These Highlights Do Not Include All The Information Needed To Use Enhertu Safely And Effectively. See Full Prescribing Information For Enhertu.
7e67e73e-ddf4-4e4d-8b50-09d7514910b6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Interstitial Lung Disease (ILD) and pneumonitis, including fatal cases, have been reported with ENHERTU. Monitor for and promptly investigate signs and symptoms including cough, dyspnea, fever, and other new or worsening respiratory symptoms. Permanently discontinue ENHERTU in all patients with Grade 2 or higher ILD/pneumonitis. Advise patients of the risk and the need to immediately report symptoms [see Dosage and Administration (2.3) , Warnings and Precautions (5.1) ] . Embryo-Fetal Toxicity: Exposure to ENHERTU during pregnancy can cause embryo-fetal harm. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.4) , Use in Specific Populations (8.1 , 8.3) ] .
Indications and Usage
ENHERTU is a HER2-directed antibody and topoisomerase inhibitor conjugate indicated: HER2-Positive Metastatic Breast Cancer in combination with pertuzumab as first-line treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer, as determined by an FDA-approved test. ( 1.1 ) as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer who have received a prior anti-HER2-based regimen either in the metastatic setting, or, in the neoadjuvant or adjuvant setting and have developed disease recurrence during or within six months of completing therapy. ( 1.1 ) HER2-Low and HER2-Ultralow Metastatic Breast Cancer as monotherapy for the treatment of adult patients with unresectable or metastatic hormone receptor (HR)-positive, HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer, as determined by an FDA-approved test, that has progressed on one or more endocrine therapies in the metastatic setting. ( 1.2 ) as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer, as determined by an FDA-approved test, who have received a prior chemotherapy in the metastatic setting; or developed disease recurrence during or within 6 months of completing adjuvant chemotherapy. ( 1.2 ) HER2-Mutant Unresectable or Metastatic Non-Small Cell Lung Cancer as monotherapy for the treatment of adult patients with unresectable or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating HER2 (ERBB2) mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy* ( 1.3 ) HER2-Positive Locally Advanced or Metastatic Gastric Cancer as monotherapy for the treatment of adult patients with locally advanced or metastatic HER2-positive (IHC 3+ or IHC 2+/ISH positive) gastric or gastroesophageal junction adenocarcinoma who have received a prior trastuzumab-based regimen. ( 1.4 ) HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options* ( 1.5 ) * These indications are approved under accelerated approval based on objective response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. ( 14.3 , 14.5 )
Dosage and Administration
Do not substitute ENHERTU for or with trastuzumab or ado-trastuzumab emtansine. ( 2.2 , 2.4 ) For intravenous infusion only . Do not administer as an intravenous push or bolus. DO NOT use Sodium Chloride Injection, USP. ( 2.4 ) Premedicate for prevention of chemotherapy-induced nausea and vomiting. ( 2.2 ) HER2-positive, HER2-low, or HER2-ultralow breast cancer, HER2-mutant NSCLC, and HER2-positive (IHC 3+) solid tumors: ENHERTU 5.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. ( 2.2 , 2.3 ) HER2-Positive First-line Metastatic Breast Cancer ENHERTU 5.4 mg/kg every 3 weeks (in combination with pertuzumab until disease progression or unacceptable toxicity. Cycle 1, Day 1: ENHERTU 5.4 mg/kg followed by pertuzumab 840 mg. ( 2.2 , 2.3 ) Subsequent cycles, Day 1: ENHERTU 5.4 mg/kg followed by pertuzumab 420 mg. ( 2.2 , 2.3 ) HER2-positive gastric cancer: 6.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. ( 2.2 , 2.3 ) Management of adverse reactions (ILD, neutropenia, thrombocytopenia, or left ventricular dysfunction) may require temporary interruption, dose reduction, or discontinuation of ENHERTU. ( 2.3 )
Warnings and Precautions
Neutropenia: Monitor complete blood counts prior to initiation of ENHERTU and prior to each dose, and as clinically indicated. Manage through treatment interruption or dose reduction. ( 2.3 , 5.2 ) Left Ventricular Dysfunction: Assess left ventricular ejection fraction (LVEF) prior to initiation of ENHERTU and at regular intervals during treatment as clinically indicated. Manage through treatment interruption or discontinuation. Permanently discontinue ENHERTU in patients with symptomatic congestive heart failure (CHF). ( 2.3 , 5.3 )
Contraindications
None .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1) ] Neutropenia [see Warnings and Precautions (5.2) ] Left Ventricular Dysfunction [see Warnings and Precautions (5.3) ]
Medication Information
Warnings and Precautions
Neutropenia: Monitor complete blood counts prior to initiation of ENHERTU and prior to each dose, and as clinically indicated. Manage through treatment interruption or dose reduction. ( 2.3 , 5.2 ) Left Ventricular Dysfunction: Assess left ventricular ejection fraction (LVEF) prior to initiation of ENHERTU and at regular intervals during treatment as clinically indicated. Manage through treatment interruption or discontinuation. Permanently discontinue ENHERTU in patients with symptomatic congestive heart failure (CHF). ( 2.3 , 5.3 )
Indications and Usage
ENHERTU is a HER2-directed antibody and topoisomerase inhibitor conjugate indicated: HER2-Positive Metastatic Breast Cancer in combination with pertuzumab as first-line treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer, as determined by an FDA-approved test. ( 1.1 ) as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer who have received a prior anti-HER2-based regimen either in the metastatic setting, or, in the neoadjuvant or adjuvant setting and have developed disease recurrence during or within six months of completing therapy. ( 1.1 ) HER2-Low and HER2-Ultralow Metastatic Breast Cancer as monotherapy for the treatment of adult patients with unresectable or metastatic hormone receptor (HR)-positive, HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer, as determined by an FDA-approved test, that has progressed on one or more endocrine therapies in the metastatic setting. ( 1.2 ) as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer, as determined by an FDA-approved test, who have received a prior chemotherapy in the metastatic setting; or developed disease recurrence during or within 6 months of completing adjuvant chemotherapy. ( 1.2 ) HER2-Mutant Unresectable or Metastatic Non-Small Cell Lung Cancer as monotherapy for the treatment of adult patients with unresectable or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating HER2 (ERBB2) mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy* ( 1.3 ) HER2-Positive Locally Advanced or Metastatic Gastric Cancer as monotherapy for the treatment of adult patients with locally advanced or metastatic HER2-positive (IHC 3+ or IHC 2+/ISH positive) gastric or gastroesophageal junction adenocarcinoma who have received a prior trastuzumab-based regimen. ( 1.4 ) HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options* ( 1.5 ) * These indications are approved under accelerated approval based on objective response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. ( 14.3 , 14.5 )
Dosage and Administration
Do not substitute ENHERTU for or with trastuzumab or ado-trastuzumab emtansine. ( 2.2 , 2.4 ) For intravenous infusion only . Do not administer as an intravenous push or bolus. DO NOT use Sodium Chloride Injection, USP. ( 2.4 ) Premedicate for prevention of chemotherapy-induced nausea and vomiting. ( 2.2 ) HER2-positive, HER2-low, or HER2-ultralow breast cancer, HER2-mutant NSCLC, and HER2-positive (IHC 3+) solid tumors: ENHERTU 5.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. ( 2.2 , 2.3 ) HER2-Positive First-line Metastatic Breast Cancer ENHERTU 5.4 mg/kg every 3 weeks (in combination with pertuzumab until disease progression or unacceptable toxicity. Cycle 1, Day 1: ENHERTU 5.4 mg/kg followed by pertuzumab 840 mg. ( 2.2 , 2.3 ) Subsequent cycles, Day 1: ENHERTU 5.4 mg/kg followed by pertuzumab 420 mg. ( 2.2 , 2.3 ) HER2-positive gastric cancer: 6.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. ( 2.2 , 2.3 ) Management of adverse reactions (ILD, neutropenia, thrombocytopenia, or left ventricular dysfunction) may require temporary interruption, dose reduction, or discontinuation of ENHERTU. ( 2.3 )
Contraindications
None .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1) ] Neutropenia [see Warnings and Precautions (5.2) ] Left Ventricular Dysfunction [see Warnings and Precautions (5.3) ]
Description
Interstitial Lung Disease (ILD) and pneumonitis, including fatal cases, have been reported with ENHERTU. Monitor for and promptly investigate signs and symptoms including cough, dyspnea, fever, and other new or worsening respiratory symptoms. Permanently discontinue ENHERTU in all patients with Grade 2 or higher ILD/pneumonitis. Advise patients of the risk and the need to immediately report symptoms [see Dosage and Administration (2.3) , Warnings and Precautions (5.1) ] . Embryo-Fetal Toxicity: Exposure to ENHERTU during pregnancy can cause embryo-fetal harm. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.4) , Use in Specific Populations (8.1 , 8.3) ] .
