or body weight. (2.5)
2.6 Administration Overview
- Administer XOLAIR by subcutaneous injection.
- XOLAIR is intended for use under the guidance of a healthcare provider.
- Initiate therapy in a healthcare setting and once therapy has been safely established, the healthcare provider may determine whether self-administration of XOLAIR prefilled syringe or autoinjector by the patient or caregiver is appropriate, based on careful assessment of risk for anaphylaxis and mitigation strategies.
5.5 Eosinophilic Conditions
In rare cases, patients with asthma on therapy with XOLAIR may present with serious systemic eosinophilia sometimes presenting with clinical features of vasculitis consistent with Churg-Strauss syndrome, a condition which is often treated with systemic corticosteroid therapy. These events usually, but not always, have been associated with the reduction of oral corticosteroid therapy. Physicians should be alert to eosinophilia, vasculitic rash, worsening pulmonary symptoms, cardiac complications, and/or neuropathy presenting in their patients. A causal association between XOLAIR and these underlying conditions has not been established.
3 Dosage Forms and Strengths
Injection:
- 75 mg/0.5 mL is a clear to slightly opalescent and colorless to pale brownish-yellow solution in a single-dose prefilled syringe with needle shield or single-dose prefilled autoinjector
- 150 mg/mL is a clear to slightly opalescent and colorless to pale brownish-yellow solution in a single-dose prefilled syringe with needle shield or single-dose prefilled autoinjector
- 300 mg/2 mL is a clear to slightly opalescent and colorless to pale brownish-yellow solution in a single-dose prefilled syringe with needle shield or single-dose prefilled autoinjector
- For injection: 150 mg white lyophilized powder in a single-dose vial for reconstitution
5.4 Corticosteroid Reduction
Do not discontinue systemic or inhaled corticosteroids abruptly upon initiation of XOLAIR therapy for asthma or CRSwNP. Decrease corticosteroids gradually under the direct supervision of a physician. In CSU patients, the use of XOLAIR in combination with corticosteroids has not been evaluated.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of XOLAIR. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
1.3 Ige Mediated Food Allergy
XOLAIR is indicated for the reduction of allergic reactions (Type I), including anaphylaxis, that may occur with accidental exposure to one or more foods in adult and pediatric patients aged 1 year and older with IgE-mediated food allergy.
XOLAIR is to be used in conjunction with food allergen avoidance.
14.3 Ige Mediated Food Allergy
The safety and efficacy of XOLAIR was evaluated in a multi-center, randomized, double-blind, placebo-controlled Food Allergy (FA) trial [NCT03881696] in 168 adult patients and pediatric patients 1 year of age to less than 56 years who were allergic to peanut and at least two other foods, including milk, egg, wheat, cashew, hazelnut, or walnut (i.e., studied foods). The FA trial enrolled patients who experienced dose-limiting symptoms (e.g., moderate to severe skin, respiratory or gastrointestinal symptoms) to a single dose of ≤100 mg of peanut protein and ≤300 mg protein for each of the other two foods (milk, egg, wheat, cashew, hazelnut, or walnut) during the screening double-blind placebo-controlled food challenge (DBPCFC). Patients with a history of severe anaphylaxis (defined as neurological compromise or requiring intubation) were excluded from the study. Patients were randomized 2:1 to receive a subcutaneous dosage of XOLAIR or placebo based on serum total IgE level (IU/mL), measured before the start of treatment, and by body weight according to Table 4 [see Dosage and Administration (2.4)] for 16 to 20 weeks. After 16 to 20 weeks of treatment, each patient completed a DBPCFC consisting of placebo and each of their 3 studied foods. Following the DBPCFC, the first 60 patients that included 59 pediatric patients and one adult patient who completed the double-blind, placebo-controlled phase of the study could continue to receive XOLAIR in a 24 to 28 week open-label extension.
Efficacy of XOLAIR is based on 165 pediatric patients who were included in the efficacy analyses provided below. The mean age of the pediatric patients was 8 years (age range: 1 to 17 years); 37% were less than 6 years of age, 38% were 6 to less than 12 years of age, and 25% were 12 to less than 18 years of age. Patient population were 56% male, 63% White, 13% Asian, 7% Black, 16% Other, and 55% of patients had a history of asthma.
The primary efficacy endpoint was the percentage of patients who were able to consume a single dose of ≥600 mg of peanut protein without dose-limiting symptoms (e.g., moderate to severe skin, respiratory or gastrointestinal symptoms) during DBPCFC. Table 16 shows XOLAIR treatment led to a statistically higher response rate (68%) than placebo (5%).
The secondary efficacy endpoints were the percentage of patients who were able to consume a single dose of ≥1000 mg of cashew, milk, or egg protein without dose-limiting symptoms during DBPCFC. The study met the secondary endpoints and demonstrated that XOLAIR treatment led to statistically higher response rates than placebo for all three foods. See Table 16 for details.
| Food, Challenge Dose | Response Rate Response defined as consumption of a single dose of the specified amount of food without dose-limiting symptoms. (%)(n/N) |
Treatment Difference (%) (XOLAIR-Placebo) (95% CI) |
|
|---|---|---|---|
| XOLAIR | Placebo | ||
| CI = Confidence interval; DBPCFC = Double-blind placebo-controlled food challenge; n = Number of responders; N = Total number of patients receiving food, challenge dose. Notes: Subjects without an exit DBPCFC or evaluable exit DBPCFC were counted as non-responders; P-values from two-sided Fisher's exact tests were <0.0001 for all the food challenge doses. |
|||
| Peanut, ≥600 mg | 68% (75/110) |
5% (3/55) |
63% (50%, 73%) |
| Peanut, ≥1000 mg Consumption of a single dose of ≥1000 mg of peanut protein was an additional secondary endpoint. The key secondary efficacy endpoints were the percentage of patients who were able to consume a single dose of ≥1000 mg of cashew, milk, or egg protein.
|
65% (72/110) |
0% (0/55) |
65% (56%, 74%) |
| Cashew, ≥1000 mg | 42% (27/64) |
3% (1/30) |
39% (20%, 53%) |
| Milk, ≥1000 mg | 66% (25/38) |
11% (2/19) |
55% (29%, 73%) |
| Egg, ≥1000 mg | 67% (31/46) |
0% (0/19) |
67% (49%, 80%) |
Seventeen percent of XOLAIR treated patients were not able to consume >100 mg of peanut protein without moderate to severe dose-limiting symptoms. Eighteen, 22, and 41 percent of XOLAIR-treated patients were not able to consume >300 mg of milk, egg, or cashew protein, respectively, without moderate to severe dose-limiting symptoms.
Additional secondary analyses included the percentage of patients who were able to consume at least two or all three foods during DBPCFC. For two foods, 71% of XOLAIR treated patients were able to consume a single dose of ≥600 mg versus 5% in the placebo group and 67% were able to consume a single dose of ≥1000 mg versus 4% in the placebo group. For a single dose of ≥600 mg of three foods, the response rates were 48% in the XOLAIR group versus 4% in the placebo group and for a single dose of ≥1000 mg of three foods, the response rate in the XOLAIR group was 39% while none of the placebo patients were able to consume the challenge dose without symptoms.
The effectiveness of XOLAIR in adults is supported by the adequate and well-controlled trial of XOLAIR in pediatric patients, disease similarity in pediatric and adult patients, and pharmacokinetic (PK) similarity [see Clinical Pharmacology (12.3)].
