Loratidine Orally Disintegrating Tablets, Usp
7ed2aec0-4da7-4edc-9998-b96c67c82738
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Antihistamine
Medication Information
Purpose
Antihistamine
Description
Drug Facts
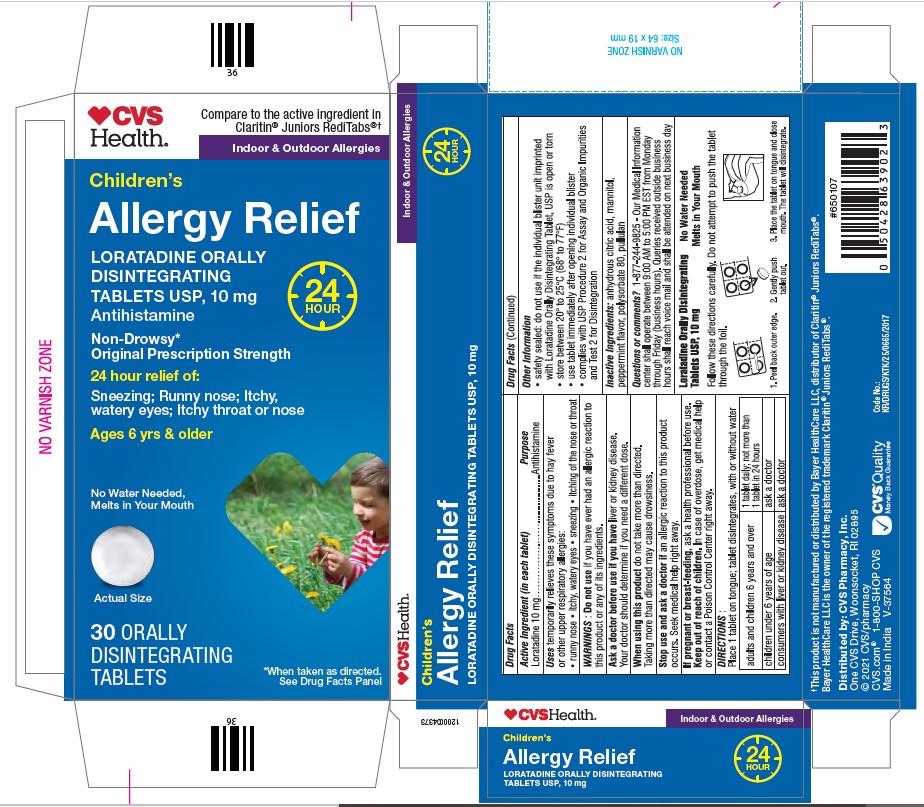
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
Section 50567-7
When using this product do not take more than directed. Taking more than directed may cause drowsiness.
Section 50569-3
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Section 50570-1
Do not use if you have ever had an allergic reaction to this product or any of its ingredients.
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Warnings
Directions
- place 1 tablet on tongue; tablet disintegrates, with or without water
| adults and children 6 years and over | 1 tablet daily; not more than 1 tablet in 24 hours |
| children under 6 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other Information
- safety sealed: do not use if the individual blister unit imprinted with Loratadine Orally Disintegrating Tablet, USP is open or torn
- store between 20° to 25°C (68° to 77°F)
- use tablet immediately after opening individual blister
- complies with USP Procedure 2 for Assay and Organic Impurities and Test 2 for Disintegration
Inactive Ingredients
anhydrous citric acid, mannitol, peppermint flavor, polysorbate 80, pullulan
Questions Or Comments?
1-877-244-9825
Active Ingredient (in Each Tablet)
Loratadine 10 mg
Package Label.principal Display Panel
CVS Health
Indoor & Outdoor Allergies
Children’s
LORATADINE ORALLY
DISINTEGRATING
TABLETS USP, 10 mg
Antihistamine
Allergy Relief
Non-Drowsy*
Original Prescription Strength
24 hour relief of:
Sneezing; Runny nose; Itchy,
watery eyes; Itchy throat or nose
Ages 6 yrs & older
30 ORALLY
DISINTEGRATING
TABLETS
Structured Label Content
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if an allergic reaction to this product occurs. Seek medical help right away.
Section 50567-7 (50567-7)
When using this product do not take more than directed. Taking more than directed may cause drowsiness.
Section 50569-3 (50569-3)
Ask a doctor before use if you have liver or kidney disease. Your doctor should determine if you need a different dose.
Section 50570-1 (50570-1)
Do not use if you have ever had an allergic reaction to this product or any of its ingredients.
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Antihistamine
Warnings
Directions
- place 1 tablet on tongue; tablet disintegrates, with or without water
| adults and children 6 years and over | 1 tablet daily; not more than 1 tablet in 24 hours |
| children under 6 years of age | ask a doctor |
| consumers with liver or kidney disease | ask a doctor |
Other Information (Other information)
- safety sealed: do not use if the individual blister unit imprinted with Loratadine Orally Disintegrating Tablet, USP is open or torn
- store between 20° to 25°C (68° to 77°F)
- use tablet immediately after opening individual blister
- complies with USP Procedure 2 for Assay and Organic Impurities and Test 2 for Disintegration
Inactive Ingredients (Inactive ingredients)
anhydrous citric acid, mannitol, peppermint flavor, polysorbate 80, pullulan
Questions Or Comments? (Questions or comments?)
1-877-244-9825
Active Ingredient (in Each Tablet) (Active ingredient (in each tablet))
Loratadine 10 mg
Package Label.principal Display Panel (PACKAGE LABEL.PRINCIPAL DISPLAY PANEL)
CVS Health
Indoor & Outdoor Allergies
Children’s
LORATADINE ORALLY
DISINTEGRATING
TABLETS USP, 10 mg
Antihistamine
Allergy Relief
Non-Drowsy*
Original Prescription Strength
24 hour relief of:
Sneezing; Runny nose; Itchy,
watery eyes; Itchy throat or nose
Ages 6 yrs & older
30 ORALLY
DISINTEGRATING
TABLETS
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:38.088225 · Updated: 2026-03-14T23:02:18.218337