Section 42229-5
HER2-Positive Metastatic Breast Cancer
Select patients for treatment of unresectable or metastatic HER2-positive breast cancer with ENHERTU in combination with pertuzumab based on confirmed HER2-positive status or HER2 gene amplification (IHC 3+ or ISH+) [see Clinical Studies (14.1)].
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 12/2025 | ||
|
Medication Guide
ENHERTU® (en-HER-too) (fam-trastuzumab deruxtecan-nxki) for injection |
|||
|
What is the most important information I should know about ENHERTU? ENHERTU can cause serious side effects, including:
|
|||
|
|
||
Your healthcare provider will check you for these side effects during your treatment with ENHERTU. Your healthcare provider may reduce your dose, delay treatment or completely stop treatment with ENHERTU if you have severe side effects.
|
|||
|
What is ENHERTU?
ENHERTU is a prescription medicine used to treat adults who have:
|
|||
Before you receive ENHERTU, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
How will I receive ENHERTU?
|
|||
|
What are the possible side effects of ENHERTU? ENHERTU can cause serious side effects. See "What is the most important information I should know about ENHERTU?" The most common side effects of ENHERTU, when used in people with metastatic breast cancer, HER2-mutant non-small cell lung cancer, and other HER2-positive solid tumors include: |
|||
|
|
||
| The most common side effects of ENHERTU in combination with pertuzumab, when used in people with HER2-positive breast cancer include: | |||
|
|
||
| The most common side effects of ENHERTU, when used in people with HER2-positive gastric or GEJ adenocarcinoma, include: | |||
|
|
||
| ENHERTU may cause fertility problems in males, which may affect the ability to father children. Talk to your healthcare provider if you have concerns about fertility. These are not all of the possible side effects of ENHERTU. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General information about the safe and effective use of ENHERTU.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about ENHERTU that is written for healthcare professionals. |
|||
|
What are the ingredients in ENHERTU?
Active Ingredient: fam-trastuzumab deruxtecan-nxki. Inactive Ingredients: L-histidine, L-histidine hydrochloride monohydrate, polysorbate 80, and sucrose. Manufactured by: Daiichi Sankyo, Inc., Basking Ridge, NJ 07920 U.S. License No. 2128 Marketed by: Daiichi Sankyo, Inc., Basking Ridge, NJ 07920 and AstraZeneca Pharmaceuticals LP, Wilmington, DE 19850 ENHERTU® is a registered trademark of Daiichi Sankyo Company, Ltd. © 2025 Daiichi Sankyo Co., Ltd. USMG-ENH-C19.1+DB09-1225-r009 For more information, call 1-877-437-7763 or go to https://www.ENHERTU.com. |
Section 43683-2
Section 44425-7
How Supplied/Storage
ENHERTU (fam-trastuzumab deruxtecan-nxki) for injection is a white to yellowish white lyophilized powder supplied as:
| Carton Contents | NDC |
|---|---|
| One 100 mg single-dose vial | NDC 65597-406-01 |
Store vials in a refrigerator at 2ºC to 8ºC (36ºF to 46ºF) in the original carton to protect from light until time of reconstitution. Do not freeze. Do not shake the reconstituted or diluted solution [see Dosage and Administration (2.4)].
15 References
- OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description
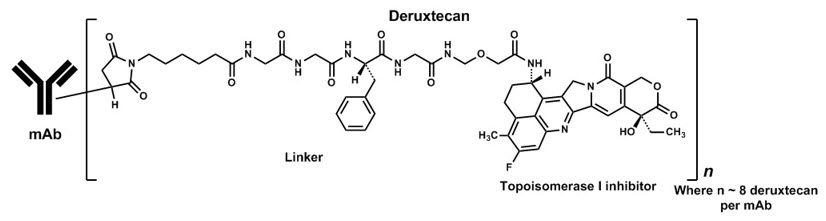
Fam-trastuzumab deruxtecan-nxki is a HER2-directed antibody and topoisomerase inhibitor conjugate. Fam-trastuzumab deruxtecan-nxki is an antibody-drug conjugate (ADC) composed of three components: 1) a humanized anti-HER2 IgG1 monoclonal antibody (mAb), covalently linked to 2) a topoisomerase inhibitor, via 3) a tetrapeptide-based cleavable linker. Deruxtecan is composed of a protease-cleavable maleimide tetrapeptide linker and the topoisomerase inhibitor, DXd, which is an exatecan derivative.
The antibody is produced in Chinese hamster ovary cells by recombinant DNA technology, and the topoisomerase inhibitor and linker are produced by chemical synthesis. Approximately 8 molecules of deruxtecan are attached to each antibody molecule. Fam-trastuzumab deruxtecan-nxki has the following structure:
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a sterile, white to yellowish white, preservative-free lyophilized powder in single-dose vials. Each vial delivers 100 mg of fam-trastuzumab deruxtecan-nxki, L-histidine (4.45 mg), L-histidine hydrochloride monohydrate (20.2 mg), polysorbate 80 (1.5 mg), and sucrose (450 mg). Following reconstitution with 5 mL of Sterile Water for Injection, USP, the resulting concentration of fam-trastuzumab deruxtecan-nxki is 20 mg/mL with a pH of 5.5. The resulting solution is administered by intravenous infusion following dilution.
5.2 Neutropenia
Severe neutropenia, including febrile neutropenia, can occur in patients treated with ENHERTU monotherapy or ENHERTU in combination with pertuzumab.
Monitor complete blood counts prior to initiation of ENHERTU and prior to each dose, and as clinically indicated. Based on the severity of neutropenia, ENHERTU may require dose interruption or reduction [see Dosage and Administration (2.3)].
8.4 Pediatric Use
Safety and effectiveness of ENHERTU have not been established in pediatric patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other studies, including those of fam-trastuzumab deruxtecan-nxki or of other fam-trastuzumab deruxtecan products.
Among patients who received ENHERTU as a single agent over a 6 to 9 month treatment period in 13 clinical trials, anti-fam-trastuzumab deruxtecan-nxki antibodies developed in 2.2% (49/2,231) of patients who received ENHERTU 5.4 mg/kg every three weeks and in 2.6% (21/793) of patients who received ENHERTU 6.4 mg/kg every three weeks. Among patients who received ENHERTU (5.4 mg/kg every three weeks) in combination with pertuzumab for a median of 16 months in 2 clinical trials, anti-fam-trastuzumab deruxtecan-nxki antibodies developed in 7.7% (33/426) of patients.
These anti-drug antibodies have no clinically significant effect on the pharmacokinetics or safety of fam-trastuzumab deruxtecan-nxki. Because of the low occurrence of anti-drug antibodies, the effect of antibodies on the effectiveness of fam-trastuzumab deruxtecan-nxki products is unknown.
Among patients with anti-drug antibodies, neutralizing antibodies against fam-trastuzumab deruxtecan-nxki were detected in 6% (4/70) of patients who received ENHERTU as a single agent (5.4 mg/kg or 6.4 mg/kg every 3 weeks) and in 18% (6/33) of patients who received ENHERTU (5.4 mg/kg every 3 weeks) in combination with pertuzumab.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1)]
- Neutropenia [see Warnings and Precautions (5.2)]
- Left Ventricular Dysfunction [see Warnings and Precautions (5.3)]
8.6 Renal Impairment
No dose adjustment of ENHERTU is required in patients with mild (creatinine clearance [CLcr] ≥60 and <90 mL/min) or moderate (CLcr ≥30 and <60 mL/min) renal impairment [see Clinical Pharmacology (12.3)]. A higher incidence of Grade 1 and 2 ILD/pneumonitis has been observed in patients with moderate renal impairment [see Warnings and Precautions (5.1)]. Monitor patients with moderate renal impairment more frequently. The recommended dosage of ENHERTU has not been established for patients with severe renal impairment (CLcr <30 mL/min) [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of fam-trastuzumab deruxtecan-nxki was evaluated in patients with cancer. Following a single dose, exposures (Cmax and AUC) of fam-trastuzumab deruxtecan-nxki and released topoisomerase inhibitor (DXd) increased proportionally over a dose range of 3.2 mg/kg to 8 mg/kg (approximately 0.6 to 1.5 times the recommended dose in breast cancer, NSCLC, and HER2-positive (IHC 3+) solid tumors and 0.5 to 1.25 times the recommended dose in gastric cancer).
At the recommended dosage of ENHERTU for patients with metastatic breast cancer, NSCLC, and HER2-positive (IHC 3+) solid tumors, the geometric mean (coefficient of variation [CV]%) Cmax of fam-trastuzumab deruxtecan-nxki and DXd were 132 µg/mL (20%) and 4.7 ng/mL (48%), respectively, and the AUC of fam-trastuzumab deruxtecan-nxki and DXd were 772 µg∙day/mL (27%) and 29 ng∙day/mL (48%), respectively. Accumulation of fam-trastuzumab deruxtecan-nxki was approximately 35% at steady-state (Cycle 3).
