4483-333 - motion time
7f3ce1e6-c0d6-4b56-8f7a-6368eb46901f
34390-5
HUMAN OTC DRUG LABEL
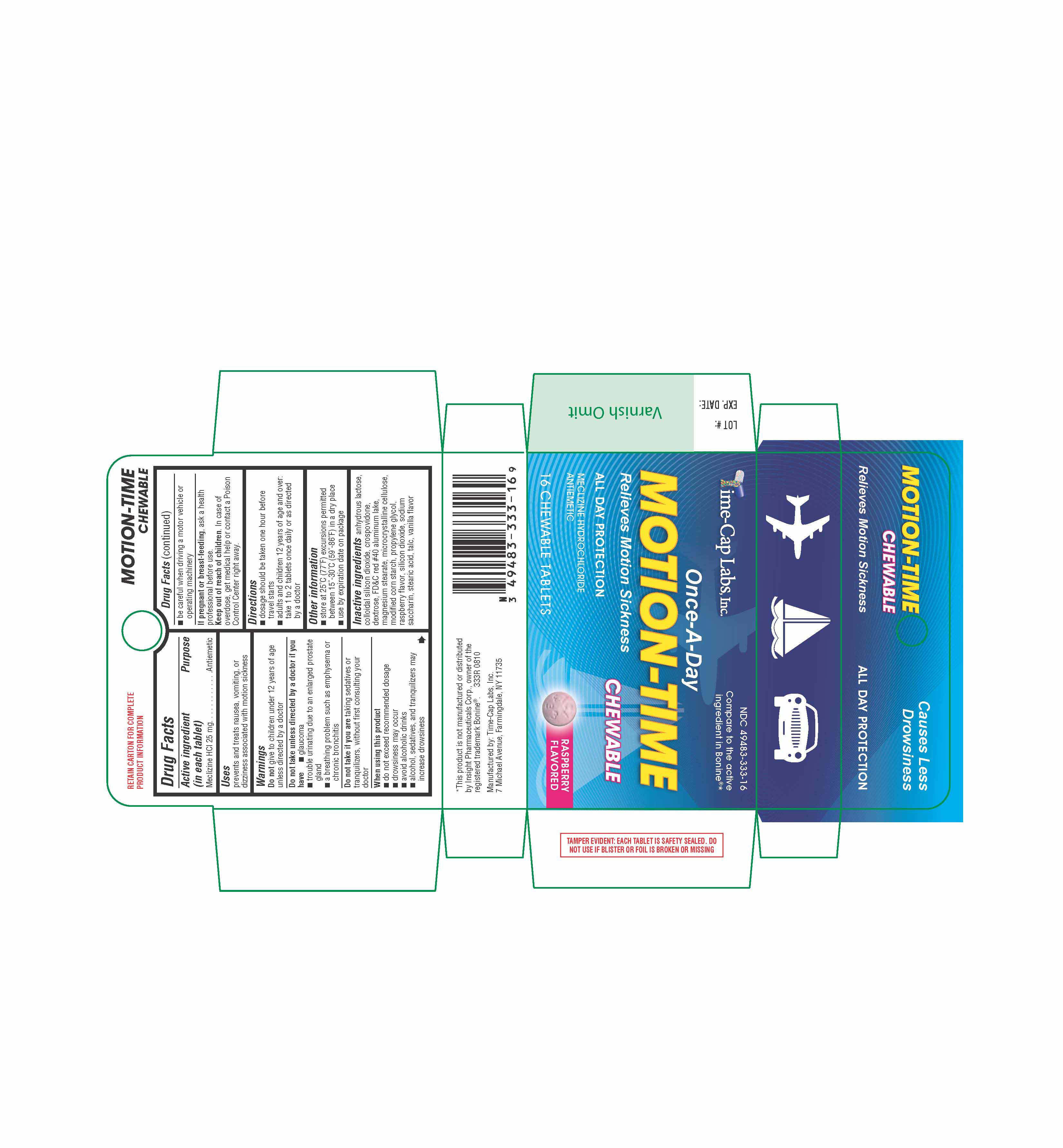
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient (in each tablet) Meclizine HCl 25 mg
Medication Information
Warnings and Precautions
|
Warnings
Do not give to children under 12 years of age unless directed by a doctor |
Active Ingredient
|
Active ingredient (in each tablet)
Meclizine HCl 25 mg |
Indications and Usage
|
Uses
prevents and treats nausea, vomiting, or dizziness associated with motion sickness |
Dosage and Administration
|
Directions
|
Description
Active ingredient (in each tablet) Meclizine HCl 25 mg
Section 44425-7
|
Other information
|
Section 50565-1
|
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. |
Section 50567-7
|
When using this product
|
Section 50569-3
|
Do not take unless directed by a doctor if you have
|
Section 50570-1
|
Do not take if you are taking sedatives or tranquilizers, without first consulting your doctor |
Section 51727-6
|
Inactive ingredients anhydrous lactose, colloidal silicon dioxide, crospovidone, dextrose, FD-C red 40 aluminum lake, magnesium stearate, microcrystalline cellulose, modified corn starch, propylene glycol, raspberry flavor, silicon dioxide, sodium saccharin, stearic acid, talc, vanilla flavor |
Section 51945-4
Meclizine HCL Chewable Tablets
Section 53414-9
|
If pregnant or breast-feeding, ask a health professional before use. |
Section 55105-1
|
Purpose
Antiemetic |
Structured Label Content
Warnings and Precautions (34071-1)
|
Warnings
Do not give to children under 12 years of age unless directed by a doctor |
Active Ingredient (55106-9)
|
Active ingredient (in each tablet)
Meclizine HCl 25 mg |
Indications and Usage (34067-9)
|
Uses
prevents and treats nausea, vomiting, or dizziness associated with motion sickness |
Dosage and Administration (34068-7)
|
Directions
|
Section 44425-7 (44425-7)
|
Other information
|
Section 50565-1 (50565-1)
|
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. |
Section 50567-7 (50567-7)
|
When using this product
|
Section 50569-3 (50569-3)
|
Do not take unless directed by a doctor if you have
|
Section 50570-1 (50570-1)
|
Do not take if you are taking sedatives or tranquilizers, without first consulting your doctor |
Section 51727-6 (51727-6)
|
Inactive ingredients anhydrous lactose, colloidal silicon dioxide, crospovidone, dextrose, FD-C red 40 aluminum lake, magnesium stearate, microcrystalline cellulose, modified corn starch, propylene glycol, raspberry flavor, silicon dioxide, sodium saccharin, stearic acid, talc, vanilla flavor |
Section 51945-4 (51945-4)
Meclizine HCL Chewable Tablets
Section 53414-9 (53414-9)
|
If pregnant or breast-feeding, ask a health professional before use. |
Section 55105-1 (55105-1)
|
Purpose
Antiemetic |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:56:42.587420 · Updated: 2026-03-14T22:55:22.018235