7f415a47-5240-41a0-b9df-217aa9a212dd
34390-5
HUMAN OTC DRUG LABEL
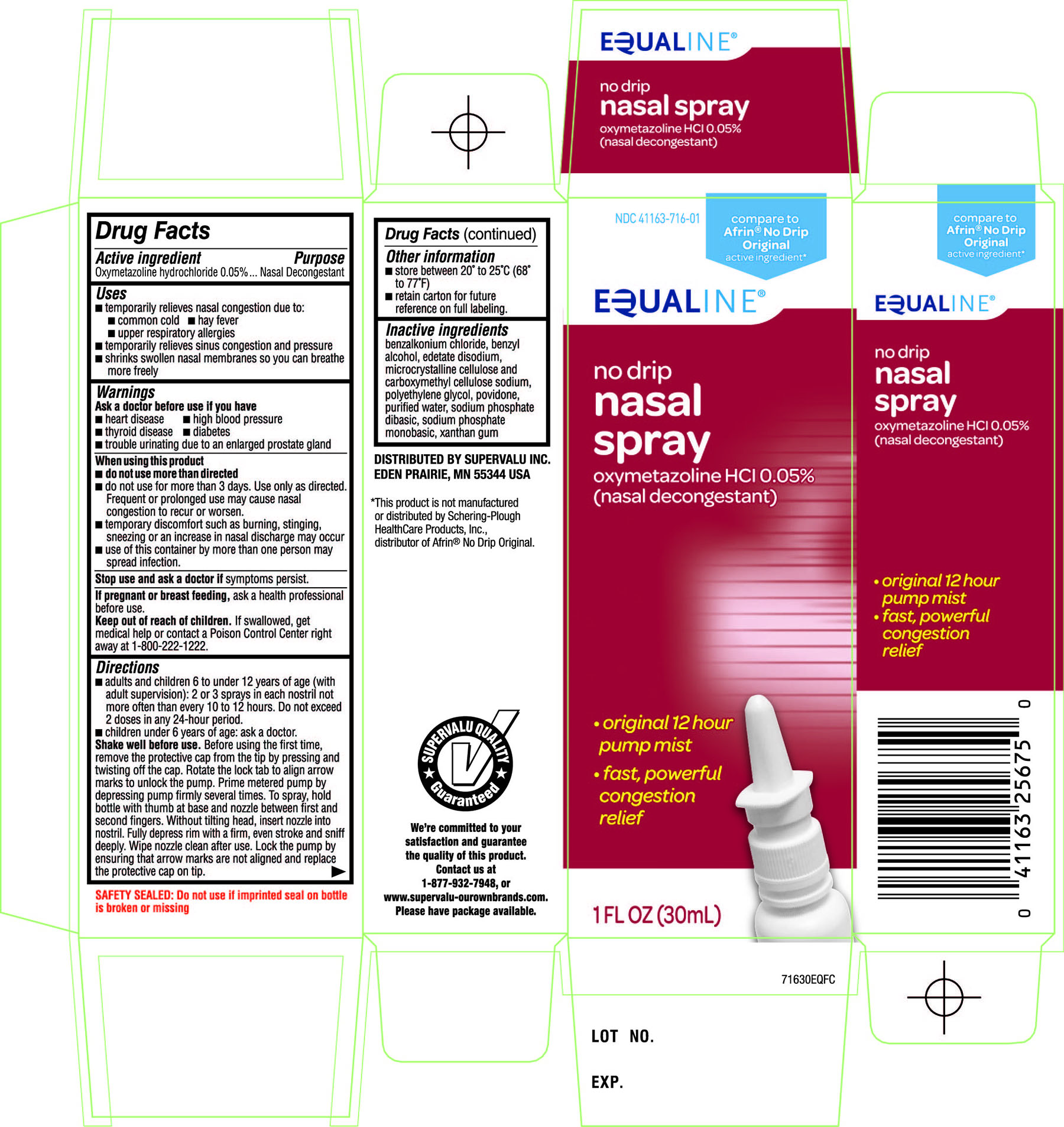
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Oxymetazoline hydrochloride 0.05%
Purpose
Nasal Decongestant
Medication Information
Purpose
Nasal Decongestant
Description
Oxymetazoline hydrochloride 0.05%
Uses
-
•temporarily relieves nasal congestion due to:
-
•common cold
-
•hay fever
-
•upper respiratory allergies
-
•temporarily relieves sinus congestion and pressure
-
•shrinks swollen nasal membranes so you can breathe more freely.
Directions
-
•adults and children 6 to under 12 years of age (with adult supervision): 2 or 3 sprays in each nostril not more often than every 10 to 12 hours. Do not exceed 2 doses in any 24-hour period.
-
•children under 6 years of age: ask a doctor.