At the recommended dosage of ENHERTU for patients with HER2-positive gastric cancer, the geometric mean Cmax,ss of fam-trastuzumab deruxtecan-nxki and DXd were 126 µg/mL (18%) and 5.2 ng/mL (42%), respectively, and the AUCss of fam-trastuzumab deruxtecan-nxki and DXd were 743 µg∙day/mL (26%) and 33 ng∙day/mL (43%), respectively. Accumulation of fam-trastuzumab deruxtecan-nxki was approximately 39% at steady-state (Cycle 3).
8.7 Hepatic Impairment
No dose adjustment of ENHERTU is required in patients with mild (total bilirubin ≤ULN and any AST >ULN or total bilirubin >1 to 1.5 times ULN and any AST) or moderate (total bilirubin >1.5 to 3 times ULN and any AST) hepatic impairment. In patients with moderate hepatic impairment, due to potentially increased exposure, closely monitor for increased toxicities related to the topoisomerase inhibitor, DXd [see Dosage and Administration (2.3)]. The recommended dosage of ENHERTU has not been established for patients with severe hepatic impairment (total bilirubin >3 times ULN and any AST) [see Clinical Pharmacology (12.3)].
1 Indications and Usage
ENHERTU is a HER2-directed antibody and topoisomerase inhibitor conjugate indicated:
HER2-Positive Metastatic Breast Cancer
- in combination with pertuzumab as first-line treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer, as determined by an FDA-approved test. (1.1)
- as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer who have received a prior anti-HER2-based regimen either in the metastatic setting, or, in the neoadjuvant or adjuvant setting and have developed disease recurrence during or within six months of completing therapy. (1.1)
HER2-Low and HER2-Ultralow Metastatic Breast Cancer
- as monotherapy for the treatment of adult patients with unresectable or metastatic hormone receptor (HR)-positive, HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer, as determined by an FDA-approved test, that has progressed on one or more endocrine therapies in the metastatic setting. (1.2)
- as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer, as determined by an FDA-approved test, who have received a prior chemotherapy in the metastatic setting; or developed disease recurrence during or within 6 months of completing adjuvant chemotherapy. (1.2)
HER2-Mutant Unresectable or Metastatic Non-Small Cell Lung Cancer
- as monotherapy for the treatment of adult patients with unresectable or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating HER2 (ERBB2) mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy* (1.3)
HER2-Positive Locally Advanced or Metastatic Gastric Cancer
- as monotherapy for the treatment of adult patients with locally advanced or metastatic HER2-positive (IHC 3+ or IHC 2+/ISH positive) gastric or gastroesophageal junction adenocarcinoma who have received a prior trastuzumab-based regimen. (1.4)
HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors
- as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options* (1.5)
* These indications are approved under accelerated approval based on objective response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. (14.3, 14.5)
12.1 Mechanism of Action
Fam-trastuzumab deruxtecan-nxki is a HER2-directed antibody-drug conjugate. The antibody is a humanized anti-HER2 IgG1. The small molecule, DXd, is a topoisomerase I inhibitor attached to the antibody by a cleavable linker. Following binding to HER2 on tumor cells, fam-trastuzumab deruxtecan-nxki undergoes internalization and intracellular linker cleavage by lysosomal enzymes. Upon release, the membrane-permeable DXd causes DNA damage and apoptotic cell death.
2.3 Dosage Modifications
Management of adverse reactions may require temporary interruption, dose reduction, or treatment discontinuation of ENHERTU as described in Tables 2 and 3. Refer to the Prescribing Information for pertuzumab for dose modification recommendations. Pertuzumab is not to be administered as a single agent.
5.4 Embryo Fetal Toxicity
Based on its mechanism of action, ENHERTU can cause fetal harm when administered to a pregnant woman. In postmarketing reports, use of a HER2-directed antibody during pregnancy resulted in cases of oligohydramnios manifesting as fatal pulmonary hypoplasia, skeletal abnormalities, and neonatal death. Based on its mechanism of action, the topoisomerase inhibitor component of ENHERTU, DXd, can also cause embryo-fetal harm when administered to a pregnant woman because it is genotoxic and targets actively dividing cells [see Use in Specific Populations (8.1), Clinical Pharmacology (12.1), Nonclinical Toxicology (13.1)]. Advise patients of the potential risks to a fetus.
Verify the pregnancy status of females of reproductive potential prior to the initiation of ENHERTU. Advise females of reproductive potential to use effective contraception during treatment and for 7 months after the last dose of ENHERTU. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ENHERTU and for 4 months after the last dose of ENHERTU [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions
- Neutropenia: Monitor complete blood counts prior to initiation of ENHERTU and prior to each dose, and as clinically indicated. Manage through treatment interruption or dose reduction. (2.3, 5.2)
- Left Ventricular Dysfunction: Assess left ventricular ejection fraction (LVEF) prior to initiation of ENHERTU and at regular intervals during treatment as clinically indicated. Manage through treatment interruption or discontinuation. Permanently discontinue ENHERTU in patients with symptomatic congestive heart failure (CHF). (2.3, 5.3)
2 Dosage and Administration
- Do not substitute ENHERTU for or with trastuzumab or ado-trastuzumab emtansine. (2.2, 2.4)
- For intravenous infusion only. Do not administer as an intravenous push or bolus. DO NOT use Sodium Chloride Injection, USP. (2.4)
- Premedicate for prevention of chemotherapy-induced nausea and vomiting. (2.2)
- HER2-positive, HER2-low, or HER2-ultralow breast cancer, HER2-mutant NSCLC, and HER2-positive (IHC 3+) solid tumors: ENHERTU 5.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. (2.2, 2.3)
- HER2-Positive First-line Metastatic Breast Cancer ENHERTU 5.4 mg/kg every 3 weeks (in combination with pertuzumab until disease progression or unacceptable toxicity.
- HER2-positive gastric cancer: 6.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. (2.2, 2.3)
- Management of adverse reactions (ILD, neutropenia, thrombocytopenia, or left ventricular dysfunction) may require temporary interruption, dose reduction, or discontinuation of ENHERTU. (2.3)
3 Dosage Forms and Strengths
For injection: 100 mg of fam-trastuzumab deruxtecan-nxki as a white to yellowish white lyophilized powder in a single-dose vial for reconstitution and further dilution
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.3 Left Ventricular Dysfunction
Patients treated with ENHERTU may be at increased risk of developing left ventricular dysfunction. Left ventricular ejection fraction (LVEF) decrease has been observed with anti-HER2 therapies, including ENHERTU.
Assess LVEF prior to initiation of ENHERTU and at regular intervals during treatment as clinically indicated. Manage LVEF decrease through treatment interruption. Permanently discontinue ENHERTU if LVEF of less than 40% or absolute decrease from baseline of greater than 20% is confirmed. Permanently discontinue ENHERTU in patients with symptomatic congestive heart failure (CHF) [see Dosage and Administration (2.3)].
Treatment with ENHERTU has not been studied in patients with a history of clinically significant cardiac disease or LVEF less than 50% prior to initiation of treatment.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.4 Preparation and Administration
In order to prevent medication errors, check the vial labels to ensure that the drug being prepared and administered is ENHERTU (fam-trastuzumab deruxtecan-nxki) and not trastuzumab or ado-trastuzumab emtansine.
Reconstitute and further dilute ENHERTU prior to intravenous infusion. Use appropriate aseptic technique.
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a hazardous drug. Follow applicable special handling and disposal procedures.1
2.2 Recommended Dosage and Schedules
Do not substitute ENHERTU for or with trastuzumab or ado-trastuzumab emtansine.
Slow or interrupt the infusion rate if the patient develops infusion-related symptoms.
Permanently discontinue ENHERTU in case of severe infusion reactions.
5.1 Interstitial Lung Disease/pneumonitis
Severe, life-threatening, or fatal interstitial lung disease (ILD), including pneumonitis, can occur in patients treated with ENHERTU monotherapy or ENHERTU in combination with pertuzumab [see Adverse Reactions (6.1)]. A higher incidence of Grade 1 and 2 ILD/pneumonitis has been observed in patients with moderate renal impairment.