While efficacy cannot be established from uncontrolled, open-label studies, for 38 pediatric patients who continued XOLAIR for 24-28 weeks in an open-label extension, the percentage of patients who were able to consume ≥600 mg of peanut protein and ≥1000 mg of egg, milk, and/or cashew protein without moderate to severe dose-limiting symptoms was maintained.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
5.6 Fever, Arthralgia, and Rash
In post-approval use, some patients have experienced a constellation of signs and symptoms including arthritis/arthralgia, rash, fever, and lymphadenopathy with an onset 1 to 5 days after the first or subsequent injections of XOLAIR. These signs and symptoms have recurred after additional doses in some patients. Although circulating immune complexes or a skin biopsy consistent with a Type III reaction were not seen with these cases, these signs and symptoms are similar to those seen in patients with serum sickness. Physicians should stop XOLAIR if a patient develops this constellation of signs and symptoms [see Adverse Reactions (6.2)].
1.4 Chronic Spontaneous Urticaria
XOLAIR is indicated for the treatment of adults and adolescents 12 years of age and older with chronic spontaneous urticaria (CSU) who remain symptomatic despite H1 antihistamine treatment.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
2.2 Recommended Dosage for Asthma
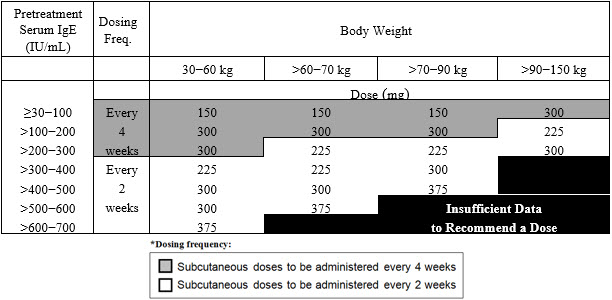
The recommended dosage for asthma is XOLAIR 75 mg to 375 mg by subcutaneous injection every 2 or 4 weeks based on serum total IgE level (IU/mL) measured before the start of treatment and by body weight (kg) [see Dosage and Administration (2.1)].
- Adult and adolescent patients 12 years of age and older: Initiate dosing according to Table 1.
- Pediatric patients 6 to <12 years of age: Initiate dosing according to Table 2.
5.7 Parasitic (helminth) Infection
Monitor patients at high risk of geohelminth infection while on XOLAIR therapy. Insufficient data are available to determine the length of monitoring required for geohelminth infections after stopping XOLAIR treatment.
In a one-year clinical trial conducted in Brazil in adult and adolescent patients at high risk for geohelminthic infections (roundworm, hookworm, whipworm, threadworm), 53% (36/68) of XOLAIR-treated patients experienced an infection, as diagnosed by standard stool examination, compared to 42% (29/69) of placebo controls. The point estimate of the odds ratio for infection was 1.96, with a 95% confidence interval (0.88, 4.36) indicating that in this study a patient who had an infection was anywhere from 0.88 to 4.36 times as likely to have received XOLAIR than a patient who did not have an infection. Response to appropriate anti-geohelminth treatment of infection as measured by stool egg counts was not different between treatment groups.
1.2 Chronic Rhinosinusitis With Nasal Polyps
XOLAIR is indicated for add-on maintenance treatment of chronic rhinosinusitis with nasal polyps (CRSwNP) in adult patients 18 years of age and older with inadequate response to nasal corticosteroids.
Principal Display Panel 150 Mg Vial Carton
NDC 50242-040-62
SINGLE-DOSE VIAL
150 mg
Xolair®
Omalizumab
FOR SUBCUTANEOUS USE
KEEP REFRIGERATED. DO NOT FREEZE.
Genentech
NOVARTIS
10198215
2.7 Xolair Prefilled Syringe and Autoinjector
XOLAIR injection doses are available as a prefilled syringe or as an autoinjector. Instruct patients or caregivers to follow the directions provided in the "Instructions for Use" for preparation and administration of XOLAIR prefilled syringe or autoinjector [see Instructions for Use].
5.3 Acute Asthma Symptoms and Deteriorating Disease
XOLAIR has not been shown to alleviate asthma exacerbations acutely. Do not use XOLAIR to treat acute bronchospasm or status asthmaticus. Patients should seek medical advice if their asthma remains uncontrolled or worsens after initiation of treatment with XOLAIR.
2.4 Recommended Dosage for Ige Mediated Food Allergy
The recommended dosage for IgE-mediated food allergy is XOLAIR 75 mg to 600 mg by subcutaneous injection every 2 or 4 weeks based on serum total IgE level (IU/mL), measured before the start of treatment, and by body weight [see Dosage and Administration (2.1)]. Refer to Table 4 for recommended dosage based on serum IgE level and body weight for patients with IgE-mediated food allergy.
Principal Display Panel 300 Mg/2 Ml Syringe Carton
Xolair®
(omalizumab) Injection
300 mg/2 mL
For Subcutaneous Use.
1 Single-Dose Prefilled Syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-227-01
Dispense the accompanying Medication Guide to each patient.
Rx only
Must be refrigerated
Genentech
NOVARTIS
11011496
Principal Display Panel 150 Mg/ml Autoinjector Carton
Xolair®
(omalizumab) Injection
150 mg/mL
For Subcutaneous Use.
1 Single-Dose Autoinjector
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-215-55
Dispense the accompanying Medication Guide to each patient.
Genentech
NOVARTIS
11011532
2.5 Recommended Dosage for Chronic Spontaneous Urticaria
The recommended dosage for chronic spontaneous urticaria (CSU) is XOLAIR 150 mg or 300 mg by subcutaneous injection every 4 weeks.
- The 300 mg dose may be administered as one subcutaneous injection of 300 mg/2 mL or as two subcutaneous injections of 150 mg/mL.
- Dosing of XOLAIR in CSU patients is not dependent on serum IgE (free or total) level or body weight.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies have been performed in animals to evaluate the carcinogenic potential of XOLAIR.
There were no effects on fertility and reproductive performance in male and female Cynomolgus monkeys that received XOLAIR at subcutaneous doses up to 75 mg/kg/week (approximately 5 times the maximum recommended human dose on a mg/kg basis).
Principal Display Panel 300 Mg/2 Ml Autoinjector Carton
Xolair®
(omalizumab) Injection
300 mg/2 mL
For Subcutaneous Use.
1 Single-Dose Autoinjector
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-227-55
Dispense the accompanying Medication Guide to each patient.
Genentech
NOVARTIS
11011548
Principal Display Panel 75 Mg/0.5 Ml Autoinjector Carton
Xolair®
(omalizumab) Injection
75 mg/0.5 mL
For Subcutaneous Use.
1 Single-Dose Autoinjector
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-214-55
Dispense the accompanying Medication Guide to each patient.
Genentech
NOVARTIS
11011505
Principal Display Panel 150 Mg/ml Syringe Carton 215 01
Xolair®
(omalizumab) Injection
150 mg/mL
For Subcutaneous Use. Single-Dose Prefilled Syringe.
Dispense the accompanying
Medication Guide to each patient.
1 prefilled syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-215-01
Caution: The needle cap may contain natural rubber latex
which may cause allergic reactions in latex sensitive individuals.
Rx only
Genentech
NOVARTIS
11011498
Principal Display Panel 150 Mg/ml Syringe Carton 215 03
Xolair®
(omalizumab) Injection
150 mg/mL
For Subcutaneous Use.
1 Single-Dose Prefilled Syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-215-03
Dispense the accompanying Medication Guide to each patient.
Rx only
Must be refrigerated
Genentech
NOVARTIS
11011522
Principal Display Panel 75 Mg/0.5 Ml Syringe Carton 214 01
Xolair®
(omalizumab) Injection
75 mg/0.5 mL
For Subcutaneous Use. Single-Dose Prefilled Syringe.
Dispense the accompanying
Medication Guide to each patient.
1 prefilled syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-214-01
Caution: The needle cap may contain natural rubber latex
which may cause allergic reactions in latex sensitive individuals.