Shake well before use. Before using the first time, remove the protective cap from the tip by pressing and twisting off the cap. Rotate the lock tab to align arrow marks to unlock the pump. Prime metered pump by depressing pump firmly several times. To spray, hold bottle with thumb at base and nozzle between first and second fingers. Without tilting head, insert nozzle into nostril. Fully depress rim with a firm, even stroke and sniff deeply. Wipe nozzle clean after use. Lock the pump by ensuring that arrow marks are not aligned and replace the protective cap on tip.
Active Ingredient
Oxymetazoline hydrochloride 0.05%
Other Information
-
•store between 20°to 25°C (68°to77°F)
-
•retain carton for future reference on full labeling.
Inactive Ingredients
benzalkonium chloride, benzyl alcohol, edetate disodium, microcrystalline cellulose and carboxymethyl cellulose sodium, polyethylene glycol, povidone, purified water, sodium phosphate dibasic, sodium phosphate monobasic, xanthan gum.
Principal Display Panel
NDC 41163-716-01
compare to Afrin® No-Drip Original active ingredient*
EQUALINE®
no drip
nasal spray
Oxymetazoline HCl 0.05%
(nasal decongestant)
-
•original 12 hour pump mist
-
•fast, powerful congestion relief
1 FL OZ (30 mL)
SAFETY SEALED: Do not use if imprinted seal on bottle is broken or missing
DISTRIBUTED BY SUPERVALU INC.
EDEN PRAIRIE, MN 55344 USA
*This product is not manufactured or distributed by Schering-Plough Healthcare Products, Inc., distributor of Afrin®No Drip Original.
SUPERVALU QUALITY Guaranteed
We’re committed to your satisfaction and guarantee the quality of this product.
Contact us at 1-877-932-7948, or
www.supervalu-ourownbrands.com.
Please have package available.
When Using This Product
-
•do not use more than directed
-
•do not use for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen.
-
•temporary discomfort such as burning, stinging, sneezing or an increase in nasal discharge may occur
-
•use of this container by more than one person may spread infection.
Stop Use and Ask A Doctor If
symptoms persist.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
Ask A Doctor Before Use If You Have
-
•heart disease
-
•high blood pressure
-
•thyroid disease
-
•diabetes
-
•trouble urinating due to an enlarged prostate gland
Structured Label Content
Uses
-
•temporarily relieves nasal congestion due to:
-
•common cold
-
•hay fever
-
•upper respiratory allergies
-
•temporarily relieves sinus congestion and pressure
-
•shrinks swollen nasal membranes so you can breathe more freely.
Purpose
Nasal Decongestant
Directions
-
•adults and children 6 to under 12 years of age (with adult supervision): 2 or 3 sprays in each nostril not more often than every 10 to 12 hours. Do not exceed 2 doses in any 24-hour period.
-
•children under 6 years of age: ask a doctor.
Shake well before use. Before using the first time, remove the protective cap from the tip by pressing and twisting off the cap. Rotate the lock tab to align arrow marks to unlock the pump. Prime metered pump by depressing pump firmly several times. To spray, hold bottle with thumb at base and nozzle between first and second fingers. Without tilting head, insert nozzle into nostril. Fully depress rim with a firm, even stroke and sniff deeply. Wipe nozzle clean after use. Lock the pump by ensuring that arrow marks are not aligned and replace the protective cap on tip.
Active Ingredient (Active ingredient)
Oxymetazoline hydrochloride 0.05%
Other Information (Other information)
-
•store between 20°to 25°C (68°to77°F)
-
•retain carton for future reference on full labeling.
Inactive Ingredients (Inactive ingredients)
benzalkonium chloride, benzyl alcohol, edetate disodium, microcrystalline cellulose and carboxymethyl cellulose sodium, polyethylene glycol, povidone, purified water, sodium phosphate dibasic, sodium phosphate monobasic, xanthan gum.
Principal Display Panel
NDC 41163-716-01
compare to Afrin® No-Drip Original active ingredient*
EQUALINE®
no drip
nasal spray
Oxymetazoline HCl 0.05%
(nasal decongestant)
-
•original 12 hour pump mist
-
•fast, powerful congestion relief
1 FL OZ (30 mL)
SAFETY SEALED: Do not use if imprinted seal on bottle is broken or missing
DISTRIBUTED BY SUPERVALU INC.
EDEN PRAIRIE, MN 55344 USA
*This product is not manufactured or distributed by Schering-Plough Healthcare Products, Inc., distributor of Afrin®No Drip Original.
SUPERVALU QUALITY Guaranteed
We’re committed to your satisfaction and guarantee the quality of this product.
Contact us at 1-877-932-7948, or
www.supervalu-ourownbrands.com.
Please have package available.
When Using This Product (When using this product)
-
•do not use more than directed
-
•do not use for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen.
-
•temporary discomfort such as burning, stinging, sneezing or an increase in nasal discharge may occur
-
•use of this container by more than one person may spread infection.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
symptoms persist.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away at 1-800-222-1222.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
-
•heart disease
-
•high blood pressure
-
•thyroid disease
-
•diabetes
-
•trouble urinating due to an enlarged prostate gland
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:28.545454 · Updated: 2026-03-14T22:54:57.914892