Advise patients to immediately report cough, dyspnea, fever, and/or any new or worsening respiratory symptoms. Monitor patients for signs and symptoms of ILD. Promptly investigate evidence of ILD. Evaluate patients with suspected ILD by radiographic imaging. Consider consultation with a pulmonologist. For asymptomatic (Grade 1) ILD, consider corticosteroid treatment (e.g., ≥0.5 mg/kg/day prednisolone or equivalent). Withhold ENHERTU until recovery [see Dosage and Administration (2.3)]. In cases of symptomatic ILD (Grade 2 or greater), promptly initiate systemic corticosteroid treatment (e.g., ≥1 mg/kg/day prednisolone or equivalent) and continue for at least 14 days followed by gradual taper for at least 4 weeks. Permanently discontinue ENHERTU in patients who are diagnosed with symptomatic (Grade 2 or greater) ILD [see Dosage and Administration (2.3)].
1.1 Her2 Positive Metastatic Breast Cancer
- ENHERTU, in combination with pertuzumab, is indicated for the first-line treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer, as determined by an FDA-approved test [see Dosage and Administration (2.1)].
- ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer who have received a prior anti-HER2-based regimen either in the metastatic setting, or, in the neoadjuvant or adjuvant setting and have developed disease recurrence during or within six months of completing therapy.
Principal Display Panel 100 Mg Vial Carton
NDC 65597-406-01
Rx only
ENHERTU®
(fam-trastuzumab deruxtecan-nxki)
For Injection
100 mg per vial
For Intravenous Infusion Only
Dispense the enclosed Medication Guide to each patient.
Reconstitute and Dilute prior to administration
Single-Dose Vial
Discard Unused Portion
Hazardous Drug
KEEP REFRIGERATED
1 vial
Daiichi-Sankyo
AstraZeneca
1.2 Her2 Low and Her2 Ultralow Metastatic Breast Cancer
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic
- Hormone receptor (HR)-positive HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer, as determined by an FDA-approved test, that has progressed on one or more endocrine therapies in the metastatic setting [see Dosage and Administration (2.1)].
- HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer, as determined by an FDA-approved test, who have received a prior chemotherapy in the metastatic setting or developed disease recurrence during or within 6 months of completing adjuvant chemotherapy [see Dosage and Administration (2.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with fam-trastuzumab deruxtecan-nxki.
The topoisomerase inhibitor component of fam-trastuzumab deruxtecan-nxki, DXd, was clastogenic in both an in vivo rat bone marrow micronucleus assay and an in vitro Chinese hamster lung chromosome aberration assay and was not mutagenic in an in vitro bacterial reverse mutation assay.
Fertility studies have not been conducted with fam-trastuzumab deruxtecan-nxki. In a six-week repeat-dose toxicity study in rats, intravenous administration of fam-trastuzumab deruxtecan-nxki resulted in spermatid retention at 20 mg/kg and 60 mg/kg (approximately 4 and 9 times the human recommended dose of 5.4 mg/kg based on AUC, respectively). Decreased testes and epididymides weights, tubular atrophy/degeneration in testes, and reduced sperm count in epididymides were observed at a dose of 197 mg/kg (19 times the human recommended dose of 5.4 mg/kg based on AUC). In a three-month repeat-dose toxicity study in monkeys, intravenous administration of fam-trastuzumab deruxtecan-nxki resulted in decreased numbers of round spermatids in the testes at seminiferous tubule stages V to VI at ≥30 mg/kg (≥7 times the human recommended dose of 5.4 mg/kg based on AUC). Evidence of reversibility was observed in monkeys by the end of a three-month recovery period.
Warning: Interstitial Lung Disease and Embryo Fetal Toxicity
- Interstitial Lung Disease (ILD) and pneumonitis, including fatal cases, have been reported with ENHERTU. Monitor for and promptly investigate signs and symptoms including cough, dyspnea, fever, and other new or worsening respiratory symptoms. Permanently discontinue ENHERTU in all patients with Grade 2 or higher ILD/pneumonitis. Advise patients of the risk and the need to immediately report symptoms [see Dosage and Administration (2.3), Warnings and Precautions (5.1)].
- Embryo-Fetal Toxicity: Exposure to ENHERTU during pregnancy can cause embryo-fetal harm. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.4), Use in Specific Populations (8.1, 8.3)].
1.4 Her2 Positive Locally Advanced Or Metastatic Gastric Cancer
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with locally advanced or metastatic HER2-positive (IHC 3+ or IHC 2+/ISH positive) gastric or gastroesophageal junction (GEJ) adenocarcinoma who have received a prior trastuzumab-based regimen.
14.4 Her2 Positive Locally Advanced Or Metastatic Gastric Cancer
The efficacy of ENHERTU was evaluated in study DESTINY-Gastric01 (NCT03329690), a multicenter, open-label, randomized trial conducted in Japan and South Korea that enrolled 188 adult patients with HER2-positive (IHC 3+ or IHC 2+/ISH positive), locally advanced or metastatic gastric or GEJ adenocarcinoma who had progressed on at least two prior regimens including trastuzumab, a fluoropyrimidine- and a platinum-containing chemotherapy. HER2 expression was determined by a central lab on tissue obtained either before or after prior trastuzumab treatment. Patients were excluded for a history of treated or current ILD, a history of clinically significant cardiac disease, active brain metastases, or ECOG performance status >1.
Patients were randomized 2:1 to receive ENHERTU (N=126) 6.4 mg/kg intravenously every 3 weeks or physician's choice of chemotherapy: irinotecan monotherapy (N=55) 150 mg/m2 intravenously every 2 weeks or paclitaxel monotherapy (N=7) 80 mg/m2 intravenously weekly. Randomization was stratified by HER2 status (IHC 3+ or IHC 2+/ISH+), ECOG performance status (0 or 1), and region (Japan or South Korea). Tumor imaging assessments were performed at screening and every 6 weeks from the first treatment dose. Treatment was administered until unacceptable toxicity or disease progression. The major efficacy outcomes were ORR assessed by ICR according to RECIST v1.1 and OS in the intent-to-treat population. Additional efficacy outcomes were PFS and DOR.
The median age was 66 years (range 28 to 82); 76% were male; and 100% were Asian. All patients received a trastuzumab product. Patients had an ECOG performance status of either 0 (49%) or 1 (51%); 87% had gastric adenocarcinoma and 13% had GEJ adenocarcinoma; 76% were IHC 3+ and 23% were IHC 2+/ISH+; 65% had inoperable advanced cancer; 35% had postoperative recurrent cancer; 54% had liver metastases; 29% had lung metastases; 45% had three or more prior regimens in the locally advanced or metastatic setting. A total of 30% of patients were identified as HER2-positive using tissue obtained following prior treatment with a trastuzumab product.
Efficacy results are summarized in Table 29, and the Kaplan-Meier curve for OS is shown in Figure 9.
| Efficacy Parameter | ENHERTU N=126 |
Irinotecan or Paclitaxel N=62 |
|---|---|---|
| CI = confidence interval; NR = not reached | ||
|
Overall Survival (OS) OS was evaluated following a statistically significant outcome of ORR.
|
||
| Median, months (95% CI) Median based on Kaplan-Meier estimate; 95% CI for median calculated using Brookmeyer-Crowley method
|
12.5 (9.6, 14.3) | 8.4 (6.9,10.7) |
| Hazard ratio (95% CI) Based on the stratified Cox proportional hazards regression model (stratified by region)
|
0.59 (0.39, 0.88) | |
| p-value Based on the stratified log-rank test (stratified by region)
|
0.0097 | |
|
Progression-Free Survival (PFS) Assessed by independent central review
|
||
| Median, months (95% CI) | 5.6 (4.3, 6.9) | 3.5 (2.0, 4.3) |
| Hazard ratio (95% CI) | 0.47 (0.31, 0.71) | |
| Confirmed Objective Response Rate (ORR) | ||
| n (%) | 51 (40.5) | 7 (11.3) |
| 95% CI 95% exact binomial confidence interval
|
(31.8, 49.6) | (4.7, 21.9) |
| p-value Based on the stratified Cochran-Mantel-Haenszel test (stratified by region)
|
<0.0001 | |
| Complete Response n (%) | 10 (7.9) | 0 (0.0) |
| Partial Response n (%) | 41 (32.5) | 7 (11.3) |
| Duration of Response (DOR) | ||
| Median, months (95% CI) | 11.3 (5.6, NR) | 3.9 (3.0, 4.9) |
| Figure 9: Kaplan-Meier Plot of Overall Survival |
|---|
1.5 Her2 Positive (ihc 3+) Unresectable Or Metastatic Solid Tumors
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options.