Rx only
Genentech
NOVARTIS
11011555
Principal Display Panel 75 Mg/0.5 Ml Syringe Carton 214 03
Xolair®
(omalizumab) Injection
75 mg/0.5 mL
For Subcutaneous Use.
1 Single-Dose Prefilled Syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-214-03
Dispense the accompanying Medication Guide to each patient.
Rx only
Must be refrigerated
Genentech
NOVARTIS
11011485
2.8 Preparation for Use and Injection of Xolair Lyophilized Powder
XOLAIR lyophilized powder should only be prepared and injected by a healthcare provider. The supplied XOLAIR lyophilized powder must be reconstituted with Sterile Water for Injection (SWFI) USP, using the following instructions:
-
1)Before reconstitution, determine the number of vials that will need to be reconstituted (each vial delivers 150 mg of XOLAIR in 1.2 mL) (see Table 6).
| XOLAIR Dose The 75 mg, 150 mg, 225 mg, 300 mg, and 375 mg XOLAIR doses are approved for use in asthma patients. All doses in the table are approved for use in CRSwNP and IgE-mediated food allergy patients. The 150 mg and 300 mg XOLAIR doses are also approved for use in CSU patients.
|
Number of Vials | Number of Injections | Total Volume Injected |
|---|---|---|---|
| 75 mg | 1 | 1 | 0.6 mL |
| 150 mg | 1 | 1 | 1.2 mL |
| 225 mg | 2 | 2 | 1.8 mL |
| 300 mg | 2 | 2 | 2.4 mL |
| 375 mg | 3 | 3 | 3.0 mL |
| 450 mg | 3 | 3 | 3.6 mL |
| 525 mg | 4 | 4 | 4.2 mL |
| 600 mg | 4 | 4 | 4.8 mL |
-
2)Draw 1.4 mL of SWFI, USP, into a 3 mL syringe equipped with a 1-inch, 18-gauge needle.
-
3)Place the vial upright on a flat surface and using standard aseptic technique, insert the needle and inject the SWFI, USP, directly onto the product.
-
4)Keeping the vial upright, gently swirl the upright vial for approximately 1 minute to evenly wet the powder. Do not shake.
-
5)Gently swirl the vial for 5 to 10 seconds approximately every 5 minutes in order to dissolve any remaining solids. The lyophilized product takes 15 to 20 minutes to dissolve. If it takes longer than 20 minutes to dissolve completely, gently swirl the vial for 5 to 10 seconds approximately every 5 minutes until there are no visible gel-like particles in the solution. Do not use if the contents of the vial do not dissolve completely by 40 minutes.
-
6)After reconstitution, XOLAIR solution is somewhat viscous and will appear clear or slightly opalescent. It is acceptable if there are a few small bubbles or foam around the edge of the vial; there should be no visible gel-like particles in the reconstituted solution. Do not use if foreign particles are present.
-
7)Invert the vial for 15 seconds in order to allow the solution to drain toward the stopper.
-
8)Use the XOLAIR solution within 8 hours following reconstitution when stored in the vial at 2ºC to 8ºC (36ºF to 46ºF), or within 4 hours of reconstitution when stored at room temperature. Reconstituted XOLAIR vials should be protected from sunlight.
-
9)Using a new 3 mL syringe equipped with a 1-inch, 18-gauge needle, insert the needle into the inverted vial. Position the needle tip at the very bottom of the solution in the vial stopper when drawing the solution into the syringe. The reconstituted product is somewhat viscous. Withdraw all of the product from the vial before expelling any air or excess solution from the syringe. Before removing the needle from the vial, pull the plunger all the way back to the end of the syringe barrel in order to remove all of the solution from the inverted vial.
-
10)Replace the 18-gauge needle with a 25-gauge needle for subcutaneous injection.
-
11)Expel air, large bubbles, and any excess solution in order to obtain a volume of 1.2 mL corresponding to a dose of 150 mg of XOLAIR. To obtain a volume of 0.6 mL corresponding to a dose of 75 mg of XOLAIR, expel air, large bubbles and discard 0.6 mL from the syringe. A thin layer of small bubbles may remain at the top of the solution in the syringe.
-
12)Administer XOLAIR by subcutaneous injection. The injection may take 5-10 seconds to administer because the solution is slightly viscous. Do not administer more than 150 mg (contents of one vial) per injection site. Divide doses of more than 150 mg between two or more injection sites. Choose a different injection site for each new injection at least 1 inch from the area used for other injections.
2.3 Recommended Dosage for Chronic Rhinosinusitis With Nasal Polyps
The recommended dosage for chronic rhinosinusitis with nasal polyps (CRSwNP) is XOLAIR 75 mg to 600 mg by subcutaneous injection every 2 or 4 weeks based on serum total IgE level (IU/mL) measure before the start of treatment and by body weight (kg) [see Dosage and Administration (2.1)]. Refer to Table 3 for recommended dosage based on serum total IgE level and body weight for patients with CRSwNP.
5.9 Potential Medication Error Related to Emergency Treatment of Anaphylaxis
XOLAIR should not be used for the emergency treatment of allergic reactions, including anaphylaxis. In studies to simulate use, some patients and caregivers did not understand that XOLAIR is not intended for the emergency treatment of allergic reactions, including anaphylaxis. The safety and effectiveness of XOLAIR for emergency treatment of allergic reactions, including anaphylaxis, have not been established. Instruct patients that XOLAIR is for maintenance use to reduce allergic reactions, including anaphylaxis, while avoiding food allergens.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use:
XOLAIR is not indicated for the relief of acute bronchospasm or status asthmaticus.
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Revised:2/2024 | |
| MEDICATION GUIDE | ||
|
XOLAIR® (ZOHL-air)
(omalizumab) injection, for subcutaneous use |
XOLAIR® (ZOHL-air)
(omalizumab) for injection, for subcutaneous use |
|
|
What is the most important information I should know about XOLAIR? XOLAIR may cause serious side effects, including: Severe allergic reaction. A severe allergic reaction called anaphylaxis can happen when you receive XOLAIR. The reaction can occur after the first dose, or after many doses. It may also occur right after a XOLAIR injection or days later. Anaphylaxis is a life-threatening condition and can lead to death. Go to the nearest emergency room right away if you have any of these symptoms of an allergic reaction:
|
||
|
What is XOLAIR?
XOLAIR is an injectable prescription medicine used to treat:
|
||
|
Who should not receive and use XOLAIR? Do not receive and use XOLAIR if you:
|
||
|
What should I tell my healthcare provider before receiving XOLAIR? Before receiving XOLAIR, tell your healthcare provider about all of your medical conditions, including if you:
|
||
How should I receive and use XOLAIR?
|
||
|
What are the possible side effects of XOLAIR? XOLAIR may cause serious side effects, including:
|
||
|
|
|
|
||
How should I store XOLAIR?
|
||
|
General information about the safe and effective use of XOLAIR.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use XOLAIR for a condition for which it was not prescribed. Do not give XOLAIR to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about XOLAIR that is written for health professionals. For more information, go to www.xolair.com or call 1-866-4XOLAIR (1-866-496-5247). |
||
|
What are the ingredients in XOLAIR? Active ingredient: omalizumab Inactive ingredients: Prefilled syringe or Autoinjector: arginine hydrochloride, histidine, L-histidine hydrochloride monohydrate, and polysorbate 20 Vial: histidine, L-histidine hydrochloride monohydrate, polysorbate 20 and sucrose Manufactured by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990. U.S. License No.: 1048 Jointly marketed by: Genentech USA, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 Novartis Pharmaceuticals Corporation, One Health Plaza, East Hanover, NJ 07936-1080 XOLAIR® is a registered trademark of Novartis AG. ©2024 Genentech USA, Inc. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage
XOLAIR prefilled syringe and autoinjector should be shipped and stored under refrigerated conditions 2°C to 8°C (36°F to 46°F) in the original carton. Protect from direct sunlight. XOLAIR prefilled syringe and autoinjector can be removed from and placed back in the refrigerator if needed. The total combined time out of the refrigerator may not be more than 2 days. Do not use if prefilled syringe or autoinjector is left at temperatures above 25°C (77°F).
