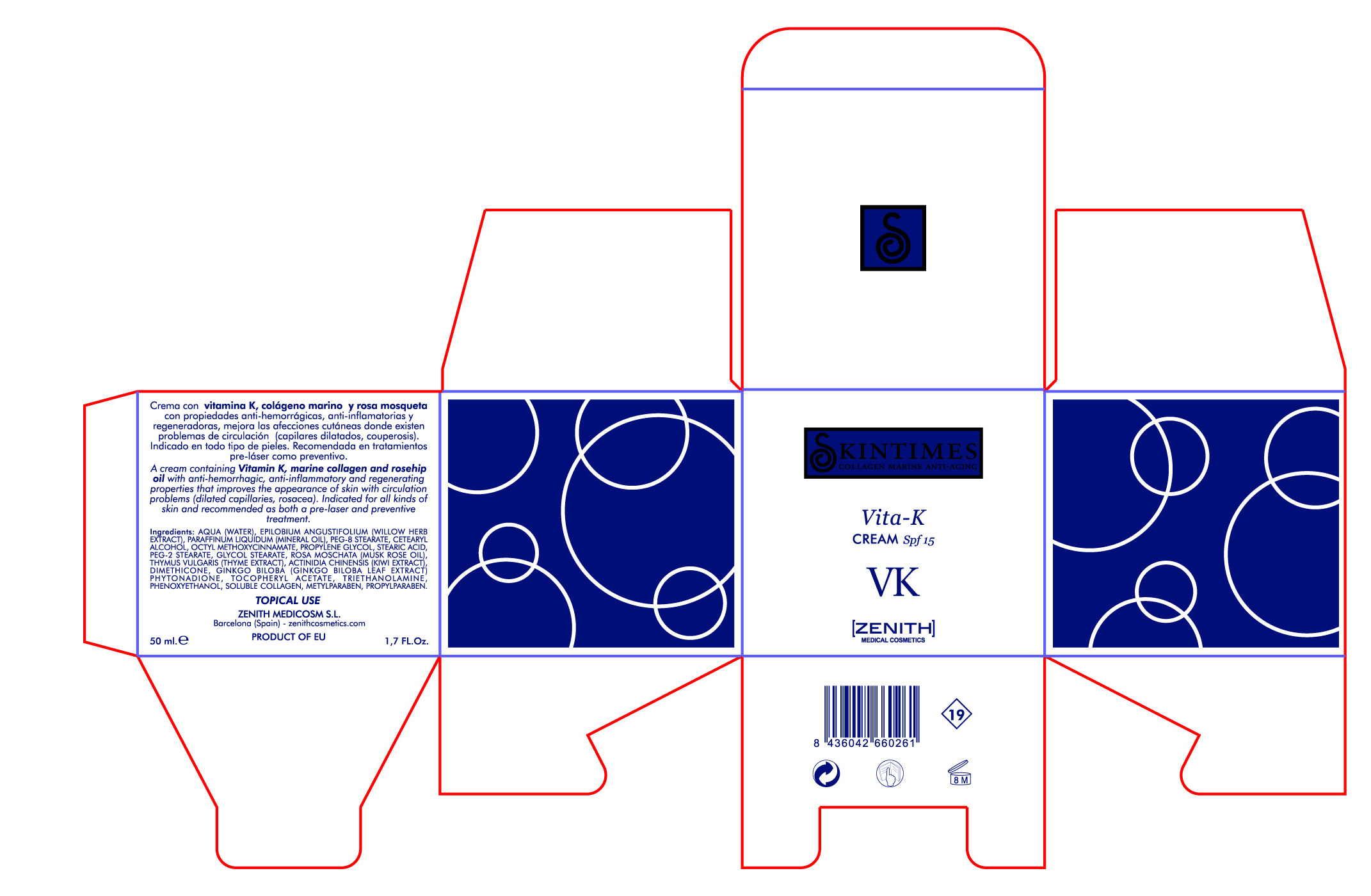
[{"name": "PEG-2 STEARATE", "unii": "94YQ11Y95F", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PROPYLPARABEN", "unii": "Z8IX2SC1OH", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALPHA-TOCOPHEROL ACETATE", "unii": "9E8X80D2L0", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "DIMETHICONE", "unii": "92RU3N3Y1O", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCOL STEARATE", "unii": "0324G66D0E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ROSA MOSCHATA OIL", "unii": "J99W255AWF", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "MINERAL OIL", "unii": "T5L8T28FGP", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CETOSTEARYL ALCOHOL", "unii": "2DMT128M1S", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PEG-8 STEARATE", "unii": "2P9L47VI5E", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TROLAMINE", "unii": "9O3K93S3TK", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "THYME", "unii": "CW657OBU4N", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "KIWI FRUIT", "unii": "71ES77LGJC", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PHYTONADIONE", "unii": "A034SE7857", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "METHYLPARABEN", "unii": "A2I8C7HI9T", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "COLLAGEN, SOLUBLE, FISH SKIN", "unii": "8JC99XGU4W", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "OCTINOXATE", "unii": "4Y5P7MUD51", "classCode": "ACTIB", "strengthNumerator": "3.75", "strengthDenominator": "50", "strengthNumeratorUnit": "mL", "strengthDenominatorUnit": "mL"}, {"name": "STEARIC ACID", "unii": "4ELV7Z65AP", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PROPYLENE GLYCOL", "unii": "6DC9Q167V3", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "EPILOBIUM ANGUSTIFOLIUM LEAF", "unii": "7NV86426N2", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GINKGO", "unii": "19FUJ2C58T", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PHENOXYETHANOL", "unii": "HIE492ZZ3T", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}]