This indication is approved under accelerated approval based on objective response rate and duration of response [see Clinical Studies (14.5)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
14.5 Her2 Positive (ihc 3+) Unresectable Or Metastatic Solid Tumors
The efficacy of ENHERTU was evaluated in 192 adult patients with previously treated unresectable or metastatic HER2-positive (IHC 3+) solid tumors who were enrolled in one of three multicenter trials: DESTINY-PanTumor02, DESTINY-Lung01, and DESTINY-CRC02. All three studies excluded patients with a history of ILD/pneumonitis requiring treatment with steroids or ILD/pneumonitis at screening and clinically significant cardiac disease. Patients were also excluded for active brain metastases or ECOG performance status >1. Patients received ENHERTU 5.4 mg/kg by intravenous infusion every three weeks. Treatment was administered until disease progression, death, withdrawal of consent, or unacceptable toxicity. The major efficacy outcome measure in all three of the studies was confirmed objective response rate (ORR) and an additional efficacy outcome measure was duration of response (DOR). All outcomes were assessed by independent central review (ICR) based on RECIST v1.1.
1.3 Her2 Mutant Unresectable Or Metastatic Non Small Cell Lung Cancer
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating HER2 (ERBB2) mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy.
This indication is approved under accelerated approval based on objective response rate and duration of response [see Clinical Studies (14.3)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
14.3 Her2 Mutant Unresectable Or Metastatic Non Small Cell Lung Cancer
ENHERTU was evaluated in DESTINY-Lung01 (NCT03505710) and at two dose levels in DESTINY-Lung02 (NCT04644237). Patients were prospectively selected for treatment with ENHERTU based on the presence of activating HER2 (ERBB2) mutations by local testing using tissue. Samples from DESTINY-Lung01 were retrospectively tested using Oncomine™ Dx Target Test (Life Technologies Corporation, Tissue-test) and Guardant360® CDx test (Guardant Health Inc., Plasma test). Demographic and baseline disease characteristics were similar for patients in DESTINY-Lung01 and DESTINY-Lung02, except for race (34% Asian vs 79% Asian, respectively). Response rates were consistent across dose levels. Increased rates of ILD/pneumonitis were observed at the higher dose. The approved recommended dose of 5.4 mg/kg intravenously every 3 weeks in the DESTINY-Lung02 study is described below [see Adverse Reactions (6.1)].
The efficacy of ENHERTU was evaluated in DESTINY-Lung02, a multicenter, multicohort, randomized, blinded, dose-optimization trial. Eligible patients were required to have unresectable or metastatic HER2-mutant non-squamous NSCLC with disease progression after one prior systemic therapy. Patients with a history of steroid dependent ILD/pneumonitis, clinically significant cardiac disease, clinically active brain metastases, and ECOG performance status >1 were excluded. Patients received ENHERTU 5.4 mg/kg by intravenous infusion every 3 weeks until disease progression or unacceptable toxicity. Tumor imaging was obtained every 6 weeks and CT/MRI of the brain was mandatory for patients with stable brain metastases at baseline.
Results from an interim efficacy analysis in a pre-specified patient cohort are described below. The major efficacy outcomes were confirmed ORR as assessed by BICR using RECIST v1.1 and DOR.
The median age was 58 years (range 30 to 78); 69% were female; 79% were Asian, 12% were White, and 10% were other races; 29% had an ECOG performance status of 0 and 71% had 1; 33% had stable brain metastases; 94% had a mutation in the ERBB2 kinase domain and 6% had a mutation in the extracellular domain. The median number of prior regimens was 2 (range: 1 to 12); 100% of patients received prior platinum therapy, 71% received prior immunotherapy, and 44% received both in combination. Fifty percent of patients were never-smokers and 50% were former smokers; 96% of patients had adenocarcinoma histology.
Efficacy results are provided in Table 28.
| Efficacy Parameter | DESTINY-Lung02 N=52 |
|---|---|
| ORR 95% CI calculated using Clopper-Pearson method NE=not estimable |
|
| Confirmed Objective Response Rate (95% CI) | 57.7% (43.2, 71.3) |
| Complete Response | 1.9% |
| Partial Response | 55.8% |
|
Duration of Response
Median, months (95% CI) Median DOR based on Kaplan-Meier estimate; 95% CI calculated using Brookmeyer-Crowley method
|
8.7 (7.1, NE) |
Structured Label Content
Section 42229-5 (42229-5)
HER2-Positive Metastatic Breast Cancer
Select patients for treatment of unresectable or metastatic HER2-positive breast cancer with ENHERTU in combination with pertuzumab based on confirmed HER2-positive status or HER2 gene amplification (IHC 3+ or ISH+) [see Clinical Studies (14.1)].
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 12/2025 | ||
|
Medication Guide
ENHERTU® (en-HER-too) (fam-trastuzumab deruxtecan-nxki) for injection |
|||
|
What is the most important information I should know about ENHERTU? ENHERTU can cause serious side effects, including:
|
|||
|
|
||
Your healthcare provider will check you for these side effects during your treatment with ENHERTU. Your healthcare provider may reduce your dose, delay treatment or completely stop treatment with ENHERTU if you have severe side effects.
|
|||
|
What is ENHERTU?
ENHERTU is a prescription medicine used to treat adults who have:
|
|||
Before you receive ENHERTU, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
How will I receive ENHERTU?
|
|||
|
What are the possible side effects of ENHERTU? ENHERTU can cause serious side effects. See "What is the most important information I should know about ENHERTU?" The most common side effects of ENHERTU, when used in people with metastatic breast cancer, HER2-mutant non-small cell lung cancer, and other HER2-positive solid tumors include: |
|||
|
|
||
| The most common side effects of ENHERTU in combination with pertuzumab, when used in people with HER2-positive breast cancer include: | |||
|
|
||
| The most common side effects of ENHERTU, when used in people with HER2-positive gastric or GEJ adenocarcinoma, include: | |||
|
|
||
| ENHERTU may cause fertility problems in males, which may affect the ability to father children. Talk to your healthcare provider if you have concerns about fertility. These are not all of the possible side effects of ENHERTU. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
|
General information about the safe and effective use of ENHERTU.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. You can ask your pharmacist or healthcare provider for information about ENHERTU that is written for healthcare professionals. |
|||
|
What are the ingredients in ENHERTU?
Active Ingredient: fam-trastuzumab deruxtecan-nxki. Inactive Ingredients: L-histidine, L-histidine hydrochloride monohydrate, polysorbate 80, and sucrose. Manufactured by: Daiichi Sankyo, Inc., Basking Ridge, NJ 07920 U.S. License No. 2128 Marketed by: Daiichi Sankyo, Inc., Basking Ridge, NJ 07920 and AstraZeneca Pharmaceuticals LP, Wilmington, DE 19850 ENHERTU® is a registered trademark of Daiichi Sankyo Company, Ltd. © 2025 Daiichi Sankyo Co., Ltd. USMG-ENH-C19.1+DB09-1225-r009 For more information, call 1-877-437-7763 or go to https://www.ENHERTU.com. |
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
How Supplied/Storage
ENHERTU (fam-trastuzumab deruxtecan-nxki) for injection is a white to yellowish white lyophilized powder supplied as:
| Carton Contents | NDC |
|---|---|
| One 100 mg single-dose vial | NDC 65597-406-01 |
Store vials in a refrigerator at 2ºC to 8ºC (36ºF to 46ºF) in the original carton to protect from light until time of reconstitution. Do not freeze. Do not shake the reconstituted or diluted solution [see Dosage and Administration (2.4)].
15 References (15 REFERENCES)
- OSHA Hazardous Drugs. OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
11 Description (11 DESCRIPTION)
Fam-trastuzumab deruxtecan-nxki is a HER2-directed antibody and topoisomerase inhibitor conjugate. Fam-trastuzumab deruxtecan-nxki is an antibody-drug conjugate (ADC) composed of three components: 1) a humanized anti-HER2 IgG1 monoclonal antibody (mAb), covalently linked to 2) a topoisomerase inhibitor, via 3) a tetrapeptide-based cleavable linker. Deruxtecan is composed of a protease-cleavable maleimide tetrapeptide linker and the topoisomerase inhibitor, DXd, which is an exatecan derivative.
The antibody is produced in Chinese hamster ovary cells by recombinant DNA technology, and the topoisomerase inhibitor and linker are produced by chemical synthesis. Approximately 8 molecules of deruxtecan are attached to each antibody molecule. Fam-trastuzumab deruxtecan-nxki has the following structure:
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a sterile, white to yellowish white, preservative-free lyophilized powder in single-dose vials. Each vial delivers 100 mg of fam-trastuzumab deruxtecan-nxki, L-histidine (4.45 mg), L-histidine hydrochloride monohydrate (20.2 mg), polysorbate 80 (1.5 mg), and sucrose (450 mg). Following reconstitution with 5 mL of Sterile Water for Injection, USP, the resulting concentration of fam-trastuzumab deruxtecan-nxki is 20 mg/mL with a pH of 5.5. The resulting solution is administered by intravenous infusion following dilution.
5.2 Neutropenia
Severe neutropenia, including febrile neutropenia, can occur in patients treated with ENHERTU monotherapy or ENHERTU in combination with pertuzumab.