Do not freeze. Do not use if the prefilled syringe or autoinjector has been frozen.
Section 59845-8 (59845-8)
| This Instructions for Use has been approved by the U.S. Food and Drug Administration. | Revised: 2/2024 |
|
Instructions for Use Xolair® (ZOHL-air) (omalizumab) injection, for subcutaneous use Prefilled Syringe |
|
| Read this Instructions for Use before you start using the XOLAIR prefilled syringe and each time you get a refill. Before you use the XOLAIR prefilled syringe for the first time, make sure your healthcare provider shows you the right way to use it. Contact your healthcare provider if you have any questions. | |
| Do not use XOLAIR for the emergency treatment of any allergic reactions, including anaphylaxis, hives, or sudden breathing problems. | |
|
|
|
Supplies needed to give your injection
|
|
|
Choose the correct prefilled syringe or combination of prefilled syringes
|
|
|
|
|
|
|
|
How should I store XOLAIR?
|
|
Important Information
|
|
| Preparing for the Injection | |
| 1 Take the carton containing the prefilled syringe out of the refrigerator. | |
|
|
| 2 Check the expiration date on the XOLAIR carton. | |
|
|
|
| 3 Place the carton on a clean, flat surface. | |
|
|
| 4 Open the carton. | |
|
|
|
|
|
|
|
|
5 Inspect the prefilled syringe closely.
|
|
| 6 Choose an injection site. | |
|
|
| 7 Wipe the injection site with an alcohol swab in a circular motion and let it air dry for 10 seconds. | |
|
|
| Giving the Injection | |
| 8 Hold the prefilled syringe firmly by the center with 1 hand and pull the needle cap straight off with your other hand. | |
|
|
|
|
|
| 9 Use your other hand and gently pinch the area of skin that was cleaned. Hold the pinched skin tight. | |
|
|
| 10 Continue holding the prefilled syringe by the center and use a quick, dart-like motion to insert the needle all the way into the pinched skin at an angle between 45 degrees to 90 degrees as shown. | |
|
|
| 11 Slowly inject all of the medicine by gently pushing the plunger all the way down until the needle-shield wings are pushed apart. | |
|
|
| 12 Release the plunger and allow the needle to be covered by the needle-shield. | |
|
|
| 13 There may be a little bleeding at the injection site. You can press a cotton ball or gauze over the injection site. | |
|
|
| If your prescribed dose requires more than 1 injection: | |
|
|
| After the Injection | |
|
14 Throw away (dispose of) your used XOLAIR prefilled syringes in an FDA-cleared sharps disposal container right away after use. |
|
|
|
| If you do not have an FDA-cleared sharps disposal container, you may use a household container that is: | |
|
|
| Manufactured by: Genentech, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 U.S. License No.: 1048 |
|
| Jointly marketed by: Genentech USA, Inc., A Member of the Roche Group, 1 DNA Way, South San Francisco, CA 94080-4990 Novartis Pharmaceuticals Corporation, One Health Plaza, East Hanover, NJ 07936-1080 |
|
| XOLAIR® is a registered trademark of Novartis AG. ©2024 Genentech USA, Inc. |
1.1 Asthma
XOLAIR is indicated for adults and pediatric patients 6 years of age and older with moderate to severe persistent asthma who have a positive skin test or in vitro reactivity to a perennial aeroallergen and whose symptoms are inadequately controlled with inhaled corticosteroids.
11 Description (11 DESCRIPTION)
Omalizumab is a recombinant DNA-derived humanized IgG1κ monoclonal antibody that selectively binds to human immunoglobulin E (IgE). The antibody has a molecular weight of approximately 149 kiloDaltons. XOLAIR is produced by a Chinese hamster ovary cell suspension culture.
XOLAIR (omalizumab) is administered as a subcutaneous (SC) injection and is available in prefilled syringe, autoinjector and in vials.
5.2 Malignancy
Malignant neoplasms were observed in 20 of 4127 (0.5%) XOLAIR-treated patients compared with 5 of 2236 (0.2%) control patients in clinical studies of adults and adolescents ≥12 years of age with asthma and other allergic disorders. The observed malignancies in XOLAIR-treated patients were a variety of types, with breast, non-melanoma skin, prostate, melanoma, and parotid occurring more than once, and five other types occurring once each. The majority of patients were observed for less than 1 year. The impact of longer exposure to XOLAIR or use in patients at higher risk for malignancy (e.g., elderly, current smokers) is not known.
In a subsequent observational study of 5007 XOLAIR-treated and 2829 non-XOLAIR-treated adolescent and adult patients with moderate to severe persistent asthma and a positive skin test reaction or in vitro reactivity to a perennial aeroallergen, patients were followed for up to 5 years. In this study, the incidence rates of primary malignancies (per 1000 patient years) were similar among XOLAIR-treated (12.3) and non-XOLAIR-treated patients (13.0) [see Adverse Reactions (6.1)]. However, study limitations preclude definitively ruling out a malignancy risk with XOLAIR. Study limitations include: the observational study design, the bias introduced by allowing enrollment of patients previously exposed to XOLAIR (88%), enrollment of patients (56%) while a history of cancer or a premalignant condition were study exclusion criteria, and the high study discontinuation rate (44%).
5.1 Anaphylaxis
Anaphylaxis has been reported to occur after administration of XOLAIR in premarketing clinical trials and in postmarketing spontaneous reports [see Boxed Warning and Adverse Reactions (6.2)]. Signs and symptoms in these reported cases have included bronchospasm, hypotension, syncope, urticaria, and/or angioedema of the throat or tongue. Some of these events have been life-threatening. In premarketing clinical trials in patients with asthma, anaphylaxis was reported in 3 of 3507 (0.1%) patients. Anaphylaxis occurred with the first dose of XOLAIR in two patients and with the fourth dose in one patient. The time to onset of anaphylaxis was 90 minutes after administration in two patients and 2 hours after administration in one patient.
A case-control study in asthma patients showed that, among XOLAIR users, patients with a history of anaphylaxis to foods, medications, or other causes were at increased risk of anaphylaxis associated with XOLAIR, compared to those with no prior history of anaphylaxis [see Adverse Reactions (6.1)].
In postmarketing spontaneous reports, the frequency of anaphylaxis attributed to XOLAIR use was estimated to be at least 0.2% of patients based on an estimated exposure of about 57,300 patients from June 2003 through December 2006. Anaphylaxis has occurred as early as after the first dose of XOLAIR, but also has occurred beyond one year after beginning regularly scheduled treatment. Approximately 60% to 70% of anaphylaxis cases have been reported to occur within the first three doses of XOLAIR, with additional cases occurring sporadically beyond the third dose.
Initiate XOLAIR only in a healthcare setting equipped to manage anaphylaxis, which can be life-threatening. Observe patients closely for an appropriate period of time after administration of XOLAIR, taking into account the time to onset of anaphylaxis seen in premarketing clinical trials and postmarketing spontaneous reports [see Adverse Reactions (6.1, 6.2)]. Inform patients of the signs and symptoms of anaphylaxis, and instruct them to seek immediate medical care should signs or symptoms occur.