Monitor complete blood counts prior to initiation of ENHERTU and prior to each dose, and as clinically indicated. Based on the severity of neutropenia, ENHERTU may require dose interruption or reduction [see Dosage and Administration (2.3)].
8.4 Pediatric Use
Safety and effectiveness of ENHERTU have not been established in pediatric patients.
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other studies, including those of fam-trastuzumab deruxtecan-nxki or of other fam-trastuzumab deruxtecan products.
Among patients who received ENHERTU as a single agent over a 6 to 9 month treatment period in 13 clinical trials, anti-fam-trastuzumab deruxtecan-nxki antibodies developed in 2.2% (49/2,231) of patients who received ENHERTU 5.4 mg/kg every three weeks and in 2.6% (21/793) of patients who received ENHERTU 6.4 mg/kg every three weeks. Among patients who received ENHERTU (5.4 mg/kg every three weeks) in combination with pertuzumab for a median of 16 months in 2 clinical trials, anti-fam-trastuzumab deruxtecan-nxki antibodies developed in 7.7% (33/426) of patients.
These anti-drug antibodies have no clinically significant effect on the pharmacokinetics or safety of fam-trastuzumab deruxtecan-nxki. Because of the low occurrence of anti-drug antibodies, the effect of antibodies on the effectiveness of fam-trastuzumab deruxtecan-nxki products is unknown.
Among patients with anti-drug antibodies, neutralizing antibodies against fam-trastuzumab deruxtecan-nxki were detected in 6% (4/70) of patients who received ENHERTU as a single agent (5.4 mg/kg or 6.4 mg/kg every 3 weeks) and in 18% (6/33) of patients who received ENHERTU (5.4 mg/kg every 3 weeks) in combination with pertuzumab.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.1)]
- Neutropenia [see Warnings and Precautions (5.2)]
- Left Ventricular Dysfunction [see Warnings and Precautions (5.3)]
8.6 Renal Impairment
No dose adjustment of ENHERTU is required in patients with mild (creatinine clearance [CLcr] ≥60 and <90 mL/min) or moderate (CLcr ≥30 and <60 mL/min) renal impairment [see Clinical Pharmacology (12.3)]. A higher incidence of Grade 1 and 2 ILD/pneumonitis has been observed in patients with moderate renal impairment [see Warnings and Precautions (5.1)]. Monitor patients with moderate renal impairment more frequently. The recommended dosage of ENHERTU has not been established for patients with severe renal impairment (CLcr <30 mL/min) [see Clinical Pharmacology (12.3)].
12.3 Pharmacokinetics
The pharmacokinetics of fam-trastuzumab deruxtecan-nxki was evaluated in patients with cancer. Following a single dose, exposures (Cmax and AUC) of fam-trastuzumab deruxtecan-nxki and released topoisomerase inhibitor (DXd) increased proportionally over a dose range of 3.2 mg/kg to 8 mg/kg (approximately 0.6 to 1.5 times the recommended dose in breast cancer, NSCLC, and HER2-positive (IHC 3+) solid tumors and 0.5 to 1.25 times the recommended dose in gastric cancer).
At the recommended dosage of ENHERTU for patients with metastatic breast cancer, NSCLC, and HER2-positive (IHC 3+) solid tumors, the geometric mean (coefficient of variation [CV]%) Cmax of fam-trastuzumab deruxtecan-nxki and DXd were 132 µg/mL (20%) and 4.7 ng/mL (48%), respectively, and the AUC of fam-trastuzumab deruxtecan-nxki and DXd were 772 µg∙day/mL (27%) and 29 ng∙day/mL (48%), respectively. Accumulation of fam-trastuzumab deruxtecan-nxki was approximately 35% at steady-state (Cycle 3).
At the recommended dosage of ENHERTU for patients with HER2-positive gastric cancer, the geometric mean Cmax,ss of fam-trastuzumab deruxtecan-nxki and DXd were 126 µg/mL (18%) and 5.2 ng/mL (42%), respectively, and the AUCss of fam-trastuzumab deruxtecan-nxki and DXd were 743 µg∙day/mL (26%) and 33 ng∙day/mL (43%), respectively. Accumulation of fam-trastuzumab deruxtecan-nxki was approximately 39% at steady-state (Cycle 3).
8.7 Hepatic Impairment
No dose adjustment of ENHERTU is required in patients with mild (total bilirubin ≤ULN and any AST >ULN or total bilirubin >1 to 1.5 times ULN and any AST) or moderate (total bilirubin >1.5 to 3 times ULN and any AST) hepatic impairment. In patients with moderate hepatic impairment, due to potentially increased exposure, closely monitor for increased toxicities related to the topoisomerase inhibitor, DXd [see Dosage and Administration (2.3)]. The recommended dosage of ENHERTU has not been established for patients with severe hepatic impairment (total bilirubin >3 times ULN and any AST) [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
ENHERTU is a HER2-directed antibody and topoisomerase inhibitor conjugate indicated:
HER2-Positive Metastatic Breast Cancer
- in combination with pertuzumab as first-line treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer, as determined by an FDA-approved test. (1.1)
- as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer who have received a prior anti-HER2-based regimen either in the metastatic setting, or, in the neoadjuvant or adjuvant setting and have developed disease recurrence during or within six months of completing therapy. (1.1)
HER2-Low and HER2-Ultralow Metastatic Breast Cancer
- as monotherapy for the treatment of adult patients with unresectable or metastatic hormone receptor (HR)-positive, HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer, as determined by an FDA-approved test, that has progressed on one or more endocrine therapies in the metastatic setting. (1.2)
- as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer, as determined by an FDA-approved test, who have received a prior chemotherapy in the metastatic setting; or developed disease recurrence during or within 6 months of completing adjuvant chemotherapy. (1.2)
HER2-Mutant Unresectable or Metastatic Non-Small Cell Lung Cancer
- as monotherapy for the treatment of adult patients with unresectable or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating HER2 (ERBB2) mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy* (1.3)
HER2-Positive Locally Advanced or Metastatic Gastric Cancer
- as monotherapy for the treatment of adult patients with locally advanced or metastatic HER2-positive (IHC 3+ or IHC 2+/ISH positive) gastric or gastroesophageal junction adenocarcinoma who have received a prior trastuzumab-based regimen. (1.4)
HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors
- as monotherapy for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options* (1.5)
* These indications are approved under accelerated approval based on objective response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial. (14.3, 14.5)
12.1 Mechanism of Action
Fam-trastuzumab deruxtecan-nxki is a HER2-directed antibody-drug conjugate. The antibody is a humanized anti-HER2 IgG1. The small molecule, DXd, is a topoisomerase I inhibitor attached to the antibody by a cleavable linker. Following binding to HER2 on tumor cells, fam-trastuzumab deruxtecan-nxki undergoes internalization and intracellular linker cleavage by lysosomal enzymes. Upon release, the membrane-permeable DXd causes DNA damage and apoptotic cell death.
2.3 Dosage Modifications
Management of adverse reactions may require temporary interruption, dose reduction, or treatment discontinuation of ENHERTU as described in Tables 2 and 3. Refer to the Prescribing Information for pertuzumab for dose modification recommendations. Pertuzumab is not to be administered as a single agent.
5.4 Embryo Fetal Toxicity (5.4 Embryo-Fetal Toxicity)
Based on its mechanism of action, ENHERTU can cause fetal harm when administered to a pregnant woman. In postmarketing reports, use of a HER2-directed antibody during pregnancy resulted in cases of oligohydramnios manifesting as fatal pulmonary hypoplasia, skeletal abnormalities, and neonatal death. Based on its mechanism of action, the topoisomerase inhibitor component of ENHERTU, DXd, can also cause embryo-fetal harm when administered to a pregnant woman because it is genotoxic and targets actively dividing cells [see Use in Specific Populations (8.1), Clinical Pharmacology (12.1), Nonclinical Toxicology (13.1)]. Advise patients of the potential risks to a fetus.
Verify the pregnancy status of females of reproductive potential prior to the initiation of ENHERTU. Advise females of reproductive potential to use effective contraception during treatment and for 7 months after the last dose of ENHERTU. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ENHERTU and for 4 months after the last dose of ENHERTU [see Use in Specific Populations (8.1, 8.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Neutropenia: Monitor complete blood counts prior to initiation of ENHERTU and prior to each dose, and as clinically indicated. Manage through treatment interruption or dose reduction. (2.3, 5.2)
- Left Ventricular Dysfunction: Assess left ventricular ejection fraction (LVEF) prior to initiation of ENHERTU and at regular intervals during treatment as clinically indicated. Manage through treatment interruption or discontinuation. Permanently discontinue ENHERTU in patients with symptomatic congestive heart failure (CHF). (2.3, 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Do not substitute ENHERTU for or with trastuzumab or ado-trastuzumab emtansine. (2.2, 2.4)
- For intravenous infusion only. Do not administer as an intravenous push or bolus. DO NOT use Sodium Chloride Injection, USP. (2.4)
- Premedicate for prevention of chemotherapy-induced nausea and vomiting. (2.2)
- HER2-positive, HER2-low, or HER2-ultralow breast cancer, HER2-mutant NSCLC, and HER2-positive (IHC 3+) solid tumors: ENHERTU 5.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. (2.2, 2.3)
- HER2-Positive First-line Metastatic Breast Cancer ENHERTU 5.4 mg/kg every 3 weeks (in combination with pertuzumab until disease progression or unacceptable toxicity.
- HER2-positive gastric cancer: 6.4 mg/kg every 3 weeks until disease progression or unacceptable toxicity. (2.2, 2.3)
- Management of adverse reactions (ILD, neutropenia, thrombocytopenia, or left ventricular dysfunction) may require temporary interruption, dose reduction, or discontinuation of ENHERTU. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
For injection: 100 mg of fam-trastuzumab deruxtecan-nxki as a white to yellowish white lyophilized powder in a single-dose vial for reconstitution and further dilution
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
5.3 Left Ventricular Dysfunction
Patients treated with ENHERTU may be at increased risk of developing left ventricular dysfunction. Left ventricular ejection fraction (LVEF) decrease has been observed with anti-HER2 therapies, including ENHERTU.
Assess LVEF prior to initiation of ENHERTU and at regular intervals during treatment as clinically indicated. Manage LVEF decrease through treatment interruption. Permanently discontinue ENHERTU if LVEF of less than 40% or absolute decrease from baseline of greater than 20% is confirmed. Permanently discontinue ENHERTU in patients with symptomatic congestive heart failure (CHF) [see Dosage and Administration (2.3)].
Treatment with ENHERTU has not been studied in patients with a history of clinically significant cardiac disease or LVEF less than 50% prior to initiation of treatment.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
2.4 Preparation and Administration
In order to prevent medication errors, check the vial labels to ensure that the drug being prepared and administered is ENHERTU (fam-trastuzumab deruxtecan-nxki) and not trastuzumab or ado-trastuzumab emtansine.
Reconstitute and further dilute ENHERTU prior to intravenous infusion. Use appropriate aseptic technique.
ENHERTU (fam-trastuzumab deruxtecan-nxki) is a hazardous drug. Follow applicable special handling and disposal procedures.1
2.2 Recommended Dosage and Schedules
Do not substitute ENHERTU for or with trastuzumab or ado-trastuzumab emtansine.
Slow or interrupt the infusion rate if the patient develops infusion-related symptoms.
Permanently discontinue ENHERTU in case of severe infusion reactions.
5.1 Interstitial Lung Disease/pneumonitis (5.1 Interstitial Lung Disease/Pneumonitis)
Severe, life-threatening, or fatal interstitial lung disease (ILD), including pneumonitis, can occur in patients treated with ENHERTU monotherapy or ENHERTU in combination with pertuzumab [see Adverse Reactions (6.1)]. A higher incidence of Grade 1 and 2 ILD/pneumonitis has been observed in patients with moderate renal impairment.
Advise patients to immediately report cough, dyspnea, fever, and/or any new or worsening respiratory symptoms. Monitor patients for signs and symptoms of ILD. Promptly investigate evidence of ILD. Evaluate patients with suspected ILD by radiographic imaging. Consider consultation with a pulmonologist. For asymptomatic (Grade 1) ILD, consider corticosteroid treatment (e.g., ≥0.5 mg/kg/day prednisolone or equivalent). Withhold ENHERTU until recovery [see Dosage and Administration (2.3)]. In cases of symptomatic ILD (Grade 2 or greater), promptly initiate systemic corticosteroid treatment (e.g., ≥1 mg/kg/day prednisolone or equivalent) and continue for at least 14 days followed by gradual taper for at least 4 weeks. Permanently discontinue ENHERTU in patients who are diagnosed with symptomatic (Grade 2 or greater) ILD [see Dosage and Administration (2.3)].
1.1 Her2 Positive Metastatic Breast Cancer (1.1 HER2-Positive Metastatic Breast Cancer)
- ENHERTU, in combination with pertuzumab, is indicated for the first-line treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer, as determined by an FDA-approved test [see Dosage and Administration (2.1)].
- ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+ or ISH+) breast cancer who have received a prior anti-HER2-based regimen either in the metastatic setting, or, in the neoadjuvant or adjuvant setting and have developed disease recurrence during or within six months of completing therapy.
Principal Display Panel 100 Mg Vial Carton (PRINCIPAL DISPLAY PANEL - 100 mg Vial Carton)
NDC 65597-406-01
Rx only
ENHERTU®
(fam-trastuzumab deruxtecan-nxki)
For Injection
100 mg per vial
For Intravenous Infusion Only
Dispense the enclosed Medication Guide to each patient.
Reconstitute and Dilute prior to administration
Single-Dose Vial
Discard Unused Portion
Hazardous Drug
KEEP REFRIGERATED
1 vial
Daiichi-Sankyo
AstraZeneca
1.2 Her2 Low and Her2 Ultralow Metastatic Breast Cancer (1.2 HER2-Low and HER2-Ultralow Metastatic Breast Cancer)
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic
- Hormone receptor (HR)-positive HER2-low (IHC 1+ or IHC 2+/ISH-) or HER2-ultralow (IHC 0 with membrane staining) breast cancer, as determined by an FDA-approved test, that has progressed on one or more endocrine therapies in the metastatic setting [see Dosage and Administration (2.1)].
- HER2-low (IHC 1+ or IHC 2+/ISH-) breast cancer, as determined by an FDA-approved test, who have received a prior chemotherapy in the metastatic setting or developed disease recurrence during or within 6 months of completing adjuvant chemotherapy [see Dosage and Administration (2.1)].
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with fam-trastuzumab deruxtecan-nxki.
The topoisomerase inhibitor component of fam-trastuzumab deruxtecan-nxki, DXd, was clastogenic in both an in vivo rat bone marrow micronucleus assay and an in vitro Chinese hamster lung chromosome aberration assay and was not mutagenic in an in vitro bacterial reverse mutation assay.
Fertility studies have not been conducted with fam-trastuzumab deruxtecan-nxki. In a six-week repeat-dose toxicity study in rats, intravenous administration of fam-trastuzumab deruxtecan-nxki resulted in spermatid retention at 20 mg/kg and 60 mg/kg (approximately 4 and 9 times the human recommended dose of 5.4 mg/kg based on AUC, respectively). Decreased testes and epididymides weights, tubular atrophy/degeneration in testes, and reduced sperm count in epididymides were observed at a dose of 197 mg/kg (19 times the human recommended dose of 5.4 mg/kg based on AUC). In a three-month repeat-dose toxicity study in monkeys, intravenous administration of fam-trastuzumab deruxtecan-nxki resulted in decreased numbers of round spermatids in the testes at seminiferous tubule stages V to VI at ≥30 mg/kg (≥7 times the human recommended dose of 5.4 mg/kg based on AUC). Evidence of reversibility was observed in monkeys by the end of a three-month recovery period.
Warning: Interstitial Lung Disease and Embryo Fetal Toxicity (WARNING: INTERSTITIAL LUNG DISEASE and EMBRYO-FETAL TOXICITY)
- Interstitial Lung Disease (ILD) and pneumonitis, including fatal cases, have been reported with ENHERTU. Monitor for and promptly investigate signs and symptoms including cough, dyspnea, fever, and other new or worsening respiratory symptoms. Permanently discontinue ENHERTU in all patients with Grade 2 or higher ILD/pneumonitis. Advise patients of the risk and the need to immediately report symptoms [see Dosage and Administration (2.3), Warnings and Precautions (5.1)].
- Embryo-Fetal Toxicity: Exposure to ENHERTU during pregnancy can cause embryo-fetal harm. Advise patients of these risks and the need for effective contraception [see Warnings and Precautions (5.4), Use in Specific Populations (8.1, 8.3)].
1.4 Her2 Positive Locally Advanced Or Metastatic Gastric Cancer (1.4 HER2-Positive Locally Advanced or Metastatic Gastric Cancer)
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with locally advanced or metastatic HER2-positive (IHC 3+ or IHC 2+/ISH positive) gastric or gastroesophageal junction (GEJ) adenocarcinoma who have received a prior trastuzumab-based regimen.