Once XOLAIR therapy has been established, administration of XOLAIR prefilled syringe or autoinjector outside of a healthcare setting by a patient or a caregiver may be appropriate for selected patients. Patient selection, determined by the healthcare provider in consultation with the patient, should take into account the pattern of anaphylaxis events seen in premarketing clinical trials and postmarketing spontaneous reports, as well as individual patient risk factors (e.g., prior history of anaphylaxis), ability to recognize signs and symptoms of anaphylaxis, and ability to perform subcutaneous injections with XOLAIR prefilled syringe or autoinjector with proper technique according to the prescribed dosing regimen and Instructions for Use [see Dosage and Administration (2.6), Adverse Reactions (6.1, 6.2)].
Discontinue XOLAIR in patients who experience a severe hypersensitivity reaction [see Contraindications (4)].
8.5 Geriatric Use
In clinical studies, 134 asthma patients, 20 CRSwNP patients, 37 CSU patients and no IgE-mediated food allergy patients 65 years of age or older were treated with XOLAIR. Although there were no apparent age-related differences observed in these studies, the number of patients aged 65 and over is not sufficient to determine whether they respond differently from younger patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of XOLAIR or of other omalizumab products.
Antibodies to XOLAIR were detected in approximately 1/1723 (<0.1%) of patients treated with XOLAIR in the clinical studies evaluated for asthma in patients 12 years of age and older. In three pediatric studies, antibodies to XOLAIR were detected in one patient out of 581 patients 6 to <12 years of age treated with XOLAIR and evaluated for antibodies. There were no detectable antibodies in the patients treated in the CSU clinical trials, but due to levels of XOLAIR at the time of anti-therapeutic antibody sampling and missing samples for some patients, antibodies to XOLAIR could only have been determined in 88% of the 733 patients treated in these clinical studies. The data reflect the percentage of patients whose test results were considered positive for antibodies to XOLAIR in ELISA assays and are highly dependent on the sensitivity and specificity of the assays.
Anti-drug antibodies were not measured in the CRSwNP or IgE-mediated food allergy trials.
4 Contraindications (4 CONTRAINDICATIONS)
XOLAIR is contraindicated in patients with severe hypersensitivity reaction to XOLAIR or any ingredient of XOLAIR [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Anaphylaxis [see Boxed Warning and Warnings and Precautions (5.1)]
- Malignancies [see Warnings and Precautions (5.2)]
7 Drug Interactions (7 DRUG INTERACTIONS)
No formal drug interaction studies have been performed with XOLAIR.
In patients with asthma, CRSwNP, and IgE-mediated food allergy the concomitant use of XOLAIR and allergen immunotherapy has not been evaluated.
In patients with CSU, the use of XOLAIR in combination with immunosuppressive therapies has not been studied.
5.8 Laboratory Tests
Serum total IgE levels increase following administration of XOLAIR due to formation of XOLAIR:IgE complexes [see Clinical Pharmacology (12.2)]. Elevated serum total IgE levels may persist for up to 1 year following discontinuation of XOLAIR. Do not use serum total IgE levels obtained less than 1 year following discontinuation to reassess the dosing regimen for asthma, CRSwNP or IgE-mediated food allergy patients, because these levels may not reflect steady-state free IgE levels [see Dosage and Administration (2.2, 2.3, 2.4)].
Warning: Anaphylaxis (WARNING: ANAPHYLAXIS)
Anaphylaxis presenting as bronchospasm, hypotension, syncope, urticaria, and/or angioedema of the throat or tongue, has been reported to occur after administration of XOLAIR. Anaphylaxis has occurred as early as after the first dose of XOLAIR, but also has occurred beyond 1 year after beginning regularly administered treatment. Because of the risk of anaphylaxis, initiate XOLAIR therapy in a healthcare setting and closely observe patients for an appropriate period of time after XOLAIR administration. Health care providers administering XOLAIR should be prepared to manage anaphylaxis which can be life-threatening. Inform patients of the signs and symptoms of anaphylaxis and instruct them to seek immediate medical care should symptoms occur. Selection of patients for self-administration of XOLAIR should be based on criteria to mitigate risk from anaphylaxis [see Dosage and Administration (2.6), Warnings and Precautions (5.1) and Adverse Reactions (6.1, 6.2)].
12.3 Pharmacokinetics
After SC administration, omalizumab was absorbed with an average absolute bioavailability of 62%. Following a single SC dose in adult and adolescent patients with asthma, omalizumab was absorbed slowly, reaching peak serum concentrations after an average of 7–8 days. In patients with CSU, the peak serum concentration was reached at a similar time after a single SC dose. The pharmacokinetics of omalizumab was linear at doses greater than 0.5 mg/kg. In patients with asthma, following multiple doses of XOLAIR, areas under the serum concentration-time curve from Day 0 to Day 14 at steady state were up to 6-fold of those after the first dose. In patients with CSU, omalizumab exhibited linear pharmacokinetics across the dose range of 75 mg to 600 mg given as single subcutaneous dose. Following repeat dosing from 75 to 300 mg every 4 weeks, trough serum concentrations of omalizumab increased proportionally with the dose levels.
In vitro, omalizumab formed complexes of limited size with IgE. Precipitating complexes and complexes larger than 1 million daltons in molecular weight were not observed in vitro or in vivo. Tissue distribution studies in Cynomolgus monkeys showed no specific uptake of 125I-omalizumab by any organ or tissue. The apparent volume of distribution of omalizumab in patients with asthma following SC administration was 78 ± 32 mL/kg. In patients with CSU, based on population pharmacokinetics, distribution of omalizumab was similar to that in patients with asthma.
Clearance of omalizumab involved IgG clearance processes as well as clearance via specific binding and complex formation with its target ligand, IgE. Liver elimination of IgG included degradation in the liver reticuloendothelial system (RES) and endothelial cells. Intact IgG was also excreted in bile. In studies with mice and monkeys, omalizumab:IgE complexes were eliminated by interactions with Fcγ receptors within the RES at rates that were generally faster than IgG clearance. In asthma patients omalizumab serum elimination half-life averaged 26 days, with apparent clearance averaging 2.4 ± 1.1 mL/kg/day. Doubling body weight approximately doubled apparent clearance. In CSU patients, at steady state, based on population pharmacokinetics, omalizumab serum elimination half-life averaged 24 days and apparent clearance averaged 240 mL/day (corresponding to 3.0 mL/kg/day for an 80 kg patient).
1 Indications and Usage (1 INDICATIONS AND USAGE)
XOLAIR is an anti-IgE antibody indicated for:
- Moderate to severe persistent asthma in adults and pediatric patients 6 years of age and older with a positive skin test or in vitro reactivity to a perennial aeroallergen and symptoms that are inadequately controlled with inhaled corticosteroids (1.1)
- Chronic rhinosinusitis with nasal polyps (CRSwNP) in adult patients 18 years of age and older with inadequate response to nasal corticosteroids, as add-on maintenance treatment (1.2)
- IgE-mediated food allergy in adult and pediatric patients aged 1 year and older for the reduction of allergic reactions (Type I), including anaphylaxis, that may occur with accidental exposure to one or more foods. To be used in conjunction with food allergen avoidance (1.3)
- Chronic spontaneous urticaria (CSU) in adults and adolescents 12 years of age and older who remain symptomatic despite H1 antihistamine treatment (1.4)
Limitations of Use:
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Anaphylaxis: Initiate XOLAIR therapy in a healthcare setting prepared to manage anaphylaxis which can be life-threatening and observe patients for an appropriate period of time after administration. (5.1)
- Malignancy: Malignancies have been observed in clinical studies. (5.2)
- Acute Asthma Symptoms: Do not use for the treatment of acute bronchospasm or status asthmaticus. (5.3)
- Corticosteroid Reduction: Do not abruptly discontinue corticosteroids upon initiation of XOLAIR therapy. (5.4)
- Eosinophilic Conditions: Be alert to eosinophilia, vasculitic rash, worsening pulmonary symptoms, cardiac complications, and/or neuropathy, especially upon reduction of oral corticosteroids. (5.5)
- Fever, Arthralgia, and Rash: Stop XOLAIR if patients develop signs and symptoms similar to serum sickness. (5.6)
- Potential Medication Error Related to Emergency Treatment of Anaphylaxis: XOLAIR should not be used for emergency treatment of allergic reactions, including anaphylaxis. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
For subcutaneous (SC) administration only. (2.2, 2.3, 2.4, 2.5)
See full prescribing information for administration instructions (2.6, 2.7, 2.8).
- Asthma: XOLAIR 75 to 375 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination charts. (2.2)
- Chronic Rhinosinusitis with Nasal Polyps: XOLAIR 75 to 600 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination charts. (2.3)
- IgE-Mediated Food Allergy: XOLAIR 75 mg to 600 mg SC every 2 or 4 weeks. Determine dose (mg) and dosing frequency by serum total IgE level (IU/mL), measured before the start of treatment, and body weight (kg). See the dose determination chart. (2.4)
- Chronic Spontaneous Urticaria: XOLAIR 150 or 300 mg SC every 4 weeks. Dosing in CSU is not dependent on serum IgE level or body weight. (2.5)
2.6 Administration Overview
- Administer XOLAIR by subcutaneous injection.
- XOLAIR is intended for use under the guidance of a healthcare provider.
- Initiate therapy in a healthcare setting and once therapy has been safely established, the healthcare provider may determine whether self-administration of XOLAIR prefilled syringe or autoinjector by the patient or caregiver is appropriate, based on careful assessment of risk for anaphylaxis and mitigation strategies.
5.5 Eosinophilic Conditions
In rare cases, patients with asthma on therapy with XOLAIR may present with serious systemic eosinophilia sometimes presenting with clinical features of vasculitis consistent with Churg-Strauss syndrome, a condition which is often treated with systemic corticosteroid therapy. These events usually, but not always, have been associated with the reduction of oral corticosteroid therapy. Physicians should be alert to eosinophilia, vasculitic rash, worsening pulmonary symptoms, cardiac complications, and/or neuropathy presenting in their patients. A causal association between XOLAIR and these underlying conditions has not been established.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection:
- 75 mg/0.5 mL is a clear to slightly opalescent and colorless to pale brownish-yellow solution in a single-dose prefilled syringe with needle shield or single-dose prefilled autoinjector
- 150 mg/mL is a clear to slightly opalescent and colorless to pale brownish-yellow solution in a single-dose prefilled syringe with needle shield or single-dose prefilled autoinjector
- 300 mg/2 mL is a clear to slightly opalescent and colorless to pale brownish-yellow solution in a single-dose prefilled syringe with needle shield or single-dose prefilled autoinjector
- For injection: 150 mg white lyophilized powder in a single-dose vial for reconstitution
5.4 Corticosteroid Reduction
Do not discontinue systemic or inhaled corticosteroids abruptly upon initiation of XOLAIR therapy for asthma or CRSwNP. Decrease corticosteroids gradually under the direct supervision of a physician. In CSU patients, the use of XOLAIR in combination with corticosteroids has not been evaluated.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of XOLAIR. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
1.3 Ige Mediated Food Allergy (1.3 IgE-Mediated Food Allergy)
XOLAIR is indicated for the reduction of allergic reactions (Type I), including anaphylaxis, that may occur with accidental exposure to one or more foods in adult and pediatric patients aged 1 year and older with IgE-mediated food allergy.
XOLAIR is to be used in conjunction with food allergen avoidance.
14.3 Ige Mediated Food Allergy (14.3 IgE-Mediated Food Allergy)
The safety and efficacy of XOLAIR was evaluated in a multi-center, randomized, double-blind, placebo-controlled Food Allergy (FA) trial [NCT03881696] in 168 adult patients and pediatric patients 1 year of age to less than 56 years who were allergic to peanut and at least two other foods, including milk, egg, wheat, cashew, hazelnut, or walnut (i.e., studied foods). The FA trial enrolled patients who experienced dose-limiting symptoms (e.g., moderate to severe skin, respiratory or gastrointestinal symptoms) to a single dose of ≤100 mg of peanut protein and ≤300 mg protein for each of the other two foods (milk, egg, wheat, cashew, hazelnut, or walnut) during the screening double-blind placebo-controlled food challenge (DBPCFC). Patients with a history of severe anaphylaxis (defined as neurological compromise or requiring intubation) were excluded from the study. Patients were randomized 2:1 to receive a subcutaneous dosage of XOLAIR or placebo based on serum total IgE level (IU/mL), measured before the start of treatment, and by body weight according to Table 4 [see Dosage and Administration (2.4)] for 16 to 20 weeks. After 16 to 20 weeks of treatment, each patient completed a DBPCFC consisting of placebo and each of their 3 studied foods. Following the DBPCFC, the first 60 patients that included 59 pediatric patients and one adult patient who completed the double-blind, placebo-controlled phase of the study could continue to receive XOLAIR in a 24 to 28 week open-label extension.
Efficacy of XOLAIR is based on 165 pediatric patients who were included in the efficacy analyses provided below. The mean age of the pediatric patients was 8 years (age range: 1 to 17 years); 37% were less than 6 years of age, 38% were 6 to less than 12 years of age, and 25% were 12 to less than 18 years of age. Patient population were 56% male, 63% White, 13% Asian, 7% Black, 16% Other, and 55% of patients had a history of asthma.
The primary efficacy endpoint was the percentage of patients who were able to consume a single dose of ≥600 mg of peanut protein without dose-limiting symptoms (e.g., moderate to severe skin, respiratory or gastrointestinal symptoms) during DBPCFC. Table 16 shows XOLAIR treatment led to a statistically higher response rate (68%) than placebo (5%).
The secondary efficacy endpoints were the percentage of patients who were able to consume a single dose of ≥1000 mg of cashew, milk, or egg protein without dose-limiting symptoms during DBPCFC. The study met the secondary endpoints and demonstrated that XOLAIR treatment led to statistically higher response rates than placebo for all three foods. See Table 16 for details.
| Food, Challenge Dose | Response Rate Response defined as consumption of a single dose of the specified amount of food without dose-limiting symptoms. (%)(n/N) |
Treatment Difference (%) (XOLAIR-Placebo) (95% CI) |
|
|---|---|---|---|
| XOLAIR | Placebo | ||
| CI = Confidence interval; DBPCFC = Double-blind placebo-controlled food challenge; n = Number of responders; N = Total number of patients receiving food, challenge dose. Notes: Subjects without an exit DBPCFC or evaluable exit DBPCFC were counted as non-responders; P-values from two-sided Fisher's exact tests were <0.0001 for all the food challenge doses. |
|||
| Peanut, ≥600 mg | 68% (75/110) |
5% (3/55) |
63% (50%, 73%) |
| Peanut, ≥1000 mg Consumption of a single dose of ≥1000 mg of peanut protein was an additional secondary endpoint. The key secondary efficacy endpoints were the percentage of patients who were able to consume a single dose of ≥1000 mg of cashew, milk, or egg protein.
|
65% (72/110) |
0% (0/55) |
65% (56%, 74%) |
| Cashew, ≥1000 mg | 42% (27/64) |
3% (1/30) |
39% (20%, 53%) |
| Milk, ≥1000 mg | 66% (25/38) |
11% (2/19) |
55% (29%, 73%) |
| Egg, ≥1000 mg | 67% (31/46) |
0% (0/19) |
67% (49%, 80%) |
Seventeen percent of XOLAIR treated patients were not able to consume >100 mg of peanut protein without moderate to severe dose-limiting symptoms. Eighteen, 22, and 41 percent of XOLAIR-treated patients were not able to consume >300 mg of milk, egg, or cashew protein, respectively, without moderate to severe dose-limiting symptoms.