14.4 Her2 Positive Locally Advanced Or Metastatic Gastric Cancer (14.4 HER2-Positive Locally Advanced or Metastatic Gastric Cancer)
The efficacy of ENHERTU was evaluated in study DESTINY-Gastric01 (NCT03329690), a multicenter, open-label, randomized trial conducted in Japan and South Korea that enrolled 188 adult patients with HER2-positive (IHC 3+ or IHC 2+/ISH positive), locally advanced or metastatic gastric or GEJ adenocarcinoma who had progressed on at least two prior regimens including trastuzumab, a fluoropyrimidine- and a platinum-containing chemotherapy. HER2 expression was determined by a central lab on tissue obtained either before or after prior trastuzumab treatment. Patients were excluded for a history of treated or current ILD, a history of clinically significant cardiac disease, active brain metastases, or ECOG performance status >1.
Patients were randomized 2:1 to receive ENHERTU (N=126) 6.4 mg/kg intravenously every 3 weeks or physician's choice of chemotherapy: irinotecan monotherapy (N=55) 150 mg/m2 intravenously every 2 weeks or paclitaxel monotherapy (N=7) 80 mg/m2 intravenously weekly. Randomization was stratified by HER2 status (IHC 3+ or IHC 2+/ISH+), ECOG performance status (0 or 1), and region (Japan or South Korea). Tumor imaging assessments were performed at screening and every 6 weeks from the first treatment dose. Treatment was administered until unacceptable toxicity or disease progression. The major efficacy outcomes were ORR assessed by ICR according to RECIST v1.1 and OS in the intent-to-treat population. Additional efficacy outcomes were PFS and DOR.
The median age was 66 years (range 28 to 82); 76% were male; and 100% were Asian. All patients received a trastuzumab product. Patients had an ECOG performance status of either 0 (49%) or 1 (51%); 87% had gastric adenocarcinoma and 13% had GEJ adenocarcinoma; 76% were IHC 3+ and 23% were IHC 2+/ISH+; 65% had inoperable advanced cancer; 35% had postoperative recurrent cancer; 54% had liver metastases; 29% had lung metastases; 45% had three or more prior regimens in the locally advanced or metastatic setting. A total of 30% of patients were identified as HER2-positive using tissue obtained following prior treatment with a trastuzumab product.
Efficacy results are summarized in Table 29, and the Kaplan-Meier curve for OS is shown in Figure 9.
| Efficacy Parameter | ENHERTU N=126 |
Irinotecan or Paclitaxel N=62 |
|---|---|---|
| CI = confidence interval; NR = not reached | ||
|
Overall Survival (OS) OS was evaluated following a statistically significant outcome of ORR.
|
||
| Median, months (95% CI) Median based on Kaplan-Meier estimate; 95% CI for median calculated using Brookmeyer-Crowley method
|
12.5 (9.6, 14.3) | 8.4 (6.9,10.7) |
| Hazard ratio (95% CI) Based on the stratified Cox proportional hazards regression model (stratified by region)
|
0.59 (0.39, 0.88) | |
| p-value Based on the stratified log-rank test (stratified by region)
|
0.0097 | |
|
Progression-Free Survival (PFS) Assessed by independent central review
|
||
| Median, months (95% CI) | 5.6 (4.3, 6.9) | 3.5 (2.0, 4.3) |
| Hazard ratio (95% CI) | 0.47 (0.31, 0.71) | |
| Confirmed Objective Response Rate (ORR) | ||
| n (%) | 51 (40.5) | 7 (11.3) |
| 95% CI 95% exact binomial confidence interval
|
(31.8, 49.6) | (4.7, 21.9) |
| p-value Based on the stratified Cochran-Mantel-Haenszel test (stratified by region)
|
<0.0001 | |
| Complete Response n (%) | 10 (7.9) | 0 (0.0) |
| Partial Response n (%) | 41 (32.5) | 7 (11.3) |
| Duration of Response (DOR) | ||
| Median, months (95% CI) | 11.3 (5.6, NR) | 3.9 (3.0, 4.9) |
| Figure 9: Kaplan-Meier Plot of Overall Survival |
|---|
1.5 Her2 Positive (ihc 3+) Unresectable Or Metastatic Solid Tumors (1.5 HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors)
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic HER2-positive (IHC 3+) solid tumors who have received prior systemic treatment and have no satisfactory alternative treatment options.
This indication is approved under accelerated approval based on objective response rate and duration of response [see Clinical Studies (14.5)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
14.5 Her2 Positive (ihc 3+) Unresectable Or Metastatic Solid Tumors (14.5 HER2-Positive (IHC 3+) Unresectable or Metastatic Solid Tumors)
The efficacy of ENHERTU was evaluated in 192 adult patients with previously treated unresectable or metastatic HER2-positive (IHC 3+) solid tumors who were enrolled in one of three multicenter trials: DESTINY-PanTumor02, DESTINY-Lung01, and DESTINY-CRC02. All three studies excluded patients with a history of ILD/pneumonitis requiring treatment with steroids or ILD/pneumonitis at screening and clinically significant cardiac disease. Patients were also excluded for active brain metastases or ECOG performance status >1. Patients received ENHERTU 5.4 mg/kg by intravenous infusion every three weeks. Treatment was administered until disease progression, death, withdrawal of consent, or unacceptable toxicity. The major efficacy outcome measure in all three of the studies was confirmed objective response rate (ORR) and an additional efficacy outcome measure was duration of response (DOR). All outcomes were assessed by independent central review (ICR) based on RECIST v1.1.
1.3 Her2 Mutant Unresectable Or Metastatic Non Small Cell Lung Cancer (1.3 HER2-Mutant Unresectable or Metastatic Non-Small Cell Lung Cancer)
ENHERTU, as monotherapy, is indicated for the treatment of adult patients with unresectable or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating HER2 (ERBB2) mutations, as detected by an FDA-approved test, and who have received a prior systemic therapy.
This indication is approved under accelerated approval based on objective response rate and duration of response [see Clinical Studies (14.3)]. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory trial.
14.3 Her2 Mutant Unresectable Or Metastatic Non Small Cell Lung Cancer (14.3 HER2-Mutant Unresectable or Metastatic Non-Small Cell Lung Cancer)
ENHERTU was evaluated in DESTINY-Lung01 (NCT03505710) and at two dose levels in DESTINY-Lung02 (NCT04644237). Patients were prospectively selected for treatment with ENHERTU based on the presence of activating HER2 (ERBB2) mutations by local testing using tissue. Samples from DESTINY-Lung01 were retrospectively tested using Oncomine™ Dx Target Test (Life Technologies Corporation, Tissue-test) and Guardant360® CDx test (Guardant Health Inc., Plasma test). Demographic and baseline disease characteristics were similar for patients in DESTINY-Lung01 and DESTINY-Lung02, except for race (34% Asian vs 79% Asian, respectively). Response rates were consistent across dose levels. Increased rates of ILD/pneumonitis were observed at the higher dose. The approved recommended dose of 5.4 mg/kg intravenously every 3 weeks in the DESTINY-Lung02 study is described below [see Adverse Reactions (6.1)].
The efficacy of ENHERTU was evaluated in DESTINY-Lung02, a multicenter, multicohort, randomized, blinded, dose-optimization trial. Eligible patients were required to have unresectable or metastatic HER2-mutant non-squamous NSCLC with disease progression after one prior systemic therapy. Patients with a history of steroid dependent ILD/pneumonitis, clinically significant cardiac disease, clinically active brain metastases, and ECOG performance status >1 were excluded. Patients received ENHERTU 5.4 mg/kg by intravenous infusion every 3 weeks until disease progression or unacceptable toxicity. Tumor imaging was obtained every 6 weeks and CT/MRI of the brain was mandatory for patients with stable brain metastases at baseline.
Results from an interim efficacy analysis in a pre-specified patient cohort are described below. The major efficacy outcomes were confirmed ORR as assessed by BICR using RECIST v1.1 and DOR.
The median age was 58 years (range 30 to 78); 69% were female; 79% were Asian, 12% were White, and 10% were other races; 29% had an ECOG performance status of 0 and 71% had 1; 33% had stable brain metastases; 94% had a mutation in the ERBB2 kinase domain and 6% had a mutation in the extracellular domain. The median number of prior regimens was 2 (range: 1 to 12); 100% of patients received prior platinum therapy, 71% received prior immunotherapy, and 44% received both in combination. Fifty percent of patients were never-smokers and 50% were former smokers; 96% of patients had adenocarcinoma histology.
Efficacy results are provided in Table 28.
| Efficacy Parameter | DESTINY-Lung02 N=52 |
|---|---|
| ORR 95% CI calculated using Clopper-Pearson method NE=not estimable |
|
| Confirmed Objective Response Rate (95% CI) | 57.7% (43.2, 71.3) |
| Complete Response | 1.9% |
| Partial Response | 55.8% |
|
Duration of Response
Median, months (95% CI) Median DOR based on Kaplan-Meier estimate; 95% CI calculated using Brookmeyer-Crowley method
|
8.7 (7.1, NE) |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:37.060408 · Updated: 2026-03-14T22:41:00.111917