Additional secondary analyses included the percentage of patients who were able to consume at least two or all three foods during DBPCFC. For two foods, 71% of XOLAIR treated patients were able to consume a single dose of ≥600 mg versus 5% in the placebo group and 67% were able to consume a single dose of ≥1000 mg versus 4% in the placebo group. For a single dose of ≥600 mg of three foods, the response rates were 48% in the XOLAIR group versus 4% in the placebo group and for a single dose of ≥1000 mg of three foods, the response rate in the XOLAIR group was 39% while none of the placebo patients were able to consume the challenge dose without symptoms.
The effectiveness of XOLAIR in adults is supported by the adequate and well-controlled trial of XOLAIR in pediatric patients, disease similarity in pediatric and adult patients, and pharmacokinetic (PK) similarity [see Clinical Pharmacology (12.3)].
While efficacy cannot be established from uncontrolled, open-label studies, for 38 pediatric patients who continued XOLAIR for 24-28 weeks in an open-label extension, the percentage of patients who were able to consume ≥600 mg of peanut protein and ≥1000 mg of egg, milk, and/or cashew protein without moderate to severe dose-limiting symptoms was maintained.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
5.6 Fever, Arthralgia, and Rash
In post-approval use, some patients have experienced a constellation of signs and symptoms including arthritis/arthralgia, rash, fever, and lymphadenopathy with an onset 1 to 5 days after the first or subsequent injections of XOLAIR. These signs and symptoms have recurred after additional doses in some patients. Although circulating immune complexes or a skin biopsy consistent with a Type III reaction were not seen with these cases, these signs and symptoms are similar to those seen in patients with serum sickness. Physicians should stop XOLAIR if a patient develops this constellation of signs and symptoms [see Adverse Reactions (6.2)].
1.4 Chronic Spontaneous Urticaria
XOLAIR is indicated for the treatment of adults and adolescents 12 years of age and older with chronic spontaneous urticaria (CSU) who remain symptomatic despite H1 antihistamine treatment.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
2.2 Recommended Dosage for Asthma
The recommended dosage for asthma is XOLAIR 75 mg to 375 mg by subcutaneous injection every 2 or 4 weeks based on serum total IgE level (IU/mL) measured before the start of treatment and by body weight (kg) [see Dosage and Administration (2.1)].
- Adult and adolescent patients 12 years of age and older: Initiate dosing according to Table 1.
- Pediatric patients 6 to <12 years of age: Initiate dosing according to Table 2.
5.7 Parasitic (helminth) Infection (5.7 Parasitic (Helminth) Infection)
Monitor patients at high risk of geohelminth infection while on XOLAIR therapy. Insufficient data are available to determine the length of monitoring required for geohelminth infections after stopping XOLAIR treatment.
In a one-year clinical trial conducted in Brazil in adult and adolescent patients at high risk for geohelminthic infections (roundworm, hookworm, whipworm, threadworm), 53% (36/68) of XOLAIR-treated patients experienced an infection, as diagnosed by standard stool examination, compared to 42% (29/69) of placebo controls. The point estimate of the odds ratio for infection was 1.96, with a 95% confidence interval (0.88, 4.36) indicating that in this study a patient who had an infection was anywhere from 0.88 to 4.36 times as likely to have received XOLAIR than a patient who did not have an infection. Response to appropriate anti-geohelminth treatment of infection as measured by stool egg counts was not different between treatment groups.
1.2 Chronic Rhinosinusitis With Nasal Polyps (1.2 Chronic Rhinosinusitis with Nasal Polyps)
XOLAIR is indicated for add-on maintenance treatment of chronic rhinosinusitis with nasal polyps (CRSwNP) in adult patients 18 years of age and older with inadequate response to nasal corticosteroids.
Principal Display Panel 150 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 150 mg Vial Carton)
NDC 50242-040-62
SINGLE-DOSE VIAL
150 mg
Xolair®
Omalizumab
FOR SUBCUTANEOUS USE
KEEP REFRIGERATED. DO NOT FREEZE.
Genentech
NOVARTIS
10198215
2.7 Xolair Prefilled Syringe and Autoinjector (2.7 XOLAIR Prefilled Syringe and Autoinjector)
XOLAIR injection doses are available as a prefilled syringe or as an autoinjector. Instruct patients or caregivers to follow the directions provided in the "Instructions for Use" for preparation and administration of XOLAIR prefilled syringe or autoinjector [see Instructions for Use].
5.3 Acute Asthma Symptoms and Deteriorating Disease
XOLAIR has not been shown to alleviate asthma exacerbations acutely. Do not use XOLAIR to treat acute bronchospasm or status asthmaticus. Patients should seek medical advice if their asthma remains uncontrolled or worsens after initiation of treatment with XOLAIR.
2.4 Recommended Dosage for Ige Mediated Food Allergy (2.4 Recommended Dosage for IgE-Mediated Food Allergy)
The recommended dosage for IgE-mediated food allergy is XOLAIR 75 mg to 600 mg by subcutaneous injection every 2 or 4 weeks based on serum total IgE level (IU/mL), measured before the start of treatment, and by body weight [see Dosage and Administration (2.1)]. Refer to Table 4 for recommended dosage based on serum IgE level and body weight for patients with IgE-mediated food allergy.
Principal Display Panel 300 Mg/2 Ml Syringe Carton (PRINCIPAL DISPLAY PANEL - 300 mg/2 mL Syringe Carton)
Xolair®
(omalizumab) Injection
300 mg/2 mL
For Subcutaneous Use.
1 Single-Dose Prefilled Syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-227-01
Dispense the accompanying Medication Guide to each patient.
Rx only
Must be refrigerated
Genentech
NOVARTIS
11011496
Principal Display Panel 150 Mg/ml Autoinjector Carton (PRINCIPAL DISPLAY PANEL - 150 mg/mL Autoinjector Carton)
Xolair®
(omalizumab) Injection
150 mg/mL
For Subcutaneous Use.
1 Single-Dose Autoinjector
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-215-55
Dispense the accompanying Medication Guide to each patient.
Genentech
NOVARTIS
11011532
2.5 Recommended Dosage for Chronic Spontaneous Urticaria
The recommended dosage for chronic spontaneous urticaria (CSU) is XOLAIR 150 mg or 300 mg by subcutaneous injection every 4 weeks.
- The 300 mg dose may be administered as one subcutaneous injection of 300 mg/2 mL or as two subcutaneous injections of 150 mg/mL.
- Dosing of XOLAIR in CSU patients is not dependent on serum IgE (free or total) level or body weight.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies have been performed in animals to evaluate the carcinogenic potential of XOLAIR.
There were no effects on fertility and reproductive performance in male and female Cynomolgus monkeys that received XOLAIR at subcutaneous doses up to 75 mg/kg/week (approximately 5 times the maximum recommended human dose on a mg/kg basis).
Principal Display Panel 300 Mg/2 Ml Autoinjector Carton (PRINCIPAL DISPLAY PANEL - 300 mg/2 mL Autoinjector Carton)
Xolair®
(omalizumab) Injection
300 mg/2 mL
For Subcutaneous Use.
1 Single-Dose Autoinjector
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-227-55
Dispense the accompanying Medication Guide to each patient.
Genentech
NOVARTIS
11011548
Principal Display Panel 75 Mg/0.5 Ml Autoinjector Carton (PRINCIPAL DISPLAY PANEL - 75 mg/0.5 mL Autoinjector Carton)
Xolair®
(omalizumab) Injection
75 mg/0.5 mL
For Subcutaneous Use.
1 Single-Dose Autoinjector
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-214-55
Dispense the accompanying Medication Guide to each patient.
Genentech
NOVARTIS
11011505
Principal Display Panel 150 Mg/ml Syringe Carton 215 01 (PRINCIPAL DISPLAY PANEL - 150 mg/mL Syringe Carton - 215-01)
Xolair®
(omalizumab) Injection
150 mg/mL
For Subcutaneous Use. Single-Dose Prefilled Syringe.
Dispense the accompanying
Medication Guide to each patient.
1 prefilled syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-215-01
Caution: The needle cap may contain natural rubber latex
which may cause allergic reactions in latex sensitive individuals.
Rx only
Genentech
NOVARTIS
11011498
Principal Display Panel 150 Mg/ml Syringe Carton 215 03 (PRINCIPAL DISPLAY PANEL - 150 mg/mL Syringe Carton - 215-03)
Xolair®
(omalizumab) Injection
150 mg/mL
For Subcutaneous Use.
1 Single-Dose Prefilled Syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-215-03
Dispense the accompanying Medication Guide to each patient.
Rx only
Must be refrigerated
Genentech
NOVARTIS
11011522
Principal Display Panel 75 Mg/0.5 Ml Syringe Carton 214 01 (PRINCIPAL DISPLAY PANEL - 75 mg/0.5 mL Syringe Carton - 214-01)
Xolair®
(omalizumab) Injection
75 mg/0.5 mL
For Subcutaneous Use. Single-Dose Prefilled Syringe.
Dispense the accompanying
Medication Guide to each patient.
1 prefilled syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-214-01
Caution: The needle cap may contain natural rubber latex
which may cause allergic reactions in latex sensitive individuals.
Rx only
Genentech
NOVARTIS
11011555
Principal Display Panel 75 Mg/0.5 Ml Syringe Carton 214 03 (PRINCIPAL DISPLAY PANEL - 75 mg/0.5 mL Syringe Carton - 214-03)
Xolair®
(omalizumab) Injection
75 mg/0.5 mL
For Subcutaneous Use.
1 Single-Dose Prefilled Syringe
Do not use for emergency treatment.
Your dose may require more than 1 injection.
For questions, contact your healthcare provider.
NDC 50242-214-03
Dispense the accompanying Medication Guide to each patient.
Rx only
Must be refrigerated
Genentech
NOVARTIS
11011485
2.8 Preparation for Use and Injection of Xolair Lyophilized Powder (2.8 Preparation for Use and Injection of XOLAIR Lyophilized Powder)
XOLAIR lyophilized powder should only be prepared and injected by a healthcare provider. The supplied XOLAIR lyophilized powder must be reconstituted with Sterile Water for Injection (SWFI) USP, using the following instructions:
-
1)Before reconstitution, determine the number of vials that will need to be reconstituted (each vial delivers 150 mg of XOLAIR in 1.2 mL) (see Table 6).
| XOLAIR Dose The 75 mg, 150 mg, 225 mg, 300 mg, and 375 mg XOLAIR doses are approved for use in asthma patients. All doses in the table are approved for use in CRSwNP and IgE-mediated food allergy patients. The 150 mg and 300 mg XOLAIR doses are also approved for use in CSU patients.
|
Number of Vials | Number of Injections | Total Volume Injected |
|---|---|---|---|
| 75 mg | 1 | 1 | 0.6 mL |
| 150 mg | 1 | 1 | 1.2 mL |
| 225 mg | 2 | 2 | 1.8 mL |
| 300 mg | 2 | 2 | 2.4 mL |
| 375 mg | 3 | 3 | 3.0 mL |
| 450 mg | 3 | 3 | 3.6 mL |
| 525 mg | 4 | 4 | 4.2 mL |
| 600 mg | 4 | 4 | 4.8 mL |
-
2)Draw 1.4 mL of SWFI, USP, into a 3 mL syringe equipped with a 1-inch, 18-gauge needle.
-
3)Place the vial upright on a flat surface and using standard aseptic technique, insert the needle and inject the SWFI, USP, directly onto the product.
-
4)Keeping the vial upright, gently swirl the upright vial for approximately 1 minute to evenly wet the powder. Do not shake.
-
5)Gently swirl the vial for 5 to 10 seconds approximately every 5 minutes in order to dissolve any remaining solids. The lyophilized product takes 15 to 20 minutes to dissolve. If it takes longer than 20 minutes to dissolve completely, gently swirl the vial for 5 to 10 seconds approximately every 5 minutes until there are no visible gel-like particles in the solution. Do not use if the contents of the vial do not dissolve completely by 40 minutes.
-
6)After reconstitution, XOLAIR solution is somewhat viscous and will appear clear or slightly opalescent. It is acceptable if there are a few small bubbles or foam around the edge of the vial; there should be no visible gel-like particles in the reconstituted solution. Do not use if foreign particles are present.
-
7)Invert the vial for 15 seconds in order to allow the solution to drain toward the stopper.
-
8)Use the XOLAIR solution within 8 hours following reconstitution when stored in the vial at 2ºC to 8ºC (36ºF to 46ºF), or within 4 hours of reconstitution when stored at room temperature. Reconstituted XOLAIR vials should be protected from sunlight.
-
9)Using a new 3 mL syringe equipped with a 1-inch, 18-gauge needle, insert the needle into the inverted vial. Position the needle tip at the very bottom of the solution in the vial stopper when drawing the solution into the syringe. The reconstituted product is somewhat viscous. Withdraw all of the product from the vial before expelling any air or excess solution from the syringe. Before removing the needle from the vial, pull the plunger all the way back to the end of the syringe barrel in order to remove all of the solution from the inverted vial.
-
10)Replace the 18-gauge needle with a 25-gauge needle for subcutaneous injection.
-
11)Expel air, large bubbles, and any excess solution in order to obtain a volume of 1.2 mL corresponding to a dose of 150 mg of XOLAIR. To obtain a volume of 0.6 mL corresponding to a dose of 75 mg of XOLAIR, expel air, large bubbles and discard 0.6 mL from the syringe. A thin layer of small bubbles may remain at the top of the solution in the syringe.
-
12)Administer XOLAIR by subcutaneous injection. The injection may take 5-10 seconds to administer because the solution is slightly viscous. Do not administer more than 150 mg (contents of one vial) per injection site. Divide doses of more than 150 mg between two or more injection sites. Choose a different injection site for each new injection at least 1 inch from the area used for other injections.
2.3 Recommended Dosage for Chronic Rhinosinusitis With Nasal Polyps (2.3 Recommended Dosage for Chronic Rhinosinusitis with Nasal Polyps)
The recommended dosage for chronic rhinosinusitis with nasal polyps (CRSwNP) is XOLAIR 75 mg to 600 mg by subcutaneous injection every 2 or 4 weeks based on serum total IgE level (IU/mL) measure before the start of treatment and by body weight (kg) [see Dosage and Administration (2.1)]. Refer to Table 3 for recommended dosage based on serum total IgE level and body weight for patients with CRSwNP.
5.9 Potential Medication Error Related to Emergency Treatment of Anaphylaxis
XOLAIR should not be used for the emergency treatment of allergic reactions, including anaphylaxis. In studies to simulate use, some patients and caregivers did not understand that XOLAIR is not intended for the emergency treatment of allergic reactions, including anaphylaxis. The safety and effectiveness of XOLAIR for emergency treatment of allergic reactions, including anaphylaxis, have not been established. Instruct patients that XOLAIR is for maintenance use to reduce allergic reactions, including anaphylaxis, while avoiding food allergens.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:58.757026 · Updated: 2026-03-14T22:39:05.457483